Abstract
The algorithm for the Dexcom G6 CGM System was enhanced to retain accuracy while reducing the frequency and duration of sensor error. The new algorithm was evaluated by post-processing raw signals collected from G6 pivotal trials (NCT02880267) and by assessing the difference in data availability after a limited, real-world launch. Accuracy was comparable with the new algorithm—the overall %20/20 was 91.7% before and 91.8% after the algorithm modification; MARD was unchanged. The mean data gap due to sensor error nearly halved and total time spent in sensor error decreased by 59%. A limited field launch showed similar results, with a 43% decrease in total time spent in sensor error. Increased data availability may improve patient experience and CGM data integration into insulin delivery systems.
Introduction
Real-time continuous glucose monitoring (CGM) systems transmit glucose data at regular intervals from a body-worn sensor and transmitter to a nearby display—a receiver, smart device, and/or integrated insulin pump—providing actionable information about current glucose and the direction and rate of glucose change. Real-time access to CGM data allows users to take immediate action based on current glucose and its trajectory; retrospective review of CGM data permits behavior modification according to historic trends. CGM data are increasingly incorporated into automated insulin delivery (AID) systems where they are used to predict glucose levels and adjust insulin delivery rates accordingly. 1 Realizing glycemic benefits from standalone CGM or AID systems requires consistent and sustained access to CGM data.2-4
Recent developments in CGM technology have enabled factory-calibrated systems, such as the Dexcom G6 CGM System, to provide accurate readings without the need for blood glucose calibrations. 5 As a safeguard, the G6 algorithm can limit data transmission if glucose values are deemed to be aberrant, leaving users without glucose data temporarily; in some cases, the system may be shut off before its expected 10-day wear due to aberrations. A new algorithm was developed to increase G6 data availability by improving noise filtering and handling of data originally mislabeled as noise with the intention of better classifying clean data that followed periods of brief sensor noise and recover periods of true high rates of change. We assessed the impact of this new algorithm on system accuracy and data availability by reprocessing raw sensor data from the G6 pivotal studies. Additionally, we evaluated changes in real-world data availability among sensor sessions before and after a limited launch of transmitters including the new algorithm in Canada.
Methods
Methods used to assess the performance of the Dexcom G6 CGM System (G6, Dexcom, Inc. San Diego, CA) have been published previously.6,7 Data presented here are combined from both studies; data from participants ages 2 to 5 years of age are not presented as they included capillary glucose comparator values instead of venous YSI samples. The studies were approved by the FDA through the Investigational Device Exemption process and registered at ClinicalTrials.gov (NCT02880267).
Methods of Data Analysis
The algorithm was enhanced using pre-pivotal G6 feasibility data and optimized to achieve improved data availability while maintaining accuracy. To assess its performance, raw sensor data from the pivotal studies were re-processed using the new algorithm. Each YSI value was paired with the CGM value that immediately followed (within 5 minutes). CGM readings between 40 and 400 mg/dL and their associated YSI values were evaluated as matched pairs.
Accuracy analyses were performed using SAS® software, version 9.3 (SAS Institute, Inc., Cary, NC) and included the proportion of the CGM system values that were within ±20% of paired YSI values ≥70 mg/dL or ±20 mg/dL of YSI values <70 mg/dL (hereafter referred to as %20/20), overall and on the first day of sensor wear. The overall accuracy of the study data was calculated as the mean absolute relative difference (MARD) between the CGM and YSI values in each of the paired points.
Data availability metrics from the clinical studies included total time spent in sensor error as well as the frequency and duration of data gaps due to sensor error. Missing data due to sensor error were identified as 5-minute samples that did not generate a displayable glucose value, excluding warm-up (first 2 hours of each session). Data gaps in each session were identified as periods of continuous missing data. Total time spent in sensor error was computed as the number of missing data samples divided by the total number of samples in the session from the end of warm-up to session end. Data availability analyses were performed using Matlab® version R2019b (The Mathworks, Inc, Natick, MA).
Transmitters including the enhanced algorithm started to ship in Canada in April/May 2020. Differences in data availability after the limited launch were determined using deidentified and anonymized data from Canadian G6 users who had successful sessions using the transmitters with the enhanced algorithm. Total time spent in sensor error was computed as above. Field data analyses were performed using Python and SQL (within BigQuery).
Results
There were 380 participants that enrolled (meaning a sensor insertion was attempted) in the clinical studies. Detailed participant demographics have been described previously.6,7 The vast majority (99%) had type 1 diabetes and the remainder (1%) had type 2 diabetes. Of the 380 participants, 66 were excluded from the accuracy assessment—14 were ages 2 to 5 years and did not have YSI measurements collected and 42 were adults who did not have at least one CGM-YSI matched pair. A total of 325 subjects were included in the accuracy evaluation of the enhanced algorithm—159 adults and 166 pediatric participants—who together contributed 25,128 CGM-YSI matched pairs. The data availability evaluation included 374 sessions that produced at least one displayable glucose reading.
Integrated CGM (iCGM) performance 8 was preserved with the enhanced algorithm; negligible or no differences in performance were observed across multiple accuracy metrics (Table 1). Overall MARD remained unchanged between the original and enhanced algorithms and was 9.8% (overall), 9.9% (adults), and 9.6% (pediatrics). While there were few YSI values obtained within periods of sensor error, the %20/20, %30/30, and %40/40 for the 120 matched pairs obtained with the enhanced algorithm but not the original were 63.3%, 81.7%, and 90.8%, respectively. The %20/20, %30/30, and %40/40 for the 93 matched pairs obtained with the original algorithm but not the enhanced were 49.5%, 63.4%, and 80.6%, respectively. The retuned algorithm retained or improved hypo- and hyperglycemia alert detection and accuracy for both pediatric and adult participants (data not shown).
Accuracy of the G6 CGM System with the Original and Enhanced Algorithms.

There was 1 pediatric subject who had 1 clinic session on Day 10. The subject’s system failed under the original algorithm prior to Day 10, whereas it failed on Day 10 with the enhanced algorithm. Hence, this subject had no CGM-YSI matched pairs under the original algorithm but had at least 1 CGM-YSI matched pair under the enhanced algorithm.
Systems employing the enhanced algorithm resumed displaying data faster after brief sensor noise as depicted by a representative CGM tracing in Figure 1A. The mean ± SD data gap length decreased by 47% from 46 ± 42 to 24 ± 17 minutes with the enhanced algorithm and the proportion of all data gaps that were 60 minutes or longer decreased by 76% from 25% to 6% (Figure 1B). The distribution of data gaps shifted—with more frequent short data gaps (5-25 min), fewer long data gaps (30+ min), and no data gaps over 2 hours with the enhanced algorithm. Reliable data were displayed when possible, splitting long data gaps and eliminating some short data gaps. The frequency of data gaps was low with both algorithms, with most data gaps of any duration occurring less than once per session. Percent time in sensor error, during which CGM data are not shown, also decreased by 59% with the enhanced algorithm—from 1.18% to 0.49% of the total wear time.
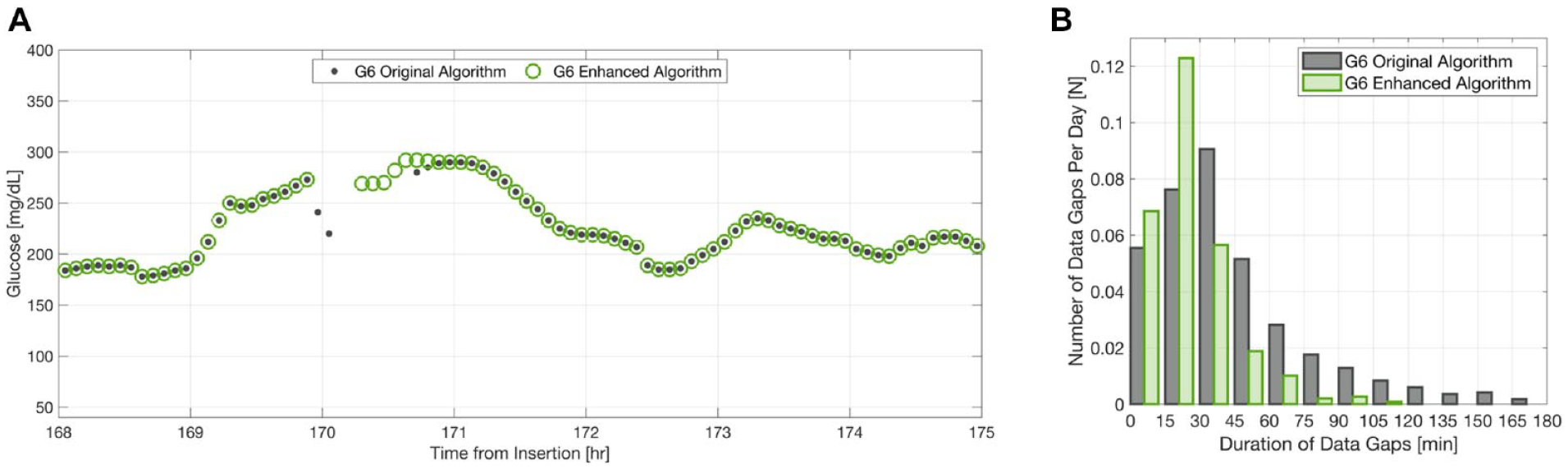
(A) Representative CGM tracing of a data gap with the original vs enhanced algorithm showing a reduction in gap duration. (B) Duration and frequency of data gaps across the 10-day sensor wear, grouped in bins of 15 min (high-end exclusive) (
The reduction in percent time in sensor error was confirmed in a limited, real-world launch in Canada. Among 110,375 sensor sessions utilizing the original algorithm, total time spent in sensor error was 1.14%. By contrast, it was only 0.65% among 60,918 sensor sessions that utilized the enhanced algorithm, indicating a 43% reduction in total time spent in sensor error.
Discussion/Conclusion
Realization of glycemic benefits from CGM systems requires consistent and sustained access to glucose data. However, factory-calibrated CGM systems must include safeguards to prevent anomalous data from being displayed, presenting a tradeoff between data availability and accuracy. The algorithm of a factory-calibrated CGM system was enhanced to improve data availability while maintaining system performance. The goals of the algorithm modification were met—the average data gap length decreased by 47% and the total time spent in sensor error decreased by 59% while percent and point accuracy were preserved across the participant age range. Similar reductions in time spent in sensor error were observed after a real-world deployment of the enhanced algorithm. Shorter and less frequent data gaps should improve user experience and decrease the number of unannounced hypo- or hyperglycemic events. Further, more consistent data availability should improve AID systems, where precise insulin delivery depends on a steady and reliable stream of CGM data.
The strengths of this study are the large number of enrolled and analyzed participants—children, adolescents, and adults—and the substantial number of CGM:YSI matched pairs evaluated. Additional studies are warranted to assess safety, user experience and/or AID system efficacy before and after the algorithm retuning.
Footnotes
Acknowledgements
We thank Drs. Sarah Puhr and John B. Welsh for their help in preparing the manuscript. Dexcom and Dexcom G6 are registered trademarks of Dexcom, Inc. in the United States and/or other countries.
Abbreviations
AID, automated insulin delivery; CGM, continuous glucose monitoring; iCGM, integrated continuous glucose monitoring; MARD, mean absolute relative difference; T1D, type 1 diabetes; T2D, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Rebecca Harvey Towers, Xiaohe Zhang, Liang Wang, Rasoul Yousefi, Ghazaleh Esmaili, Arturo Garcia, and Stayce Beck are full-time employees of Dexcom, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Dexcom, Inc.
