Abstract
Presented is a series of narrative reviews that summarize published information regarding the effect or potential effect of interfering substances on the accuracy of continuous glucose monitoring (CGM) devices. While drawing together what is currently known regarding this topic, the future direction in this field and clinical implications posed by polypharmacy on CGM performance are considered. This first in a series of four review articles classifies commercially available CGMs by glucose measurement principle before reviewing what is currently known regarding substance interference mechanisms and design approaches that may serve to reduce interfering effects. Points covered include the following: minimally invasive electrochemical CGMs, which may be classified by first-, second-, or third-generational design (these models are at risk of interference from electroactive substances, or substances that can interfere with the enzymatic biorecognition process); non-invasive fluid sampling CGMs, which draw glucose across the skin barrier but are similarly reliant on the electrochemical measurement of an enzymatic reaction product; and minimally invasive implantable CGMs, which exhibit different interfering substance behaviors to other CGM classes, using a non-enzyme-based glucose-recognition agent coupled to optical detection. An understanding of substance-interfering mechanisms allows consideration of the potential impact on clinical accuracy of substances that are routinely prescribed, can be purchased over the counter, or are new to market.
Introduction
The magnitude and directionality of interfering substance contributions to CGM readings has implications for people who rely on glucose readings for diabetes management, but especially so for people who require insulin, many of whom may also be taking multiple medications. With an incomplete knowledge of the effect of interfering substances on device performance, management decisions and insulin dosing calculations may be based on potentially inaccurate readings. This is a particular issue in automated insulin delivery systems, in which control algorithms make autonomous insulin dosing decisions based on CGM data.
Commentators have highlighted the issue of incomplete information regarding interfering substance effects on CGM performance, despite manufacturers being required to perform interference studies during product development and system design verification. 1 This deficiency is partly attributable to the fact that most regulator-approved CGM devices operate within interstitial fluid (ISF), a medium that is challenging to extract at sufficient volumes to allow the quantification of interfering substances. Therefore, despite CGMs being in market for more than two decades, knowledge regarding the identity of many potentially interfering substances, and their effect on CGM clinical accuracy, remains limited.
Sample availability is less of an issue with blood glucose monitoring (BGM) systems—episodic measurement devices that have been established in market for more than four decades. The larger volume blood samples used for reference measurements, whether venous, capillary, or arterial, can be accessed with relative ease. BGM guidance has evolved over many years, with the establishment of clear guidelines from both regulators and national/international bodies regarding standardized test procedures and acceptance criteria for defining interfering substance effects on BGM systems. Of particular importance are the US Food and Drug Administration (FDA) guidance on over-the-counter self-monitoring blood glucose test systems, 2 the International Standards Organization (ISO) 15197 In vitro diagnostic test systems requirements for BGM devices 3 and the Clinical and Laboratory Standards Institute (CLSI) EP07 Interference Testing in Clinical Chemistry guidelines. 4 For example, ISO 15197:2013 considers that instructions for use should report a substance as interfering when the in vitro testing of a blood sample containing a high therapeutic/physiological level of the substance, results in an average glucose bias between the test and control sample that exceeds ±0.55 mmol/L (±10 mg/dL) at glucose concentrations of <5.55 mmol/L (<100 mg/dL), or exceeds ±10% at glucose concentrations of ≥5.55 mmol/L (≥100 mg/dL).
These BGM guidelines and standards require the manufacturer to determine the effect of potentially interfering endogenous and exogenous substances, including common medications, on device performance, with lists of known or potentially interfering substances described, alongside requirements to undertake an interference risk analysis. CLSI guidelines require consideration of the patient specimen, chemical knowledge of the procedure and intended use of the device. CLSI also publishes supplemental tables listing potential interfering substances and suggested test concentrations. 5 CLSI POCT05—Performance Metrics for Continuous Interstitial Glucose Monitoring recognizes the potential effect of interfering substances on CGM performance but does not provide detailed guidance regarding recommended CGM performance evaluation. 6
In recognition of this, the International Federation of Clinical Chemists (IFCC) has created a workgroup to address current deficiencies in CGM legislation (IFSS WG-CGM). The scope includes (1) the establishment of metrics for the evaluation of the analytical performance of CGMs; (2) to work with ISO on a new CGM guideline, analogous to ISO 15197, for the establishment of standardized procedures and acceptance criteria for CGM; and (3) the establishment of metrological traceability. 7 While this working group has a broad remit, they have identified the need for CGM study designs to encompass inclusion/exclusion criteria for possible interfering medications. 8
This review focuses on regulatory-approved products in major markets at the time of writing, which includes the market-dominant minimally invasive electrochemical CGMs and the minimally invasive implantable Senseonics Eversense CGM design (Senseonics Inc., Germantown, MD). One non-invasive fluid sampling CGM, the Nemaura SugarBEAT (Nemaura Medical, Loughborough, UK), is also in market and will be briefly reviewed. References to historical systems are made where the underpinning glucose measurement mechanism provides insight into potential interfering mechanisms that may be common to currently marketed devices. This review contains a brief introduction to minimally invasive first-, second-, and third-generation electrochemical CGMs, implantable CGMs, and non-invasive fluid sampling CGMs. The mechanisms by which interfering substances may potentially affect these different types of CGM devices are reviewed, as are the main CGM design factors that have been implemented in efforts to reduce interfering substance effects. In parts 2 and 3 of this review series, the main interfering substances that have been associated with the market-leading CGMs are reviewed, as are other classes of substances that have been identified as influencing CGM readings. Part 4 includes a tabular summary of interfering substances that have been identified as influencing the performance of CGM systems and concludes with a discussion on the implications of these findings for real-world usage of CGMs and the need for standardization. Sources of CGM interference that are not substance-specific, such as environmental factors, radiologic, electronic, or human-body-mediated (tissue growth, sensor surface blockage/biofouling, immunologic), are broadly outside the scope of this review.
Distribution of Interfering Substances in Fluid Compartments Within the Body
Given that the main classes of commercial CGMs operate within ISF, a brief consideration of the composition of this matrix is useful. ISF is a water-based matrix that surrounds cells and tissue and, similar to blood plasma, contains multiple components including ions, smaller molecules, biological macromolecules, and cellular materials.9,10 Evidence suggests that most analytes present in blood can be assumed to be present in ISF, including almost all proteins and medications.11,12 This is due to the ready passage of species between capillary blood and ISF, with many molecules migrating either by paracellular diffusion, via junctions between capillary endothelial cells, or transcellular transport via cellular vesicles. 13 Inflammation may also facilitate the passage of molecular species.13,14 However, whilst CGM is a powerful tool for monitoring glucose levels in ISF, the physiological concentrations of most analytes in ISF remain poorly characterized relative to blood.13,15
While it is reasonable to assume that an interfering substance present in blood will also be found in ISF, absolute concentration levels may be significantly different. Given challenges in extracting ISF in volumes sufficient for independent reference instrument analyses, 16 researchers and CGM developers often report in vitro CGM interfering effects in terms of analyte levels in blood plasma, 17 or spiked into surrogate ISF. 18 Estimates of in vivo substance concentrations in ISF are often taken to be the same as in blood, but this takes no account of physiological differences between the two compartments. 19 The use of microdialysis catheters for direct in vivo sampling of ISF has been reported and may be considered closer to clinical truth. 20
Classification of Continuous Glucose Monitors
CGM systems may be classified according to their degree of invasiveness within the human body, as shown in Figure 1. While significantly invasive CGMs are primarily intended for maintaining glucose control in critically ill patients through intravascular glucose monitoring, 21 greatest progress, both in terms of patient care and commercial success, has been made with minimally invasive CGMs. These devices are designed to penetrate the body, but to a limited extent.
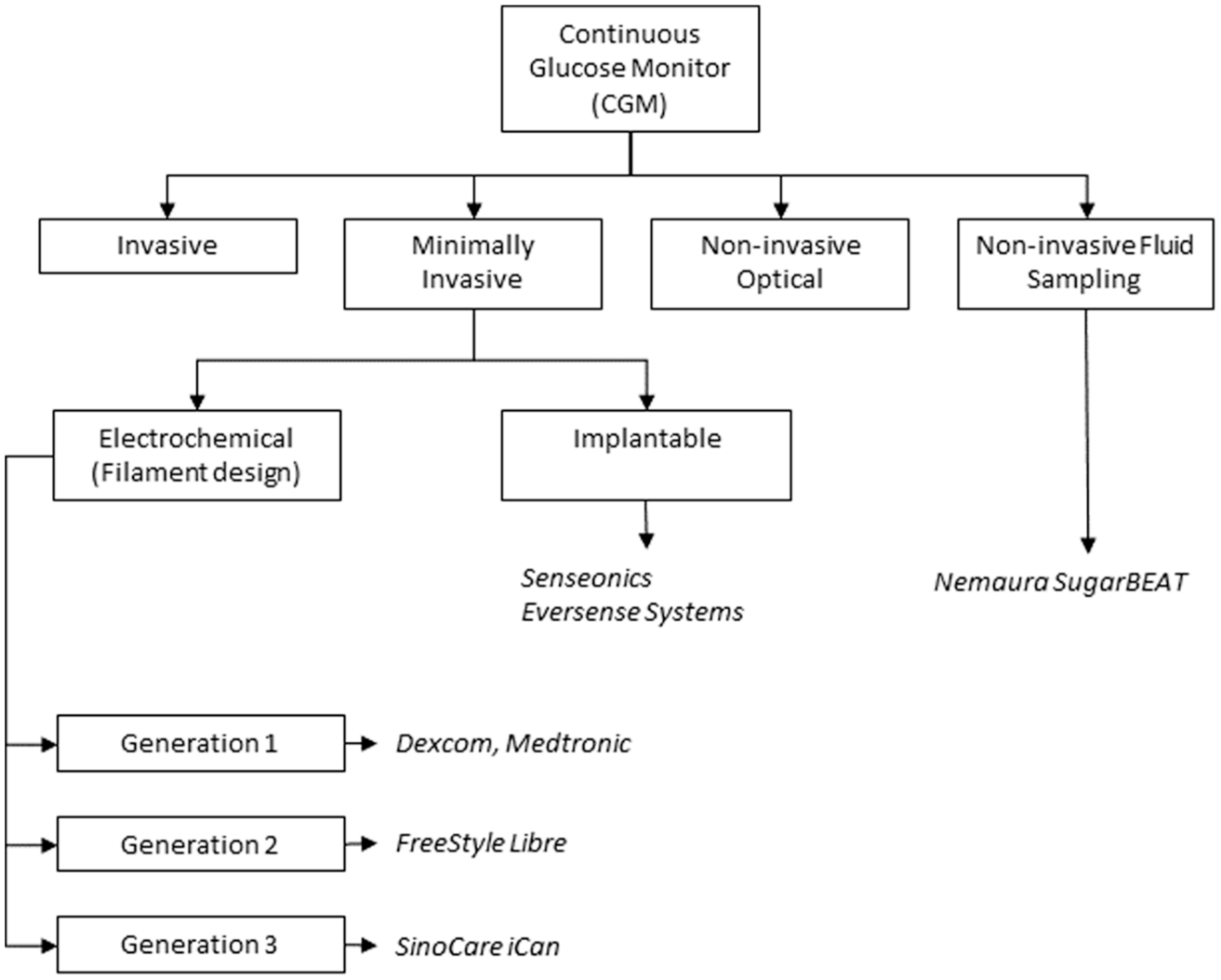
Classification of commercially available CGM systems with commercial examples.
Non-invasive fluid sampling CGMs represent a unique class of CGM device. While they measure glucose in the body, they do not penetrate the skin, being reliant on processes such as reverse iontophoresis (RI) to actively draw body fluids and hence glucose across the skin. Non-invasive CGMs, like non-invasive fluid sampling CGMs, are body-worn devices that do not require physical placement of a sensing element within the body. However, unlike non-invasive fluid-sampling devices, these systems use optical or electrical energy to cross the skin barrier and interact with tissue by optical or electrical means. The resultant responses are complex and highly temperature-dependent and require significant algorithmic interrogation to generate responses that correlate to glucose levels. To date, no non-invasive devices have been approved by regulatory bodies for diabetes management.
This review will focus on commercialized minimally invasive CGMs, which will be subdivided into (1) electrochemical CGMs that sit on the skin with a slender electrode filament that penetrates the subcutaneous tissue; and (2) implantable CGMs, which require placement under the skin via a minor surgical procedure. A further class of minimally invasive CGM based on microneedle technology, while being of significant interest, has not reached commercial realization, so it will not be reviewed here. All classes of minimally invasive CGM measure glucose within ISF. For a full appreciation of the breadth of CGM devices in market and in development, the reader is referred to the comprehensive review of Shang et al. 22
Minimally Invasive Electrochemical CGMs
Classification of Minimally Invasive Electrochemical CGMs
These devices incorporate a slender flexible sensing filament containing an active chemistry that samples ISF within the subcutaneous tissue. The literature describes four generations of electrochemical biosensor depending on the sensing mechanism used (a fifth-generation approach based on wearable biosensors integrated with artificial intelligence/Internet of Medical Things technology has also been described). 23 Examples of CGMs of the first-, second-, and third-generation are available commercially, all relying on redox enzymes for the specific biocatalytic oxidation of glucose, coupled to a reduction in the associated enzyme cofactor (Figure 2). Table 1 provides details of some current and historic first, second and third-generation CGM models and, where known, some of the primary design modifications that have been made to the glucose-sensing module to improve system accuracy. This list is not exhaustive and is intended to provide information limited to several of the more commercially successful CGMs, past and present.
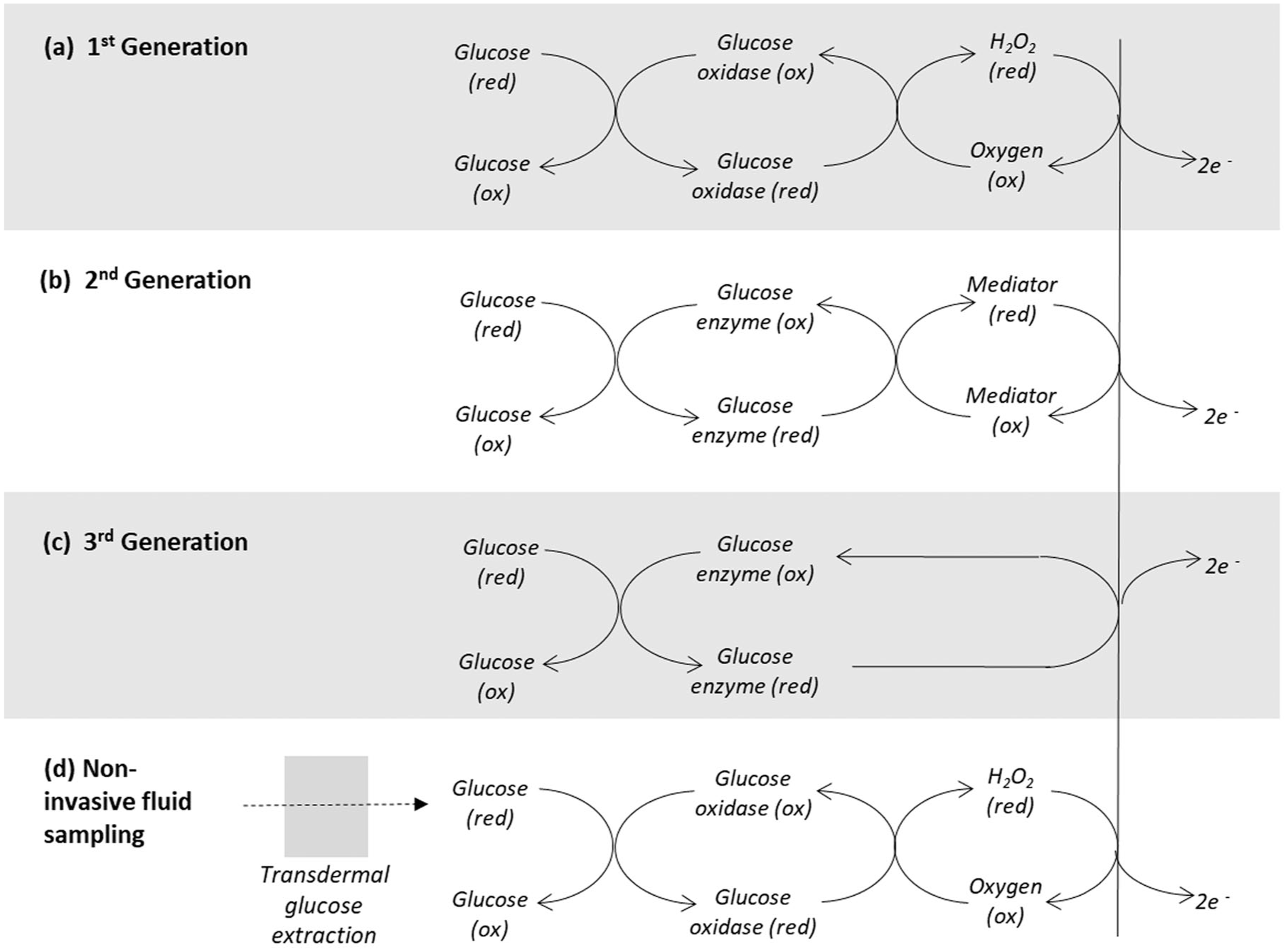
Classification of electrochemical CGMs. (a) First generation: oxygen acts as the electron acceptor from the reduced form of the enzyme cofactor, forming hydrogen peroxide which is oxidized at a suitably poised working electrode; (b) second generation: a mediator species is used in place of oxygen, allowing reduced operating potentials to be applied; (c) third generation: the CGM design is engineered to facilitate direct electron transfer from the embedded enzyme cofactor directly to the electrode surface; (d) non-invasive fluid sampling devices may operate on similar principles, but do not penetrate the skin surface, requiring a suitable non-invasive extraction methodology, such as reverse iontophoresis to sample glucose.
Classification of Currently or Previously Commercially Available CGM Models of the First-, Second-, and Third-Generational Biosensor Design, Regarding Interferences From Chemical Substances.
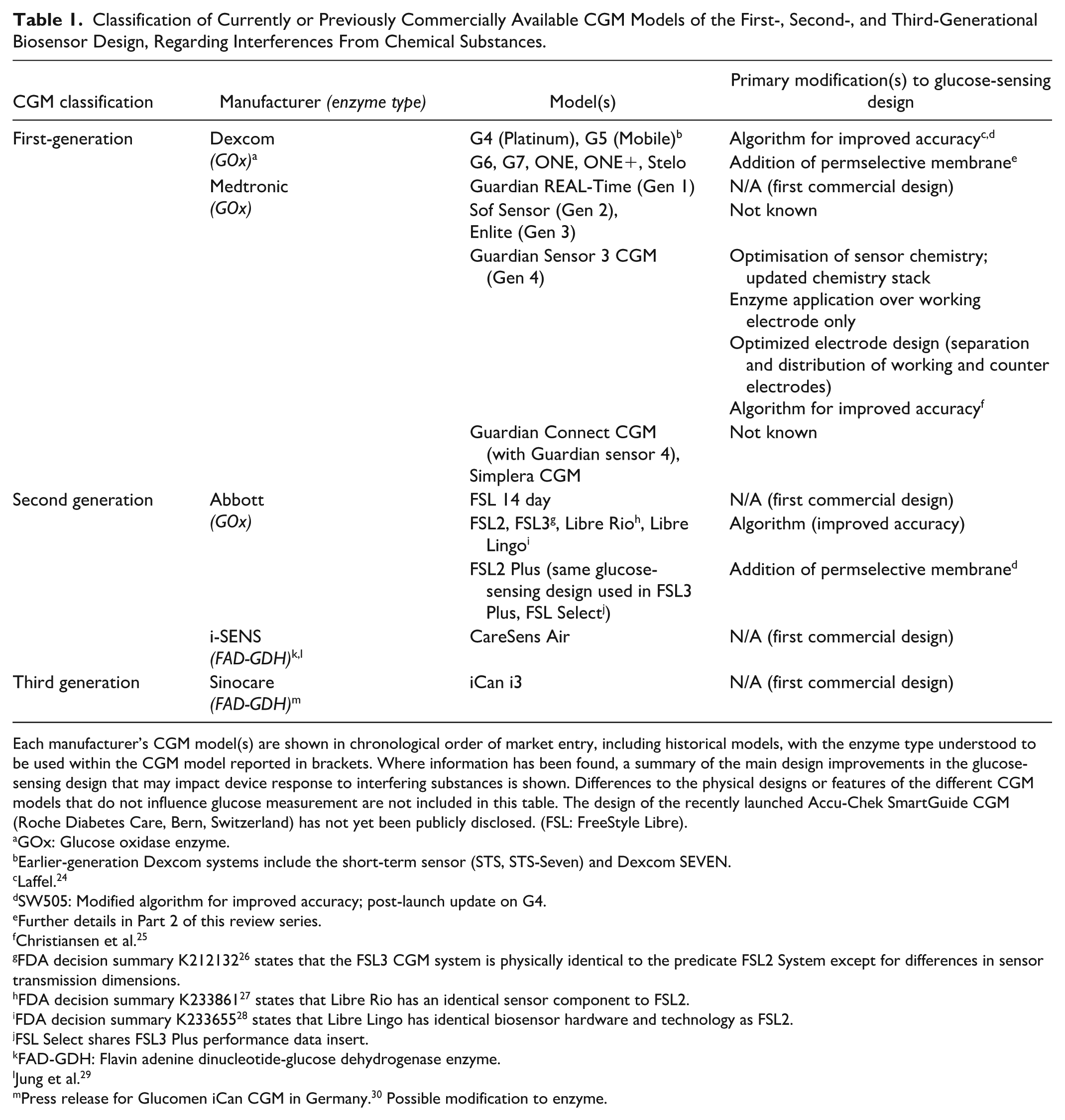
Each manufacturer’s CGM model(s) are shown in chronological order of market entry, including historical models, with the enzyme type understood to be used within the CGM model reported in brackets. Where information has been found, a summary of the main design improvements in the glucose-sensing design that may impact device response to interfering substances is shown. Differences to the physical designs or features of the different CGM models that do not influence glucose measurement are not included in this table. The design of the recently launched Accu-Chek SmartGuide CGM (Roche Diabetes Care, Bern, Switzerland) has not yet been publicly disclosed. (FSL: FreeStyle Libre).
GOx: Glucose oxidase enzyme.
Earlier-generation Dexcom systems include the short-term sensor (STS, STS-Seven) and Dexcom SEVEN.
Laffel. 24
SW505: Modified algorithm for improved accuracy; post-launch update on G4.
Further details in Part 2 of this review series.
Christiansen et al. 25
FDA decision summary K212132 26 states that the FSL3 CGM system is physically identical to the predicate FSL2 System except for differences in sensor transmission dimensions.
FDA decision summary K233861 27 states that Libre Rio has an identical sensor component to FSL2.
FDA decision summary K233655 28 states that Libre Lingo has identical biosensor hardware and technology as FSL2.
FSL Select shares FSL3 Plus performance data insert.
FAD-GDH: Flavin adenine dinucleotide-glucose dehydrogenase enzyme.
Jung et al. 29
Press release for Glucomen iCan CGM in Germany. 30 Possible modification to enzyme.
CGMs Based on First-Generation Electrochemical Biosensor Design
First-generation CGMs employ glucose oxidase enzyme (GOx) as the glucose-recognition agent and measure the hydrogen peroxide that is formed as oxygen, naturally present in ISF, accepts electrons from the reduced form of the enzyme cofactor (Figure 2a). Device designs typically incorporate a working electrode formed from a precious metallic wire and a silver/silver chloride (Ag/AgCl) counter electrode. Under physiologic conditions, the onset of hydrogen peroxide electro-oxidation can occur at relatively modest potentials, with the signal (current) magnitude increasing with the potential applied across the electrode assembly. A plateau effect may be seen in the region ~+400-600 mV, where the current output versus applied potential is less steep, thus effectively providing a “window” for collection of more stable signals of acceptable magnitude. While offering design simplicity, there is a cost:
Oxygen is sparingly soluble in ISF. For a first-generation CGM to be capable of measuring across a clinically relevant glucose range, sufficient oxygen must be available to the glucose oxidase (GOx) enzyme to provide a non-limiting biosensor response under conditions of high glucose and/or low ISF oxygen. Correspondingly, designs typically incorporate a glucose flux-limiting membrane which ideally should not impede oxygen transfer, thus reducing the risk of oxygen depletion and hence sensor inaccuracy.
The optimum potential for the electro-oxidation of hydrogen peroxide is relatively high. This increases the number of compounds in ISF that can be oxidized at the electrode, thus contributing to the overall sensor signal. Design solutions have included the use of selective membranes that impede the passage of interfering species to the electrode based on molecular charge and size.
An increasing number of first-generation CGMs have received regulatory clearance in recent years. Given commonality in the measurement principles involved, discussion here will mainly focus on the market-leading first-generation CGMs manufactured by Dexcom Inc. (San Diego, CA) and Medtronic Inc. (Minneapolis, MN).
CGMs Based on Second-Generation Electrochemical Biosensor Design
Second-generation CGMs have a similar mode of operation to first-generation systems but employ an artificial mediator to transfer electrons from the reduced enzyme cofactor to the electrode (Figure 2b). This approach offers benefits in that the mediator is formulated to (1) undergo oxidation at low potential, thereby reducing non–glucose-specific contributions to the output signal; and (2) have acceptable solubility in ISF such that, unlike oxygen, it does not become a potential limiting factor within the sensing mechanism. The FreeStyle Libre (FSL) family of products, manufactured by Abbott Diabetes Care (Alameda, CA), are second-generation CGMs, using GOx as the recognition enzyme but formulated with a mediator that is incorporated into an osmium redox polymer. The CareSens Air system, manufactured by i-SENS (Seoul, Korea) uses a FAD-GDH enzyme, where the flavin adenine dinucleotide co-factor is embedded within the glucose dehydrogenase structure. 29 GDH enzymes used within second-generation glucose monitoring systems are oxygen insensitive. 31
CGMs Based on Third-Generation Electrochemical Biosensor Design
This category of CGM is designed to facilitate electron transfer directly from the cofactor of the glucose recognition enzyme, typically a GDH enzyme, to the electrode without the requirement for a mediator (Figure 2c).30,31 This principle of direct electron transfer (DET) within CGMs is not recent, as evidenced by the work of Sode and co-workers.32-34 Strategies to realize third-generation systems include the selective protein engineering of enzymes, the use of redox polymer “wiring elements” and the creation of appropriately nano-scaled structures to facilitate DET. 35 The removal of mediator dependency addresses complications related to poor oxygen solubility in ISF and artificial mediator immobilization. Third-generation systems reported in the literature operate at potentials lower than those of first-generation systems, with Sode describing an electrochemical CGM operating a potential of +150 mV. 36
Commentators have reviewed potential limitations of the third-generation approach, citing risks of enzyme leaching, biocompatibility issues, 37 and the challenge of developing bespoke nanomaterials to access deeply embedded enzyme cofactor complexes. 38 The Sinocare iCan i3 CGM (Changsha Sinocare Inc., Changsha, China) has obtained regulatory approval in both China and Europe, and is described by the manufacturer as the first commercial factory-calibrated CGM system employing DET.
Non-Invasive Fluid Sampling CGMs
Several nascent non-invasive fluid sampling CGM designs, in which body fluids are accessed without the requirement for skin puncture have been reported.22,39-41 The SugarBEAT device (Nemaura Medical Ltd., Loughborough, UK) is the only system currently in market, with a CE class IIb designation when used for diabetes management. 42 ISF is extracted from the body by RI, and glucose is collected within an enzyme-hydrogel pad placed over an electrode assembly (Figure 2d). The GlucoWatch Biographer, generally considered the first commercially available CGM, operated on a similar principle but was withdrawn from the FDA premarket approval database in 2010. 43
Tierney studied the effect of 1000 mg acetaminophen challenges on the GlucoWatch Biographer, finding no effect on system accuracy. 44 Other than this, there is scant published data regarding interfering substance effects on these types of RI systems. However, given the detection mechanism is of the first-generation biosensor design, these devices are potentially subject to the same interference profiles common to first-generation CGM devices, although the transdermal migration of some substances to the sensing element may be inhibited by the skin barrier. Given the paucity of interfering substance data, coupled to the limited market presence of this class of system, they will be considered as having similar interfering characteristics to first-generation systems and will not be further reviewed.
Minimally Invasive Implanted CGMs
Several minimally invasive implanted CGMs have been reported in the literature, 22 but to date, only the Senseonics Eversense Implantable system has received regulatory approval. The long-term implantable nature of these devices requires a synthetic ligand, rather than an enzyme, to recognize and reversibly bind glucose. This binding process is measured optically, the intensity of the signal being related to the concentration of glucose in the surrounding tissue. Potential interference mechanisms will be distinct to electrochemical CGMs, being related to the specific ligand chemistry and the absorption/emission of light of similar wavelength to the optical indicator.
CGM Interference Mechanisms and Design Mitigations
As described in the introduction, regulatory guidance documents and standards provide descriptions of what constitutes a substance interfering effect for a BGM device. The approach is pragmatic, allowing the in vitro identification of individual substances that have a marked effect on the accuracy of BGM systems. However, the approach has limitations. For example, in the case of the ISO 15197:2013 acceptance criteria for BGMs, a substance that results in a 9% mean glucose bias from the control condition does not require reporting in the instructions for use, but a substance that yields an 11% mean glucose bias does require reporting. Thus, situations may arise in which an individual on a particular dietary or medication regime may be at risk of receiving inaccurate readings due to the cumulative interfering effect of substances that individually do not breach the acceptance criteria. It is therefore important for the manufacturer, whether of CGM or BGM, to develop product designs that minimize or eliminate the effect of non-glucose species on device performance.
Electrochemical Interfering Effects in Minimally Invasive CGMs
As shown in Figure 2, minimally invasive electrochemical CGMs employ a working electrode to receive electrons generated by the enzyme-catalyzed oxidation of glucose, with the resultant current translated into an equivalent glucose concentration. Electrochemical interfering mechanisms that may influence the glucose responses of CGM devices are summarized in Figure 3 and may be contrasted to Figure 2, which illustrates glucose-sensing mechanisms in the absence of interfering species. Whilst representing state-of-the-art for the non-laboratory measurement of glucose levels in blood and ISF, the signal response generated by this type of design approach may potentially be influenced by several factors, including:
Direct electrochemical oxidation. When present in ISF, certain endogenous and exogenous substances may undergo direct oxidation at the working electrode and thus contribute a non–glucose-specific signal to the CGM reading (Figure 3a). First-generation CGMs with the relatively high operating potentials required to electro-oxidise hydrogen peroxide, will be more susceptible to this effect.
Interferent/mediator interactions in second-generation CGM designs. Possible interference mechanisms include: (i) direct transfer of electrons from electron-donating substances in ISF to the mediator, resulting in a non-specific glucose response (Figure 3b); and (ii) direct transfer of electrons from the reduced form of the mediator to electron-accepting compounds present in the ISF (Figure 3c), thereby reducing the CGM signal. Ascorbic acid, a commonly used nutritional supplement with antioxidant properties, has been implicated in electron donation to certain types of mediators as will be reviewed in Part 2 of this review, as will be oxygen which is known to act as an acceptor of electrons from certain mediator compounds (Part 3).
Oxygen interference in second-generation CGM designs featuring glucose oxidase + mediator. Dissolved oxygen in the ISF may compete with an artificial mediator for electrons held within the enzyme cofactor (Figure 3d). The hydrogen peroxide generated during this reaction will not undergo electro-oxidation at the lower working electrode potential of second-generation systems. Thus, a systematic bias may be introduced to the glucose response in an individual with a dissolved oxygen level significantly different to that of the general user population, as will be discussed in Part 3.
Electrode fouling. This may occur due to the electro-oxidation and subsequent deposition of interfering substance residues onto sensor surfaces (Figure 3e). This in turn will impede glucose migration to the sensing surface, resulting in a decrease in the magnitude of the glucose-specific electrochemical signal. For example, the progressive passivation of platinum electrodes by sulfhydryl and phenolic compounds, characterized by a progressive decrease in current due to formation of a polymeric film over the electrode interface, has been well documented.45-50 This factor is a potentially more challenging issue for electrochemical CGMs compared to their BGM counterparts in that CGM devices are indicated for use over multiple days and thus will be subject to repeated exposure to fouling species. However, impacts may potentially be mitigated using compensatory algorithmic approaches, as will briefly be discussed.
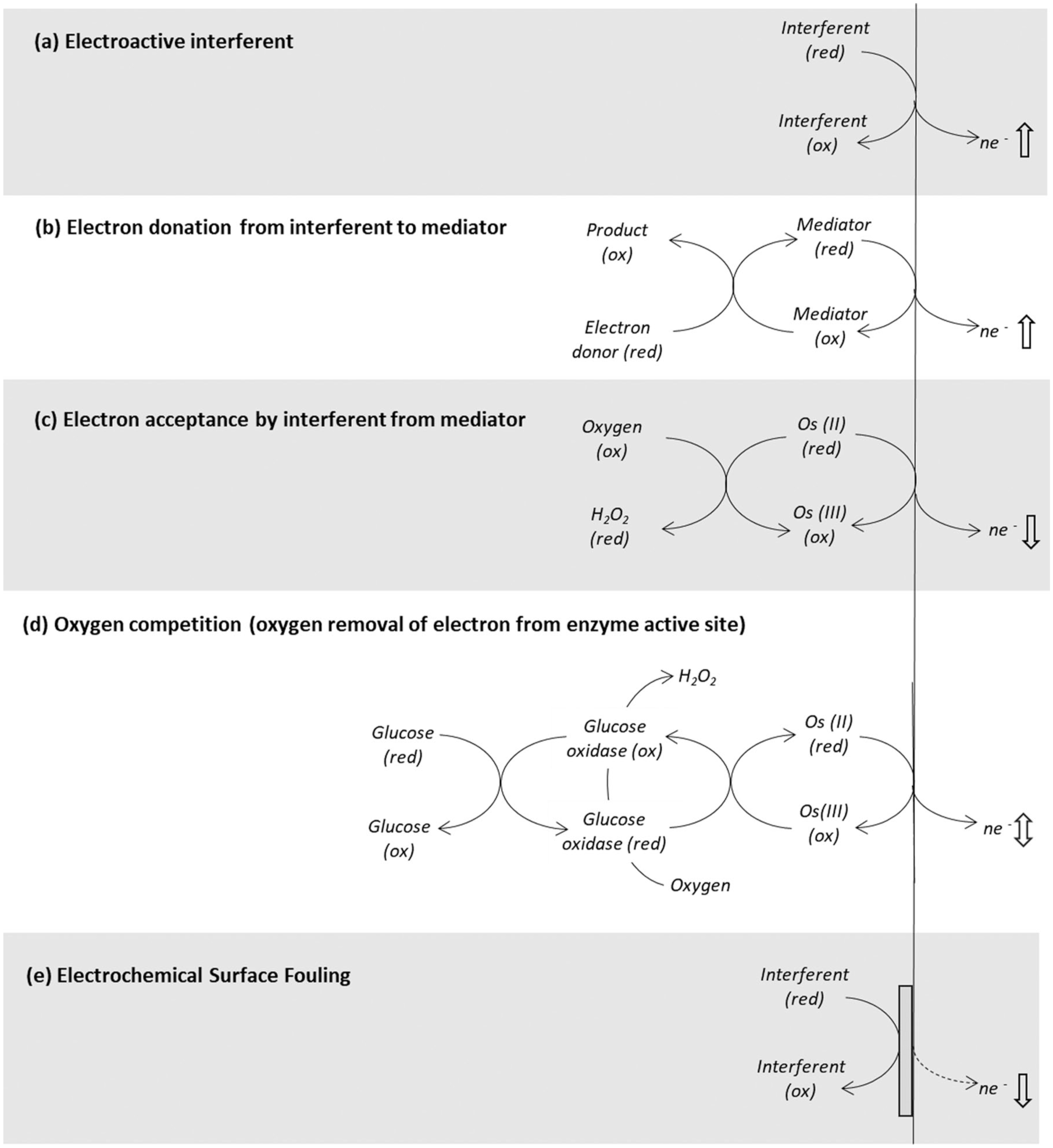
Electrochemical interference mechanisms. Some substances within ISF may undergo direct electrochemical oxidation at a suitably poised electrode (a) or readily donate or accept an electron with the mediating species in second-generation biosensors (b and c), resulting in a non-glucose-specific change in signal. A higher or lower oxygen tension in ISF can result in a systematic bias to the generated signal (d). Certain classes of electroactive compound are known to passivate the electrode surface due chemisorption, resulting in a progressive increase in electron transfer resistance and a reduction in measurement sensitivity (e). (ne-: transfer of one or more electrons).
Design Mitigations for Electrochemical Interference Effects in Minimally Invasive CGMs
Membrane designs to reduce impacts from interfering substances
A stacked series of membrane layers is a common design characteristic of minimally invasive CGMs, with each layer serving a specific purpose, such as biocompatibility, provision of optimal measurement conditions within the sensor assembly, immobilization or entrapment of sensing reagents, control of glucose flux, oxygen permeability, or reduction in interfering substance effects.51,52 Each CGM manufacturer will develop their own device-specific membrane formulations and manufacturing methodologies based on their specific CGM design and product requirements.
Many membrane designs have been developed, targeted at limiting interfering effects.50,51 Size exclusion may be used to prevent or reduce the flux of larger molecules, 52 while membranes featuring a network of positive and/or negative ionic charges have been formulated to impede the transfer of ionized species under physiologic conditions.53-55 Membrane structures immediately adjacent to the transducer interface provide optimal conditions for electrochemical performance and should minimally impede the passage of the target analyte. 52 For example, in first-generation electrochemical systems, such membranes should allow the passage of hydrogen peroxide from the active chemistry region to the electrode surface whilst limiting the flux of non-specific electroactive compounds, while in second-generation systems, membrane designs should be non-limiting with respect to the passage of reduced mediator but offer the benefit of lower operating potentials. 51
The inclusion of multiple membrane layers in a sensor design comes at a cost: each membrane formation and drying step within the manufacturing process will be subject to variability dependent on the specific process and degree of control applied. 56 This inevitably results in membranes with subtly different physical structures, pore sizes, and thicknesses, adding to overall system imprecision. The specific design of the membrane stack will also influence sensor response times due to increased mass-transfer resistance. 37
Where relevant, the membrane designs used in CGM constructions for reducing specific interfering effects will be reviewed in Part 2. An in-depth description of membrane designs used in the construction of commercial CGMs is available elsewhere.51,52
Mediators
As described, second-generation electrochemical sensor designs seek to reduce extraneous signals arising from electroactive species present within the sample matrix by incorporating an artificial mediator to facilitate electron transfer from the enzyme-embedded cofactor to the electrode transducer at reduced operating potentials. Whilst this approach is widely adopted within BGM designs, competition from oxygen within GOx systems and the possibility of direct electron transfer between substances and the artificial mediating redox species may influence glucose signals (Figure 3b and c) and should be considered at the product design stage. The enzyme-mediator design of the second-generation FSL systems will be reviewed in Part 2.
Electrode design
Some BGM systems incorporate an additional working electrode that is essentially identical to the primary working electrode, but without the analyte-sensing enzyme.57,58 The Nova Statstrip, a BGM system designed for use in a hospital setting, is described by the manufacturer as incorporating a Multi-Well system that corrects for interfering effects from multiple substances. 59 The purpose of this “interference” electrode is to provide a non-glucose measurement signal that responds primarily to the presence of electroactive interferences. Subtraction of the non-glucose signal from the enzyme-active electrode signal provides a potentially more accurate glucose response. In the CGM field, Dexcom patent US7896809 describes a continuous analyte sensor featuring a dual working electrode design and shows differing signals arising from enzyme and non-enzyme coated electrodes in the presence of glucose and acetaminophen. 60 The design was also said to account for changes in host metabolism and the maturation of tissue at the biointerface of the sensor, although this design approach does not appear to be implemented in currently available CGM models from this manufacturer.
Minimally Invasive Electrochemical CGMs: Enzymatic Interferences
Enzymatic Interference in Minimally Invasive CGMs
Electrochemical CGMs employ an immobilized enzyme as the glucose biorecognition agent, with the active site of the enzyme able to recognize specific structural features on the glucose molecule. Whilst having greatest specificity for glucose, interference effects can arise due to structurally similar saccharides either undergoing reaction with the enzyme (Figure 4) or otherwise interfering with the active site of the enzyme and impeding glucose entry. 61 This mechanism of interference is well known in the BGM field, with the FDA among others providing guidance regarding interference testing of saccharides and sugar alcohols. Other sources of saccharide interference on electrochemical CGMs include membrane resistance, analyte diffusion, GOx reactivity and purity, and the presence of enzymatic impurities within the GOx formulation itself. 62
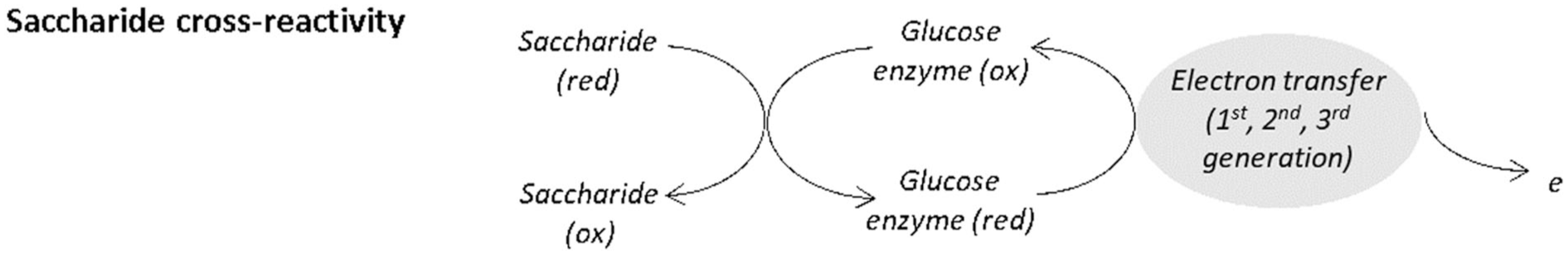
Certain non-glucose saccharides, due to structural similarities, may react with the glucose-sensing enzyme, resulting in a non–glucose-specific increase in CGM signal.
Glucose molecules are aldohexoses, having a 6-carbon monosaccharide aldehyde form. Several saccharides have been implicated in influencing electrochemical glucose sensors, including the aldohexoses galactose and mannose. Maltose, xylose, lactose, ribose, and arabinose may also influence CGM readings.63,64
Design Mitigations for Enzymatic Interference Effects in Minimally Invasive CGMs
GOx has thus far been the enzyme of choice in the construction of most commercialized enzymatic CGMs but are susceptible to oxygen interference effects. While this issue has possibly been overstated in the BGM field, 65 many BGM and CGM designs incorporate alternative dehydrogenase and quinoprotein-based dehydrogenase enzymes that are oxygen insensitive. Lee et al 66 have described a third-generation electrochemical CGM incorporating an engineered FAD-GDH enzyme, providing oxygen insensitivity coupled to direct transfer of electrons from the FAD-GDH active site to the transducing electrode. However, FAD-GDH systems, like GOx systems, also have cross-reactivity to certain saccharides, most notably xylose.67,68
Implantable CGM Interfering Substance Mechanisms
Since longer term implantable CGMs use synthetic binding ligands to recognize glucose, they are not subject to the enzymatic interference mechanism illustrated in Figure 4. However, there remains a risk that substances with structural similarity to glucose may interact, generating an interfering effect. Similarly, whilst these specific device designs rely on optical transduction, a non-glucose dependent optical response may arise should there be substances present in the sample matrix that absorb or emit light within the same spectral operating range of the sensor, as will be discussed further for the Senseonics Eversense CGM models in Part 2 of this review. 17
Algorithmic Design Approaches to Mitigate Interfering Substance Effects
The BGM field includes references to systems in which algorithms have been used to reduce interfering substance effects. 69 In electrochemical BGM systems, such algorithmic approaches have been enhanced by varying the electrode potential over the duration of the assay, which is typically several seconds in duration.70,71 This, coupled to subsequent algorithmic deconvolution of the sample signature, can aid in estimating the levels of electroactive compounds present in a sample, as well as compensate for the systematic effect of, for example, hematocrit and temperature on the blood-based sample response. Whilst CGM devices make use of algorithmic correction, e.g., in accounting for sensor-drift over the claimed lifetime of the device or using electrochemical impedance spectroscopy to proactively detect sensor faults, 25 no evidence has so far been found to suggest that pulsed electrochemical waveforms coupled to algorithmic signal deconvolution is employed in CGMs to minimize interfering substance effects.
Biofouling
Distinct to electrode fouling, the physical adsorption of chemical compounds to embedded sensor structures may also impede the free transfer of glucose, oxygen and other compounds to the sensor chemistry and underlying transducer due to membrane pore blockage. The growth of biological tissue around subcutaneous sensor structures, especially in implanted devices, may have a similar effect. 37 Trauma due to transdermal sensor placement, skin edema and erythema due to sensor wear and localized host responses due to prolonged sensor placement may further impede device performance,72,73 as may bacterial colonization. Such issues have prompted manufacturers to develop protein resistant surfaces that are electrically neutral and substantially hydrophilic.74,75 This aspect of CGM interference is outside of the scope of this review, being assessed in detail elsewhere. 51
Summary
Part 1 of this review series introduces the classification and mechanistic design features of commercially available CGM systems. An understanding of this area allows both an appreciation of the interference mechanisms that can occur in the presence of potential interfering substances, as summarized in Table 2, as well as design mitigations to reduce these non-specific glucose effects. Part 2 of this review series will provide a more in-depth consideration of the glucose-sensing design of the main CGM models currently available in market and, where applicable, the design improvements that manufacturers have made to reduce the effect of the main interferents known to affect CGM accuracy.
Potential Interfering Conditions/Mechanisms for Currently Commercially Available CGM Systems.

Whilst second- and third-generation electrochemical CGMs operate at lower potentials versus first generation systems and are thus less influenced by directly electroactive substances, empirical assessments may be required to confirm the extent of interference for a given substance. Glucose oxidase and glucose dehydrogenase based CGMs exhibit different cross-reactivities to different saccharide substrates, while strains of the same enzyme type may exhibit different affinities to given saccharide substrates.
Footnotes
Acknowledgements
The author is indebted to Dr Elizabeth Holt MD, FACE, Head of Global Clinical, Medical and Safety, Dr Mike Grady, Director of Clinical Affairs, and Mr Brian Heald, Senior Vice President, Product, for their invaluable suggestions and in-depth review of this manuscript. The author also thanks Dr Marco Cardosi for helpful discussions during the writing of this manuscript.
Abbreviations
Ag/AgCl, silver/silver chloride; BGM, blood glucose monitor; CGM, continuous glucose monitor; CLSI, Clinical and Laboratory Standards Institute; DET, direct electron transfer; FAD, flavin adenine dinucleotide; FDA, Food and Drug Administration; FSL, FreeStyle Libre; GDH, glucose dehydrogenase; GOx, glucose oxidase; IFCC, International Federation of Clinical Chemists; ISF, interstitial fluid; ISO, International Standards Organization.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SJ Setford is an employee of LifeScan Scotland Ltd.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SJ Setford is a salaried employee of LifeScan Scotland Ltd.
