Abstract
Background:
Up to one-third of people with gestational diabetes (GDM) will have persistent dysglycemia, and more than half do not complete the recommended postpartum oral glucose tolerance test (OGTT). This study assessed the use of blinded postpartum continuous glucose monitoring (CGM) to detect dysglycemia by assessing return rates, participant experience, and power to predict OGTT results.
Method:
Blinded CGM was placed on postpartum day 1 to 3 before discharge from the hospital and again at six weeks after pregnancy complicated by GDM and worn at home for up to 10 days. Participants mailed the CGM back and were encouraged to undergo standard of care six-week OGTT.
Results:
Fifty women (36 ± 6 years old; 40% non-Hispanic white, 24% non-Hispanic black, 22% Asian, 14% Hispanic; 34% Medicaid insured) were consented. First CGM was completed by 86%, second CGM was completed by 60%, and postpartum OGTT was performed by 68%. Mean first sensor glucose was 121.8 ± 14.1 mg/dL. Dysglycemia on OGTT was diagnosed in seven participants: six with impaired glucose tolerance (18%) and one with diabetes (3%). Percent time <96% in the range 70 to 180 mg/dL predicted abnormal OGTT with positive predictive value of 54% and negative predictive value of 100%. The sensitivity and specificity of CGM to predict postpartum dysglycemia were 100% and 78%, respectively. If given a choice, 94% of participants would prefer CGM over OGTT.
Conclusions:
Postpartum CGM is a reasonable and convenient initial postpartum screen for postpartum dysglycemia with high completion rates, sensitivity, and acceptability ratings. Percent time in range 70 to 180 mg/dL had strong predictive power for OGTT.
Keywords
Introduction
Diabetes mellitus is one of the leading causes of death by disease in the United States with high rates of morbidity and significant health care costs. 1 Gestational diabetes mellitus (GDM) is a significant risk factor for future diabetes and is one of the most common pregnancy complications. It affects approximately 7% of pregnancies with rising prevalence, and although for many people the dysglycemia resolves with delivery, up to one-third of patients will have persistent dysglycemia postpartum. 2 As high as 60% of those with GDM will develop type 2 diabetes later in life. 2 GDM disproportionately affects historically underserved populations with higher rates reported in Hispanic, black, Native American, and Asian or Pacific Islander populations. 3 Similarly, the development of type 2 diabetes mellitus after GDM also follows the same racial and ethnic inequities. 4
Postpartum screening for dysglycemia offers an early time point for intervention to prevent future type 2 diabetes and its associated morbidities. In order to diagnose persistent postpartum dysglycemia (IFG: impaired fasting glucose, IGT: impaired glucose tolerance, and diabetes) and future risk of type 2 diabetes, the American Diabetes Association, American College of Obstetrics and Gynecology, and World Health Organization recommend that all patients with GDM undergo an oral glucose tolerance test (OGTT) at six to 12 weeks postpartum.2,5,6 Postpartum testing is important for future reproductive, metabolic, and cardiovascular health, but is burdensome. 7 Unfortunately, this screening recommendation is achieved in less than half of patients diagnosed with GDM. 8 Previous research has tried to assess risk factors for low completion rates as well as utilize methods of promotion and incentivization of postpartum OGTT but has only achieved screening rates of up to 60% even in the setting of research studies.9-13 Even when OGTT is performed, fasting values are often missed due to issues with scheduling and ongoing breastfeeding.
Alternative testing strategies have failed to achieve the desired feasibility and predictive value needed. Hemoglobin A1C (HbA1c) is affected by peripartum red blood cell turnover. Studies have explored patterns of insulin resistance after delivery and use of early screening, prior to hospital discharge, as a strategy to improve screening rates.14,15 Early OGTT improves completion rates compared with six- to 12-week OGTT for the diagnosis of impaired glucose tolerance, and in a meta-analysis has had an overall sensitivity of 74% (ranging from 62% to 100% in individual studies), specificity of 56% (range 41%-70%), positive predictive value (PPV) of 29% to 42.6%, overall positive likelihood ratio (PLR) of 1.7, negative predictive value (NPV) of 75.3% to 100%, and overall negative likelihood ratio (NLR) of 0.4.16-20
Professional versions of continuous glucose monitors (CGMs) can be placed by health care professionals, worn at home with no data or alarms, and mailed back for download and review. CGM has been reported to be effective in the detection of dysglycemia in other populations at risk for diabetes and may detect more dysglycemia than HbA1c or OGTT.21-23 Earlier and less burdensome testing has the potential to increase diabetes and prediabetes detection. This may allow focused early intervention in order to decrease morbidity in future pregnancies and prevent future diabetes. Given its user-friendly nature and potential to be used without an additional visit, we aimed to assess the use of blinded CGM for the detection of postpartum dysglycemia and compared its results to standard of care OGTT in people with recent GDM.
Materials and Methods
This prospective, single-site observational study assessed the use of CGM for the detection of postpartum dysglycemia. This study was approved by the Institutional Review Board and conducted following this institution’s Program for the Protection of Human Subjects and the principles outlined in the Declaration of Helsinki. 24 The study was registered on clinialtrials.org (NCT05714761). Written informed consent for study participation including permission to view medical records of participant and infants was obtained from all participants. Recruitment was conducted from February 2023 to January 2024 at a single urban medical center.
Birthing people with a diagnosis of GDM in their current or recent pregnancy and aged 18 years or older were enrolled during their third trimester or within three days of delivery. Exclusion criteria included known pregestational diabetes or planned chronic postpartum glucocorticoid use. Diagnosis of gestational diabetes could be by any method. After informed consent, participant demographics and medical history were obtained. On postpartum day 1 to 3, a blinded Dexcom Professional CGM was placed, and participants were trained on its care and removal. Participants wore the sensor for up to ten days and returned the device by mail or in person. A second postpartum sensor was given to participants to take home, and they were instructed to place the sensor two to three days prior to their scheduled OGTT. Participants were instructed to undergo their OGTT as part of their usual care. However, if they were unable to complete their OGTT through routine care they were additionally offered testing as part of study procedures. Skin assessment was assessed according to the Draize’s scale. 25 Participants completed 5-point Likert scales for user satisfaction after each sensor wear and OGTT. Participant compensation was provided and was structured to incentivize OGTT completion.
Sensor sessions with at least 72 hours of data were included in analysis. For the second sensor session, the day of OGTT is not included in this analysis. Demographic data for participants who completed each portion of the study are reported. Oral glucose tolerance test results were analyzed according to American Diabetes Association diagnostic definitions: IFG defined as fasting glucose 100 to 125 mg/dL, IGT defined as two-hour glucose 140 to 199 mg/dL, and diabetes defined as fasting glucose ≥126 mg/dL or two-hour glucose ≥200 mg/dL. 6 Descriptive statistics for CGM metrics are reported. Unpaired t tests or Wilcoxon tests depending on distribution of data were used to compare data between participants who had a normal OGTT to those who had an abnormal OGTT, and paired tests were used to compare the first and second sensors for those who completed both. A P-value of <.05 was considered significant. Receiver operating characteristic (ROC) curves were created for sensitivity and specificity of mean glucose, time in range (TIR: 70-180 mg/dL), and percent time above thresholds of 140, 180, and 200 mg/dL compared with OGTT results. Area under the curve (AUC) for each ROC curve was calculated. Antenatal steroids with betamethasone for fetal pulmonary maturity or dexamethasone for anesthesia-associated antiemetic prophylaxis during C-section was assessed. A secondary CGM analysis was performed after removal of any data within four half-lives of steroid administration.
Study data were collected and managed using REDCap (Research Electronic Data Capture), a secure, web-based software platform designed to support data capture for research studies.26,27 R Studio version 2023.09.1 was used in CGM data processing.28,29
Results
Participant Characteristics
Fifty participants were enrolled with a mean age of 36 ± 5.7 years and were 40% non-Hispanic white, 24% non-Hispanic black, 22% Asian, and 14% Hispanic. Risk factors for future development of diabetes included family history of diabetes in 60%, median prepregnancy BMI of 26.6 (interquartile range [IQR]: 23.8-30.5), and 78% multigravida. Fifty-four percent used oral agents and/or insulin for management of GDM. Dexamethasone was administered during C-section delivery in 20% of the cohort (Table 1). Participants who did not return the CGM were similar in all demographic and baseline data other than lower infant birth weight (3260 g [2750-3480] vs 2740 [2345-2885]) (Supplemental Table 1).
Maternal, Antenatal and Peripartum Characteristics.

Data are presented as n (percent) unless otherwise stated.
Abbreviations: SD: standard deviation; GED: General Educational Development certification; BMI: body mass index; IQR: interquartile range; GDM: gestational diabetes.
Data available for 46 participants.
OGTT Testing Completion and Results
Oral glucose tolerance test was completed in 68% of participants (n = 34) (Figure 1). Two participants had the OGTT coordinated by study personnel because it was not planned as part of standard of care (1 participant was leaving the country and another due to provider preference). Of the OGTTs completed, fasting glucose was included in 45%. Seven participants (21%) had abnormal OGTT results with six (18%) having impaired glucose tolerance and one (3%) having diabetes. Two participants had impaired fasting glucose as well as impaired glucose tolerance.

Consort diagram.
First Postpartum Sensor
First sensors were placed on postpartum days 1 to 3. First sensors were returned by 86% of participants (n = 43), all of which had at least 72 hours of data for analysis, and 88% reported sensor data included a time of fasting for at least six hours.
Data from the first sensor had a mean CGM glucose of 121.8 ± 14.1 mg/dL. Median percent time above 180 mg/dL was 1.9%. Overall, 21% had more than 5% time above 180 mg/dL. CGM data from participants diagnosed with persistent dysglycemia on OGTT had statistically significantly higher percent time above 180 mg/dL as well as significantly lower TIR (Table 2). Removal of data potentially affected by steroid administration did not significantly change CGM metrics. There were no significant differences found based on GDM treatment or lactation.
Postpartum Continuous Glucose Monitoring Metrics From First Postpartum Sensor.
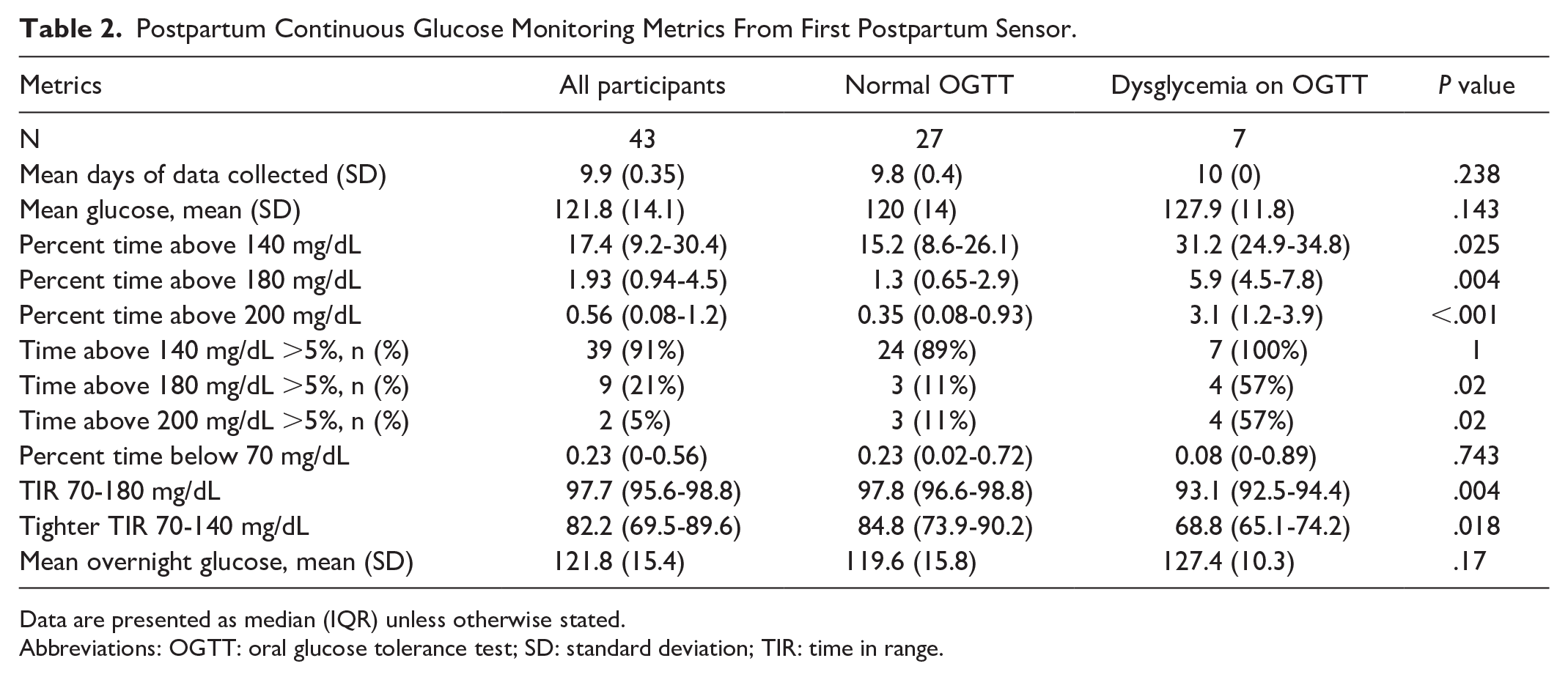
Data are presented as median (IQR) unless otherwise stated.
Abbreviations: OGTT: oral glucose tolerance test; SD: standard deviation; TIR: time in range.
Second Six-Week Sensor
A second sensor was placed at 6 ± 1.5 weeks postpartum. Sixty percent of participants wore and returned the second sensor with enough data for analysis (n=30). Percent time above 180 mg/dL was higher and TIR lower in the group with dysglycemia on OGTT (Supplemental Table 2). For those who completed both sensors, the second sensor data had similar CGM metrics to the first postpartum data other than lower time <70 mg/dL (median 0.27 [0.009-0.96] vs 0.02 [0-0.27], P = .019) and higher mean overnight glucose (119 ± 14 mg/dL vs 125 ± 14 mg/dL, P = .012) (Supplemental Table 3).
Predictive Power of CGM to Predict OGTT
Using OGTT results as the standard, the AUC for ROC curves for TIR, time above 180 mg/dL, and time above 200 mg/dL were higher than the AUC for mean glucose and percent time above 140 mg/dL (Supplemental Table 4). AUC for TIR was 0.89 (CI: 0.77-1) for the first sensor and 0.89 (CI: 0.78-1) for the second sensor (Figure 2). Removal of steroid-affected data had minimal impact on AUC (Supplemental Table 4). AUC for time above 200 mg/dL was also high, however the sensitivity declined over differences in percent time of less than a percentage point which is likely within the error for measurement so was not considered clinically useful (Supplemental Table 4).
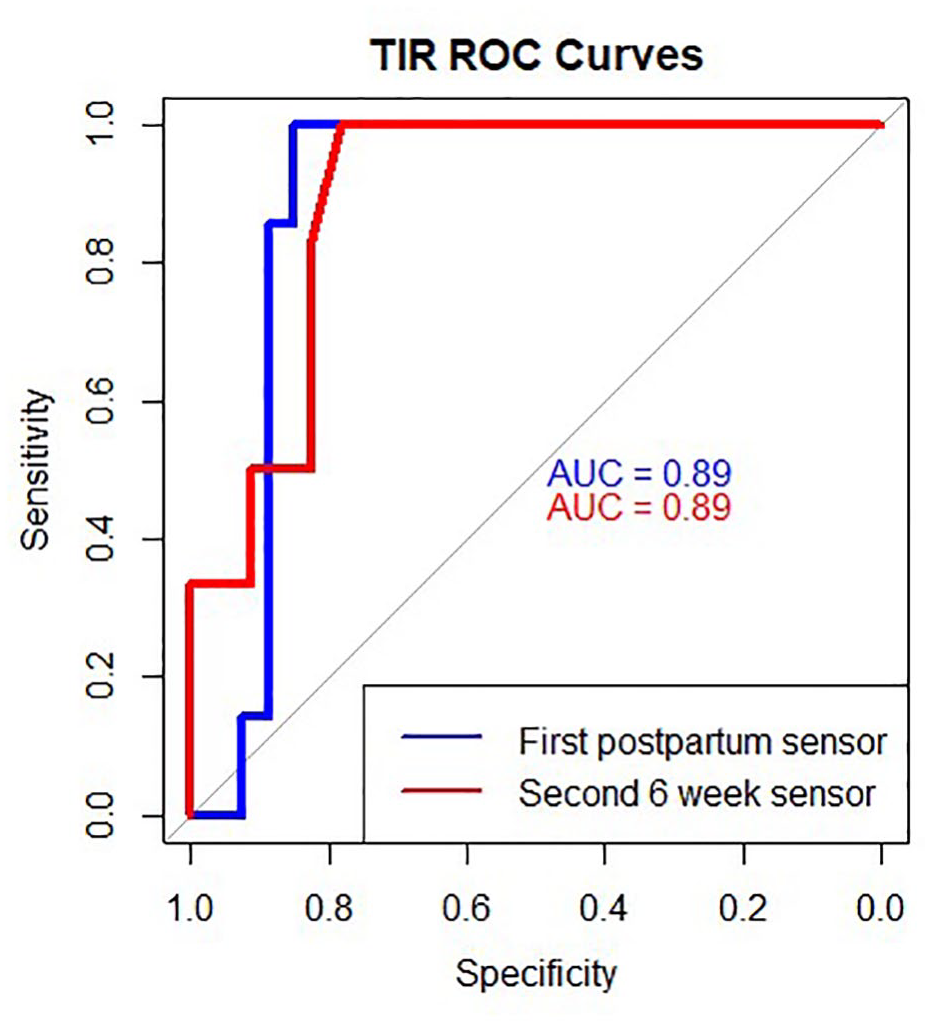
ROC curves for first and second sensors for time in range (TIR) 70 to 180 mg/dL.
Less than 96% TIR on the first postpartum sensor had 100% sensitivity, 78% specificity, PLR of 4.5 (PPV 54%), and NLR of 0 (NPV 100%). On the second sensor, TIR less than 96% had 67% sensitivity, 83% specificity, PLR of 3.83 (PPV 50%), and NLR of 0.4 (NPV 90%).
User Experience
More than very slight skin irritation occurred with 2% of the first sensors and 6% of the second sensors. Seven percent agreed or strongly agreed that the sensor was uncomfortable or painful and that wearing the first sensor was a burden, but 86% agreed or strongly agreed that wearing a sensor is worthwhile if it told them more about their health. When asked if OGTT was a burden, 52% agreed or strongly agreed, but 88% said undergoing OGTT is worth it if it told them more about their health. If given the choice, more participants said they would opt for CGM placed at discharge (55%) over CGM placed at postpartum follow-up (39%) or OGTT (6%).
Discussion
Placement of CGM at hospital discharge had high rates of device return, high rates of adequate data for analysis, high sensitivity and negative predictive value, and favorable user satisfaction. The second sensor return rate was much lower and similar to OGTT completion rates. Early CGM has a bigger potential for closing the postpartum screening care gap.
Data on postpartum glycemic metrics is limited. A study of CGM in people without diabetes found very low time above 180 mg/dL (median 0% [0%-0.2%]). 30 Even in the participants with normal OGTT, we found much higher rates of time in hyperglycemia. Similar to our study findings, CGM data from postpartum day 1 has also been reported to have higher rates of percent time above 180 mg/dL (1.0% ± 2.6% in people without GDM and 1.4% ± 3.2% in people with GDM). 31 There is a common misconception that metabolic changes from GDM are confined to pregnancy and resolve postpartum. Our study supports the need for more research on glycemic changes into the postpartum period.
An ideal test for postpartum dysglycemia screening should have a high sensitivity and NPV as well as a low burden. We found that TIR of less than 96% on the first postpartum sensor has a strong sensitivity of 100% and good specificity of 78% for predicting dysglycemia on postpartum OGTT. The NLR of 0 indicates a low likelihood of having dysglycemia on OGTT if the TIR is not less than 96%. This test performance is better than that reported for early OGTT, postpartum HbA1C, or six-week fasting serum glucose.18,32,33 The second six-week sensor had a slightly higher AUC, but the completion rate of the first sensor was higher so may be the more useful time for placement. Our findings of increased completion rates with earlier intervention mirrors findings of early OGTT performance.16-20 Timing of intervention is likely the largest determinant of completion due to the burden associated with return visits during the postpartum period.
Since performance metrics were not changed by removal of data that could be affected by steroid administration, it can be possible to use CGM in all people with GDM regardless of recent steroid exposure, which is routine at some institutions with spinal anesthesia. Further studies will be needed to explore other potential confounders of CGM metrics in the postpartum period such as preeclampsia or rate of weight loss.
Higher rates of fasting data captured by CGM use compared with OGTT may help to close a gap in our current method of routine postpartum screening with OGTT; however, rates of impaired fasting glucose were too low in our cohort for a full analysis.
The population recruited for this study was diverse and representative of the population at highest risk of persistent dysglycemia following GDM in terms of racial and ethnic background, insurance status, and medical risk factors for diabetes. Retention in the CGM portion of the study was high with only 14% withdrawal or loss to follow-up. The demographics and risk factors of participants who did not complete the sensor session were similar to those who returned the sensor. It would be important to avoid introducing a screening strategy that could contribute to health disparities instead of helping close them.
A convenience sample of 50 participants was used for this study. There was likely bias in who chose to participate. Even with consistent follow-up strategies entailing reminder texting and calling and participant stipend, only 68% of participants completed a routine postpartum OGTT and many attended their visit without fasting. However, this rate of completion is similar to what is reported in regular care and other research studies, indicating our outcomes may be generalizable to the greater population.3,8,11-13 A larger study with more fasting data will be needed to confirm the sensitivity and specificity analyses and generalizability of our findings. Subsequent research should include a large, multicenter study to establish optimal glucose thresholds and timing and include comparison to the true gold standard of future diabetes diagnosis. Whether postpartum CGM can be used as a diagnosis or a trigger to have an OGTT performed needs to be more thoroughly evaluated.
Participants preferred CGM to OGTT, however CGM can cause skin reactions and sensors can fail or fall off early. Potential inaccuracies in CGM need to be further considered since in this study we did not confirm CGM values with any fingerstick or reference value. Participants rated CGM and OGTT similarly in terms of whether the burden was worth the information the test gave in regard to health. However only those who performed the testing answered the follow-up questions so there is bias in health perspectives. Although CGM technology can have a large upfront cost, given the high cost of undiagnosed diabetes, use of CGM may be cost effective. OGTT cost includes supplies and staffing for a two-hour long test. A 2017 analysis of the economic burden of diabetes reported the annual cost per case for undiagnosed diabetes was $2950 for people less than 45 years of age, so it is possible that improvement in diagnosis can lead to overall cost savings. 34 Future studies are needed to evaluate the extent of cost savings and include cost of sensors and shipping, coordination of care for both testing strategies, and childcare needs for OGTT.
Conclusion
Our current strategy for postpartum testing is not serving our providers or patients well. Failure to obtain follow-up diabetes screening after childbirth may have significant implications for future pregnancies and eventual diabetes diagnosis. Postpartum CGM has high completion rates, sensitivity, and acceptability ratings and may help close the screening gap for postpartum dysglycemia screening and prevent future adverse health outcomes.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251351819 – Supplemental material for Postpartum Continuous Glucose Monitoring to Detect Dysglycemia After Gestational Diabetes
Supplemental material, sj-docx-1-dst-10.1177_19322968251351819 for Postpartum Continuous Glucose Monitoring to Detect Dysglycemia After Gestational Diabetes by Camila Cabrera, Selassie Ogyaadu, Camilla Levister, Leah Kaplan, Aslihan Ipek, Lauren Ferrara, Carol J. Levy and Grenye O’Malley in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors acknowledge Julia Ordway for her assistance in data analysis.
Abbreviations
AUC, area under the curve, CGM, continuous glucose monitors, GDM, gestational diabetes mellitus, HbA1c, hemoglobin A1C, IFG, impaired fasting glucose, IGT, impaired glucose tolerance, IQR, interquartile range, NLR, negative likelihood ratio, NPV, negative predictive value, OGTT, oral glucose tolerance test, PLR, positive likelihood ratio, PPV, positive predictive value, ROC, receiver operating characteristic, TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CL and GOM received research funding paid to their institution from DexCom, Abbott, Tandem, Insulet, Novonordisk, and Mannkind. CJL received research funding paid to her institution from DexCom, Abbott, Tandem, Insulet, Novonordisk, and Mannkind and served as a consultant for DexCom. The remaining authors report no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research support and product were supplied by DexCom, Inc for this investigator-initiated study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
