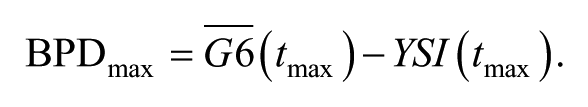Abstract
Acetaminophen (APAP) can cause erroneously high readings in real-time continuous glucose monitoring (rtCGM) systems. APAP-associated bias in an investigational rtCGM system (G6) was evaluated by taking the difference in glucose measurements between rtCGM and YSI from 1 hour before to 6 hours after a 1-g oral APAP dose in 66 subjects with type 1 or type 2 diabetes. The interference effect was defined as the average post-dose (30-90 minutes) bias minus the average baseline bias for each subject. The clinically meaningful interference effect was defined as 10 mg/dL. The G6 system’s overall mean (±SD) interference effect was 3.1 ± 4.8 mg/dL (one-sided upper 95% CI = 4.1 mg/dL), significantly lower than 10 mg/dL. The G6 system’s resistance to APAP interference should provide reassurance to those using the drug.
Acetaminophen (APAP) is a commonly used analgesic and antipyretic agent. Its phenolic moiety is easily oxidized and can interfere with electrochemical sensors used for real-time continuous glucose monitoring (rtCGM). 1 The additional signal can result in erroneously high rtCGM readings.2-6 Patients are routinely advised against basing diabetes management decisions on rtCGM data in the hours following a dose of APAP.5,6 Consequently, rtCGM systems that provide accurate readings during APAP use are of considerable interest. A novel permselective membrane coating for an investigational rtCGM system (G6, Dexcom, Inc, San Diego, CA) was designed to minimize or prevent the spurious (interference) signal generated by APAP. This study evaluated the average interference effect (additional bias) from a maximum recommended dose of acetaminophen on the G6 rtCGM’s displayed glucose value when compared to a reference glucose concentration measurement; the primary objective was to demonstrate that the overall mean interference effect was less than 10 mg/dL.
Methods
This prospective, multicenter, single-arm study (ClinicalTrials.gov identifier: NCT03087877) enrolled 70 subjects with diabetes at four investigational sites (14-19 subjects per site) in the United States between January and March 2017. The study was conducted under an abbreviated IDE 21 CFR 812.2(b) and was reviewed and approved by central IRBs. Major eligibility criteria for this analysis included age 18 years or older, diagnosis of type 1 (T1D) or type 2 diabetes (T2D), and ability to refrain from APAP use for 24 hours prior to sensor insertion and for the duration of the study. Major exclusion criteria were significant allergy to medical-grade adhesives, pregnancy, gastroparesis, hematocrit outside normal range, abnormal liver function tests, and known contraindication to taking the recommended oral dose of APAP (e.g., cirrhosis, chronic heavy ethanol use, breast feeding).
Sensor insertions were performed at the clinic by clinical staff. Sensors were placed on the abdomen and subjects were asked to use the study-assigned blood glucose meter (Bayer Contour Next EZ blood glucose meter; 510(k) K111268) for system calibrations, and for self-management decisions. On day 4 or day 5 of sensor wear, subjects returned in a fasting state to attend one, in-clinic session lasting 8 hours. An oral, 1-gram dose of APAP (the maximum recommended one-time dose) was administered during the clinic session. Venous blood samples were drawn and plasma glucose concentrations measured with the YSI reference instrument (YSI, Inc, Yellow Springs, OH) over a 7-hour period. Measurements were made at 10 ± 5-minute intervals for 1 hour pre-dose to 2 hours post-dose, then at 15 ± 5-minute intervals for remainder of the clinic session. A heating pad was applied around the IV site to “arterialize” the venous sample, allowing for a closer match between venous and capillary glucose concentrations.3,4 Capillary BG testing was also performed at every 30 ± 5-minute intervals using the study-assigned blood glucose meter. No glucose manipulations were performed.
Serum APAP concentrations were determined at two time points: 1 hour prior to APAP dosing, to verify that patients had refrained from APAP dosing prior to the study; and 1 hour after APAP dosing, to examine the APAP concentration relative to the rtCGM bias within the expected peak absorption time.
This study’s primary objective was to determine if the G6 interference effect was less than a clinically relevant threshold (defined as 10 mg/dL) after APAP ingestion. G6 sensor readings temporally matched with YSI values formed the analysis dataset. The interference effect was defined as the average bias 30 minutes to 90 minutes after APAP intake minus the average bias at baseline for each subject. For example, if a subject’s average baseline bias (0 to 60 min before APAP intake) was 1 mg/dL and increased to 3 mg/dL between 30 to 90 minutes after APAP intake, then the interference effect for that subject is 2 mg/dL. The hypothesis was that the mean interference effect was less than the clinically meaningful threshold, defined for this study as 10 mg/dL. A one-sample t-test was used to test this hypothesis at a one-sided significance level of 5%. Among the study’s secondary objectives was the maximum interference effect of the G6 sensor worn by each subject. The maximum bias post-dose (BPDmax) was calculated as the average of the three glucose values including and adjacent to the most biased value (
The maximum interference effect was defined as the maximum post-dose (30-90 minutes) bias (BPDmax) minus the average baseline bias for each subject. In a feasibility study, we found this method reduced the variability of the estimated bias over a single glucose measurement. Additional secondary endpoints included the mean interference effect of patient subgroups stratified by site, gender, BMI, and age; the mean bias of the study-assigned blood glucose meter; and the mean absolute relative difference (MARD) of the G6 system before and after APAP intake.
Results
Seventy subjects enrolled in the study; among these 70 subjects, 70% had T1D and 30% had T2D; 51% were female; the mean ± SD BMI was 28.7 ± 6.6 kg/m2; the mean ± SD HbA1c was 7.5 ± 1.2%. Data from 4 subjects were excluded because of sensor failures or accidental sensor restarts; data from 66 (94.3%) subjects were available for analysis.
The mean interference effect of the G6 system was 3.1 ± 4.8 mg/dL (Table 1) and the one-sided upper 95% CI was 4.1 mg/dL, which was significantly lower than the performance goal of 10 mg/dL (P < 0.001). The mean interference effect over time is shown in Figure 1a. The mean bias was 5.9 mg/dL at baseline and increased to 9.1 mg/dL during 30 minutes to 90 minutes after APAP intake. The mean interference effect versus the APAP concentration at 1 hour is given in Figure 1b. The mean interference effect was similar for subjects with high (10-26 mg/L) venous APAP concentration at 1 hour compared to those with low (<10 mg/L) venous APAP concentration (3.9 vs 1.6 mg/dL, respectively; Table 1). Subgroup analysis of patients stratified by site, gender, BMI, and age demonstrated that mean interference effect was below the performance goal of 10 mg/dL for each defined patient subgroup (Table 1). The maximum interference effect was also assessed for each subject; the overall mean maximum interference effect was 7.7 mg/dL (one-sided upper 95% CI was 8.8 mg/dL)—still lower than the performance goal.
Interference Effect Overall, by APAP Concentration and by Subject Characteristics.
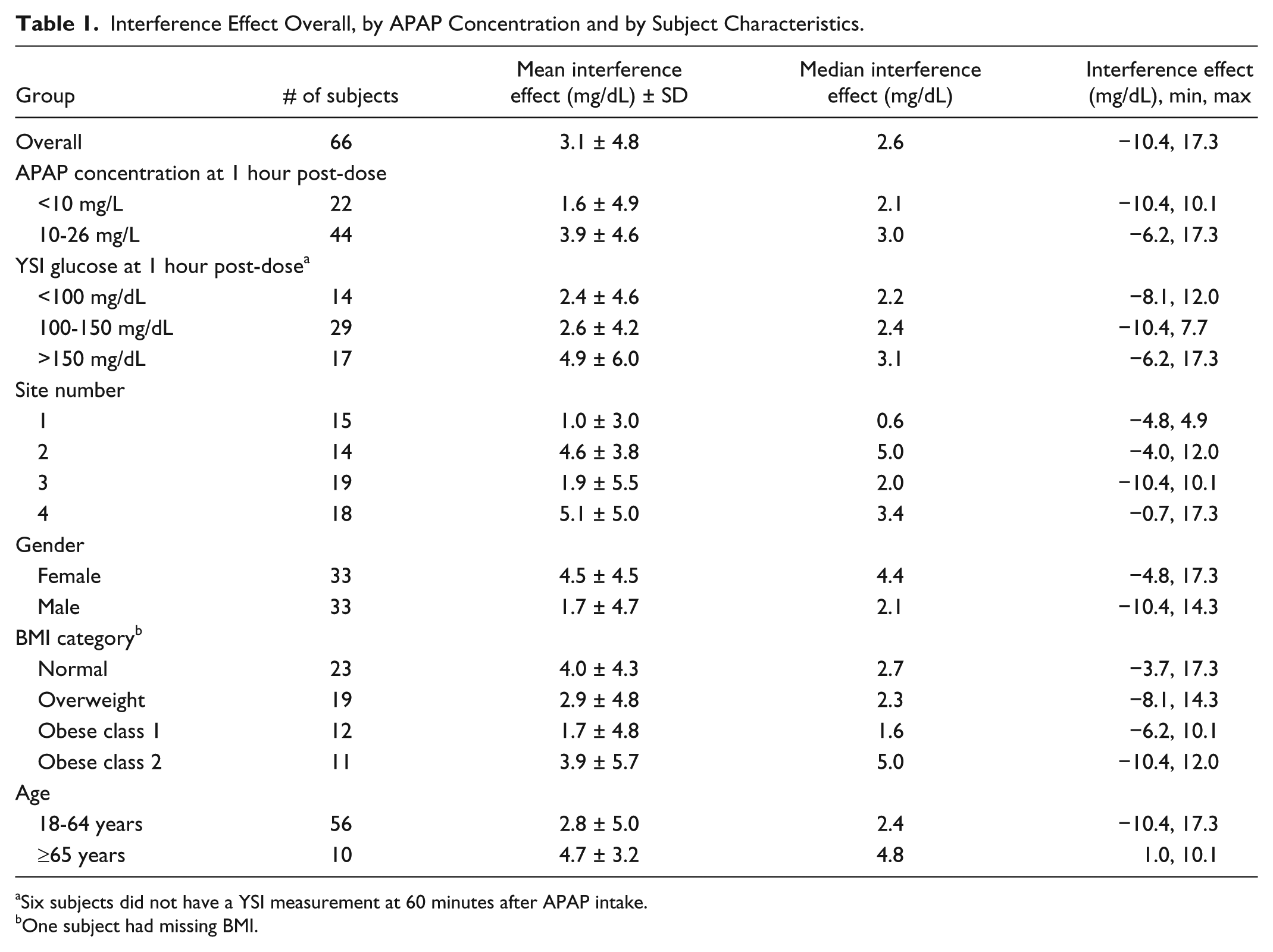
Six subjects did not have a YSI measurement at 60 minutes after APAP intake.
One subject had missing BMI.
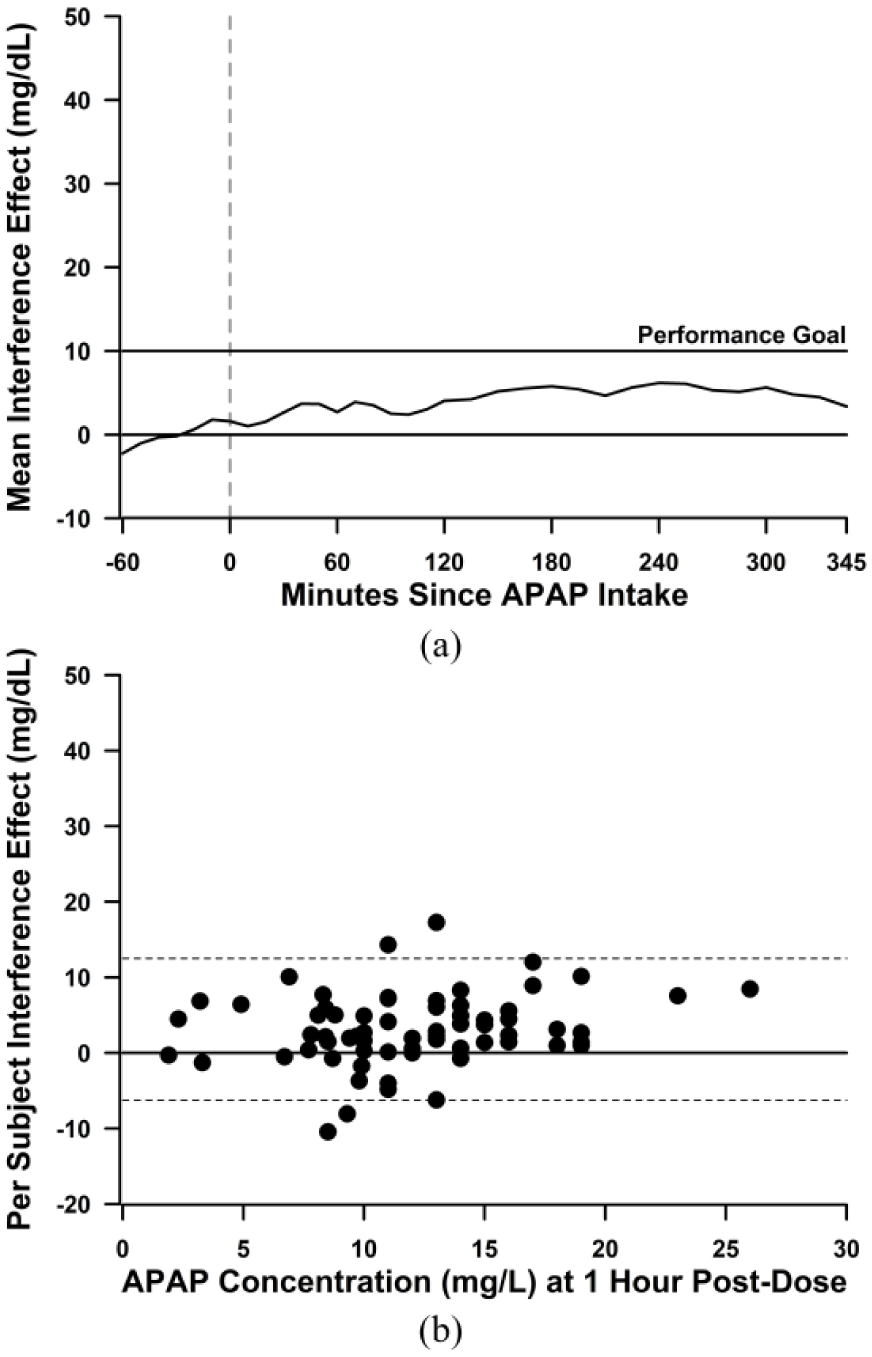
The APAP interference effect on the G6 system. (a) Mean interference effect over time of the G6 system. Performance goal was a mean interference effect (average post-dose bias minus average baseline bias) of 10 mg/dL. (b) Per subject interference effect by APAP concentration at 1 hour post-dose with 95% limits of agreement (dotted lines).
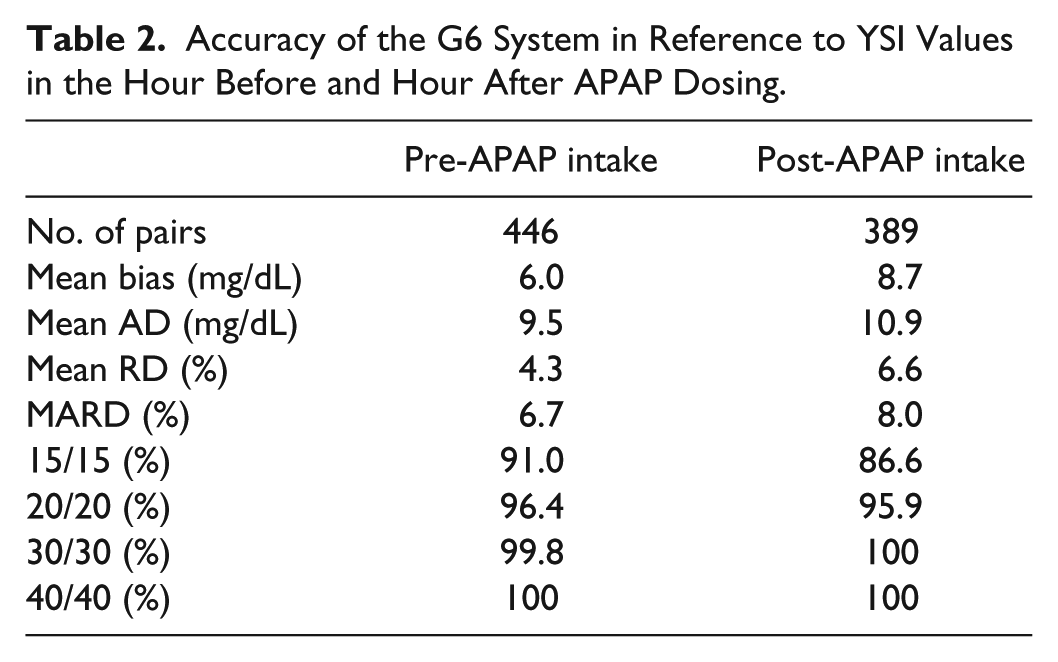
The accuracy of the G6 rtCGM system assessed in reference to YSI values is given in Table 2. The mean absolute relative difference (MARD) was 6.7% pre-APAP dose and 8.0% post-dose. Similarly, the %20/20 accuracy remained stable after APAP intake (96.4% pre-dose vs 95.9% post-dose).
Accuracy of the G6 System in Reference to YSI Values in the Hour Before and Hour After APAP Dosing.
The accuracy of the study-assigned glucose meter devices was assessed in reference to temporally matched YSI values. The meters had a mean bias of 3.6 mg/dL at baseline, which increased to 4.6 mg/dL during 30 minutes to 90 minutes after APAP intake. However, the MARD remained stable following APAP intake (4.6% pre-dose vs 4.8% post-dose).
Discussion/Conclusions
Real-time and intermittently scanned CGM systems that rely on electrochemical sensors, such as the Dexcom G4, Dexcom G5, MiniMed Sof-Sensor, Medtronic Enlite, and Abbott FreeStyle Libre, can be subject to varying degrees of interference from several medications and nondrug compounds, including acetaminophen, salicylates, and ascorbic acid.2-7 Because of the known interference effect of APAP, instructions for use and training materials highlight that users should not rely on rtCGM data when they have recently taken APAP.5,6 However, there remains a need for rtCGM systems that are resistant to APAP interference for people using rtCGM data nonadjunctively or for those using rtCGM data to govern insulin pump behavior in closed-loop systems.
The design of the current study was based on pilot in vivo studies of APAP pharmacokinetics and the dynamics of its interference with APAP-sensitive rtCGM systems. This study’s primary objective (overall mean interference effect of ≤10 mg/dL) was established based on the perceived minimal clinical risk of a 10 mg/dL positive bias in rtCGM readings and the expected variability of readings from rtCGM systems during normal day-to-day use (i.e., in the absence of APAP). The study’s objective was met in that the observed mean interference effect was 3.1 ± 4.8 mg/dL, which was statistically significantly lower than the performance goal of 10 mg/dL (one-sided upper 95% CI of 4.1 mg/dL). This small interference effect would have a negligible effect on bolus insulin administered. Even when the maximum interference effect for each subject was evaluated, the mean maximum interference effect was 7.7 mg/dL (one-sided upper 95% CI was 8.8 mg/dL)—less than the performance goal.
While there was a modest bias in the G6 throughout the study, even at baseline, the mean bias for the meters used to calibrate the G6 sensors at baseline was 3.6 mg/dL; biased SMBG calibrations may partially explain the slight bias in the rtCGM values. Blood glucose values entered for calibration are evaluated and weighted as part of a rolling average and used by a proprietary algorithm to determine the glucose value sent to the display device. Therefore, bias in meter readings can translate to bias in rtCGM data. 8
The study evaluated the effects of a single 1-g APAP dose, which represents the maximum recommended adult dose for any 6-hour period. The drug reaches peak blood concentration within 90 minutes of ingestion and has a half-life of 1.5-2.5 hours in adults.9,10 Because the temporal profile of interference with susceptible rtCGM systems follows the temporal profile of ISF and plasma APAP concentrations, 3 study of higher or repeated doses was deemed unnecessary.
The novel G6 rtCGM system and its resistance to APAP interference described here should alleviate accuracy concerns for those using the drug.
Footnotes
Acknowledgements
The authors are grateful to the investigators at the clinical sites—Drs. K. Castorino, T. Bailey, L. J. Klaff, and M. Christiansen—for contributing to data collection. The authors thank the participants for their involvement in the study.
Abbreviations
AD, absolute difference; APAP, acetaminophen; CGM, continuous glucose monitoring; MARD, mean absolute relative difference; RD, relative difference; rtCGM, real-time continuous glucose monitoring; SMBG, self-monitored blood glucose; T1D, type 1 diabetes; T2D, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PC, TKJ, JH, DP, and AKB are full-time employees of Dexcom, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Dexcom, Inc.