Abstract
Background:
Glycemic control in the hospital setting is imperative for improving outcomes among patients with diabetes. Bedside point-of-care (POC) glucose monitoring has remained the gold standard for decades, while only providing momentary glimpses into a patient’s glycemic control. Continuous glucose monitoring (CGM) has been shown to improve glycemic control in the ambulatory setting. However, a paucity of inpatient experience and data remains a barrier to US Food and Drug Administration (FDA) approval and expanded/non-research use in the hospital setting.
Method:
Amid the COVID-19 pandemic, the FDA exercised its enforcement discretion to not object to the use of CGM systems for the treatment of patients in hospital settings to support COVID-19 health care–related efforts to reduce viral exposure of health care workers. Following this announcement, Scripps Health, a large not-for-profit health care system in San Diego, California, implemented CGM as the new “standard of care” (CGM as SOC) for glucose monitoring and management in the hospital.
Results:
The present report serves to (1) detail the implementation procedures for employing this new SOC; (2) describe the patients receiving CGM as SOC, their glycemic control, and hospital outcomes; and (3) share lessons learned over two years and nearly 900 hospital encounters involving CGM.
Conclusions:
Here, we conclude that CGM is feasible in the hospital setting by using a dedicated diabetes care team and the CGM technology with remote monitoring.
Introduction
Over a quarter of patients admitted to non-intensive care unit (non-ICU) hospital settings have diabetes, 1 and nearly a third of adult patients hospitalized in California have diabetes. 2 Notably, these hospitalizations are, on average, more costly than those of patients without diabetes, regardless of the primary reason for admission. 3 Higher health care costs can be attributed, in part, to poor glycemic control in the hospital. Due to the dynamic and often unpredictable nature of the hospital environment; frequent fluctuations in sleep, appetite, nutritional status, and medications; and the physiological stress of acute illness or injury, poor glycemic control in the hospital is common and can lead to adverse outcomes. Inpatient hyperglycemia is an independent, significant contributor to hospital-acquired infection, a critical outcome for patients and health systems,4,5 while both hypoglycemia and hyperglycemia have been associated with longer lengths of stay (LOSs), increased mortality, and higher health care costs.1,5 -16
Given the consequences of poor inpatient glycemic control, hospitals have devoted significant resources to improving glucometrics. However, current approaches are impeded by the limitations of capillary point-of-care testing (POCT). For non-ICU patients with diabetes, POCT is conducted approximately every four to six hours.17,18 Outside these instances, glucose goes largely unmonitored, leaving patients at risk for undetected hypo/hyperglycemia. Retrospective (blinded) continuous glucose monitoring (CGM) studies have shown POCT to miss approximately one-third of postprandial hyperglycemic excursions, 19 and 78% 20 to 90% 19 of hypoglycemic events among hospitalized patients with known diabetes. A sizable percentage of missed hypoglycemic episodes were asymptomatic (26%), and/or occurred overnight (>50%). A 2016 expert panel agreed that CGM in the non-ICU setting has the potential to detect hypo/hyperglycemic events that would otherwise be missed with POCT. 21
Despite widespread use and effectiveness for
Due to the need to limit exposure and preserve personal protective equipment during the COVID-19 pandemic, the FDA released a statement on April 1, 2020, exercising its enforcement discretion to not object to the use of CGM systems for the treatment of hospitalized patients to support COVID-19 health care–related efforts. In May 2020, Scripps Health implemented CGM as the new “standard of care” (CGM as SOC) for inpatient glucose monitoring. As the use of this technology is novel in this setting, the present report describes implementation, the characteristics and outcomes of those who received CGM as SOC, and important, real-world lessons learned.
Methods
Institutional review board (IRB) approval was not required to implement CGM as SOC but was obtained for evaluation and reporting purposes.
Setting
Continuous glucose monitoring as SOC program is underway on the non-ICU floor units of Scripps Mercy Hospital’s San Diego, CA, campus—a disproportionate share of ~500-bed hospital, predominantly serving low-resource patients.
Teams
CGM as SOC is supported by specialized teams for each aspect of the program and a multi-disciplinary leadership team. The off-site data team provides daily patient eligibility lists with calculated priority scores each morning to inform program recruitment efforts. On a patient’s completion of the program, the data team downloads and securely stores their CGM and electronic medical record (EMR) data for future evaluation. The on-site team receives the daily eligibility lists and, with approval of the bedside nurses, enrolls patients in the program and conducts CGM initiation/placement/removal and technical troubleshooting. A remote CGM advanced practice nurse (APN) team reviews daily CGM trends and provides insulin recommendations to bedside nurses and attending physicians. The CGM APNs are certified diabetes care and education specialists trained in insulin management. Infrequently, a patient is managed directly by inpatient endocrinology, and the APN team does not manage insulin use.
Hospital bedside nurses are trained to use CGM values for glucose management. The remote 24/7 hypoglycemia monitoring team (RMT) is a multi-function non-clinical, centralized, off-site team trained in the competencies of alarm recognition who alert bedside nurses of impending or ongoing hypoglycemia that requires immediate action. Program development, oversight, and evaluation are provided by a master’s-level program director, PhD-level translational research scientists, and physician scientists specializing in adult endocrinology. Organization of the program teams is depicted in Figure 1.
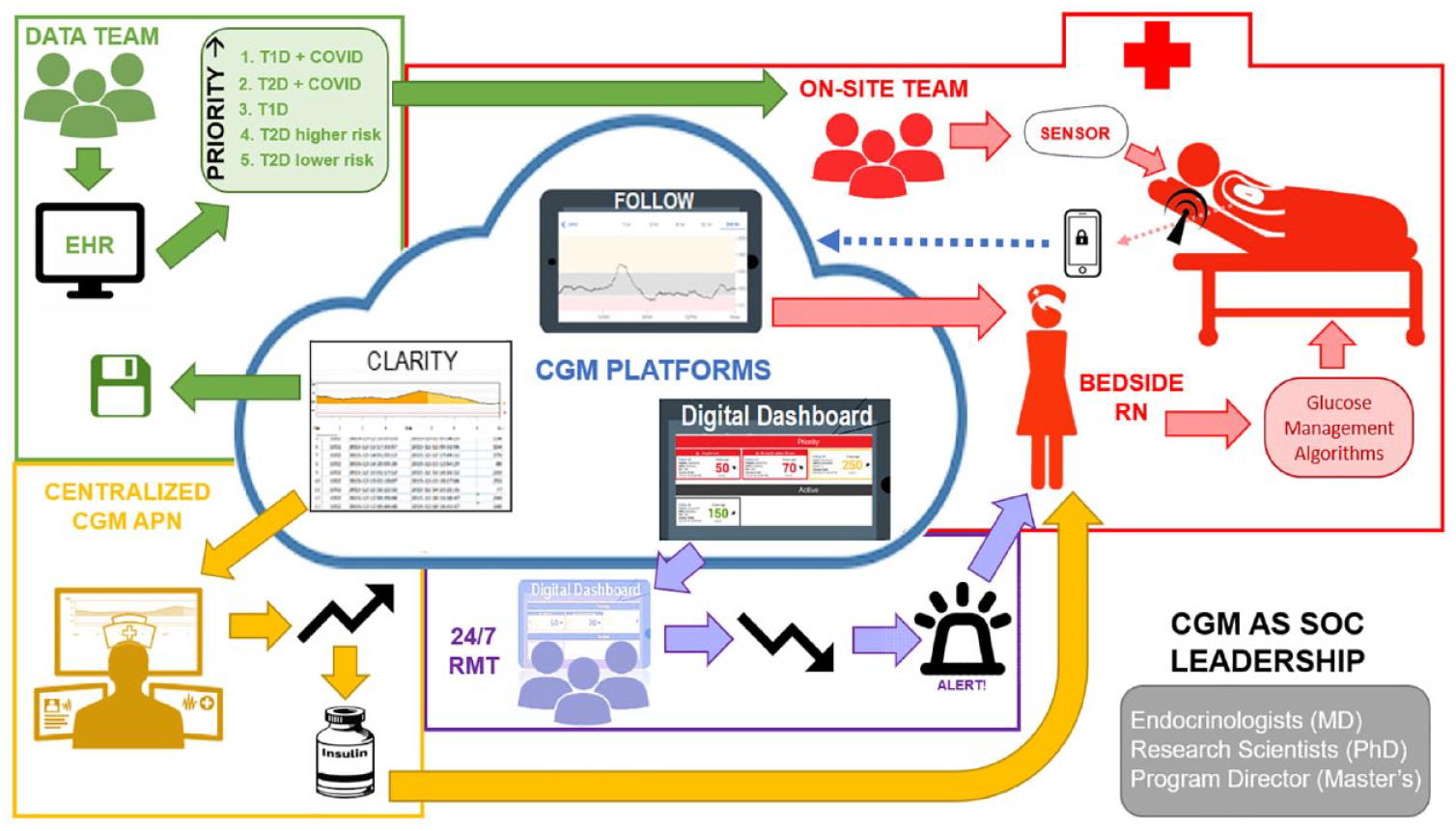
CGM data platforms and teams.
Patients
Patients ≥18 years of age with a diabetes diagnosis receiving insulin orders during their admission were eligible for CGM as SOC. Exclusion criteria include (1) discharge to home/non-CGM unit, procedure requiring CGM device removal, and/or discontinuation of insulin orders anticipated within 24 hours; (2) adhesive allergy; (3) inpatient use of personal CGM device; and (4) any condition the care team deems contraindicated. As more patients were eligible for CGM as SOC daily than could be accommodated, a clinical prioritization score system was applied (Table 1).
Risk Score Calculation for Patient Prioritization.
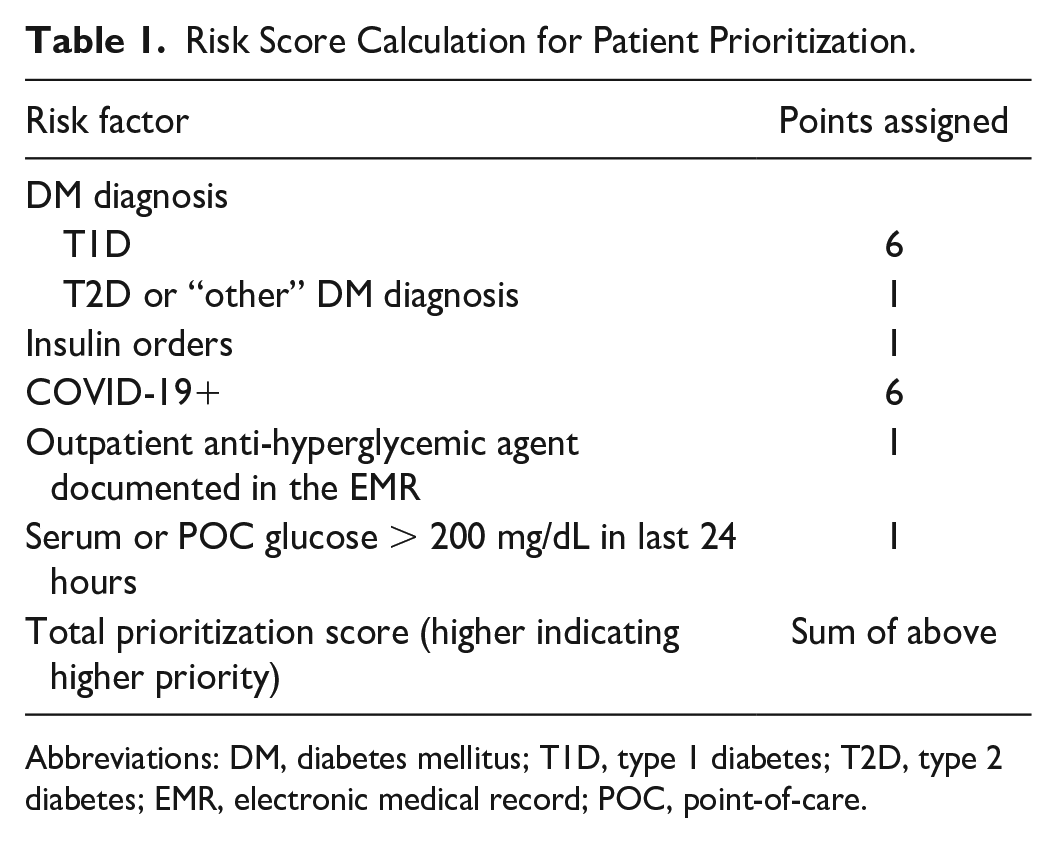
Abbreviations: DM, diabetes mellitus; T1D, type 1 diabetes; T2D, type 2 diabetes; EMR, electronic medical record; POC, point-of-care.
Devices, Platforms, and Technology Usage
The Dexcom G6 CGM System (Dexcom, Inc., San Diego, CA) was selected for its ability to transmit data for remote monitoring, factory calibration, short warm-up period, and alarm notifications. The CGM transmitter uses Bluetooth technology to send data to the G6 application on a hospital smartphone located in a lockbox at bedside (Figure 1). Continuous glucose monitoring values and alarms are not visible/audible in patient rooms. Following a two-hour warm-up, the smartphone transmits values every five minutes to three secure, Health Insurance Portability and Accountability Act (HIPAA)-compliant cloud-based platforms (Figure 1):
Dexcom FOLLOW is an app-based tool on an iPad at the nursing station for bedside nurses to use for real-time, routine monitoring of CGM data at scheduled daily checks and following any alerts from the RMT prior to potential intervention;
Digital Dashboard (not yet commercially available) is a web-based tool monitored 24/7 by the RMT that allows real-time surveillance and alerts of impending or ongoing hypoglycemia and saves monitoring time by prioritizing patients according to clinical risk for rapid/proactive intervention using real-time CGM data. The Digital Dashboard is programmed with alarms for glucose ≤80 mg/dL (to allow time for the bedside registered nurse to be notified and treat prior to <70 mg/dL) and trend arrows indicating if glucose continues to fall at the current rate, levels could decrease by up to 30 mg/dL in 15 minutes. In response, the RMT contacts the bedside nurse who consults the FOLLOW app to obtain the glucose value and treat per protocol (Table 2).
Dexcom CLARITY is a web-based data management and reporting tool where CGM trends and metrics can be viewed via easy-to-read graphs and used by the CGM APNs for clinical optimization. The data team also uses CLARITY, following a patient’s completion of the program, to download complete CGM data for retrospective evaluation.
Standardized Glucose Management Algorithms Used by CGM Nurses.
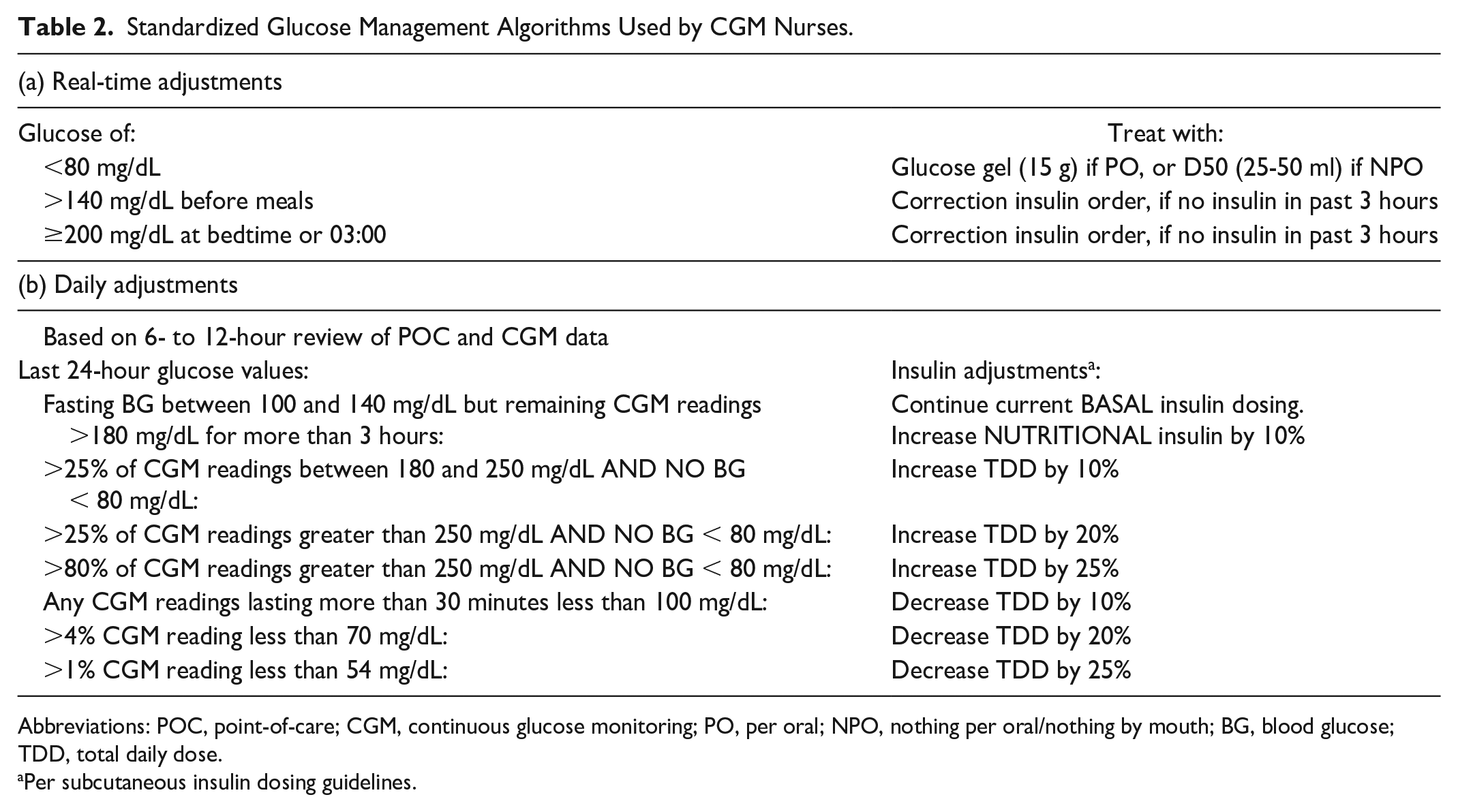
Abbreviations: POC, point-of-care; CGM, continuous glucose monitoring; PO, per oral; NPO, nothing per oral/nothing by mouth; BG, blood glucose; TDD, total daily dose.
Per subcutaneous insulin dosing guidelines.
Implementation Procedures
The CGM on-site team applies the CGM system on the abdomen or, infrequently (eg, prone patients), on the upper arm. Once inserted, the system remains in place for the duration of the hospitalization, with sensor changes every 9 days (to prevent exceeding the 10-day device limit) or more frequently as needed. The CGM device is removed for magnetic resonance imaging (MRI), computed tomography (CT), and surgical procedures using diathermy, 21 and replaced after. To monitor accuracy, validation against POCT is conducted ~six hours after device placement using the %20/20 agreement rate. 29 Once validated, CGM values are used for all glucose checks, and the device is re-validated once daily. Any time a CGM value is obtained from FOLLOW by the bedside nurse, in place of a routine POC or to guide a real-time intervention, the nurse will document this CGM value directly into the EMR (EPIC Systems, Verona, WI) in a dedicated flowsheet within the Glucose Management report that allows clear distinction of glucose value sources (eg, POCT, serum, CGM). If validation fails, the bedside nurse conducts subsequent validation checks with each routine POCT and does not use the CGM data for clinical management until validated by the %20/20 rule, or, in rare cases, replaces the device following multiple failed validations.
Daily Glucose Trend Reviews and Medication Guidance
The CGM APNs use CLARITY for daily review of glucose trends and provides insulin adjustment recommendations based on standardized algorithms (see Table 2) to the attending physician. Of note, management of basal/bolus insulin is based on overnight and post-prandial trends, which are not available under standard POCT protocols that assess pre-meal and bedtime glucose alone.
Data Sources and Analysis
Continuous glucose monitoring as SOC data are entered in Research Electronic Data Capture (REDCap).30,31 Patient data are obtained via the EMR through an automated data acquisition platform built into Snowflake Data Cloud (Snowflake Inc, Bozeman MT). Continuous glucose monitoring data for each encounter are downloaded via Dexcom CLARITY.
Data are presented overall, and separately for type 1 diabetes (T1D) and T2D. We did not aim to compare diabetes types, so data are solely presented descriptively. Categorical data are described as frequencies/percentages, and continuous data as means and standard deviations, or medians and interquartile ranges (IQRs), if skewed. Encounter-level CGM metrics included CGM duration, glucose mean and standard deviation, coefficient of variation, and several TIR, time-above-range, and time-below-range metrics, as well as hypoglycemic events (as previously described).27,32 The POCT/CGM concordance was calculated using encounter-level and overall mean absolute relative differences (MARDs) and is displayed via a Clarke Error Grid (CEG) 33 using the “ega” package 34 in R. Comorbidity data were reported based on International Classification of Diseases (ICD) code classification using the “comorbidity” package 35 in R.
All data processing and analyses were conducted in R v. 4.0.3. 36
Results
This analysis spans 26 months of data (May 2020-July 2022) and N = 891 admissions (n = 47 [5%] T1D, n = 844 [95%] T2D) from N = 800 unique individuals (n = 39 T1D, n = 761 T2D). As detailed in Table 3, patients were 64.2 years of age, on average, and nearly half (47.5%) self-identified as Hispanic. Most patients reported outpatient anti-hyperglycemic use (65.8%) and had a POC or serum glucose >200 mg/dL in the 24 hours prior to CGM initiation (68.5%). Glycated hemoglobin A1c (HbA1c) was elevated (mean: 8.6%), as was the first POC or serum glucose on admission (mean: 242 mg/dL). Most individuals had diabetes with chronic complications (57.7%), and specific comorbidity burden was high. Nearly one-fifth of patients tested positive for COVID-19 during their hospitalization.
Demographic and Clinical Characteristics.
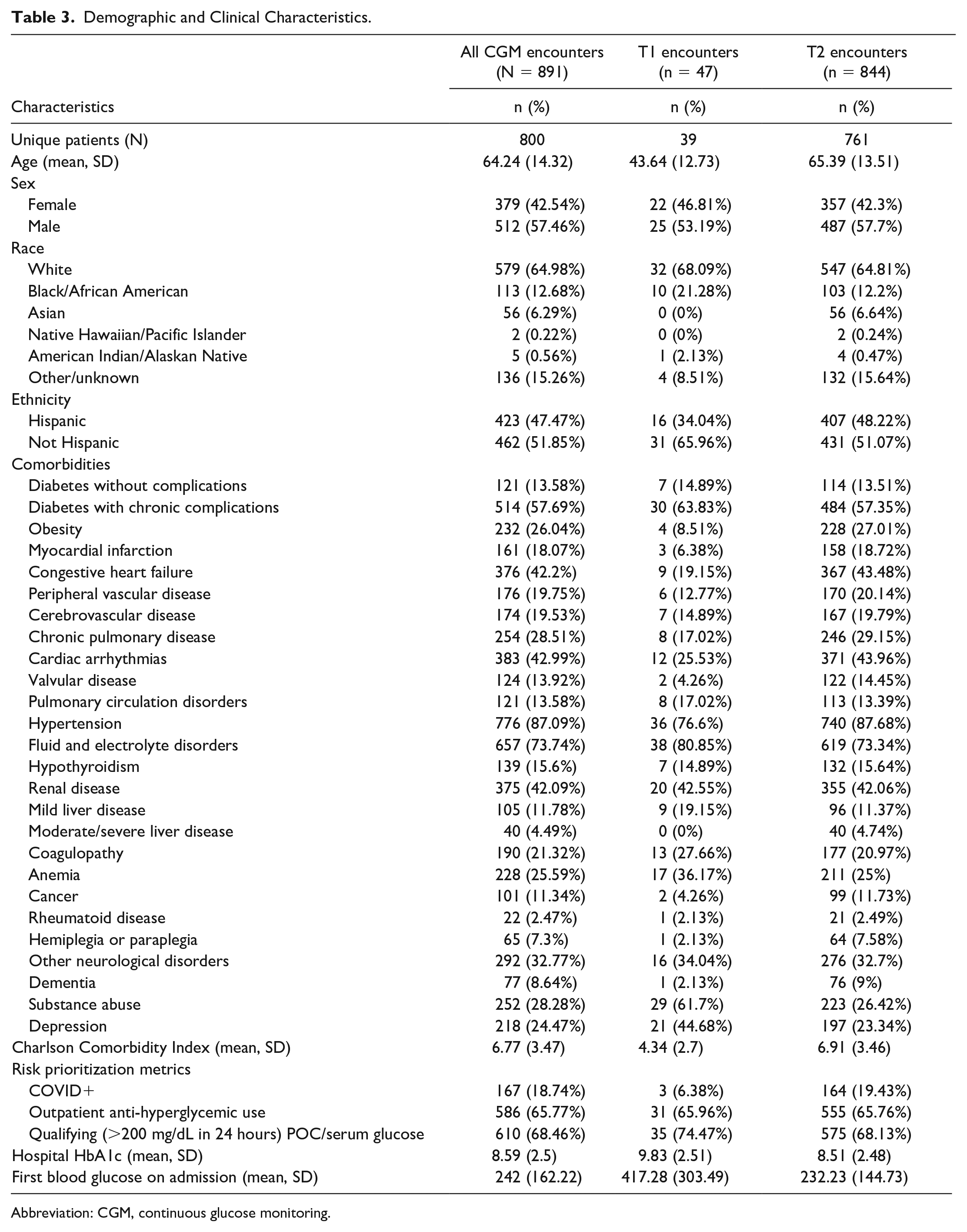
Abbreviation: CGM, continuous glucose monitoring.
Nearly all (95%) hospitalizations were urgent/emergent admissions. Most patients enrolled were admitted to the medical/surgical (60.7%) or telemetry (30.2%) units. Patients had CGM devices placed at a median of three days into their hospital stay and had a median LOS of eight days. Many patients enrolled in this program had surgery during the hospitalization (38.3%), had a diagnosis of sepsis (36.7%), or were admitted to the ICU for some portion of their hospital stay (20.1%). Patients were most frequently transferred on discharge to skilled nursing, long-term care, hospice, or other facility (51.9%); whereas 43.6% were discharged home. Ultimately 4.6% of patients were deceased on discharge, and all deceased had T2D (Table 4).
Hospitalization Characteristics.
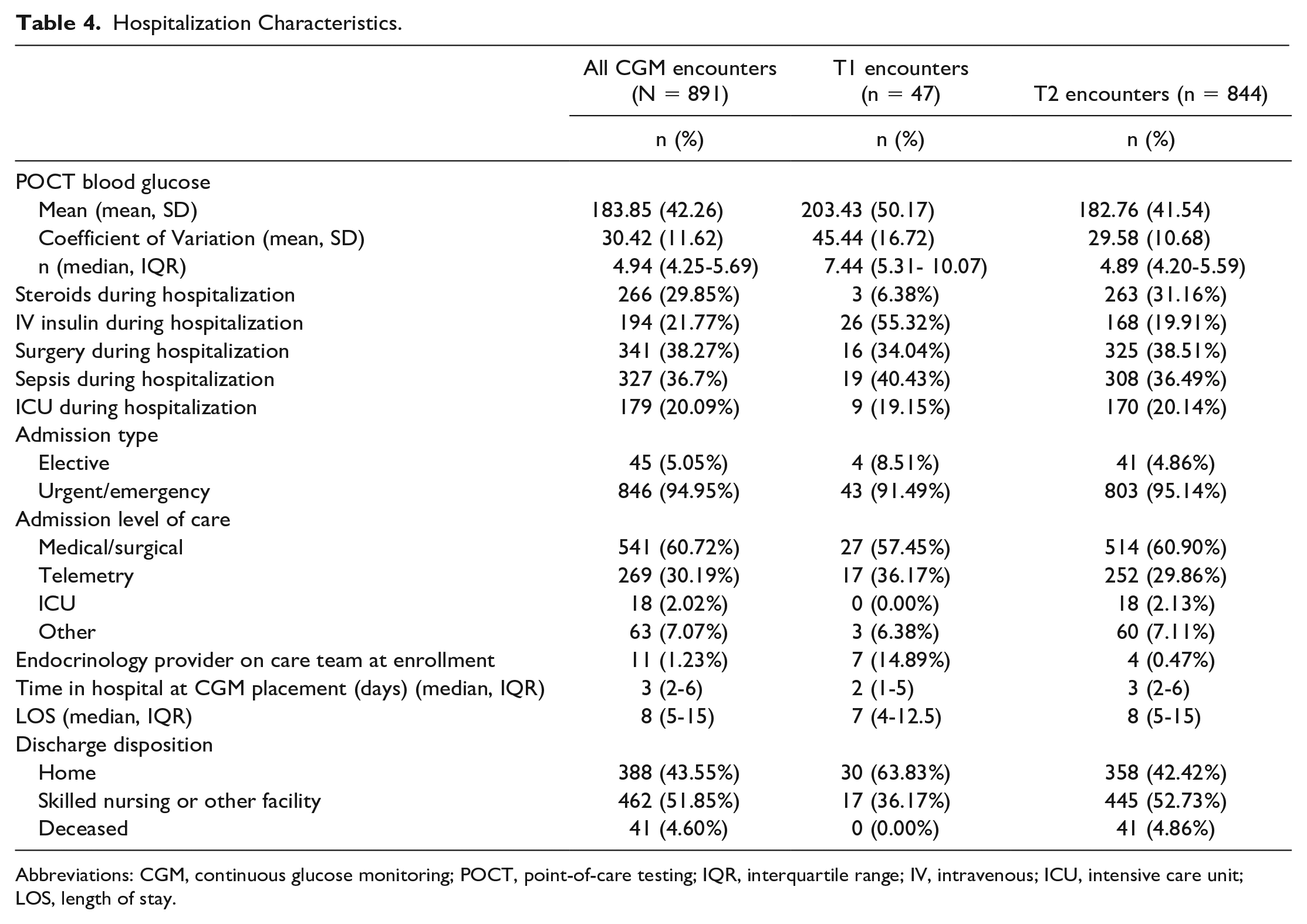
Abbreviations: CGM, continuous glucose monitoring; POCT, point-of-care testing; IQR, interquartile range; IV, intravenous; ICU, intensive care unit; LOS, length of stay.
Nearly a third of patients were administered systemic steroids during their hospitalization. Patients were on intravenous (IV) insulin in 21.8% of encounters. Over the course of the hospitalization, patients received a median of 4.9 POCTs/day, with a mean POCT glucose of 183.8 mg/dL (Table 4).
Continuous glucose monitoring metrics are presented in detail in Table 5 for both the first 24 hours that a patient was on CGM and for their entire duration of CGM device wear. Median overall wear time was 73 hours. During the first 24 hours, patients had mean glucose more than 200 mg/dL, with 39.4% TIR 100 to 180 mg/dL. Most excursions from optimal range were hyperglycemia (56% >180 mg/dL, overall) with infrequent (1% <70 mg/dL; 0.5% <54 mg/dL) hypoglycemia, including only 10% of patients having any hypoglycemic event. Examining the data over the entire CGM period shows modest descriptive improvements, with a mean glucose of 195 mg/dL and 44.4% TIR 100 to 180 mg/dL. Again, most deviations from optimal were due to hyperglycemia (52% >180 mg/dL) rather than hypoglycemia (0.6% <70 mg/dL; 0.3% <54 mg/dL), where 21% had any hypoglycemic events—among those, the median was one event lasting under one hour. Concordance between CGM and POCT measured by MARD was 16.61%, on average; and visualizing concordance within the measurable range of the G6 revealed 99.1% of matched pairs were in the benign zones A/B, whereas less than 1% of values fell within the critical zones D/E (Figure 2). Most individuals only wore one sensor during their hospitalization (78.6%) with a median wear time per sensor of 72 hours.
CGM Characteristics, 24 Hours and Entire CGM Duration.
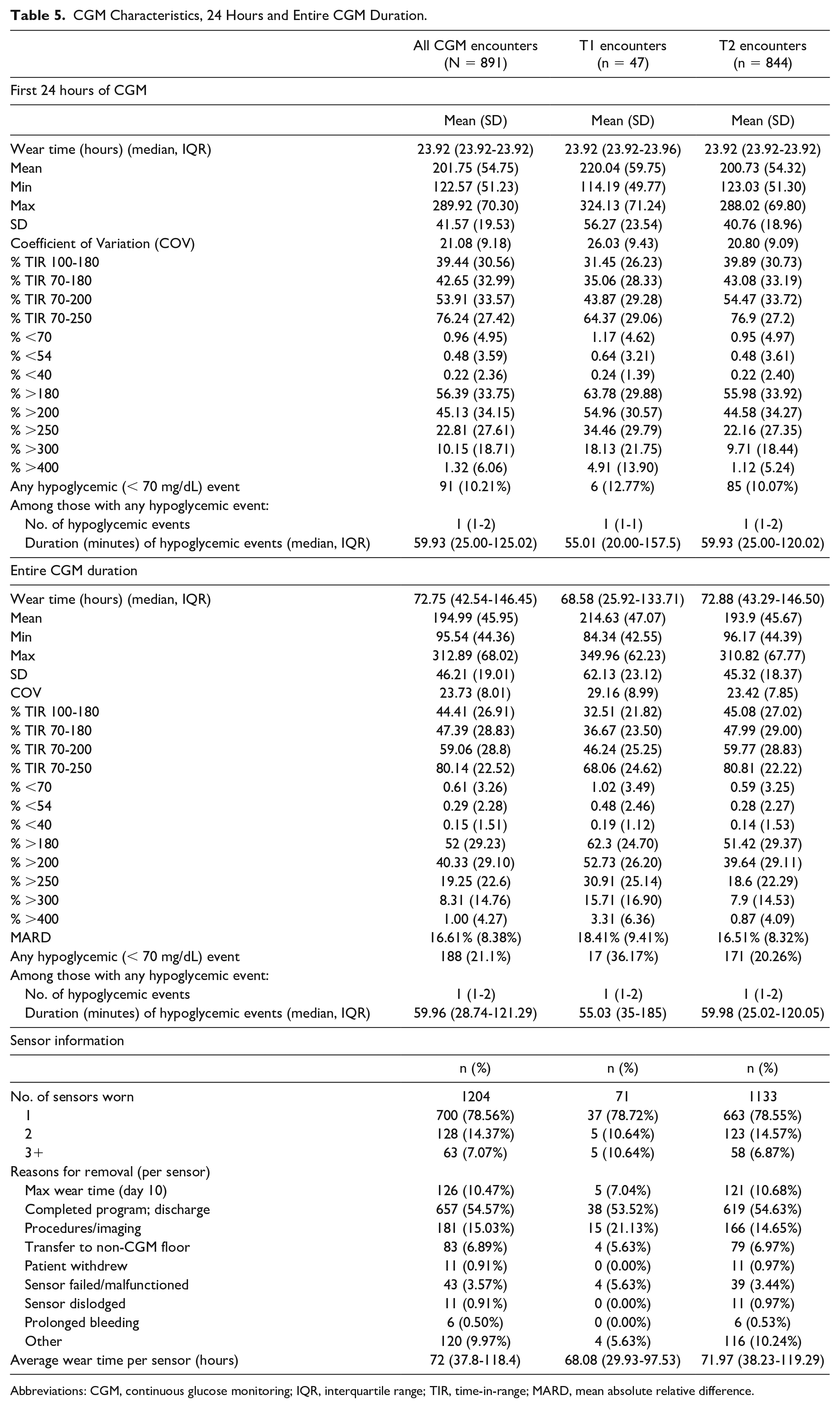
Abbreviations: CGM, continuous glucose monitoring; IQR, interquartile range; TIR, time-in-range; MARD, mean absolute relative difference.
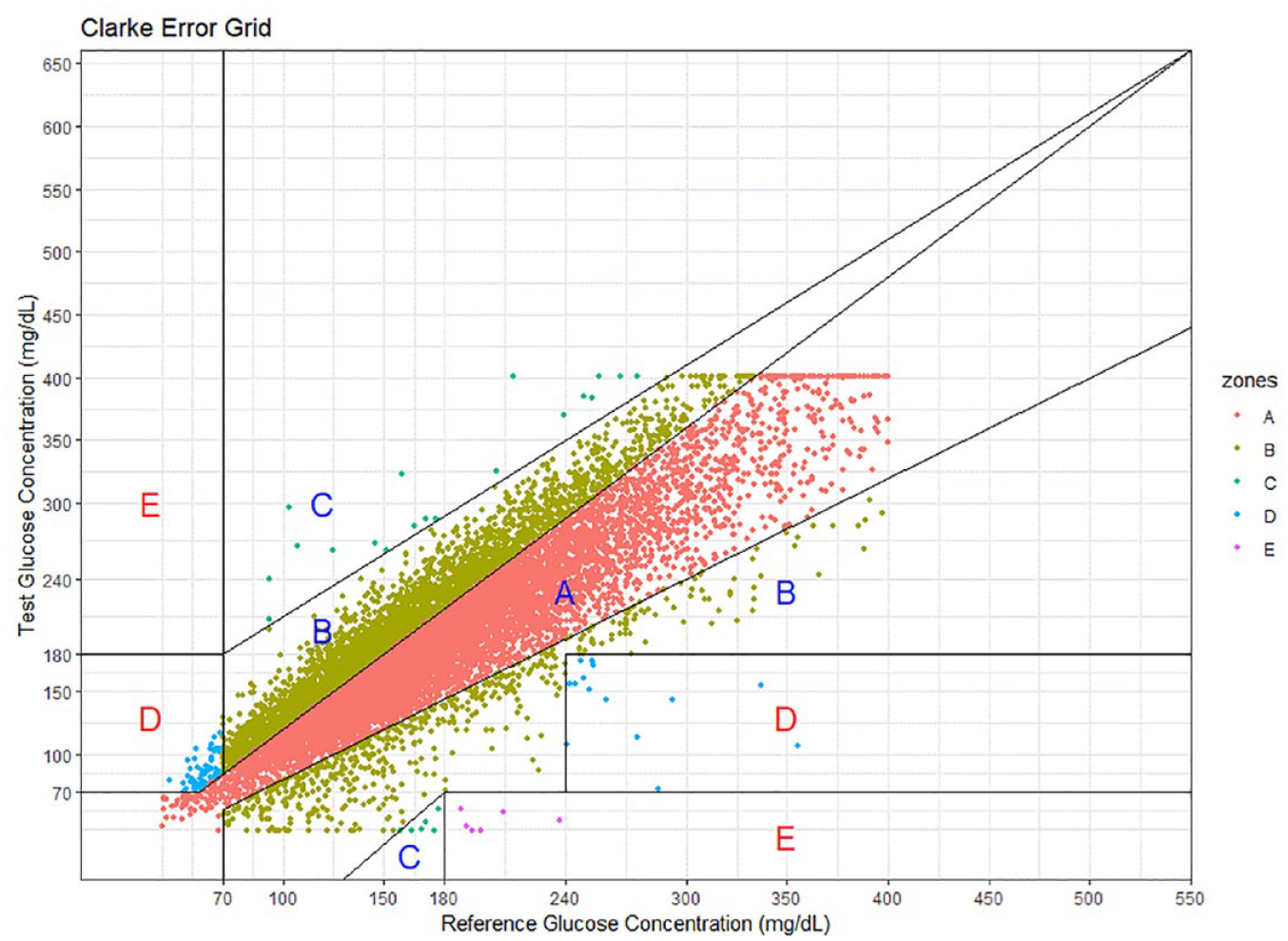
Clarke Error Grid (CEG) for all POC-CGM pairs (N = 13 202) within the CGM device measurable range.
Discussion
At the outset of the COVID-19 pandemic, our diabetes research and clinical teams rapidly pivoted from evaluating CGM in the hospital setting in the context of research, to a real-world, hospital-wide initiative to care for high-risk patients with diabetes. Informed by the implementation of the pilot RCT that was halted due to the pandemic 27 and integrating key stakeholder feedback, we adopted a similar, multi-disciplinary team–based approach for implementing CGM in the hospital. As CGM technology is new to frontline staff, having dedicated experts on hand to place devices and, importantly, troubleshoot technology, is critical. One notable change from the pilot RCT is the use of the RMT to reduce personnel in the clinical setting while continuing to have 24/7 hypoglycemia monitoring. The RMT removes the burden of monitoring from the already-busy nursing staff and prevents further alarm fatigue yet alerts these nurses to ongoing or impending hypoglycemia that may warrant action. The advantage of the RMT being located off site is that this decreases the number of people needed to be in a clinical setting—a key advantage when this program began in May 2020 at the onset of the COVID-19 pandemic. This model has proven effective in monitoring and alerting providers to act, while mitigating exposure to the patient—who in almost 20% of cases was COVID+—unless there was a need to intervene.
Located in San Diego, Scripps Mercy hospital has a diverse patient population, as evidenced by the near 50-50 split of Hispanic and non-Hispanic individuals enrolled in the CGM as SOC program, and serves an economically disadvantaged population, with more than half of the patient population insured through government-based programs. Patients of lower socioeconomic status are known to have poorer health outcomes, especially among those with chronic diseases, including diabetes. 37 Indeed, we saw that the patients included in our program had poor glycemic control with high initial glucose values, and many patients had severe comorbidity burdens, placing them at high risk for adverse outcomes. Many patients had serious conditions including sepsis and COVID-19, and more than a third required a surgical procedure during their hospital stay. While only accounting for 5% of patients enrolled in CGM as SOC, patients with T1D descriptively appeared to have poorer glycemic control, despite being younger and having a lower comorbidity burden than those with T2D; however, both groups demonstrated modest improvements in glycemic control when examining the entire wear period relative to the first 24 hours of CGM. Similar to our pilot study, 27 most deviations from target range were hyperglycemia rather than hypoglycemia given the inclusion criteria, and that remote monitoring and alarm protocols are designed for the more critical, rapid treatment of hypoglycemia. Even so, extreme hyperglycemia (>250 mg/dL) was infrequent—less than 20% of values over the entire CGM period.
An important consideration for affecting glycemic control, and more broadly, hospital outcomes, is the actionable wear time for CGM. As most patients were at least three days into their hospitalization when they received CGM, the median wear time for CGM devices was 72 hours, and our protocol to validate CGM versus POCT at least 6 hours after device insertion (typically >24 hours into a hospital stay), the actionable window for intervention remains small even though once inserted, patients tend to remain on CGM through discharge. To maximize the potential effect of CGM on hospital outcomes, our team is working to increase this actionable window through earlier system placement and use for clinical management. Further evaluation of how actionable time on CGM affects effectiveness is warranted.
To effectively use CGM in the hospital setting requires confidence in the accuracy of the device, and we routinely assess this in relation to the gold standard—POCT. Bedside nurses still conduct reduced-frequency POCT (once daily following initial validation) while a patient wears a CGM device, and cross-validation using established methods such as the %20/20 agreement rate 29 can increase provider confidence. At a larger scale, post hoc assessments of concordance provide an overall assessment of device performance. We see that our MARD is within the expected range from previous studies,26,29 and discrepancies are largely without critical impact to treatment decisions as indicated by the low frequency of zones D/E on the CEG. 33 The CEG revealed trends toward CGM devices modestly overestimating the POC-obtained glucose values, which may explain why mean POC glucose levels across the hospitalization appear to be lower compared with the CGM device–measured means. Maintaining routine, interim evaluation of CGM performance against the previous SOC and communicating those findings to the clinical and on-site teams is imperative for ensuring confidence in the devices among those using it daily in practice. Having a specialized team of CGM experts can provide education and support to overcome hurdles of using new technology, including when questions of concordance or accuracy arise.
Communication across multi-disciplinary teams is critical, particularly to educate and support providers who are less familiar with CGM. Having a specialized inpatient diabetes care team enables routine education and specialized consultation, and the technical staff on-site can assist with any troubleshooting needs that may arise. However, with a large multi-disciplinary team and the infrastructure needed to support them, the cost of employing a new SOC is an important consideration. Despite the added costs to support the personnel, devices, and technologies required to employ CGM in the hospital setting, these could be ultimately offset by improved patient outcomes from better glycemic control reducing LOS, hospital costs, and reduced nurse time required for routine POCT. A cost-effectiveness evaluation of CGM in the hospital setting is warranted and will be conducted as part of the ongoing RCT being conducted by this team.
Furthermore, the multiple technologies and data platforms used are complex, and perhaps the CGM as SOC program could be employed with fewer staff and lower costs if technologies were integrated to a streamlined system and the EMR. Beyond the challenges of cost, there remain practical barriers in working with relatively new technology in a novel setting. Many nurses and clinical providers are unfamiliar with CGM and were slow to uptake the new technology into daily use. With a specialized team of experts providing regular communication on the enhanced safety through the 24/7 RMT hypoglycemia monitoring, reduced bedside nurse burden and patient satisfaction of less POCT, and ongoing education on the implementation procedures, the health system’s confidence of using CGM has continued to grow. However, there are still hurdles including many procedures with insufficient data on the safety or accuracy of CGM, requiring the device to be removed. This ultimately requires reverting to POCT until the device can be reinserted, and critical data may be missed from both a clinical care and effectiveness evaluation standpoint. Finally, in the current CGM as SOC program, patients are blinded to the CGM data being collected in real time, and the impact of patient’s viewing their own CGM data in real time warrants further study, and likely would necessitate further patient education.
Conclusions
Following our pilot RCT suggesting a benefit of CGM in the inpatient setting, and expedited by the FDA’s non-objection to the use of CGM in the hospital amid the COVID-19 pandemic, we debuted a real-world implementation of CGM as SOC in one hospital within our health system. Given our previous (and ongoing, NCT05307237) effectiveness evaluations, the protocols and infrastructure for implementation were not novel for our diabetes care team, which likely contributes to the high enrollment and overall success of the rapidly adapted program; however, these real-world adaptations were not without challenges and lessons learned.
The team-based approach that combines bedside and remote patient monitoring in our CGM as SOC model relies on the cloud-based digital technologies described in the methods above. This multi-faceted approach, including the use of a remote data team to identify and prioritize eligible patients and the RMT for 24/7 hypoglycemia monitoring of the Digital Dashboard and its alarms, complements the on-site team running the technical implementation and the APN team optimizing the patients care. Altogether this multi-disciplinary team provides added safety and reassurance for our patients and providers using CGM in a real-world SOC setting.
Footnotes
Acknowledgements
We would like to thank the Scripps Health physicians, nursing, and executive leaders who provided input on and endorsed this program. We would like to express our appreciation for the bedside nurses, physicians, and on-site and remote CGM teams who have contributed to the implementation and success of the CGM as SOC program. Finally, we would like to thank Scripps Health and the David Winkler Endowment for the financial support and Dexcom for providing devices for the CGM as SOC program.
Abbreviations
APN, advanced practice nurse; CEG, Clarke Error Grid; CGM, continuous glucose monitoring; CGM as SOC, continuous glucose monitoring as standard of care; EMR, electronic medical record; FDA, US Food and Drug Administration; HbA1c, glycated hemoglobin A1c; ICU, intensive care unit; IQR, interquartile range; IRB, institutional review board; IV, intravenous; MARD, mean absolute relative difference; POCT, point-of-care testing; RCT, randomized-controlled trial; REDCap, Research Electronic Data Capture; RMT, remote monitoring team; T1D, type 1 diabetes; T2D, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Scripps Health.
