Abstract
Background:
There is limited experience with continuous glucose monitoring (CGM) in intensive care units (ICUs). This study examined CGM accuracy and changes during hemodynamic instability in ICU patients with COVID-19.
Methods:
We pooled data from three ICUs using CGM within a hybrid protocol combining point-of-care (POC) blood glucose testing with intermittent nonadjunctive CGM use. We compared sensor-meter agreement during lowest oxygen saturation, arterial partial pressure of oxygen (PaO2), pH, or mean arterial pressure (MAP). Linear mixed models (LMM) were used to estimate the effects of clinical condition on estimates of sensor accuracy.
Result:
Of 169 patients, >80% had a history of diabetes, mean age was 61 ± 12 years, and 82%, 93%, and 62% received corticosteroids, mechanical ventilation and vasopressors respectively. The median percent CGM time in range (TIR, 70-180 mg/dL) was 72% (64.9-81.4), 70% (54.0-76.9), and 46% (26.6-68.5) for hospitals A, B, and C. Median time below 70 mg/dL was <0.1% for all hospitals. The absolute relative difference between CGM and POC pairs did not correlate with the lowest PaO2, oxygen saturation, pH, or mean arterial pressure. In LMM adjusting for within subject and between subject variability, patients on dialysis had higher mean absolute relative difference (MARD, [coefficient = 2.39, P = .05]), while patients on mechanical ventilation had lower MARD ventilation (coefficient = −4.33, P = .05). Of the 6783 pairs 97.3% fell within Clare zones A and B.
Conclusion:
These preliminary findings suggest CGM use does not appear to be significantly affected during critical illness. Confirmatory accuracy studies are needed.
Introduction
Since the start of the COVID-19 pandemic, the introduction and emergency use of continuous glucose monitoring (CGM) devices have provided not only a way to potentially preserve personal protective equipment (PPE), but a wealth of knowledge on their safety, efficacy, and performance in the inpatient setting. Because hyperglycemia and hypoglycemia have been shown to be associated with poor outcomes, 1 particularly in patients with COVID-19 in the intensive care unit (ICU), the information CGM data provides can also facilitate the use of intravenous (IV) insulin therapy to achieve glycemic control goals.
Recent observational studies have shown the feasibility and safety of CGM use in the noncritical 2 and ICU settings3,4 in conjunction with various insulin delivery methods.5,6 To address accuracy concerns, institutions have utilized hybrid glucose monitoring approaches in which point-of-care (POC) glucose testing is used to validate CGM accuracy prior to nonadjunctive, or stand-alone, use.3,4,6 -11 Emerging randomized controlled trials also support the safety of CGM use in the hospital outside of the ICU.12,13 In a recent scoping review by Nielson et. al. (2024), CGM accuracy in the ICU varied widely across studies with an MARD of 9.7%-20.6% (median 13.2%; IQR 12.9-13.9%) reported for currently available CGM technologies. 11 Most of the studies were observational (91.7%) and to date, there are no randomized control trials investigating currently available commercial CGM in critically ill patients.3,9,11
We pooled data from three academic institutions that used CGM during the COVID-19 pandemic with the aim of examining accuracy of CGM in the critical care environment and evaluating CGM performance during periods of hemodynamic instability.
Materials And Methods
Design and Study Population
We conducted a multicenter retrospective observational study including 169 patients with COVID-19, admitted to the ICU at The Ohio State University Medical Center, Grady Memorial Hospital, and Stony Brook University Hospital (hereafter referred to as Hospitals A, B, and C, respectively). All patients were treated with insulin therapy and monitored with real-time CGM devices. The study primary aim was to examine CGM accuracy during periods of heightened critical illness with comparison of paired CGM and POC BG values. Our secondary aim was to examine factors of ICU CGM use within a hybrid protocol. This study was approved by the institutional review boards at all academic centers.
Diabetes Technology
Each hospital used the Nova Biomedical StatStrip POC meter which is U.S. Food and Drug Administration (FDA) approved for use in critical care. 14 POC measurements were obtained from arterial, venous, and capillary glucose samples. All three centers utilized the factory-calibrated Dexcom G6 CGM (Dexcom, San Diego, CA), in conjunction with the G6 App for transmission of sensor data to a smart-device (hospital B) or receiver (hospitals A and C), the Follow App for remote monitoring (hospital B), and Dexcom Clarity for data monitoring (hospitals A-C). Continuous glucose monitoring devices were placed on the upper arm by trained nursing or physician staff on patients with hyperglycemia and COVID-19 requiring insulin therapy who were admitted to the ICU. Continuous glucose monitoring sensors were Bluetooth paired to a combination of device specific receivers and Android phones. Glucose data was available after a 2-hour warm up period at which time CGM validation began. Intermittent nonadjunctive use of CGM (hourly use of CGM for insulin adjustment) occurred after sensor validation using POC devices used in routine clinical practice. While no specific manual calibration protocol was in place, manual calibration was performed at hospitals A and B at nurses’ discretion when validation was not met. Validation and ongoing use consisted of the following:
A and B: CGM could be used nonadjunctively after two consecutive hourly sensor glucose values fell within ±20% of the POC BG value (if POC BG >100 mg/dL or within ±20 mg/dL (if the POC BG <100 mg/dL). Thereafter POC BG occurred at least every 6 hours, if there were predicted low glucose or high/low threshold alerts from the CGM, or if there was a change in clinical status such as initiation of vasopressors, intubation, or nutrition. 15
C: CGM could be used nonadjunctively after 24 hours of hourly sensor glucose values, if they fell within ±20% of the POC BG value (if POC BG >100 mg/dL) or within ±20 mg/dL (if the POC BG <100 mg/dL) (and confirmed by a lead endocrinologist). For patients using IV insulin, POC BG occurred every 2 hours, alternating with CGM values every other hour. POC BG was also required if there was a change in glucose >100 mg/dL since the last reading and if there was >20% difference between POC and CGM. Outside of the ICU, POC BG was required before meals in patients who were eating or every 12 hours in those who were not. 4
Data Collection
We extracted data from the electronic health record (EHR) of 50 patients at hospital A, 71 patients at hospital B, and 48 patients at hospital C. This included clinical characteristics, inpatient medication use (including steroids, vasopressors, and insulin), sensor glucose, and timestamped POC BG values. Sensor glucose data (288 values/day for 100% CGM wear) was downloaded directly from the manufacturer’s cloud-based software. Sensor-meter pairs were constructed by matching the EHR POC values with the downloaded CGM value generated within 5 minutes and POC values marked as calibrations were excluded from analysis. In addition, we matched sensor-meter paired values obtained within 30 minutes of the patient’s single lowest arterial partial pressure of oxygen (PaO2), pulse oximetry, arterial pH, and mean arterial pressure (MAP) during CGM wear. Pairs were also matched prior to patient expiration (hospital B only, if applicable). Pairs were matched within excel and all data was uploaded into STATA 15.0 where it was cleaned and merged prior to analysis.
Data Analysis
Continuous variables with normal distribution were reported as mean (SD) while those with nonnormal distribution were reported as median (interquartile range [IQR]). Discrete CGM values were used to aggregate derived measures of glucose, including % time in target (70-180 mg/dL), above target (>180 and >250 mg/dL), hypoglycemia (<70 and <54 mg/dL), mean glucose, and coefficient of variation. The glucose range of 70 to 180 mg/dL is consistent with other studies examining time in target.16 -20 Measures of hypoglycemia and hyperglycemia are in accordance with American Diabetes Association (ADA) standards and Diabetes Technology Society (DTS) recommendations for CGM metric use in clinical trials.20,21 Elements of CGM use within the hybrid protocol were evaluated including frequency of POC measurements and time to initial validation of CGM, Univariate associations between clinical condition variables and sensor-meter pair mean absolute relative difference (MARD) were measured using two-sample t test and Pearson’s r correlation. We constructed mixed models using backward selection to included variables found to be significant in univariate analysis. A linear mixed effect modeling (LMM) for repeated measures was used to model MARD as a function of those patient characteristics, adjusting for within subject clustering from daily measures and controlling for hospital site as a covariate. A two-sided significance level of 0.05 was used for all the statistical tests. Finally, a DTS Grid was conducted to assess the clinical accuracy of continuous glucose monitoring (CGM) data compared with a reference standard. This analysis categorizes the magnitude of error into five zones based on clinical relevance: zone A (no risk), zone B (mild risk), zone C (moderate risk), zone D (high risk), and zone E (extreme risk). The percentage of data points falling into each zone was calculated to determine the overall clinical performance of the CGM system. 22
Data and Resource Availability
In accordance with institution policy on sharing data and research resources, the final research data from this study may be made available for research purposes under a limited data use agreement specifying criteria for data access, conditions for research use, privacy and confidentiality standards to ensure data security and prohibitions for manipulating data for the purposes of identifying subjects. Requests may be directed to the corresponding author (KD).
Results
There were 169 COVID-19 positive ICU patients included in this study across the three hospitals. The mean age was 60.9 ± 12.2 years and 82%, 93%, and 62% received corticosteroids, mechanical ventilation, and vasopressors respectively. The majority of the patients had preexisting type 2 diabetes mellitus, and the average A1C was over 8.8% ± 2.6% (range 4.2%-18%). The majority of patients from hospital B identified as African American (89%), whereas most patients from the other two hospitals identified as Caucasian (68% at hospital A and 54% at hospital C). Hospital C also had a substantial percentage of Hispanic/Latino patients (31.1%). There was a larger percentage of patients at hospital C who had diabetic ketoacidosis (DKA)/hyperosmolar nonketotic state compared with the other two hospitals (50%) (P < .001). The majority of the patients were on mechanical ventilation at the time of CGM placement. Patient characteristics are summarized in Table 1.
Baseline Characteristics.
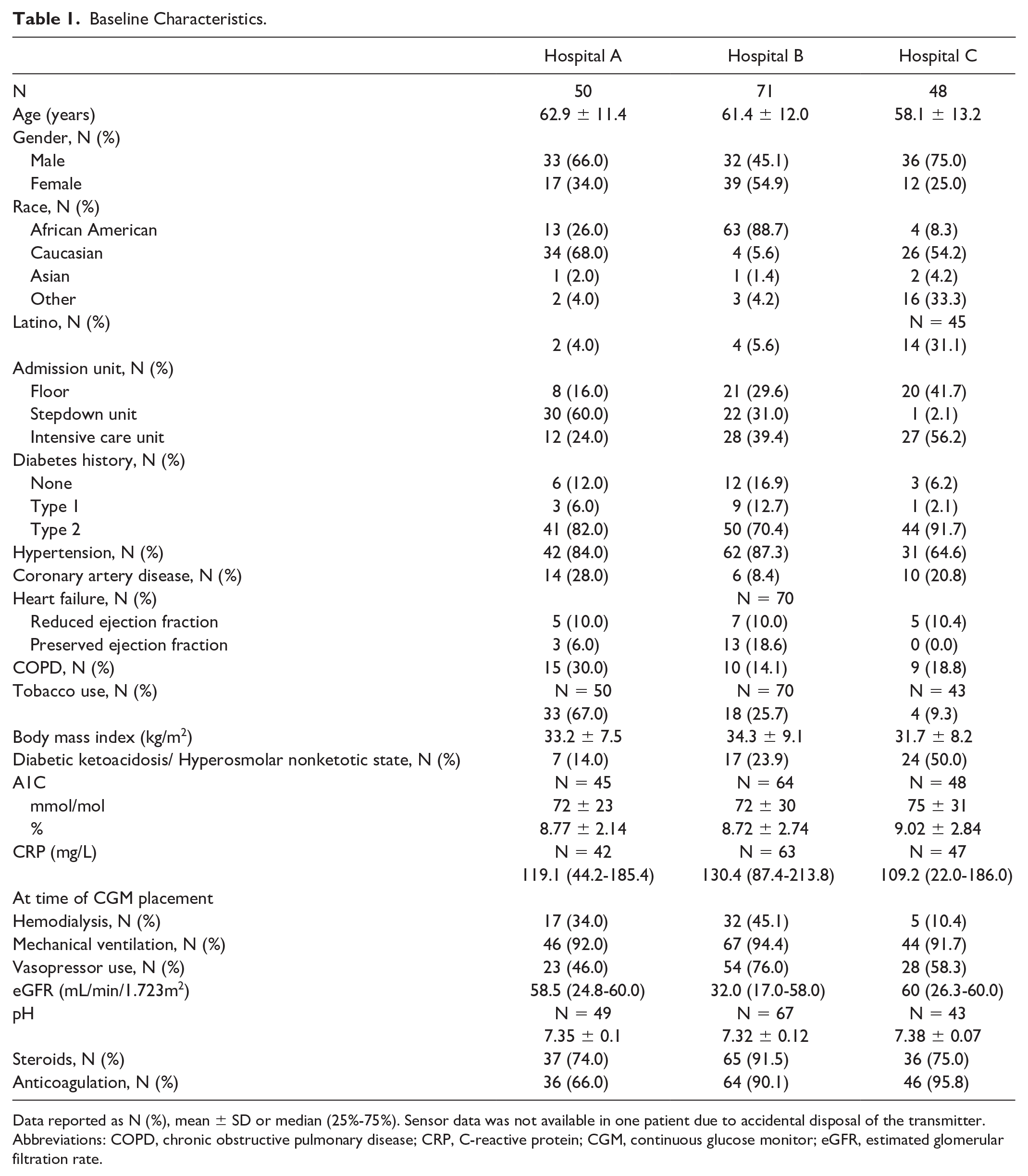
Data reported as N (%), mean ± SD or median (25%-75%). Sensor data was not available in one patient due to accidental disposal of the transmitter.
Abbreviations: COPD, chronic obstructive pulmonary disease; CRP, C-reactive protein; CGM, continuous glucose monitor; eGFR, estimated glomerular filtration rate.
The median sensor duration in hours was 224.8 (IQR 129.0-237.9) at hospital A, 129.2 (IQR 56.1-237.9) at hospital B, and 188.5 (77.8-235.3) at hospital C. The median duration of IV insulin was 119 (IQR 73-170), 117 (IQR 48-239), and 50 (IQR 23-120) hours at hospitals A, B, and C respectively. Mean POC BG frequency was 10.3 ± 6.1, 7.0 ± 5.2, and 16 ± 6.5 times per day on day 1 of IV insulin and 6.7 ± 3.1, 5.9 ± 3.5, and 12.2 ± 5.8 times per day thereafter. The time to first sensor value validation varied between the two hospitals with this reported data (20 [0-130] hours at hospital A, 87 [29-316] hours at hospital B). The percentage of first sensor values that met validation criteria used by hospitals A and B was 67% at hospital A, 69% at hospital B, and 28% at hospital C. All hospitals reported a decrease in the number of POC BG per day from day 1 of sensor use versus days 2 through 7 (Table 2).
Sensor Use.
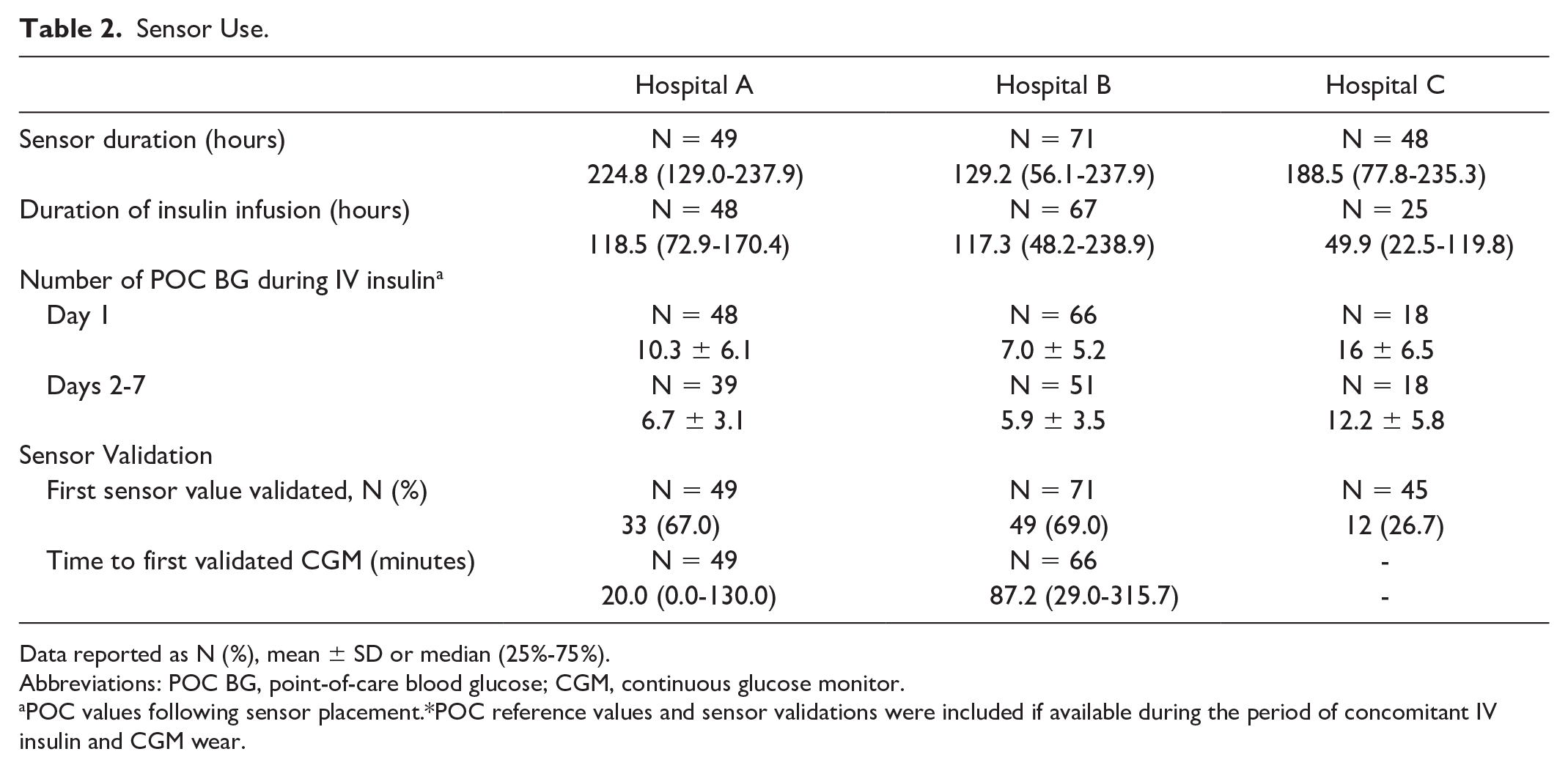
Data reported as N (%), mean ± SD or median (25%-75%).
Abbreviations: POC BG, point-of-care blood glucose; CGM, continuous glucose monitor.
POC values following sensor placement.*POC reference values and sensor validations were included if available during the period of concomitant IV insulin and CGM wear.
Sensor glucose values were categorized into glucose ranges: >250 mg/dL, >180 mg/dL, between 70 and 180 mg/dL (time in range, TIR), <70 mg/dL, and <54 mg/dL. The median percent CGM TIR (70-180 mg/dL) was 72%, 70%, and 46% for hospitals A, B, and C during IV insulin (overall P = .005, A vs C P < .001, B vs C P = .005). Median time below 70 mg/dL was <0.1% for all hospitals during IV or subcutaneous (SQ) insulin. The percentage of time spent greater than 250 mg/dL was 3.3 (0.8-7.9) at hospital A, 2.8 (0.0-14.4) at hospital B, and 13.8 (4.5-32.9) at hospital C during IV insulin with higher percentages during SQ insulin (Table 3).
Sensor Glucose Values.
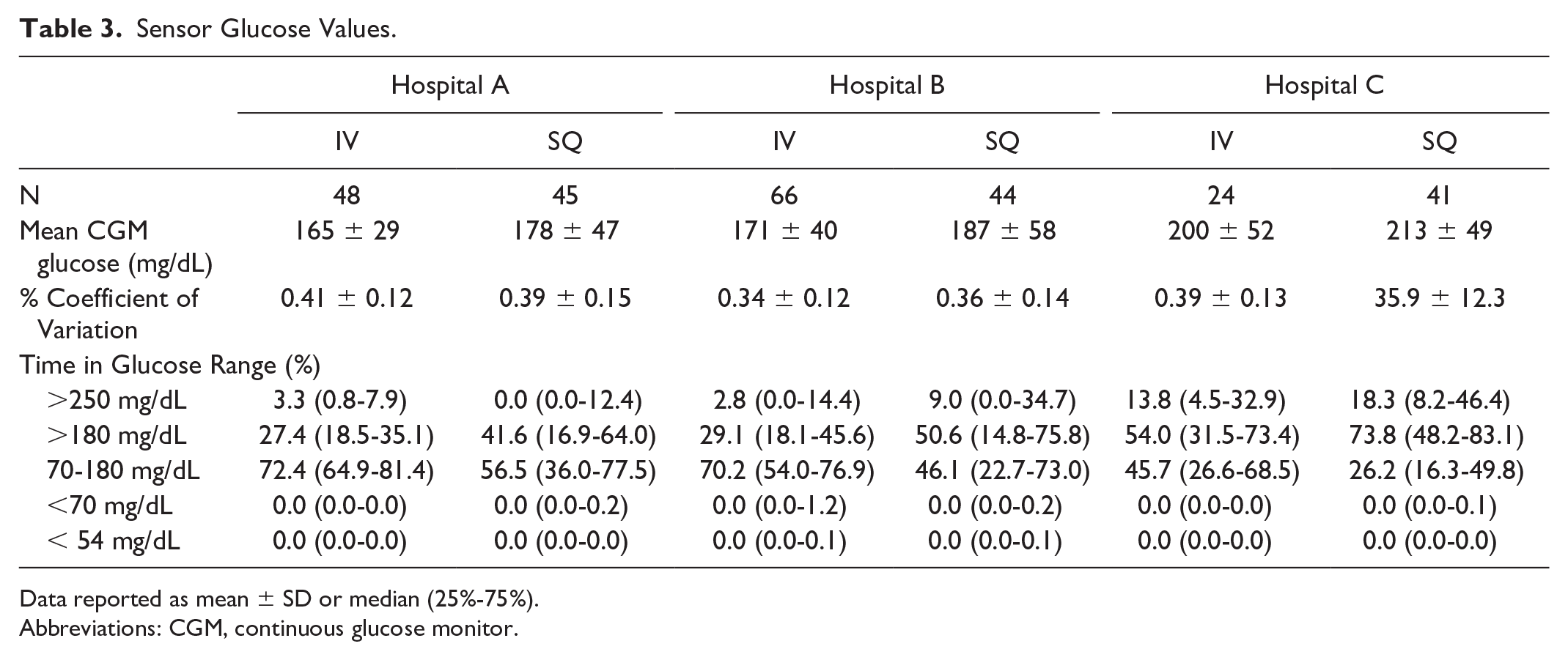
Data reported as mean ± SD or median (25%-75%).
Abbreviations: CGM, continuous glucose monitor.
Mean MARD across all hospitals was 18.4 ± 17.7, however there were significant differences in MARD across the three hospital sites, F(2,9415) = 39.11, P < .001F(2, 9415) = 39.11, p < 0.001. The mean MARD was 15.2 ± 12.5 at hospital A, 18.6 ± 19.0 at hospital B, and 19.5 ± 18.3 at hospital C. Sensor values were compared with POC BG values to calculate sensor-meter absolute relative difference during each patient’s lowest PaO2, oxygen saturation, arterial pH, and MAP. The median absolute relative difference was <20% at all hospitals (Table 4). Otherwise, the absolute relative difference between CGM and POC at did not vary consistently across thresholds of physiologic measures and hospitals (Table 4). Moreover, the absolute relative difference did not correlate with the lowest PaO2, oxygen saturation, pH, or mean arterial pressure in pooled analyses (Figure 1).
Sensor Meter Agreement.
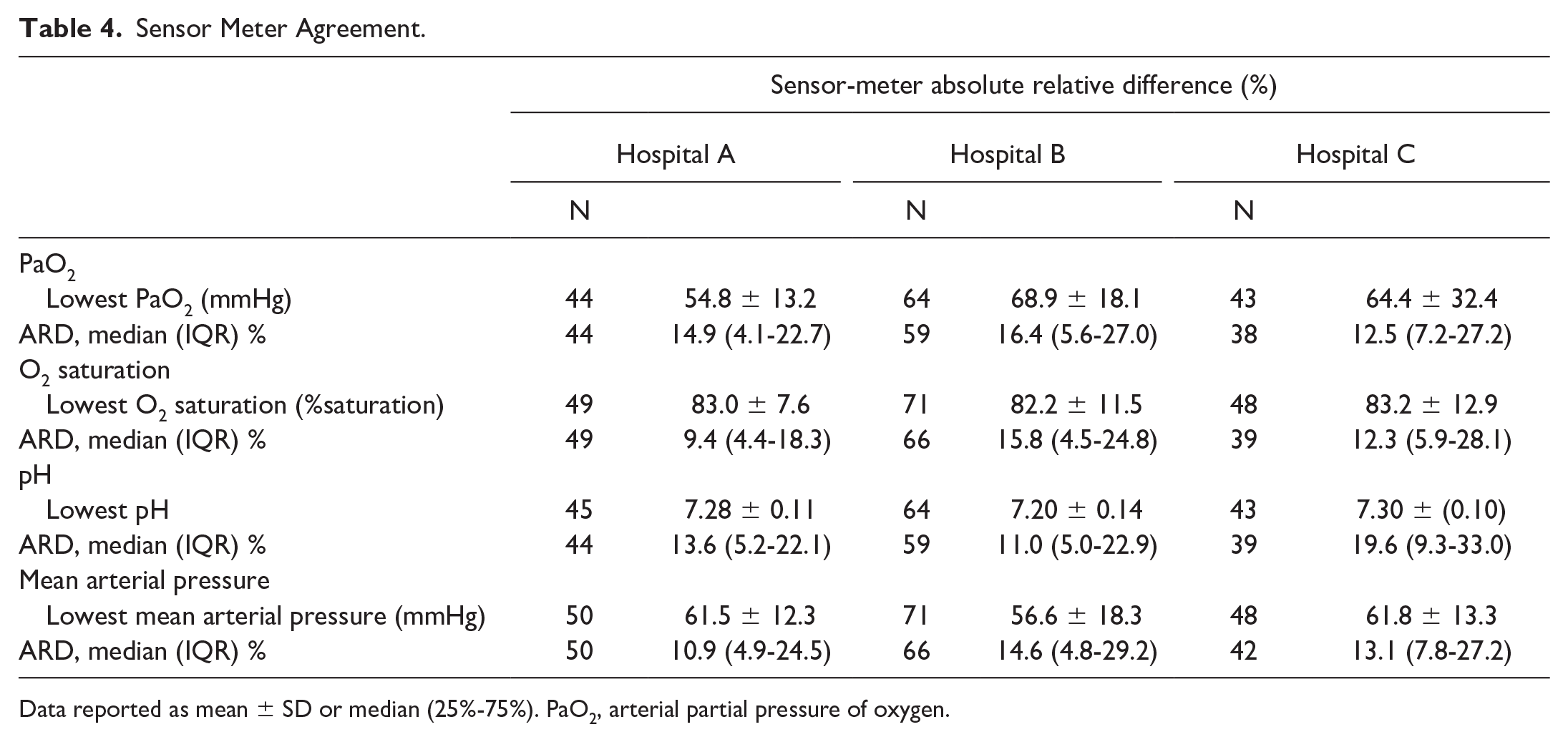
Data reported as mean ± SD or median (25%-75%). PaO2, arterial partial pressure of oxygen.
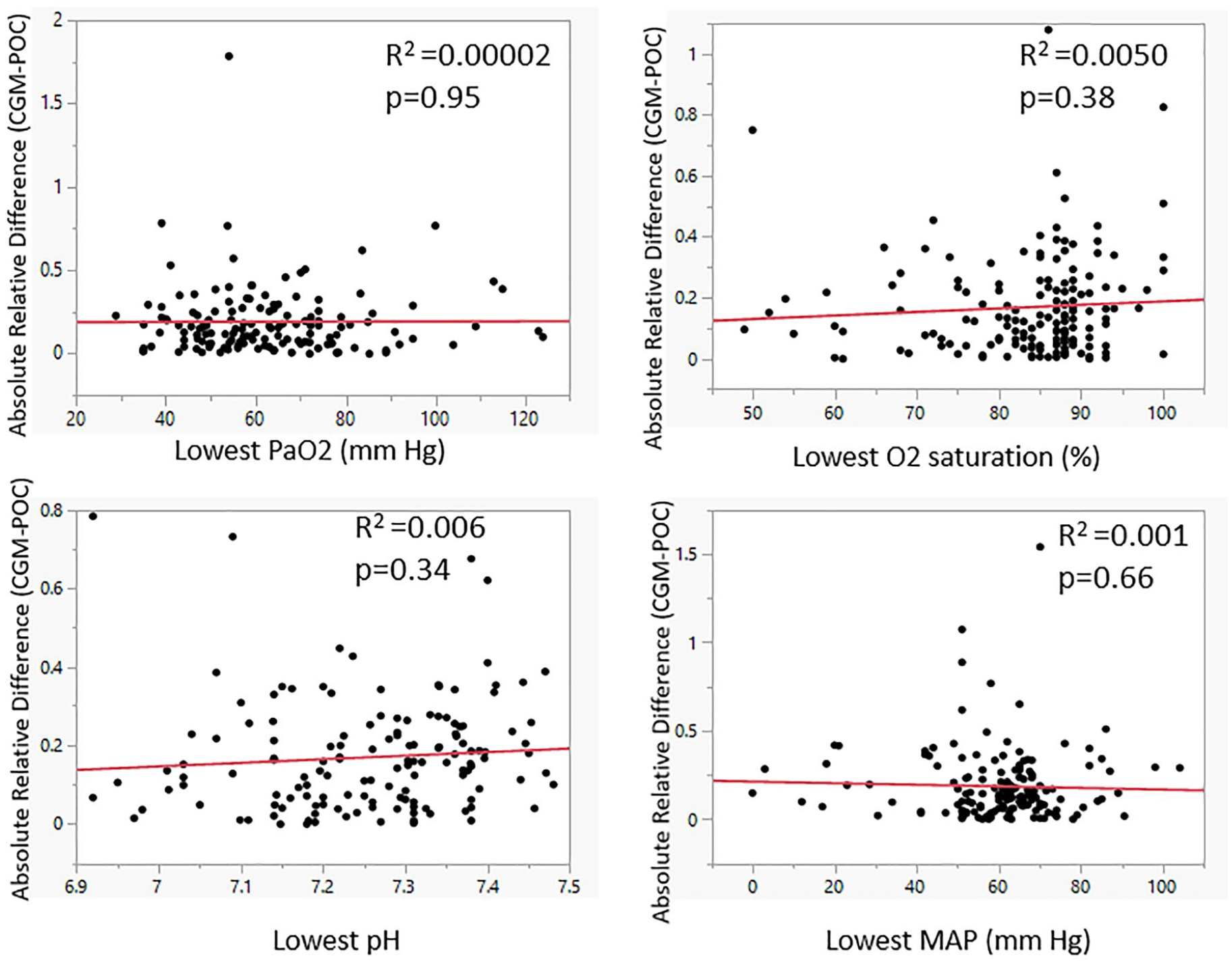
Relationship between lowest PaO2, arterial partial pressure of oxygen, O2 saturation, pH, and mean arterial pressure with CGM accuracy (absolute relative difference of CGM and POC).
At hospital B, additional analyses were performed in 11 patients who were wearing CGM at the time of expiration (Supplemental Figure). Ten patients were receiving comfort care while the eleventh had a “Do not resuscitate” order and expired after developing asystole. The mean absolute relative difference between available sensor meter pairs (N = 25) in the six hours prior to expiration was 15.4 ± 15.8%.
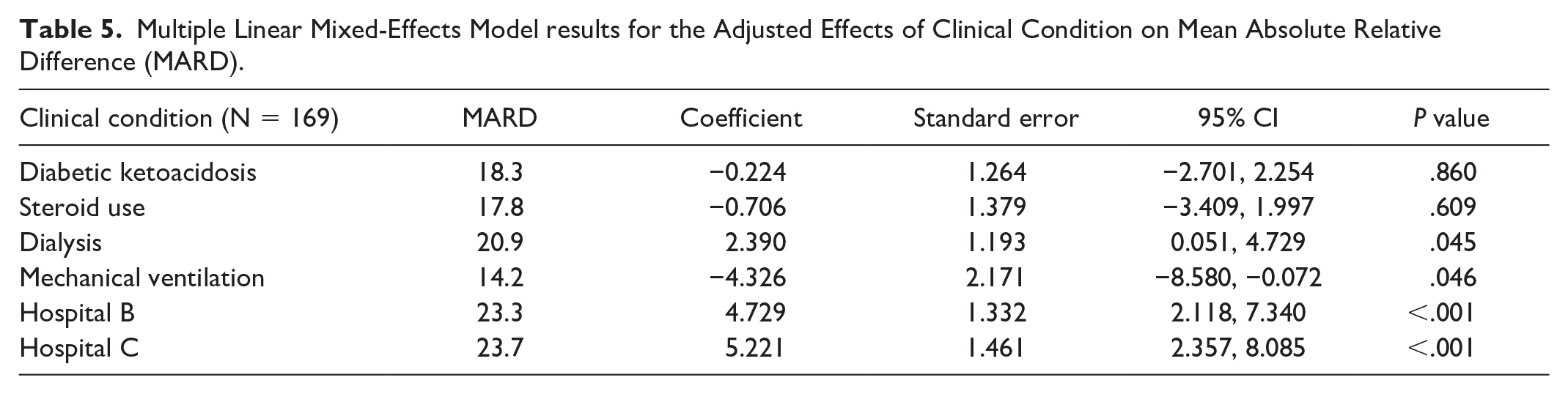
Our pooled analysis contained 9465 POC/CGM pairs with hospital A having 1785 pairs, hospital B 2964, and hospital C 4669. Patients on dialysis and those in DKA at the time of sensor placement had higher MARD (19.4 ± 19.5 vs. 18.1 ± 17.0, P = .003 and 19.4 ± 17.6 vs. 17.9 ± 17.7, P < .001, respectively). Patients who were ventilated and on steroids at the time of placement had lower MARD (18.2 ± 17.0 vs. 23.4 ± 26.4, P < .001 and 18.2 ± 17.4 vs. 19.4 ± 19.1, P = .0153). Duration of insulin infusion (hours) was associated with lower MARD (coefficient = −0.021, P ≤ 0.001). The effects of vasopressors, anticoagulants, and C-reactive protein (CRP) values on MARD were also assessed in univariate models and were not found to be significant. Clinical condition variables that were found to be significantly correlated with MARD in unadjusted analysis were included in an LMM model (Table 5). In multivariate LMM modeling adjusting for within subject variability and controlling for hospital site as a covariate, dialysis at the time of CGM placement was significantly associated higher MARD (coefficient = 2.390, P = .045), while mechanical ventilation at the time of placement was associated with lower (coefficient = −4.326, P = .046). There was significant variation in MARD across hospitals with hospital B and hospital C having higher MARD than hospital A (coefficient = 4.729, P < .001 and coefficient = 5.221, P < .001, respectively). Although MARD did vary significantly by hospital, there were no significant interactions between predictive variables included in the model and hospital site suggesting that the effects of clinical condition on MARD do not significantly vary by hospital site. In addition, there was significant variability in MARD between patients, as indicated by the random intercept variance and the mixed model provides a better fit than a model without random effects.
Multiple Linear Mixed-Effects Model results for the Adjusted Effects of Clinical Condition on Mean Absolute Relative Difference (MARD).
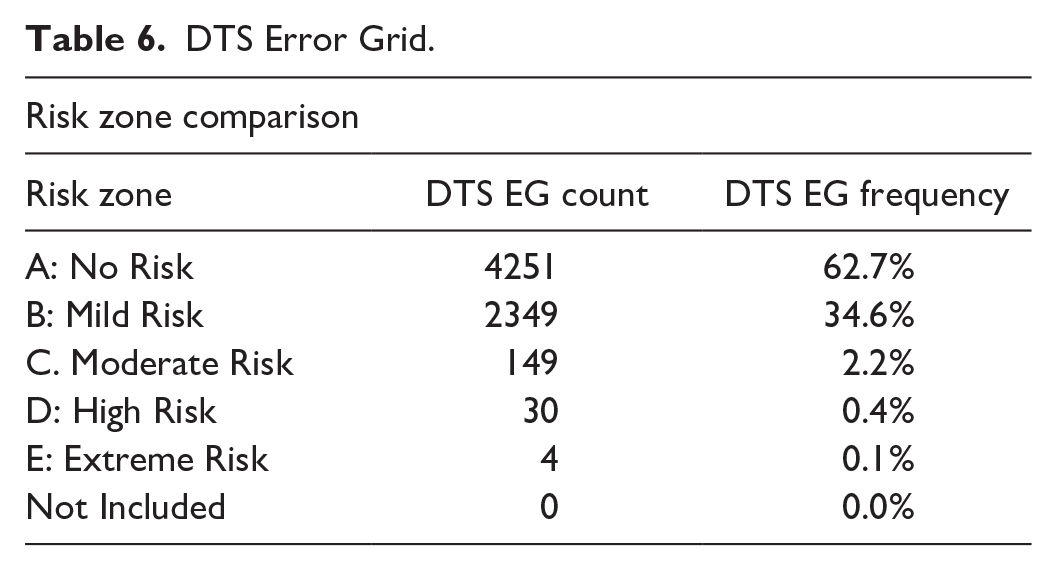
A total of 6783 pairs were analyzed using the DTS Error Grid Classification (Table 6 and Figure 2). Overall, 62.7% of pairs fell within risk zone A, indicating no clinically significant error, and 34.6% were in risk zone B, indicating mild risk. A smaller proportion of pairs were classified in risk zones C (2.2%), D (0.4%), and E (0.1%), which represent increasing levels of clinical significance. Breakdown by hospital showed that hospital A had 71.2% of pairs in zone A, while hospital B had 64.8%. Hospital C had the lowest proportion in zone A (52.1%), with a higher proportion in zone B (45.0%). The overall distribution reflects favorable performance across all sites, with the majority of pairs falling in zones A and B.
DTS Error Grid.
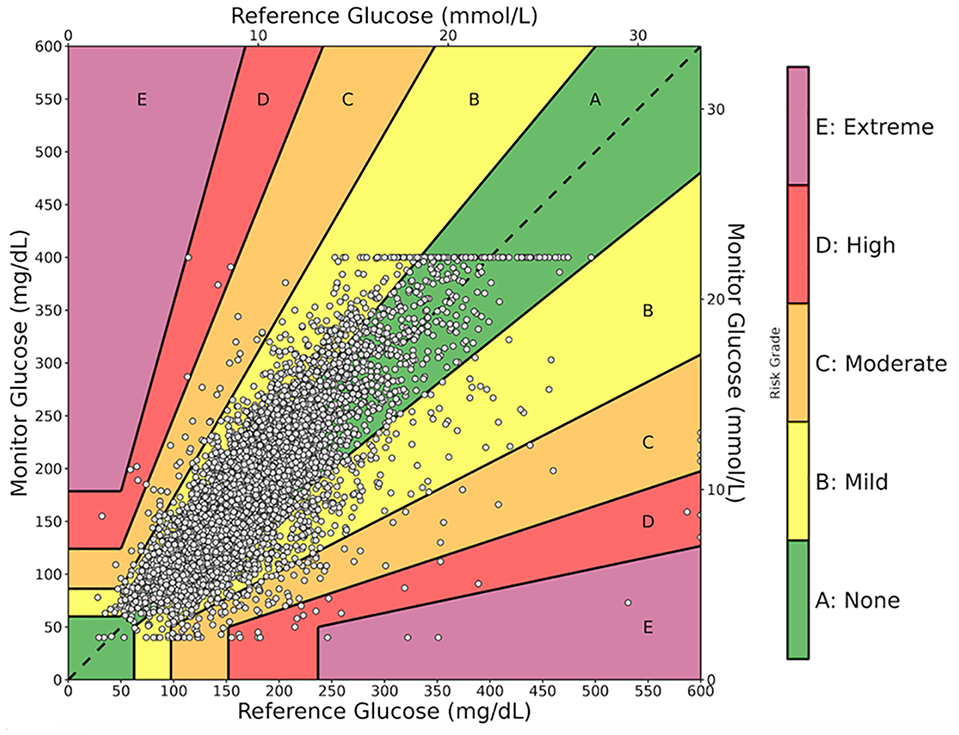
Diabetes technology society (DTS) Error grid assessing clinical accuracy of CGM glucose measurements relative to reference point-of-care (POC) values.
Discussion
Our findings from real-world clinical practice in COVID-19 ICUs show the use of real-time CGM under a hybrid glucose monitoring strategy can reduce the frequency of POC BG measurement while maintaining safe and acceptable glycemic control. In addition, sensor-meter agreement appeared to be preserved during extreme abnormalities in physiologic parameters in the setting of exposure to a myriad of critical care therapies. While changes in patient severity of illness did not appear to affect sensor-meter agreement, patient baseline clinical condition, in particular dialysis and mechanical ventilation at the time of sensor placement were associated with sensor accuracy.
Previous studies have also shown decreased CGM accuracy in peritoneal and hemodialysis patients with MARDs ranging from 11.3% to 22.7%23 -25 but not with lesser degrees of renal insufficiency. 6 Decreased sensor performance in the presence of end stage renal disease and dialysis may be attributed to enzymatic electrochemical reactions or interfering substances that may interfere with glucose oxidase sensors such as the Dexcom G6. 26 In addition, changes in glucose during dialysis could contribute to decreased sensor performance. In contrast, ventilation at sensor placement was associated with significantly lower MARD or increased sensor accuracy. While initially surprising, as these patients are generally more clinically ill, the finding may be indicative of changes in nutritional status. Patients who are not ventilated are more likely to be consuming an oral diet which may cause increased glucose variability and sensor lag time compared with nil per os or enteral feed regimens seen more often in ventilated patients. Nutritional status other than the presence of enteral nutrition was not collected. However, we did not find any interaction between enteral nutrition or hours on IV insulin and ventilation status. Continuous glucose monitoring accuracy varied significantly across hospital systems. However, there were no significant interactions between the predictive clinical condition variables and hospital site, meaning differences may be attributed to unmeasured variables such as sensor rate of change or factors specific to site implementation such as timing of sensor insertion, or calibration practices.
We and others have reported preliminary findings suggesting the use of CGM can reduce the burden of inpatient diabetes care by reducing the need for hourly glucose testing and by reducing the usage of PPE among patients under isolation precautions with a highly contagious and deadly disease.3,6,10,15 While POC frequency was reduced significantly from standard hourly testing at all institutions, it is interesting to note that at hospitals A and B, which required Q6 ongoing POC testing and validation, the mean frequency of POC testing was 6.7 ± 3.1 and 5.9 ± 3.5, respectively. Meaning that nursing was typically performing additional testing and validation in response to protocol requirements (i.e., clinical status changes, hypoglycemia threshold alarms, failure to meet ongoing validation). The analysis of this large sample of critically ill patients treated with a hybrid glucose monitoring strategy expands our knowledge on the safety, efficacy, and accuracy of CGM use in the ICU. Although COVID-19 is currently not as prevalent, the feedback from healthcare providers who used CGM during the pandemic has been positive regarding ease of use, reassurance of safety, and improved monitoring.4,15
There are very limited data on the use of CGM in the ICU on the incidence of hypoglycemia and achievement of glycemic targets. 27 The observed mean glucose (below 180 mg/dL) and TIR of over 70% in hospitals A and B during IV insulin therapy likely reflects the goals that can be achieved with current IV insulin protocols. 28 The higher mean glucose at hospital C is likely a reflection of the significantly higher proportion of patients with DKA at the time of sensor placement and shorter duration of IV insulin and may also be reflective of loco-regional trends in COVID-directed therapeutics earlier in the pandemic. The shorter duration of IV insulin could be related to protocol implementation factors. Hospital C’s hybrid protocol required ongoing POC validation every 2 hours, which may have presented higher nursing burden and exposure and led to an earlier rate of IV insulin discontinuation. The use of this technology was associated with a minimal risk of hypoglycemia. As expected, the lower TIR observed after transition to SQ insulin is consistent with the use of CGM in non-ICU settings—where achieving targets is more challenging with multiple daily injections 13 as opposed to the use of IV insulin.
Our findings are consistent with those of Nielsen et al, 11 which reported a wide range of MARD values for currently available factory-calibrated CGM devices in the critical care setting (9.7%-20.6%). Furthermore, our analysis shows stable accuracy during extremes of hemodynamic instability. Despite this, inpatient CGM accuracy is diminished when compared with the ambulatory setting where CGM systems demonstrate excellent accuracy, with an MARD of 7% to 9%.29,30 There are no concrete benchmarks for accuracy; however, Finfer et al 31 recommended 98% of values should be within 12.5% of the reference range for patients treated in the critical care setting while Danne et al 32 suggested CGM MARD ≤10% likely has little additional benefit for insulin dosing. In a modeling study conducted in the critical care setting, safe insulin dosing could be achieved with less accurate CGM systems compared with intermittent testing due to the continuous nature of the measurement. A MARD of 11% was sufficient and a higher MARD of <17.8% would avoid significant insulin dosing errors. 33 Despite decreased accuracy in the inpatient setting, the benefits of CGM therapy can still be achieved through hybrid use. We recommend confirming the accuracy of devices with POC periodically (every 4-6 hours) and during severe hemodynamic instability, during conditions that predispose to rapid rate of change, as the accuracy may be lower than that reported on the wards 34 or in the setting as a result of multiple factors. 35 Our findings, combined with prior reviews, suggest that hybrid protocols remain critical for mitigating accuracy issues, particularly during periods of glycemic instability.11,36
In the current study the “Urgent Low Soon” alert was used clinically but other trend alerts were not activated due to concern for alert fatigue. We were unable to analyze trend accuracy due to an insufficient reference sampling interval. 37 Such information would be of value as it provides vital context for the utility and safety of nonadjunctive use beyond point accuracy. Future research should consider how trend information or alerts could be used to inform treatment decisions. As automated insulin delivery devices become mainstream in the ambulatory setting, their safe use in hospitals will also rely on this information.
Limitations of this study include differences in patient population (predominance of DKA patients in hospital C), protocols and their implementation (including timing of sensor placement relative to hospital admission and initiation and duration of IV insulin) in respective critical care settings. Moreover, it is possible that the retrospective design of this study could have missed a critical exposure period for physiologic variables, particularly for those which could have a delayed effect on accuracy, such as change in nutrition or steroid use. The use of POC testing as reference is unavoidable in usual clinical practice and further accuracy studies are needed. In addition, the hybrid protocol design, which requires increased POC testing when the CGM and POC values are discordant, during hypoglycemia, and during changes in clinical condition, is likely to underestimate CGM accuracy. 11 POC glucoses was also obtained from multiple sources including arterial, venous, and capillary samples. For further accuracy evaluation future studies may consider systematic, protocolized reference testing 11 using the FDA recently cleared Nova Primary Glucose Analyzer System, as the commonly used comparator device, the YSI 2300 Analyzer (YSI 2300; YSI Incorporated, Yellow Springs, OH), cleared in 1991, is now discontinued. 38 Such data are of great relevance to continue to move forward in the rigorous evaluation of these devices during extreme disease conditions in patients with hyperglycemia who requiring close monitoring.
Conclusions
This multicenter study shows that MARD does not appear to vary during changes in clinical condition marked by extreme physiologic measures. We also observed that the use of a hybrid glucose monitoring strategy using CGM and POC BG leads to a reduction in the frequency of POC BG measures during IV insulin while maintaining glucose levels. The scope of the utilization of CGM during critical illness requires further study as we continue to look for ways to improve care while reducing staff burden.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251343108 – Supplemental material for Continuous Glucose Monitoring in the Intensive Care Unit: A Multicenter, Retrospective Hospital-Based Analysis
Supplemental material, sj-docx-1-dst-10.1177_19322968251343108 for Continuous Glucose Monitoring in the Intensive Care Unit: A Multicenter, Retrospective Hospital-Based Analysis by Eileen R. Faulds, Joi C. Hester, Yasaman Badakhshi, Joshua D. Miller, Rosalind C. Basil, Suvrat Chandra, Albert S. Chang, Marlene Garcia, Laureen Jones, Katiria A. Pintor, Andrew Boutsicaris, Danielle J. Kelly, Jillian Pattison, Nihaal Reddy, Rohan Shah, Matthew Exline, Georgia Davis, Francisco J. Pasquel and Kathleen M. Dungan in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
None.
Author’s Note
Data from this manuscript were presented at the American Diabetes Association Scientific Sessions 2023, San Diego, CA.
Abbreviations
CGM, continuous glucose monitoring; ICUs, intensive care units; PaO2, arterial partial pressure of oxygen; MAP, mean arterial pressure; LMM, linear mixed models; TIR, time in range; POC, point-of-care; MARD, mean absolute relative difference; PPE, personal protective equipment; BG, blood glucose; IV, intravenous; EHR, electronic health record; IQR, interquartile range; ADA, American Diabetes Association; DTS, diabetes technology society; SQ, subcutaneous; DKA, diabetic ketoacidosis; FDA, U.S. Food and Drug Administration; YSI, yellow springs instrument.
Author Contributions
KMD, JM, ERF, MCE, LGJ, and FJP were involved in the conception and design of the clinical protocol and study. JCH, YB, RB, SC, AC, MG, KP, AB, DJK, JP, NR, and RH collected data for the study. KMD, NH, JP, and RS analyzed the data. JCH wrote the first draft of the manuscript, and all authors reviewed, edited, and approved the final version of the manuscript. KMD had full access to all the data and takes responsibility for the integrity of the data and accuracy of the analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KMD discloses research support from Abbott, Insulet, and Dexcom, consulting with Eli Lilly, Insulet, Dexcom, and Oppenheimer, honorarium from Elsevier, Med Learning Group, Medscape, Impact Education, and Cardiometabolic Health Congress and royalties from UpToDate. At time of publication, JM is employed by Medtronic; he reports past research support from Dexcom and has previously served as an advisor for Medtronic Diabetes and MannKind, inc. ERF discloses research support from Dexcom and Insulet, consulting from Dexcom, and honorarium from Dexcom and Medscape. FJP discloses unrestricted research support from Insulet, Dexcom, Tandem Diabetes Care, Novo Nordisk, and Ideal Medical Technologies, personal fees from Dexcom for consulting activities, and fees to his institution from Insulet for consulting activities. JCH, YB, RB, SC, AC, MG, KP, AB, DJK, JP, NR, RS, LGJ, and MCE have no conflicts of interest to report.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Ohio State University Clinical and Translational Science Institute (CTSI) and the National Center for Advancing Translational Sciences of the National Institutes of Health under grant number UM1TR004548 and award number P30DK111024 from the National Institutes of Diabetes, Digestive and Kidney Diseases (NIDDK)-funded Georgia Center for Diabetes Translation Research. The content is solely the responsibility of the authors and does not represent the views of the National Institutes of Health.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
