Abstract
Background:
The CE-marked CareSens Air continuous glucose monitoring (CGM) system (CSAir) features a 15-day sensor lifetime, a 2-hour warm-up period and mandatory manual calibrations. During subsequent product development, the algorithm was updated to reduce the warm-up period to 30 minutes and make user-entered calibrations optional. This study compared the CSAir’s performance between the manual and updated algorithms.
Methods:
Thirty adults with diabetes wore three CSAir sensors on their upper arms for 15 days. The study included three in-clinic sessions with capillary comparator measurements at 15-minute intervals over seven hours and glucose manipulation in the hypo- or hyperglycemic range. Point accuracy was assessed via mean absolute relative difference (MARD), 20/20 agreement rates (AR) stratified by BG range, and sensor wear time. Further evaluations included clinical point accuracy, alert reliability, technical reliability, safety and user satisfaction.
Results:
The CSAir’s updated algorithm exhibited improved accuracy compared with the manual calibration algorithm, with a total 20/20 AR of 93.9% (vs 90.1%) and an MARD of 8.7% (vs 9.9%). Accuracy remained stable across measurement ranges and sensor lifetime. Diabetes Technology Society Error Grid analysis revealed high clinical accuracy, with 88.0% and 92.4% of data pairs in zone A for the manual and updated algorithms, respectively. The estimated survival probability was 88.8%. Participants reported positive user satisfaction. No safety concerns were identified.
Conclusions:
Both algorithms of CSAir demonstrated robust performance and reliability with improved accuracy with the updated version. The study results of the CSAir suggest its suitability for nonadjunctive use.
Keywords
Introduction
Continuous glucose monitoring (CGM) systems have become essential tools in clinical diabetes management, enabling people with diabetes to improve and maintain glycemic control. 1 These devices provide real-time glucose measurements and trend information which supports therapeutic decision-making without confirmatory blood glucose testing when approved for nonadjunctive use. This facilitates timely interventions and enhanced diabetes self-management. 2 The integration of CGM-derived metrics, such as “Time in Range,” into national and international diabetes guidelines underscores their growing importance in standard diabetes care.3-5 However, the effectiveness of these systems relies heavily on their performance and safety, making thorough evaluation of new CGM technologies and their updates crucial for ensuring optimal patient care. 6
The CareSens Air CGM system (CSAir, i-SENS Inc., Seoul, Republic of Korea) (Figure 1) represents one of the recent advances in CGM technology. The original version of the device, first approved by the South Korean Ministry of Food and Drug Safety, 7 and more recently CE-marked (February 2024) for adjunctive use, features a 15-day sensor lifetime, 2-hour warm-up period, and manual calibrations. Since then, CSAir has undergone further technological refinement and now features an updated algorithm with optional calibrations (meaning that user-entered calibration values are no longer required but optional) and a reduced warm-up period of 30 minutes, potentially offering greater flexibility and comfort in real-world use.

CareSens Air CGM system with insertion device, transmitter and integrated sensor and mobile phone as receiver.
This article presents results from a postmarket clinical follow-up (PMCF) study with the objective to validate the performance of the approved version of CSAir requiring manual calibrations. In addition, an analysis of the CGM readings from the updated algorithm, that were retrospectively obtained from the raw sensor data collected during the study, was carried out. The primary aim was to compare the performance between the manual calibration and updated algorithms, based on the data collected in the same PMCF study.
Methods
Study Design
This study was conducted in July and August 2024 as a prospective, mono-center, single-arm, open-label, interventional evaluation trial in adult participants with type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM) treated with intensive insulin therapy by the Institute for Diabetes Technology Ulm, Germany. There was a preference to enroll people with T1DM, because glucose excursions could be induced more easily than in people with T2DM. The main exclusion criteria were hypoglycemia unawareness, severe hypoglycemia in the six months before enrollment, HbA1c >10% and intake of substances or planned medical procedures known to affect performance of the examined CGM system.
Ethical approval was granted by the responsible ethics committee, the study was notified to the competent authority, and it was registered with the German Clinical Trials Register (DRKS00034614).
Study Devices
The study was carried out with the approved version of the CSAir CGM system, which was used by the participants according to its instructions for use. The system consisted of a sensor/inserter unit and a software application running on an Android-based smartphone provided by the manufacturer. The CSAir CGM system has a known interference to ascorbic acid (vitamin C) at high doses associated with intravenous injections. The sensors originated from two manufacturing lots. CGM readings from the updated calibration algorithm were generated retrospectively from the collected raw data. The updated calibration algorithm was developed and validated independently from the PMCF study through i-SENS’ clinical and real-world data. While the updated algorithm supports optional calibrations, this analysis evaluated performance using retrospectively processed raw sensor data assuming no calibrations were performed. Capillary comparator and calibration measurements were performed using the Contour Next blood glucose monitoring system (Ascensia Diabetes Care Holdings AG, Basel, Switzerland). Sporadic BG level measurements with the Cobas Integra 400 Plus laboratory analyzer were carried out in parallel to Contour Next to determine its bias and verify its accuracy.
Study Procedures
In accordance with the 15-day lifetime of the CSAir sensors, the study duration was 16 calendar days, during which participants spent most of the time in a free-living setting. Each participant simultaneously wore three CSAir sensors on the back of their upper arms. Multiple sensors were applied to improve the reliability of safety and performance results while keeping the scale of this PMCF study limited. Participants arrived to the study site on the afternoon of day 1 and inserted the sensors themselves; sensors could be replaced in case of a sensor loss within the first 24 hours. Furthermore, sensors could be affixed with additional tape if necessary. The participants carried out manual calibrations in agreement with the user instructions (~2 hours after insertion, ~14 hours after insertion and then once daily) using capillary blood glucose (BG) level measurements. Over the course of the study, three in-clinic sessions were scheduled for each participant at the beginning (study days 2-5, including the first 24 hours after insertion), middle (study days 6-10) and end (study days 11-15) of the study period. During in-clinic sessions duplicate capillary comparator measurements (two test strips from the same finger-prick) were scheduled every 15 minutes for a total of seven hours, and no additional CGM system calibrations were carried out. Simultaneously, the BG levels of the participants were manipulated by controlling food intake and insulin administration, for which participants arrived at the study site fasting and automatic insulin delivery (AID) systems were switched to manual mode until the end of the procedure. Here, the goal was to maintain BG levels either <70 mg/dL or >300 mg/dL for approximately 60 minutes. After insertion (study day 1) and removal (study day 16), participants completed a questionnaire for user satisfaction standardized for CGM performance studies (QUSS-CGM). 8
Data Analysis
The CGM readings of the approved device version with manual calibrations were directly downloaded from the smartphone applications. The CGM readings from the updated calibration algorithm were generated retrospectively by the manufacturer before receiving the comparator data and provided to the IfDT for independent performance analysis. To generate these readings, the raw sensor data were inputted as a time series into the updated calibration algorithm in the same manner as sequentially recorded raw sensor data in order to reflect real-time performance. The duplicate comparator capillary BG measurements were excluded if differences between measurements exceeded ±10% or ±10 mg/dL or otherwise averaged. Continuous glucose monitoring data recorded outside the measuring range (40-500 mg/dL) was excluded from point accuracy and stability analysis. Both versions of the CSAir automatically store CGM readings every five minutes. Thus, in-clinic capillary comparator measurements were paired with CGM readings recorded simultaneously or within the following four minutes, and data from all sensors were pooled.
Analytical point accuracy was characterized through calculation of the mean absolute relative difference (MARD), relative bias (also known as mean relative difference) and 20/20 agreement rate (20/20 AR) between CGM and comparator values (percentage of CGM-comparator pairs with ±20 mg/dL or ±20% at comparator glucose concentrations <100 or ≥100 mg/dL, respectively). Statistical differences between the two calibration algorithms in MARD and 20/20 AR were assessed using a paired
The technical reliability was characterized through a Kaplan-Meier sensor survival analysis. Safety was assessed by documenting any device-related adverse events.
Results
Comparator Data
A total of 30 adult participants with T1DM or T2DM undergoing intensive insulin therapy were enrolled in the study. Baseline participant characteristics are shown in Table 1.
Study Population Characteristics (n = 30).
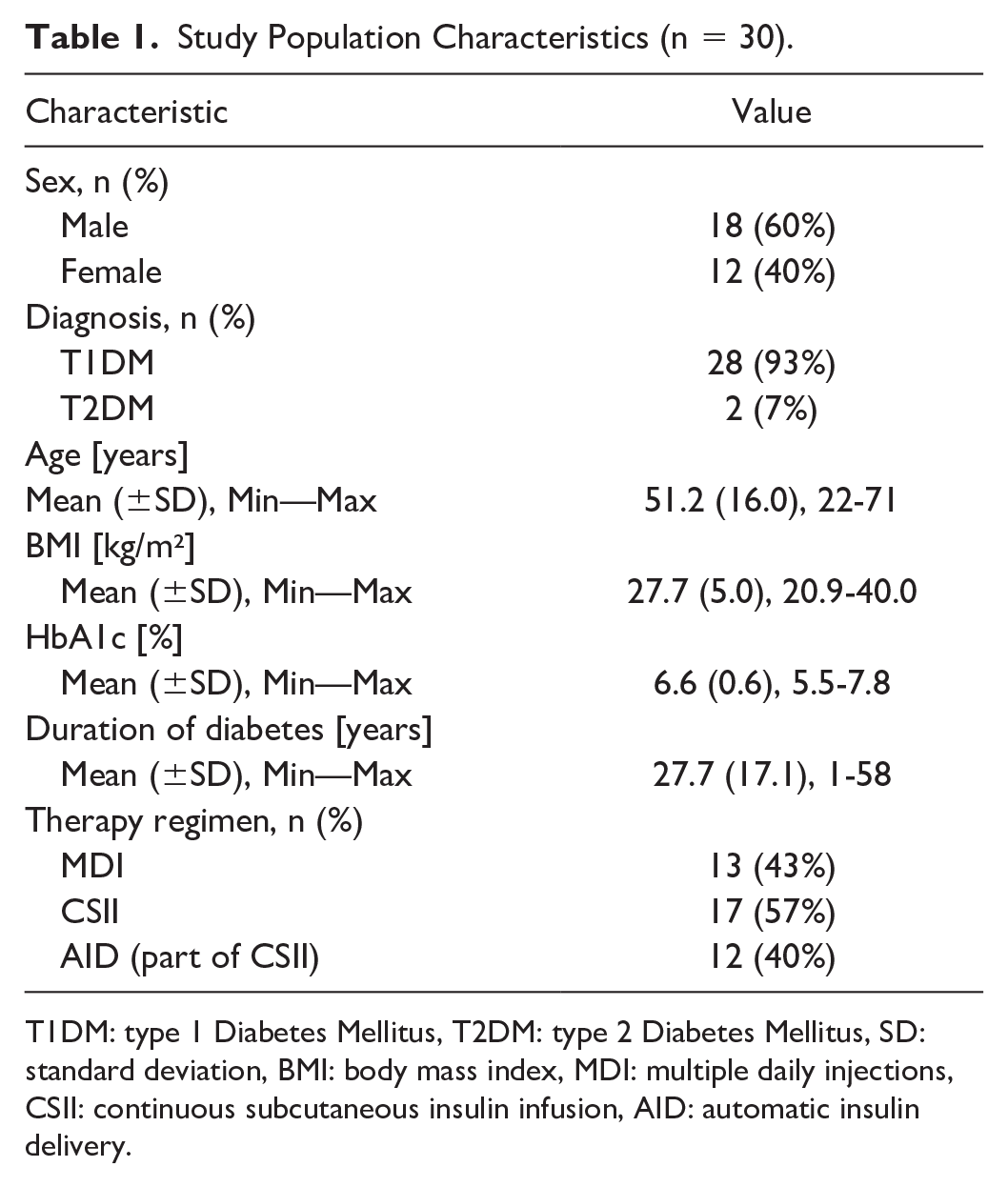
T1DM: type 1 Diabetes Mellitus, T2DM: type 2 Diabetes Mellitus, SD: standard deviation, BMI: body mass index, MDI: multiple daily injections, CSII: continuous subcutaneous insulin infusion, AID: automatic insulin delivery.
The relative bias (± standard deviation) of Contour Next to the laboratory analyzer was +0.3 ± 3.2% (n=148), thus showing sufficient accuracy for use as a comparator device. Of the 2610 scheduled comparator measurements, a total of 2599 valid capillary comparator measurements were included in the analysis. 7.4% of measurements were <70 mg/dL and 6.7% >300 mg/dL. From consecutive measurements with 10 to 20 minutes intervals, 2504 comparator BG-RoC pairs were derived with a mean absolute RoC (MARoC) of 0.85 mg/dL/min.
CGM Performance
Of the 94 sensors inserted, four were replaced within the first 24 hours after insertion. A total of 88 sensors worn by all 30 participants generated data that could be used in the performance analysis.
The analytical point accuracy for the manual calibration and the updated calibration algorithms, stratified by comparator glucose range, are shown in Table 2. A total of 7220 and 7247 CGM-comparator pairs were available from the two calibration algorithms, respectively. The difference in the number of pairs is caused by the fact that one algorithm might display a CGM reading outside the measuring range, while the other might not. These results indicate that the accuracy could be improved with the updated calibration algorithm, where both 20/20 AR and MARD are significantly better for the updated calibration algorithm (
Point Accuracy Metrics of Manual and Updated Calibration Algorithms for Different Comparator Measurement Ranges.
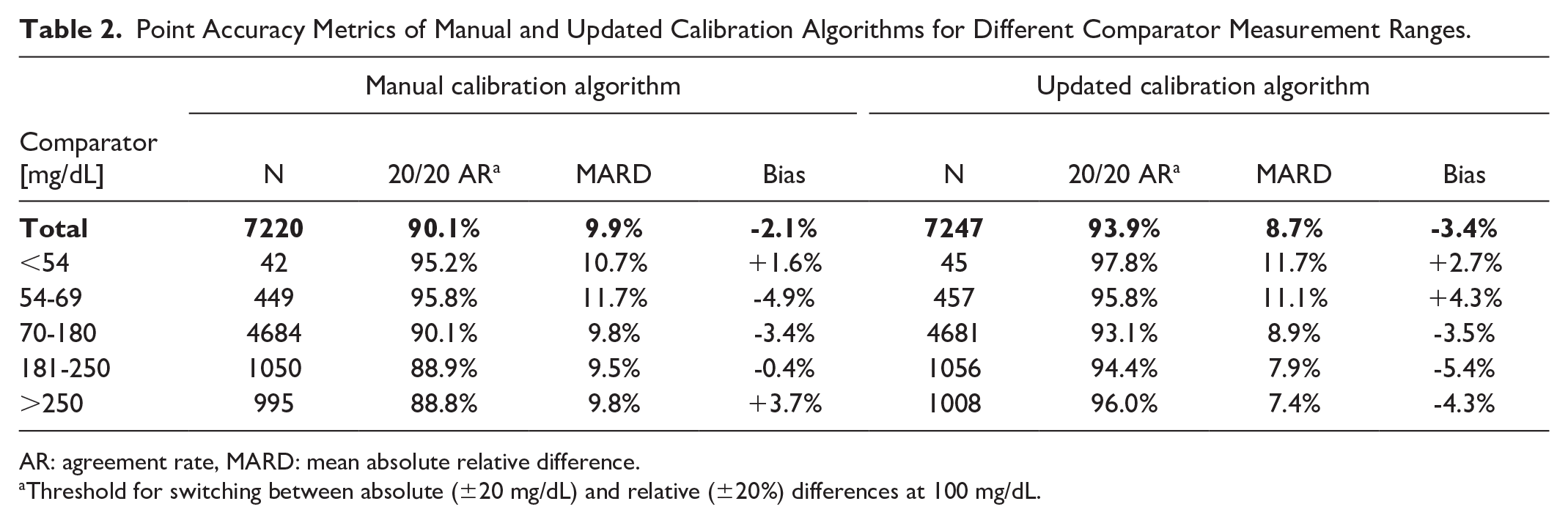
AR: agreement rate, MARD: mean absolute relative difference.
Threshold for switching between absolute (±20 mg/dL) and relative (±20%) differences at 100 mg/dL.
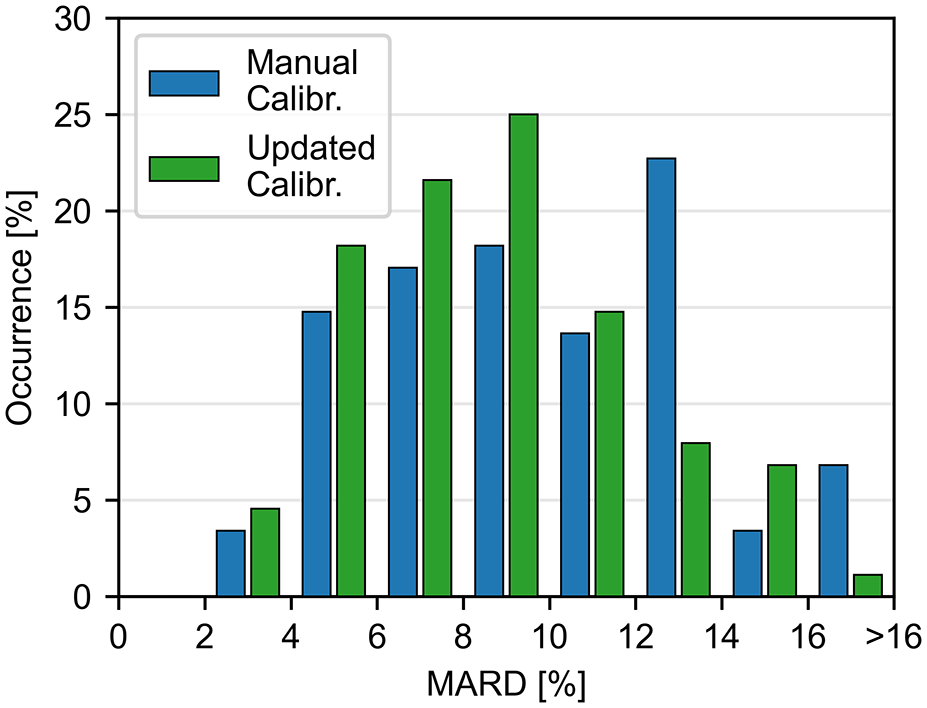
Distribution of sensor-specific mean absolute relative differences (MARD, n = 88).
Assessment of clinical accuracy using DTSEG analysis revealed that CGM-comparator pairs for both algorithms were predominantly distributed within clinically acceptable zones A and B, with only seven and nine pairs falling outside these zones for the manual and updated calibration algorithms, respectively (Figure 3). Comparative analysis of sensor stability across the wear period based on study days 2 to 5 (including the first 24 hours after insertion) demonstrated improved performance with the updated calibration versus the manual calibration algorithm (Table 3).
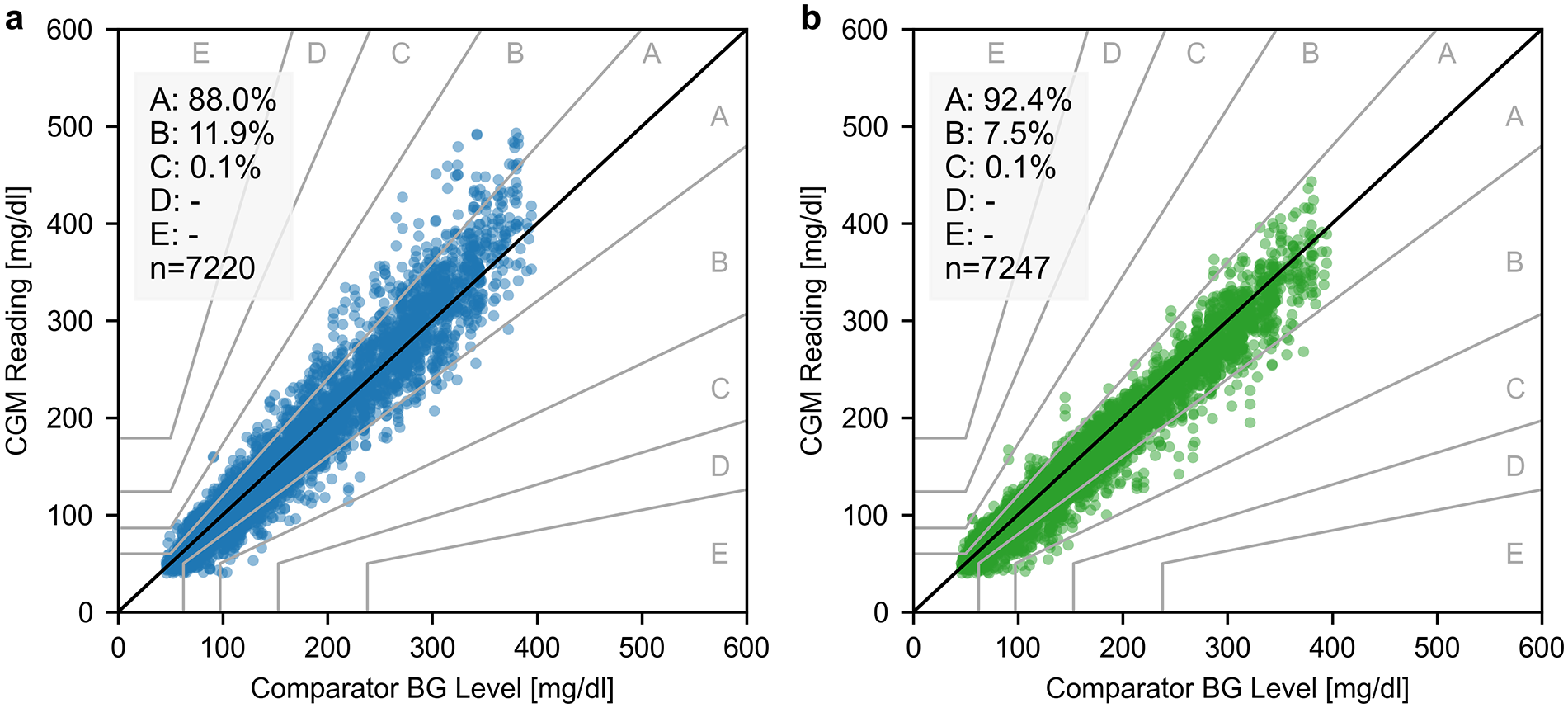
Diabetes Technology Society Error Grid analysis of the manual calibration algorithm (panel a) and updated calibration algorithm (panel b). CGM: continuous glucose monitoring, BG: blood glucose.
Stability in Terms of Point Accuracy Stratified by Sensor Lifetime Period.
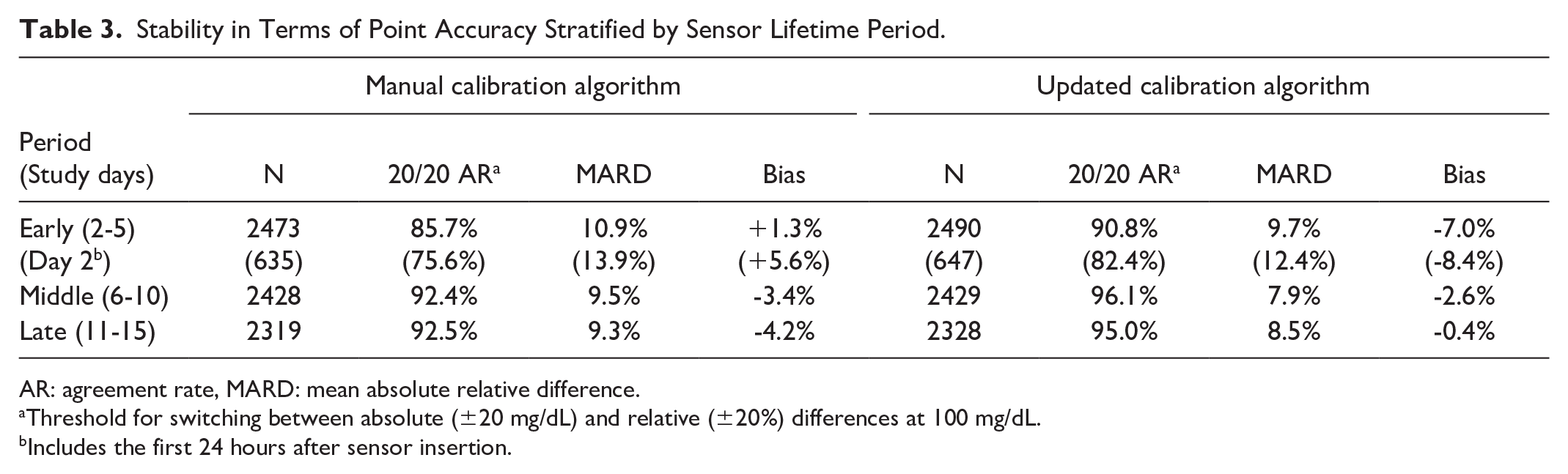
AR: agreement rate, MARD: mean absolute relative difference.
Threshold for switching between absolute (±20 mg/dL) and relative (±20%) differences at 100 mg/dL.
Includes the first 24 hours after sensor insertion.
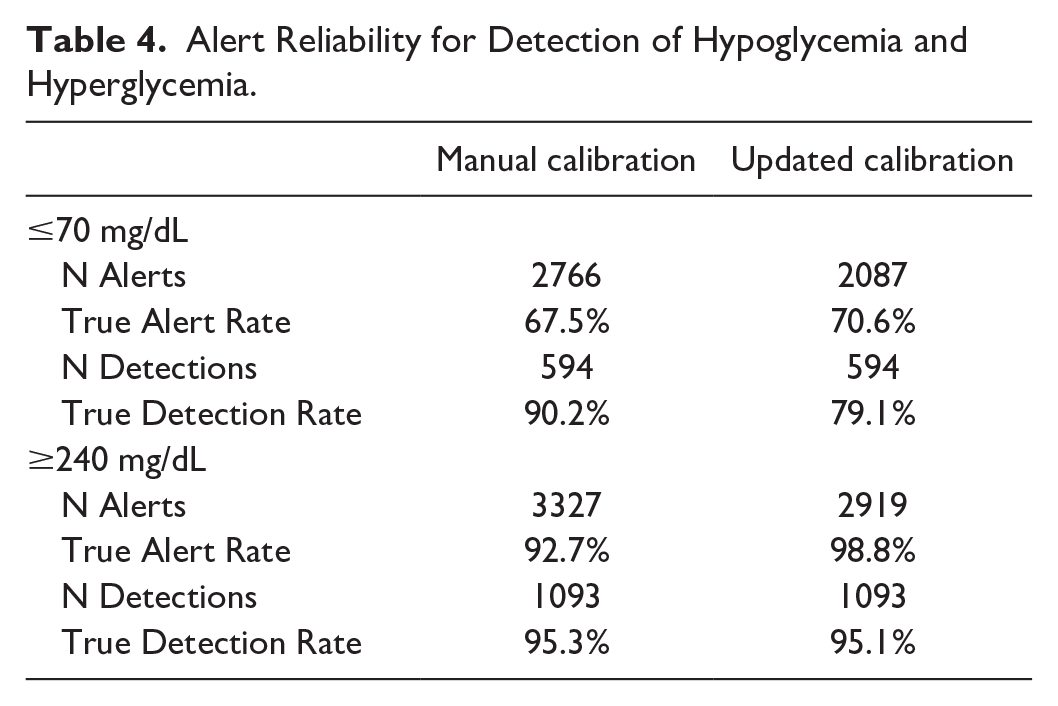
The analysis of alert reliability for hypoglycemia and hyperglycemia thresholds (≤70 mg/dL and ≥240 mg/dL, respectively) was conducted for both algorithms. The updated calibration algorithm showed an improved true alert rate while exhibiting a modest reduction in true detection rate compared with the manual calibration algorithm (Table 4).
Alert Reliability for Detection of Hypoglycemia and Hyperglycemia.
The estimated survival probability to the end of the lifetime for the 94 inserted sensors with both algorithms was 88.8% (95% confidence interval 80.2%-93.8%, Figure 4). In total, 16 sensors losses evenly distributed throughout the sensor lifetime occurred: six due to adhesive weakness, four due to a technical malfunction, and six due to a user error (censored in the survival analysis).
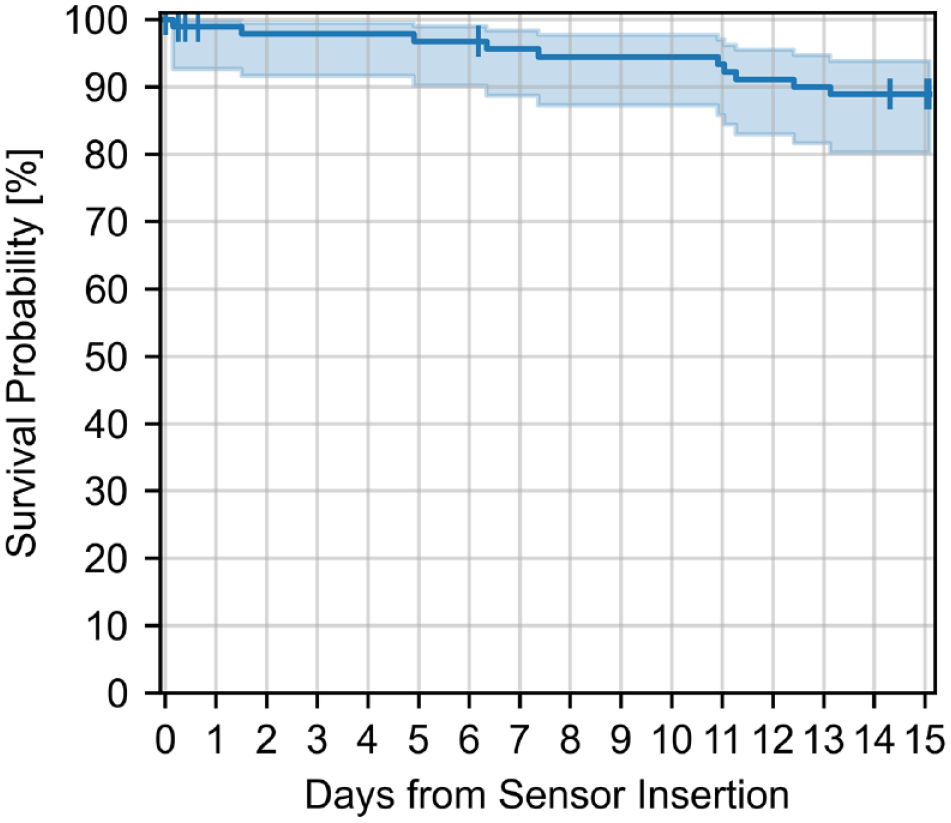
Kaplan-Meier survival plot of CSAir sensors (n = 94). The shaded area indicates the 95% confidence interval. The vertical dashes indicate censored sensor losses due to user errors.
An analysis of safety outcomes revealed that two out of 94 inserted sensors (2%) caused an adverse device effect (ADE). The observed ADEs comprised a mild skin pressure mark from the transmitter of the CSAir and a skin irritation in three places under the adhesive recorded after removal of the sensor. Both ADEs were classified as mild and are anticipated in the context of CGM system use.
User satisfaction aspects including the CSAir’s wearing comfort, sensor size, the reliability of the readings, the overall impression, as well as further recommendation of the product were positively rated by the majority of participants, a selection is shown in Figure 5.
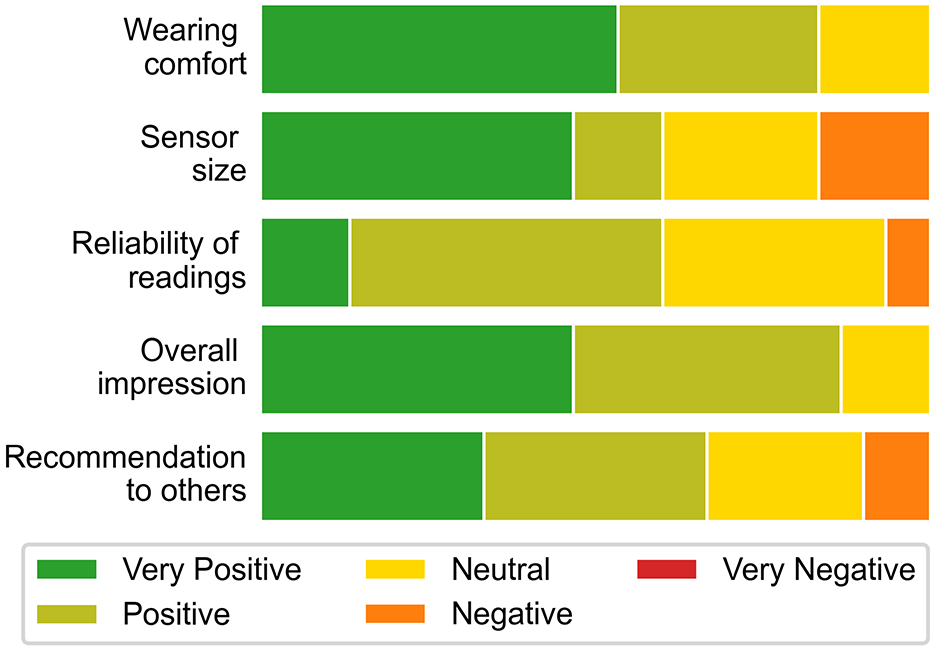
Summary results of selected items of the user satisfaction questionnaire.
Discussion
This study evaluated the performance of the CSAir using the original CE-approved manual calibration algorithm compared with an updated calibration algorithm using retrospectively processed raw sensor data obtained from the predecessor system. The manual calibration algorithm is characterized by a 15-day sensor lifetime, two-hour warm-up period and mandatory daily calibration. In contrast, the updated calibration algorithm featured a shortened 30-minute warm-up time and optional user-entered calibration. The CSAir exhibited enhanced performance metrics in terms of analytical point accuracy when utilizing the updated calibration algorithm, as evidenced by improved ARs and MARD in comparison to the manual calibration algorithm.
Overall, the analytical evaluation revealed robust accuracy metrics, achieving a MARD of 8.7% and a 20/20 AR of 93.9% with the updated calibration algorithm. Sensor accuracy remained stable throughout the measurement range, as well as over time for both algorithms, with the updated calibration algorithm presenting improved point accuracy results. In addition, the variability of sensor-specific MARDs was improved with the updated calibration algorithm, in particular the percentage of sensors with MARD >16%. Slightly improved results were also found compared with a previously published study with the CSAir with the manual calibration, where the 20/20 AR was 89.0% compared with venous measurements. 7 However, the choice of comparator sample origin (capillary vs venous) can affect observed CGM accuracy due to the physiological difference in glucose concentration between capillary and venous blood. 9
An in-silico evaluation revealed that CGM data utilization for insulin dosing decisions is feasible when the MARD remains below 10%. 10 Both the manual and updated calibration algorithms demonstrated performance consistent with this criterion, indicating a performance sufficiently robust for nonadjunctive CGM device use. The potential utility of the CSAir for therapy decisions is substantiated by its performance being comparable to established factory-calibrated CGM systems previously approved for nonadjunctive use (eg, Dexcom G7 [MARD of 8.2% and 20/20 agreement rate of 95.3%] 11 and FreeStyle Libre 3 [MARD of 7.8%]), 12 whose lack of compulsory finger pricking is associated with a reduced burden.
The robust clinical accuracy was confirmed by the results of the DTSEG analysis, where 88.0% and 92.4% of data pairs for the manual calibration and updated calibration algorithm, respectively, were found in zone A, indicating no clinically relevant measurement errors.
The reliability of hypo- and hyperglycemic threshold alerts is crucial for improving glycemic control and avoiding the burden of alert fatigue. 13 The ability of the CSAir to produce true hypo- and hyperglycemia alerts improved, while the percentage of correct hypo- and hyperglycemia detections moderately decreased with the updated calibration algorithm. The enhanced true alert rate observed with the updated calibration algorithm indicates improved reliability of glycemic excursion warnings, potentially reducing unnecessary interventions. Although this was offset by a modest decrease in true detection rate, the overall alert performance profile suggests a favorable balance for clinical applications, particularly in terms of reducing alarm fatigue while maintaining safety. This trade-off reflects a challenge in CGM design, where optimizing for specificity may slightly compromise sensitivity. In clinical practice, however, false positives are often more disruptive than occasional missed detections, especially where alarm fatigue can lead to alarm disregard or discontinuation of device use. Furthermore, 89% of CSAir sensors reaching the intended sensor wear time indicate that this system is a durable and reliable option.
There were no safety concerns during the study. The high user satisfaction scores reflected positive acceptance of the CGM system, demonstrating its reliability from both technical and operational perspectives. Given that qualitative feedback was optional in the questionnaire, comments were limited in number and scope; however, four participants reported initial concerns regarding reliability during the early sensor wear period, which likely accounts for the distribution of reliability ratings—60% positive or very positive and 40% neutral or negative. These concerns appeared to resolve with continued use.
Several limitations of this study warrant consideration. While the percentages of comparator blood glucose values in extreme ranges (>300 mg/dL and <70 mg/dL) approached the recommended threshold of 7.5%, 14 they did not exceed it. In addition, comparator glucose concentration RoCs influence the observed accuracy of a CGM system. 9 This study’s MARoC of 0.85 mg/dL/min reflected moderate glucose dynamics, which aligns with those reported in the majority of performance evaluations of other CGM systems. 9 Although the study cohort was relatively small (n=30), the use of three CGM sensors per participant provided sufficient data to validate the system’s safety and performance for market use.
Conclusions
This study evaluated the performance of the CE-marked CSAir, comparing its original manual calibration and updated calibration algorithms. Data collected from 30 participants revealed that the updated calibration algorithm achieved superior accuracy metrics, particularly in the 20/20 agreement rate. The demonstrated point accuracy and alert reliability indicate the system’s clinical safety profile for both algorithms, suggesting potential regulatory approval for nonadjunctive use. Notably, the system’s performance is similar to existing CGM systems. These findings validate the system’s readiness for clinical implementation and its potential to expand treatment options for diabetes management.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251351318 – Supplemental material for Comparative Performance Analysis of Manual and Updated Optional Calibration Algorithms for the CareSens Air CGM System
Supplemental material, sj-docx-1-dst-10.1177_19322968251351318 for Comparative Performance Analysis of Manual and Updated Optional Calibration Algorithms for the CareSens Air CGM System by Nina Jendrike, Manuel Eichenlaub, Manuela Link, Sükrü Öter, Anne Beltzer, Marta Gil Miró, Cornelia Haug, Jung Hee Seo, Moon Hwan Kim, Stefan Pleus and Guido Freckmann in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors would like to thank the volunteers who participated in this study and the staff at the Institute for Diabetes Technology.
Abbreviations
ADE, adverse device effect; AID, automatic insulin delivery; AR, agreement rate; BG, blood glucose; CE, conformité europeenne; CGM, continuous glucose monitoring; CSAir, CareSens Air CGM System; DTSEG, Diabetes Technology Society Error Grid; MARD, mean absolute relative difference; MARoC, mean absolute rate of change; MRD, mean relative difference; RoC, rate of change; T1DM, type 1 Diabetes Mellitus; T2DM, type 2 Diabetes Mellitus.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: N.J., M.E., M.L., S.Ö., A.B., M.G.M., C.H. and S.P. are employees of the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm (IfDT), which carries out clinical studies, for example, with medical devices for diabetes therapy on its own initiative and on behalf of various companies. G.F. is the general manager and medical director of IfDT. G.F. and/or its employees have received speakers’ honoraria or consulting fees in the last three years from Abbott, BOYDsense, Dexcom, Glucoset, i-SENS, Lilly Deutschland, Menarini, Novo Nordisk, Perfood, PharmaSens, Roche, Sinocare, Terumo, Vertex, and Ypsomed. J.H.S. and M.H.K. are employees of the i-SENS, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by i-SENS, Inc. (Seocho-gu, Seoul, Korea). The clinical investigation plan of this study was developed in conjunction with i-SENS and the Institut für Diabetes-Technologie Ulm.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
