Abstract
Aim:
This review aims to map the existing literature on the use of diabetes technology in people receiving dialysis, with a focus on utilization, accuracy, and effectiveness.
Methods:
A scoping review was conducted using the Joanna Briggs Institute methodology, with systematic searches of Medline, Embase, and CINAHL for studies on diabetes technologies in dialysis populations.
Results:
The search identified 1060 continuous glucose monitoring (CGM) and 1467 continuous subcutaneous insulin infusion or automated insulin delivery (CSII/AID) records, with 64 studies included. Eighteen studies assessed CGM accuracy, reporting mean absolute relative difference (MARD) values ranging from 8.1% to 29%, with over 97% of readings falling within Clarke error grid zones A or B. Thirteen studies compared glycemic markers, finding that HbA1c underestimated glucose by 7.3 mmol/mol, while glycated albumin showed a stronger correlation (r = 0.508). Four studies reported on dialysis effects, showing that people on automated peritoneal dialysis (APD) had lower mean glucose levels (181 ± 64 mg/dL) compared to continuous ambulatory peritoneal dialysis (CAPD) (238 ± 67 mg/dL;
Conclusion:
This review highlights the potential of CGM and AID to improve diabetes outcomes in people on dialysis. While their clinical utility is evident, broader access and further research are needed to optimize their use in this high-risk population.
Keywords
Introduction
Diabetes is one of the leading causes of end-stage kidney disease (ESKD) in many countries worldwide. 1 As the incidence of diabetes rises, so too does the number of individuals with both diabetes and ESKD who require kidney replacement therapy (KRT). The limited availability of organs for transplantation, coupled with the cardiovascular comorbidities associated with diabetes, often restricts eligibility for kidney transplantation. Consequently, hemodialysis (HD) or peritoneal dialysis (PD) is frequently the only viable treatment options for many individuals. Despite significant advances in diabetes care, those with diabetes undergoing dialysis experience a reduced quality of life and a high burden of hypoglycemia and hyperglycemia2 -5 and hypoglycemia unawareness.6,7
Managing diabetes in individuals with ESKD undergoing KRT presents unique challenges. Traditional glycemic management strategies can be less effective due to altered insulin pharmacokinetics and the dietary restrictions associated with ESKD. 8 The ESKD is also linked with impaired kidney gluconeogenesis, a weakened counterregulatory hormonal response, malnutrition, insulin resistance, and impaired beta-cell function. 9 As such, health care providers must navigate a complex landscape to maintain optimal glycemic levels in this population. High glucose levels can lead to increased thirst and fluid retention between dialysis sessions, contributing to fluid overload and higher mortality rates. These increased fluid gains can also affect dialysis tolerance, cause unpleasant hypotensive episodes, and prolong recovery time.10 -12
Technological advancements in diabetes management, such as continuous glucose monitoring (CGM) systems, insulin pumps or continuous subcutaneous insulin infusion (CSII) or automated insulin delivery (AID), and data management applications, offer promising solutions to improve glycemic levels among people on dialysis. These technologies provide real-time insights into glucose levels and facilitate more tailored insulin delivery. By enabling people with diabetes to respond proactively to glucose fluctuations, these tools meet the dynamic needs of those managing multiple comorbidities. However, integrating diabetes technologies into dialysis settings presents challenges. 13 Access to advanced diabetes management tools and specialized diabetes services remains limited for many individuals with ESKD.13,14 As the field of diabetes technology evolves, it is crucial to explore its application within the dialysis population.
Diabetes management in dialysis populations is complex, yet the role of diabetes technologies remains unclear. While previous reviews focus on glycemic outcomes,15 -17 gaps remain in understanding their accuracy, effectiveness, and real-world use. Given the unique metabolic challenges of KRT, a focused review is essential to optimize care, improve outcomes, and address disparities in access to advanced diabetes technology.
Methods
Aim of the Scoping Review
This review aims to map existing literature on diabetes technology use in people receiving dialysis, focusing on utilization, accuracy in the dialysis population, and effectiveness in diabetes management.
Research Questions
Search Strategy and Information Sources
Protocol and registration
This scoping review followed the Joanna Briggs Institute methodology. The reporting of items is in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) extension for Scoping Reviews (PRISMA-ScR) reporting guidelines. 18 The protocol was registered with the Open Science Framework at osf.io/tqvzg (https://doi.org/10.17605/OSF.IO/N39BP).
Information sources
The search strategy was developed in collaboration with a research librarian. The databases searched included CINAHL, Medline, and Embase. The search was conducted on May 1, 2024, updated on August 28, 2024, and further updated in May 2025 to identify any subsequent publications. The first author carried out searches using the keywords and Medical Subject Headings (MeSH) terms specified in Supplemental File 1. No restrictions were applied regarding publication date, study design, or country. The search was conducted in Medline (Ovid) and subsequently translated to the other databases. In addition, the reference lists of the included articles were reviewed to identify other potential studies. Our full search strategy is included in the Supplemental File 1.
Study Selection and Eligibility Criteria
Study selection
Articles were uploaded to the online systematic review platform, Covidence, for screening. Duplicates were detected and removed. The titles and abstracts of all studies were then screened by three primary reviewers (HHH-A, JNS, KC) to determine eligibility for full-text review. The primary reviewers conducted full-text assessments to identify studies that met the inclusion criteria. Any disagreements between the primary reviewers were discussed with a fourth, fifth, and sixth reviewer to reach a final decision.
Eligibility criteria
Inclusion criteria
Studies conducted on adult people (age ≥18 years) focused on the use of diabetes technology such as CGM or CSII or AID undergoing dialysis with diabetes:
Research articles, clinical trials, clinical audits, abstracts, conference proceedings, case studies, and observational studies comparing the efficacy of diabetes technology interventions (CGM and/or CSII or AID with usual care in people with diabetes on dialysis).
Studies published in English.
Exclusion criteria
Studies that did not specifically assess the effects of diabetes technology interventions in dialysis population.
Studies that assessed diabetes technology intervention in the kidney transplant population.
Studies evaluating diabetes technology in mixed cohorts of dialysis and transplant population were excluded from this review if data specific to the dialysis subgroup could not be distinctly separated.
Systematic reviews or literature reviews.
Editorials or opinion pieces.
Studies published in languages other than English.
Data Extraction and Synthesis
Charting the data
Three reviewers (HHH-A, JNS, KC) developed the data extraction table, which was reviewed by the senior authors (AF, DCW, JK). HHH-A guided JS and KC in the data extraction process and subsequently verified the extracted data. The senior authors (AF, JK) provided feedback on the final literature tables. The development and completion of these tables allowed us to summarize and synthesize the data effectively.
Synthesis of results
The study characteristics, methods, and outcomes were organized in a tabular format. Given the heterogeneity of the included studies, the extracted data were compiled to provide a comprehensive overview of each study, detailing the purpose and use of diabetes technologies and the reported outcomes in a standardized format. This information informed a narrative review that examined key aspects of the identified interventions, including the validation of CGM accuracy in dialysis populations, comparisons between CGM and traditional glycemic markers, the impact of dialysis on glucose profiles, the use of CGM to assess the efficacy of diabetes treatments, CGM-based evaluations of glycemic outcomes, and the application of AID systems in the dialysis population.
Results
Study Characteristics
A systematic search of electronic databases identified a total of 1060 studies on CGM/AID and 1467 studies on insulin infusion (CSII/AID). After the automatic removal of 345 duplicates related to CGM and 310 related to CSII or AID using Covidence review manager, 64 studies were retained. These were selected following a manual screening of titles, abstracts, and full texts by the reviewers. The study screening and selection process is illustrated in the PRISMA diagram (Figure 1).
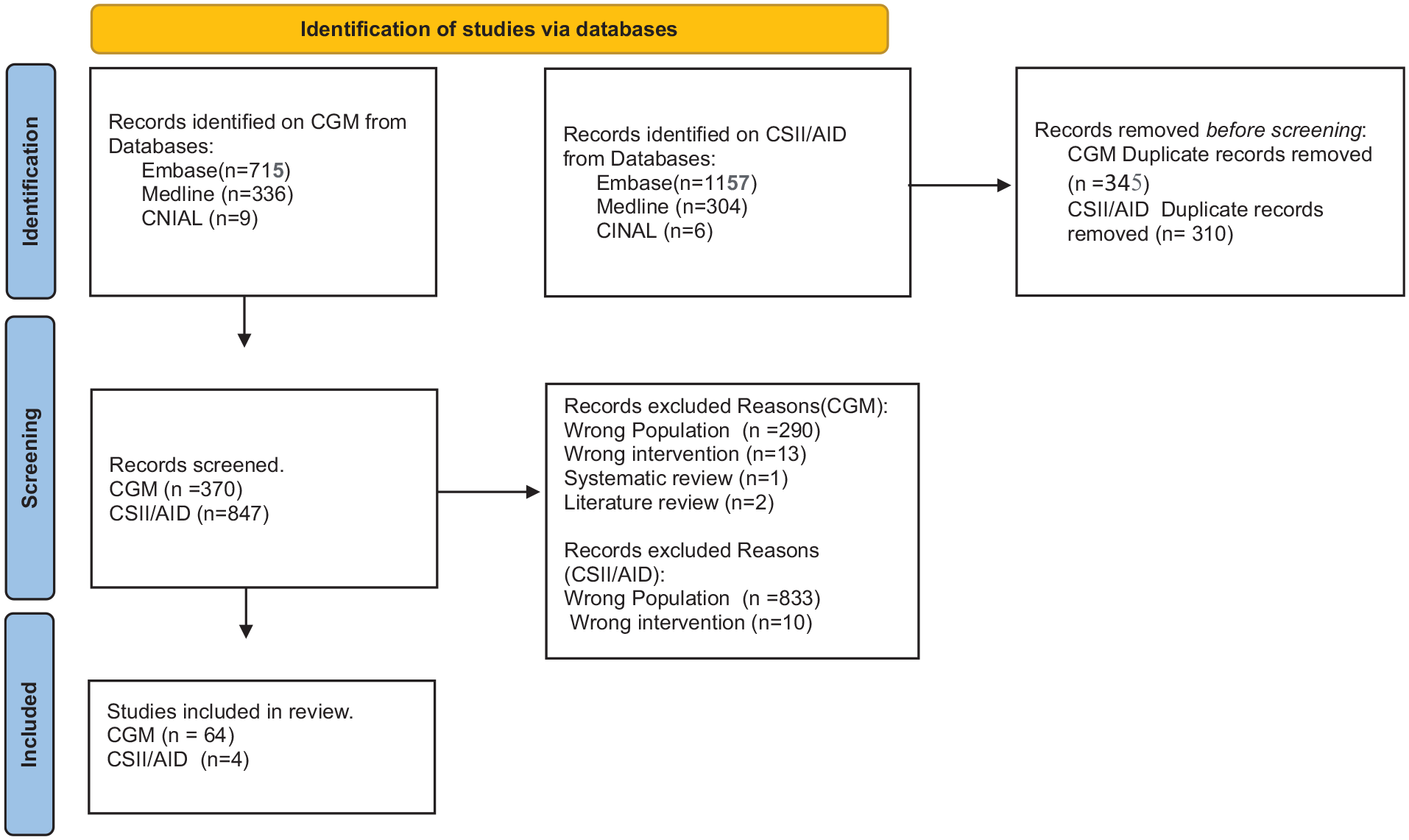
Flow diagram of study selection.
All included studies were published between 2003 and 2025, with the majority conducted in Europe (the United Kingdom, Denmark, France, Italy, Spain, Greece, Romania, Poland, Switzerland), Asia (Japan, China, India, Korea, Hong Kong, Bangladesh), North America (the United States of America), South America (Colombia, Brazil), and Australia. Samples ranged from 1 to 153 people, with follow-up durations from 4 hours to 12 months. Most participants were aged between 35 and 85 years, with 60% to 80% being male. Study designs comprised cohort (n = 27), clinical trials (n = 10), post hoc analyses (n = 5), cross-sectional (n = 9), case reports/series (n = 8), and pilot study (n = 5). Most focused on type 2 diabetes (n = 42): eight involved type 1, and 14 included mixed or unspecified types. The HD was the dominant modality (n = 46), followed by PD (n = 16) and both (n = 2). Technologies evaluated included CGMS (Dexcom G6, FreeStyle Libre, iPro2, Guardian Sensor 3) and closed-loop systems. Primary outcomes included device accuracy (Mean Absolute Relative Difference [MARD], Bland-Altman, error grids), glycemic metrics (time in range [TIR], variability, HbA1c), hypoglycemia detection, and AID safety, feasibility, and acceptability. Table 1 summarizes the study characteristics.
Study Characteristics.
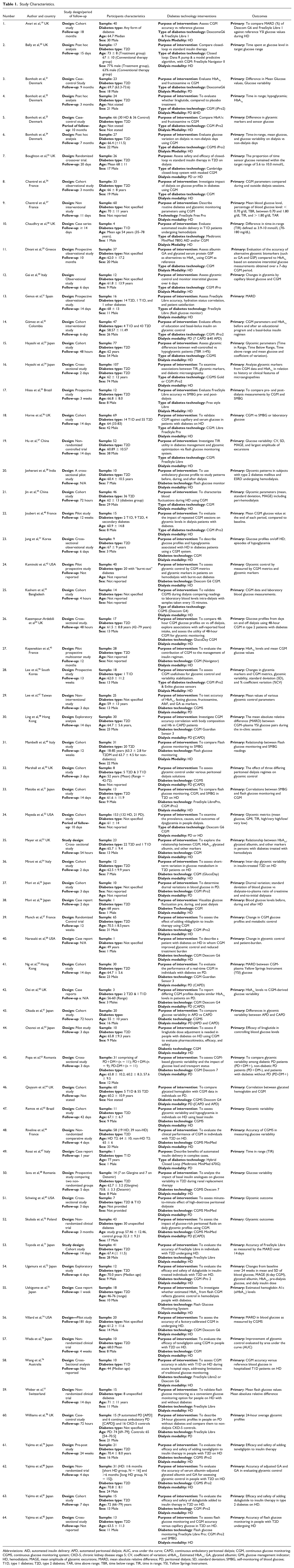
Abbreviations: AID, automated insulin delivery; APD, automated peritoneal dialysis; AUC, area under the curve; CAPD, continuous ambulatory peritoneal dialysis; CGM, continuous glucose monitoring; CGMS, continuous glucose monitoring system; CKD-5, chronic kidney disease stage 5; CV, coefficient of variation; eHbA1c, Estimated HbA1c; GA, glycated albumin; GMI, glucose management indicator; HD, hemodialysis; MAGE, mean amplitude of glycemic excursions; MARD, mean absolute relative difference; PD, peritoneal dialysis; SD, standard deviation; SMBG, self-monitoring of blood glucose; T1D, type 1 diabetes; T2D, type 2 diabetes; TAR, time above range; TBR, time below range; TIR, time in range; YSI, Yellow Springs Instrument.
Findings
The extracted data from the studies were categorized into six key themes: the validation of CGM accuracy in dialysis populations, comparisons between CGM and traditional glycemic markers, the impact of dialysis on glucose profiles, the use of CGM to assess the efficacy of diabetes treatments, CGM-based evaluations of glycemic outcomes, and the application of AID systems in the dialysis population. Tables 2 to 7 in Supplemental File 1 provide an overview of the outcomes assessed in the included studies and show how each study contributed to these thematic categories.
Validation studies: accuracy of CGM in dialysis populations
The 18 studies19,31,35,36,39,43,44,48
-51,59,66,71,74,76,77,82 reviewed included a total of 483 people undergoing dialysis, examining the accuracy and clinical utility of CGM and flash glucose monitoring devices in this population, with reference to capillary blood glucose or laboratory glucose measurements (see Table 2 in Supplemental File). Accuracy measures, such as the MARD, ranged from 8.1% to 29%,19,31,35,48,59,71,74,76,77,82 with most studies reporting MARD values between 10% and 20%. Correlation coefficients between CGM/flash glucose monitoring readings and capillary or laboratory glucose were generally strong (r2 ranging from 0.77 to 0.936). Accuracy was frequently reduced during or immediately after dialysis (
Comparisons between continuous glucose monitoring and traditional glycemic markers in dialysis populations
Thirteen studies21,23,29,47,53
-55,60,63,64,66,73,80 involving a total of approximately 397 people on dialysis, consistently show that HbA1c underestimates true glycemic level (see Table 3 in Supplemental File). Bomholt et al
23
reported a mean underestimation of 7.3 mmol/mol (95% confidence interval [CI] = –10.0 to –4.7,
Impact of dialysis on glucose profiles
Four studies involving approximately 103 participants investigated the effects of dialysis on glucose profiles in people with diabetes using CGM (see Table 4 in Supplemental File).26,61,69,70 Among those undergoing PD, individuals on automated peritoneal dialysis (APD) exhibited lower average blood glucose levels (181 ± 64 mg/dL) and less variability (SD = 36.3 ± 14.5 mg/dL) compared to those on continuous ambulatory peritoneal dialysis (CAPD), who recorded higher levels (238 ± 67 mg/dL; SD = 49.2 ± 14.1 mg/dL) (
Assessing the efficacy of diabetes treatments using continuous glucose monitoring
Eleven studies involving 257 participants with T2D (type 2 diabetes) on HD evaluated interventions including glucagon-like peptide-1 receptor agonists (GLP-1 RAs) (liraglutide, dulaglutide), dipeptidyl peptidase-4 (DPP-4) inhibitors (vildagliptin, teneligliptin), basal-bolus insulin, and basal insulin analogues (see Table 5 in Supplemental File).22,32,45,57,62,65,68,72,75,79,81 Bomholt et al
22
reported a non-significant change with liraglutide (7.5-7.4%,
Improvements in glycemic variability were also reported. Yajima et al
81
demonstrated a reduction in the coefficient of variation (CV) from 28.1% to 19.8% (
Continuous glucose monitoring-based evaluation of glycemic outcomes in the dialysis population
Twenty-two studies involving 768 people with diabetes on dialysis reported glycemic outcomes (see Table 6 in Supplemental File).24,27,30,32,33,34,37,38,40
-42,46,52,54-56,58,63,65,68,78 Glucose levels typically fell during dialysis and rebounded afterwards, with notable nocturnal variation, eg, drop from 8.8 to 8.4 mmol/L (
Automated insulin delivery in the dialysis population
Four studies involving 48 participants with diabetes undergoing dialysis (T1D, T2D) evaluated the safety and efficacy of AID systems (see Table 7 in Supplemental File).20,25,28,67 All studies reported significant improvements in glycemic levels, particularly in TIR. Bally et al
20
found that users of closed-loop systems spent 69.0% of the time within the target range, compared with 31.5% in the control group, a difference of 37.6% (95% CI = 24.4-50.8;
Mean glucose levels also declined. Boughton et al
25
reported a reduction from 11.6 to 10.1 mmol/L (
Additional outcomes included lower glycemic variability; Rossi et al
67
reported a reduction in the CV to 24% at one year and a decrease in HbA1c to 55 mmol/mol (7.2%). Chaudhry et al
28
observed a significant decrease in TAR, from 55.5% to 34.8% (
Discussion
This review highlights the potential of CGM and AID technologies to enhance diabetes management in the dialysis population. Evidence from a range of studies underscores their effectiveness in addressing the distinct challenges associated with both HD and PD. Individuals receiving dialysis are particularly susceptible to hypoglycemia, hyperglycemia, and glycemic variability, largely due to dialysis-induced metabolic changes.
Validation studies included in the review confirm that CGM systems offer acceptable accuracy in both HD and PD settings, with MARD values typically between 10% and 23%. These results align with findings in non-dialysis populations83 -88 and show strong correlations with laboratory glucose and self-monitoring of blood glucose (SMBG), supporting CGM as a reliable monitoring tool for people on dialysis. 89 However, accuracy often declines during and shortly after HD, likely due to physiological changes and sensor lag,90,91 requiring cautious interpretation. Despite this, CGM provides valuable insights such as trend data and alarms. In contrast, SMBG, while accurate, provides only snapshot measurements and lacks the continuous glucose profile offered by CGM. Furthermore, Clarke and Parkes Consensus Error Grid analyses across the included studies demonstrated high clinical accuracy of CGM and flash glucose monitoring devices in dialysis populations. Specifically, 97%–99% of glucose readings fell within Zones A and B on the Clarke Error Grid, and 98%–100% on the Parkes Consensus Error Grid, indicating no or minimal clinical risk. These results are comparable to findings in non-dialysis populations, supporting the reliability of CGM in the dialysis setting.
Traditional markers such as HbA1c consistently underestimate glycemic levels in dialysis populations due to altered red blood cell lifespan, erythropoietin use, and the uremic environment. This review reaffirms that HbA1c is less reliable in this context. Glycated albumin shows stronger correlation with CGM-derived glucose values and provides a more accurate measure of glycemic control in people receiving dialysis, 92 highlighting the need to consider alternative or additional markers. The TIR has been proposed as a complementary biomarker, offering several advantages over HbA1c. It reflects glycemic variability, captures hypoglycemia and hyperglycemia, responds quickly to treatment changes, and is not affected by pathological factors that may affect HbA1c levels. 90
The dynamic impact of dialysis on glucose profiles was evident. Differences in glucose patterns between APD and CAPD modalities further illustrate the complexity of glycemic management in this population, particularly due to the sustained hyperglycemia associated with high-glucose dialysate solutions. Moreover, mean glucose levels were significantly lower during dialysis sessions and higher post-dialysis, accompanied by increased glucose variability and a higher incidence of hypoglycemia during dialysis. These glycemic fluctuations present a clinical challenge and underscore the value of CGM in providing real-time insights into glucose trends and enabling timely interventions.
Studies evaluating the use of CGM to guide diabetes treatment have demonstrated its effectiveness in monitoring therapeutic impact on glycemic control. These findings underscore the value of CGM in optimizing medication regimens for people on dialysis by enhancing efficacy and reducing the risk of hypoglycemia. Intervention studies further support the potential of CGM-guided adjustments to improve glycemic outcomes in this population. In non-dialysis populations, CGM is also a feasible tool for comparing treatment approaches. 93 However, uncertainty regarding regulatory acceptance of CGM data may hinder wider adoption. Clear guidance from authorities such as the Conformité Européenne (CE) and Food and Drug Administration (FDA) would help establish its role in the evaluation of new therapies. 93
This review further highlights the utility of CGM in managing glycemia and glycemic variability in people on dialysis. The CGM effectively identifies intradialytic glucose lows and post-dialytic highs, supporting tailored insulin adjustments and improved glycemic level without increasing the risk of hypoglycemia. It also captures glycemic differences between dialysis and non-dialysis days, providing insights into metabolic fluctuations. These findings are supported by previous systematic reviews,15 -17, 94 further reinforcing the potential of CGM in this population. 89
The AID systems have proven effective in non-dialysis populations, improving TIR, reducing hypoglycemia, and enhancing glycemic variability compared to standard insulin therapy.95,96 Emerging evidence in dialysis people shows similar benefits despite metabolic fluctuations during HD or PD. Closed-loop insulin delivery offers a person-centered approach by lessening the need for frequent monitoring and insulin adjustments. However, altered insulin clearance, slow insulin pharmacokinetics, and potential CGM inaccuracies during dialysis present challenges. 89 Still, studies in T1D and T2D people on dialysis report improved glucose control and safety with fully closed-loop systems. Although current data are limited, larger, longer-term trials are needed to confirm these benefits and support wider use in this complex group.
Despite growing evidence and endorsement from national and international guidelines supporting the use of diabetes technologies in dialysis settings,8,97 their utilization remains limited in this vulnerable population. Historically, and in line with manufacturer guidance, the use of CGM devices (including Dexcom G5/G6/G7 and FreeStyle Libre 1/2/3) has not been recommended for people on dialysis due to limited accuracy data in this group. However, as the evidence base grows, manufacturers will need to reconsider their current guidance.
Laursen et al 98 explored the experiences of people on HD using CGM, identifying six key themes: improved management of hypo- and hyperglycemia, informed decision-making, reduced practical challenges, enhanced confidence and safety, greater effectiveness with health care professional support, and mixed opinions on alarms. Overall, participants viewed CGM as a valuable tool for diabetes management during dialysis. However, additional health care professional support or tailored education may be needed to maximize CGM use and alleviate psychosocial challenges such as anxiety related to alarms or device burden, thereby enabling more sustained use.
Both CGM and AID offer significant benefits for glycemic management in this population. Yet, barriers including cost, limited clinical capacity, and workforce readiness continue to hinder routine integration. 13 Persistent inequities in access further restrict utilization and limit optimization of diabetes care for those at greatest risk.89,99 To overcome these challenges, targeted staff training, adequate funding, and clear care pathways are essential. These measures would support effective adoption, provide ongoing clinical support, and promote equitable access to diabetes technologies in dialysis settings. Further research on implementation strategies is needed to refine treatment approaches and improve care for people with diabetes undergoing dialysis. While CGM and AID systems show promise, their high costs, lack of universal reimbursement, and limited access, particularly in low- and middle-income countries, pose significant barriers. Given the complex medical needs of this population, it is essential to raise awareness and promote equitable access to these technologies wherever feasible.
Strengths and Weaknesses
This review provides a comprehensive overview of the potential benefits of CGM and AID in dialysis populations, drawing on studies from various dialysis modalities. It offers valuable insights into efficacy, safety, and glycemic outcomes but is limited by small sample sizes, varying follow-up durations, and the inclusion of lower-level evidence such as cross-sectional analyses, audits, and case reports, which may affect consistency and weaken conclusions. While scoping reviews typically do not include formal quality appraisal, the predominance of observational data and variability in study quality should be considered when interpreting the findings. Most studies were conducted in Europe, Asia, and the Americas, with limited representation from low- and middle-income countries, potentially restricting the generalizability of the results.
Conclusion
This review highlights the promising role of CGM and AID in improving diabetes management in dialysis populations. The CGM reliably monitors glucose, offering valuable insights to guide treatment, while AID improves glucose level and safety with a personalized approach. Integrating these technologies into routine care could enhance outcomes and reduce diabetes burden in people on dialysis. Wider adoption requires clear clinical pathways, dedicated funding, and health care professional training. Further research is needed to optimize implementation and ensure optimal care.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251353811 – Supplemental material for Scoping Review—Diabetes Technology for Individuals on Kidney Replacement Therapy (Dialysis): Current Trends and Future Directions
Supplemental material, sj-docx-1-dst-10.1177_19322968251353811 for Scoping Review—Diabetes Technology for Individuals on Kidney Replacement Therapy (Dialysis): Current Trends and Future Directions by Hellena Hailu Habte-Asres, Joseph Ngmenesegre Suglo, Khuram Chaudhry, Angus Forbes, David C. Wheeler and Janaka Karalliedde in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
Nil.
Abbreviations
AID, automated insulin delivery; APD, automated peritoneal dialysis; AUC, area under the curve; CAPD, continuous ambulatory peritoneal dialysis; CE, Conformité Européenne; CGM, continuous glucose monitoring; CINAHL, Cumulative Index to Nursing and Allied Health Literature; CSII, continuous subcutaneous insulin infusion; CV, coefficient of variation; DPP-4 inhibitors, dipeptidyl peptidase-4 inhibitors; ESKD, end-stage kidney disease; FDA, Food and Drug Administration; GLP-1 RA, Glucagon-Like Peptide-1 Receptor Agonists; HbA1c, glycated hemoglobin; HD, hemodialysis; KRT, kidney replacement therapy; MARD, mean absolute relative difference; MeSH, medical subject headings; mg/dL, milligrams per deciliter; PD, peritoneal dialysis; PRISMA, preferred reporting items for systematic reviews and meta-analyses; SD, standard deviation; SMBG, self-monitoring of blood glucose; T1D, type 1 diabetes; T2D, type 2 diabetes; TAR, time above range; TBR, time below range; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HHH-A received speaker honoraria from AstraZeneca and Bayer. JNS, KC, and AF have declared no conflicts of interest. DCW has an ongoing consultancy contract with AstraZeneca and has received payments for consultancy and/or speaking activities from multiple companies, including Amgen, Astellas, Bayer, Boehringer Ingelheim, Eledon, GSK, Galderma, Gilead, Janssen, Mundipharma, Menarini, MSD, NovoNordisk, Pharmacosmos, Tricida, and Vifor. JK has received honoraria for delivering educational meetings and/or attending advisory boards from Boehringer Ingelheim, AstraZeneca, Sanofi, and NAPP and research grants from AstraZeneca and Sanofi.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
