Abstract
Traditionally, the care of critically ill patients with diabetes or stress hyperglycemia in the intensive care unit (ICU) demands the use of continuous intravenous insulin (CII) therapy to achieve narrow glycemic targets. To reduce the risk of iatrogenic hypoglycemia and to achieve glycemic targets during CII, healthcare providers (HCP) rely on hourly point-of-care (POC) arterial or capillary glucose tests obtained with glucose monitors. The burden of this approach, however, was evident during the beginning of the pandemic when the immediate reduction in close contact interactions between HCP and patients with COVID-19 was necessary to avoid potentially life-threatening exposures. Taking advantage of the advancements in current diabetes technologies, including continuous glucose monitoring (CGM) devices integrated with digital health tools for remote monitoring, HCP implemented novel protocols in the ICU to care for patients with COVID-19 and hyperglycemia. We provide an overview of research conducted in the ICU setting with the use of initial CGM technology to current devices and summarize our recent experience in the ICU.
Introduction
Improving glycemic control is paramount to prevent complications in the intensive care unit (ICU). 1 To achieve glycemic targets, the use of continuous intravenous insulin (CII) therapy is generally indicated in the ICU setting, as it allows for rapid adjustments of insulin doses to keep glucose levels within a narrow target. This approach is efficacious but demands a high burden for staff as it usually requires hourly testing with point-of-care (POC) glucose values to make therapeutic adjustments. 2 Despite close monitoring, the risk of iatrogenic hypoglycemia is still high and similar to the increased risk for poor outcomes observed with hyperglycemia, the incidence of hypoglycemic events is also associated with high morbidity and mortality.3,4 This association has been observed in ICU and non-ICU settings. 5 Hence, different strategies have been investigated to monitor glucose levels closely to guide insulin therapy decisions and maintain acceptable glucose ranges.
The first commercially available continuous glucose monitoring (CGM) devices were introduced in the 1990’s (Medtronic MiniMed, Northridge, CA), 6 and soon after their commercialization the initial experience with CGM in the ICU was published. 7 Efforts investigating CGM in the ICU are still ongoing, but the acceleration in the use of the technology in clinical practice emerged as a response to COVID-19 when the Food and Drug Administration (FDA) indicated no objection to the use of these devices in the hospital.
We present here a review of the literature related to the use of CGM to care for patients admitted to critical care units and provide an overview of the accuracy data, implementation approaches including remote monitoring, as well as a discussion of the potential barriers for implementation.
Glucose Monitoring and Glycemic Targets in the ICU
Glycemic targets for ICU patients have been debated for the last 2 decades. The initial findings by Dr. Van den Berghe and her colleagues from the University Hospital of Leuven in Belgium that showed a reduction in mortality in surgical ICU patients with intensive insulin treatment revolutionized critical care practice. The authors reported an impressive survival advantage (32% reduction, after adjustment for repeated interim analyses) among patients treated to a euglycemia target (BG 80-110 mg/dL (4.4-6.1 mmol/L), compared to those treated to a conventional target (CII started for BG >215 mg/dL (>12 mmol/L) with a CII target of 180-200 mg/dL [10.0-11.1 mmol/L]). The incidence of severe hypoglycemia (<40 mg/dL [<2.2 mmol/L]) was 5% (39/765) with intensive insulin therapy vs <1% (6/783) with conventional control. 1
The findings from the single-center Leuven trial motivated the design of additional studies aiming at reproducing such findings, with some studies stopped due to unacceptable risk of hypoglycemia. The NICE SUGAR trial, the largest multinational trial (enrolling mostly medical ICU patients) comparing intensive insulin therapy (target: 81 to 108 mg/dL [4.5 to 6.0 mmol/L]) vs a conventional target (<180 mg/dL [10 mmol/L]) showed, however, an unexpected increase in mortality with intensive insulin therapy. The treatment effect difference in mean glucose was narrower in this study (115 ± 18 vs. 144 ± 23 mg/dL [6.4 ± 1 vs. 8 ± 1.2 mmol/L]) and the incidence of severe hypoglycemia (<40 mg/dL (2.2 mmol/L), was slightly higher (6.8% vs 0.5%) than the Leuven trial. Differences in targets, types of infusion-pumps, sampling sites (capillary vs intravenous), accuracies of glucometers, and nutritional strategies were suggested as potential explanations for the discrepancy between NICE SUGAR and the Leuven trial results. 3 Since the NICE SUGAR trial, there has been a general agreement in targeting glucose levels between 140 and 180 mg/dL (7.8-10 mmol/L) in the ICU with consideration of lower targets (110-140 mg/dL [6.1-7.8 mmol/L]) for surgical patients. There is, however, lack of support for “euglycemia targets” (BG <110 mg/dL [6.1 mmol/L]). 8 The Critical Care Society recommends [quality of evidence: very low] that a BG ≥ 150 mg/dL (8.3 mmol/L) should trigger initiation of insulin therapy, titrated to keep BG < 150 mg/dL (8.3 mmol/L) for most adults ICU patients and to maintain BG values absolutely <180 mg/dL (10 mmol/L) using a protocol that achieves a low rate of hypoglycemia (BG ≤ 70 mg/dL [3.9 mmol/L]). 9
The elimination of iatrogenic hypoglycemia as a confounding factor of the potential treatment effect of intensive glucose control remains an important gap in knowledge where the use of technology may have a role. The availability of glucose levels every 1 to 5 minutes, data trends, algorithms to assess the rate of change in glucose values, alarms for prevention of hypoglycemia, and remote monitoring provide an unprecedented opportunity to design and examine protocols that could finally determine the best treatment targets in the ICU.
In 2010, Holzinger et al reported a significant reduction of severe hypoglycemia (1.6 vs. 11.5%) in critically ill patients (N = 124) using the Real-Time CGM device (discontinued) vs standard of care. Continuous glucose monitoring data were used at least every 2 hours to guide insulin infusion adjustments with glucose target goal of 80 to 110 mg/dL (4.4-6.1 mmol/L). 10 In addition, circulatory shock requiring norepinephrine therapy had no influence on the accuracy and reliability of the device. 11 In 2014, Boom et al tested the safety, efficacy, workload, and costs of using the FreeStyle Navigator device (discontinued) compared to standard of care. The investigators used a computerized algorithm for insulin dosing based on CGM data in the intervention group, targeting glucose levels between 90 and 162 mg/dL (5-10 mmol/L). Alarms were set at 90 mg/dL (5 mmol/L) and hypoglycemia defined as <40 mg/dL (2.2 mmol/L), and values were entered in the algorithm for insulin adjustment. The system required multiple calibrations during the day. The authors reported non-inferiority in glycemic control using CGM vs POC, with a potential reduction in nursing workload and costs related to glycemic control. No episodes of severe hypoglycemia were observed in either group, likely due to the higher target range and the use of a computerized algorithm. 12
Agus et al, randomized 980 pediatric patients to intensive (target 80-110 mg/dL [4.4-6.1 mmol/L]) versus standard care to assess the impact of glycemic control on clinical outcomes. The authors used CGM (Guardian REAL-TIME, discontinued) to prevent and detect severe hypoglycemic episodes early, but no decisions regarding insulin dosing or glucose rescue were made based on CGM. 13 The incidence of severe hypoglycemia (<40 mg/dL [2.2 mmol/L]) was 3% in the intensive group (16 of 490 patients) compared with 1% in the standard-care (5 of 490 patients) (P = .03). The overall rate of hypoglycemia (<60 mg/dL [3.3 mmol/L]) was 19% versus 9% (P < .001). 13 No differences in health-care-related infections (primary endpoint), mortality, length of stay, or organ failure, were observed. Although the relevance of achieved glucose targets (and difference between groups in a narrow range in patients without diabetes) is uncertain, the findings of a sub-analysis of this trial suggest severe hypoglycemia may be prevented with protocols using alarms based on the rate of glucose change, or can be treated early to reduce their duration, particularly when using higher alarm thresholds. 14 The multiple calibrations required during the protocol make the specific approach less attractive for inpatient use. An additional randomized control trial (RCT), by Agus et al, using CGM (G4 Platinum, Guardian REALTime, and Enlite) to signal impending hypoglycemia, was stopped due to futility and potential harm with a lower glycemic target, with an unexpected increase in infectious outcomes and higher risk of severe hypoglycemia (5.2% vs 2.%, P = .03). 15
More recently, Beardsall et al, compared the use of real-time CGM to optimize nutritional delivery (dextrose infusion) and guidance on using insulin delivery in preterm infants to maintain glucose levels between 2.6 and 10 mmol/L (46.8-180 mg/dL) vs standard of care. Three endpoints of equal importance were examined (time in the range between 2.6 and 10 mmol/L (46.8-180 mg/dL) and between 4 and 8 mmol/L (72-144 mg/dL), and mean sensor glucose concentration). Overall, CGM use reduced exposure to prolonged or severe hyperglycemia and hypoglycemia in this pediatric population. 16
In summary, studies using CGM appear to show the use of this technology can reduce the incidence of severe hypoglycemia when targeting lower glucose levels and may be associated with reduced costs. While we wait for additional clinical trial data (with a clinically relevant separation in glycemic targets), it appears reasonable to maintain a glucose target of 140 to 180 mg/dL (7.8-10 mmol/L) for most patients (for insulin adjustment) and to use time-in range as a metric to evaluate overall glycemic control. Considering higher thresholds for hypoglycemia prevention (eg, 85 mg/dL (4.7 mmol/L) in the non-ICU 17 and perhaps at <100 mg/dL [5.5 mmol/L] in the ICU)18 -20 may be more appropriate as there is not clear evidence yet that targeting “euglycemia” improves clinical outcomes in children or adults.
Previous and Current CGM Technologies
In 1999, the FDA approved the first CGM system for commercialization by Medtronic. 21 Today, there is a wide variety of CGM devices available in the market. There are intravascular sensors (IV-CGM), subcutaneous (SC-CGM), and transdermal CGM sensors.
In the United States, the use of CGM systems in the ICU is infrequent. The GlucoScout and OptiScanner 5000 are IV-CGM approved by FDA but are not commonly used. None of the SC-CGM have yet been approved for inpatient monitoring in the United States. Currently, the only CGM device clinically approved in Europe, for use in the ICU is the IVBG System. OptiScanner 5000 and Sentrino are also approved but for use in non-critically ill patients in Europe. 22
Newer devices have innovative features that make them more reliable, accurate, and easier to use compared with older devices. A non-exhaustive list of CGM devices examined in the last 2 decades is included in Table 1. Older CGM devices needed frequent calibration with fingerstick glucose measurements to have accurate readings. In recent years, newer CGM systems have been paired with smartphones via mobile apps that facilitate glucose monitoring, visualization of current trends, and arrows that depict the rate of glucose change, access summarized information, and data sharing.23,24 Table 2 lists characteristics of currently available subcutaneous CGM devices and their limitations.
CGM Devices, Manufacturers, Regulatory Status, and Availability.
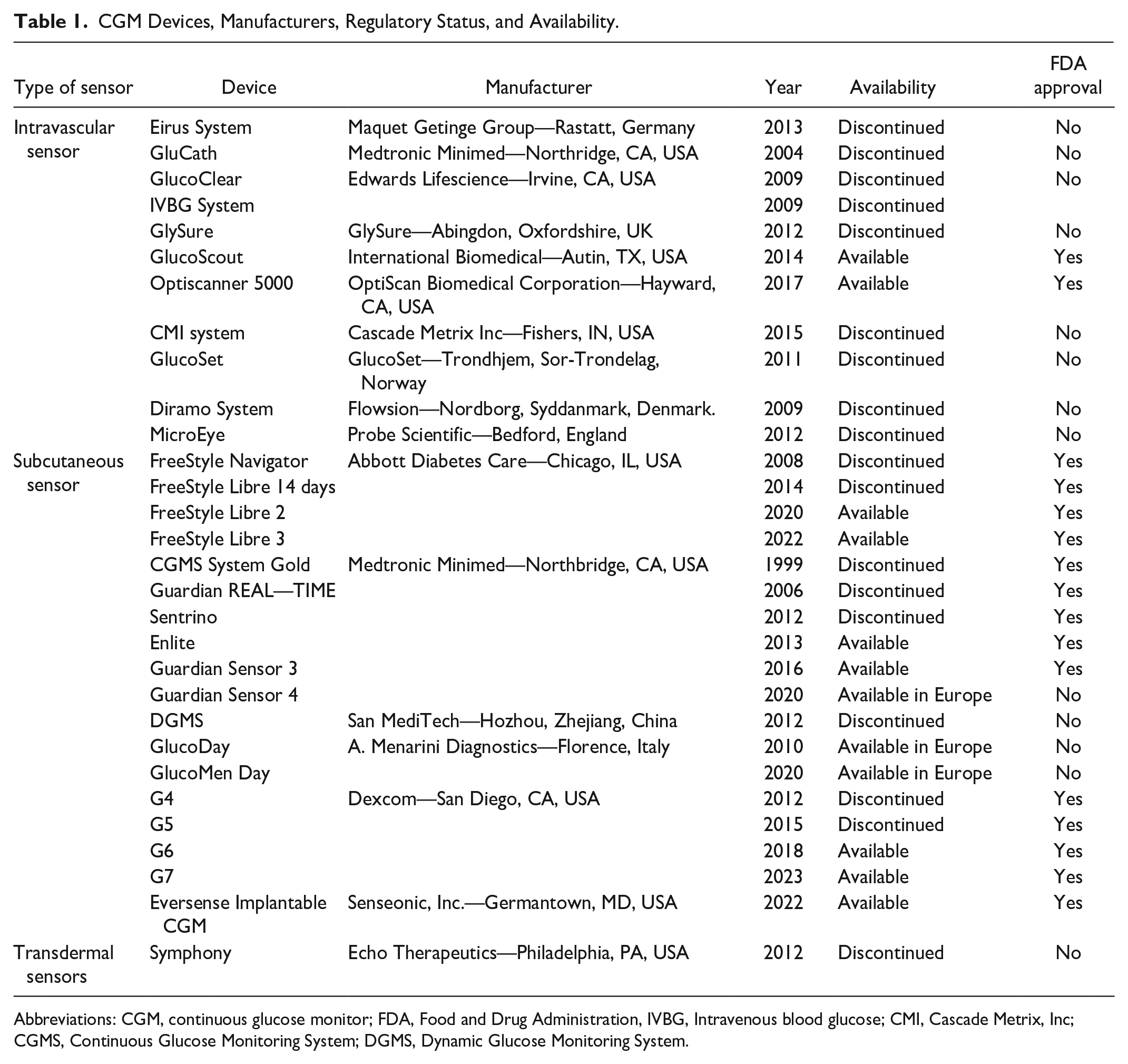
Abbreviations: CGM, continuous glucose monitor; FDA, Food and Drug Administration, IVBG, Intravenous blood glucose; CMI, Cascade Metrix, Inc; CGMS, Continuous Glucose Monitoring System; DGMS, Dynamic Glucose Monitoring System.
Characteristics of Currently Available Subcutaneous CGM Devices and Their Limitations.
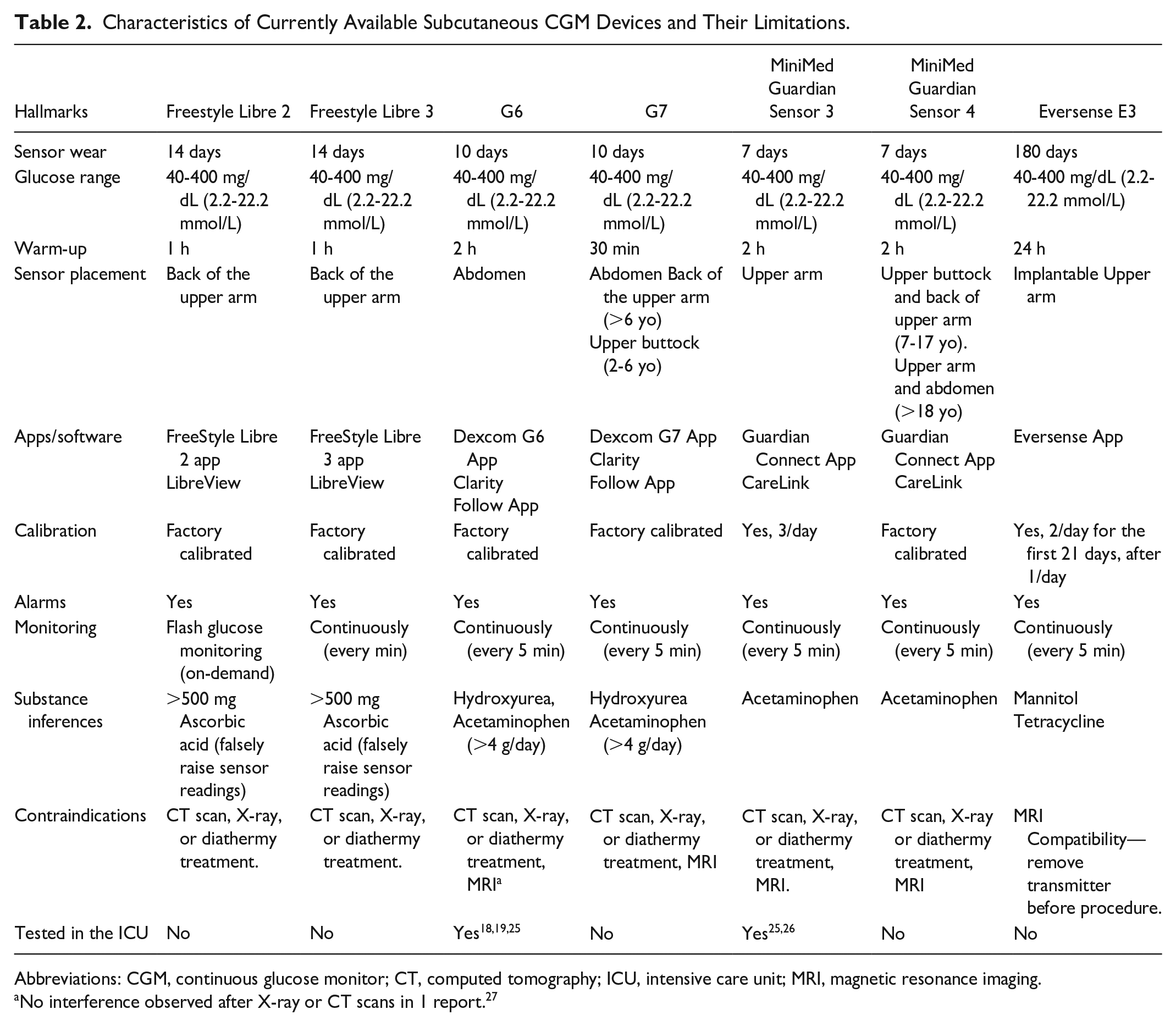
Abbreviations: CGM, continuous glucose monitor; CT, computed tomography; ICU, intensive care unit; MRI, magnetic resonance imaging.
No interference observed after X-ray or CT scans in 1 report. 27
Accuracy
The accuracy and reliability of CGM in the critical care setting has been a focus of many CGM studies. Understanding the functionality and limitations of CGM during the care of critically ill patients is a priority, as misleading glucose values could lead to inadequate or inappropriate treatment and potentially devastating consequences. Previous studies evaluating CGM use in ICU populations have provided information on accuracy metrics, most commonly reporting the mean or median absolute relative difference (MARD or MedARD) between sensor and reference glucose values, as well as error grid data (ie, Clarke or surveillance) for assessment of clinical reliability. Although there have been a small number of studies evaluating the accuracy and feasibility of IV-CGM,28 -31 most studies have evaluated SC-CGM devices given higher rates of complications (eg, thrombus formation, infection) associated with IV-CGM use. 32
Previous studies using subcutaneous CGM devices have shown differences in accuracy, with reported MARD or MedARD values ranging from 6.6% to 15.6%.12,33,34 These differences may be explained in part by variations in clinical factors, frequency of CGM calibration and type of reference glucose measurement used for comparison. Despite the variability in accuracy metrics, error grid analyses in many of these studies indicated good correlation between sensor and reference glucose pairs with ≥98% of values falling within acceptable treatment zones for clinical management.11,33 Currently, definitive accuracy studies comparing a gold standard method with available SC-CGM devices have not yet been completed. However, utilization of newer CGM devices for clinical care during the COVID-19 pandemic has provided additional accuracy data and further insight into the feasibility of CGM use in critical illness. Accuracy analyses using the Dexcom G6 in critically ill patients with COVID-19 reported MARD values of 11.1% 19 and 12.6%, 19 again with ≥98% of paired glucose values falling in Clarke Error Grid zones A and B.19,25 Although recent data continue to support the potential for CGM implementation in ICU practice, it highlights the need for ongoing assessment of sensor function in a setting where acute clinical changes may impact device reliability.
Randomized Clinical Trials Using CGM in the ICU
We reviewed systematically PubMed to identify randomized clinical trials testing the impact of CGM in the ICU on glycemic metrics. Using the following search strategy: ((((continuous glucose monitoring) OR (CGM)) AND (((critically ill) OR (intensive care unit)) OR (ICU))) AND ((((hyperglycemia) OR (glycemic control)) OR (hypoglycemia)) OR (diabetes))) AND (randomized clinical trial) we identified 54 articles (last search conducted on September 25, 2022). Out of these 54 articles, 16 were confirmed to be randomized clinical trials using GCM in the ICU. In general, studies mostly showed non-inferiority compared to standard monitoring, with several studies showing a reduction in the incidence of hypoglycemia. A summary of these studies is listed in Table 3. Additional descriptions of key studies are discussed above in the Glucose Monitoring and Glycemic Targets in the ICU section.
Clinical Trials Testing the Impact of CGM on Glycemic or Clinical Endpoints in the ICU.
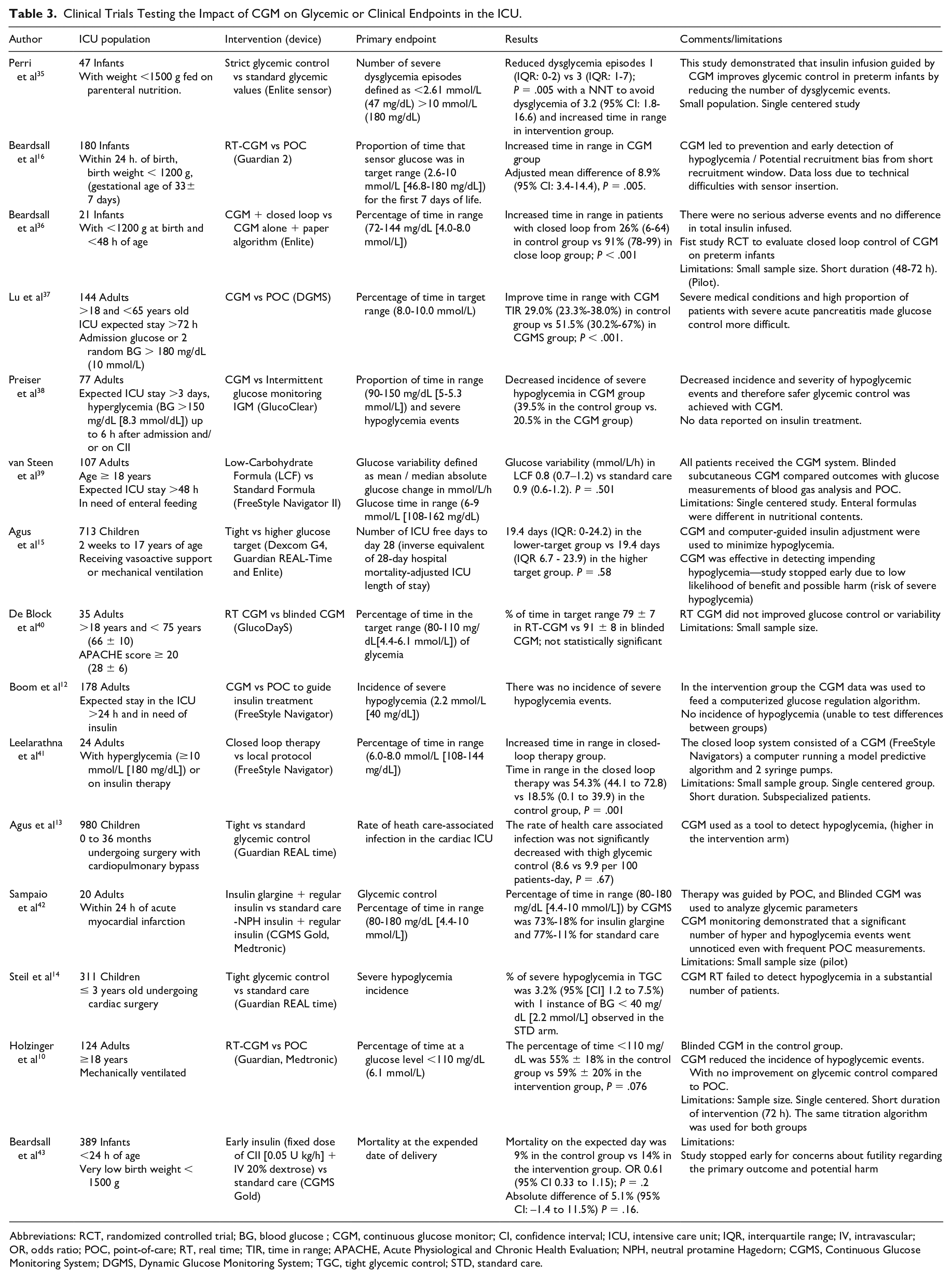
Abbreviations: RCT, randomized controlled trial; BG, blood glucose ; CGM, continuous glucose monitor; CI, confidence interval; ICU, intensive care unit; IQR, interquartile range; IV, intravascular; OR, odds ratio; POC, point-of-care; RT, real time; TIR, time in range; APACHE, Acute Physiological and Chronic Health Evaluation; NPH, neutral protamine Hagedorn; CGMS, Continuous Glucose Monitoring System; DGMS, Dynamic Glucose Monitoring System; TGC, tight glycemic control; STD, standard care.
Real World Experience During Covid-19
Early during the COVID-19 pandemic, it was clear that (1) patients with hyperglycemia/diabetes had worse outcomes and (2) there was an urgent need to find alternative strategies to monitor and treat patients with diabetes to avoid the risk of infection of healthcare workers and improve glycemic control. In April 2020, the FDA announced that they would not object to the use of non-invasive remote CGM to aid glucose monitoring during the pandemic. 2 To reduce HCP exposure to the virus, personal protective equipment (PPE) waste, and reduced staff workload multiple centers implemented the use of remote CGM in the ICU. Several studies emerged to describe the experience gained with this technology while caring for critically ill patients with COVID-19.18 -20,25,44 Studies consistently reported the feasibility of CGM use in this setting aimed at reducing the burden of critical care nurses caring for patients with COVID-19. Sadhu et al, 25 Agarwal et al, 19 Davis et al, 18 and Faulds et al 20 (except for Sadhu et al that used G6 and Guardian Connect, all other groups used a G6 CGM device only) reported an estimated reduction of POC testing of 33.1%, 60%, 68%, and 71%, respectively.
The use of remote CGM has emerged as a promising strategy to monitor patients remotely and to guide insulin infusion, particularly in extreme situations as experienced early during the COVID-19 pandemic. Additional research is needed to confirm the safety of such approaches and the impact on additional metrics, including cost-effectiveness, staff burden, and workload.19,20,25
Implementation of CGM in the ICU, Role of Hybrid Protocols
The COVID-19 pandemic created a need for rapid deployment of traditionally outpatient CGM systems in a novel inpatient environment. While the FDA provisional allowance opened the possibility of using CGM in the inpatient setting, there still existed concerns surrounding safety and implementation. Health systems worked to craft protocols that would help mitigate safety concerns and facilitate CGM use among nursing staff. Hybrid protocols combining intermittent POC glucose testing with CGM were widely used during the COVID-19 pandemic. Most protocols required more stringent initial sensor-POC pair validation before allowing intermittent non-adjunctive CGM (ie, use of CGM to make therapeutic decisions without the need of confirmatory POC testing) with less frequent ongoing POC testing and validation. Such protocols helped ensure a degree of accuracy while still facilitating reduction in required frequency of POC testing. Table 4 describes key elements of COVID-19 CGM protocols with the majority using a threshold of 20% (for glucoses >100 mg/dL [5.5 mmol/L]) or 20 mg/dL (1.1 mmol/L) (for glucose <100 mg/dL [5.5 mmol/L]) for validation.18 -20,45 Five of the 6 studies described more frequent POC testing after initial placement18,20,25,44,45 and in 3 protocols the CGM was in fact not used until 24 hours after placement.25,44,45 Figure 1 shows a summary of combined experience gained at The Ohio State University and Grady/Emory University with currently available technologies. Using a real-time CGM device, a smartphone with mobile apps can be located outside of the patient room. Data can also be shared at the nursing station and with ICU or diabetes team staff members (via an app for data sharing). Healthcare provider can customize real-time alarms (for low or high glucose or signal loss). Additional steps for implementation have been previously described in more detail.2,20,46
Hybrid CGM + POC Protocols Used During the COVID-19 Pandemic
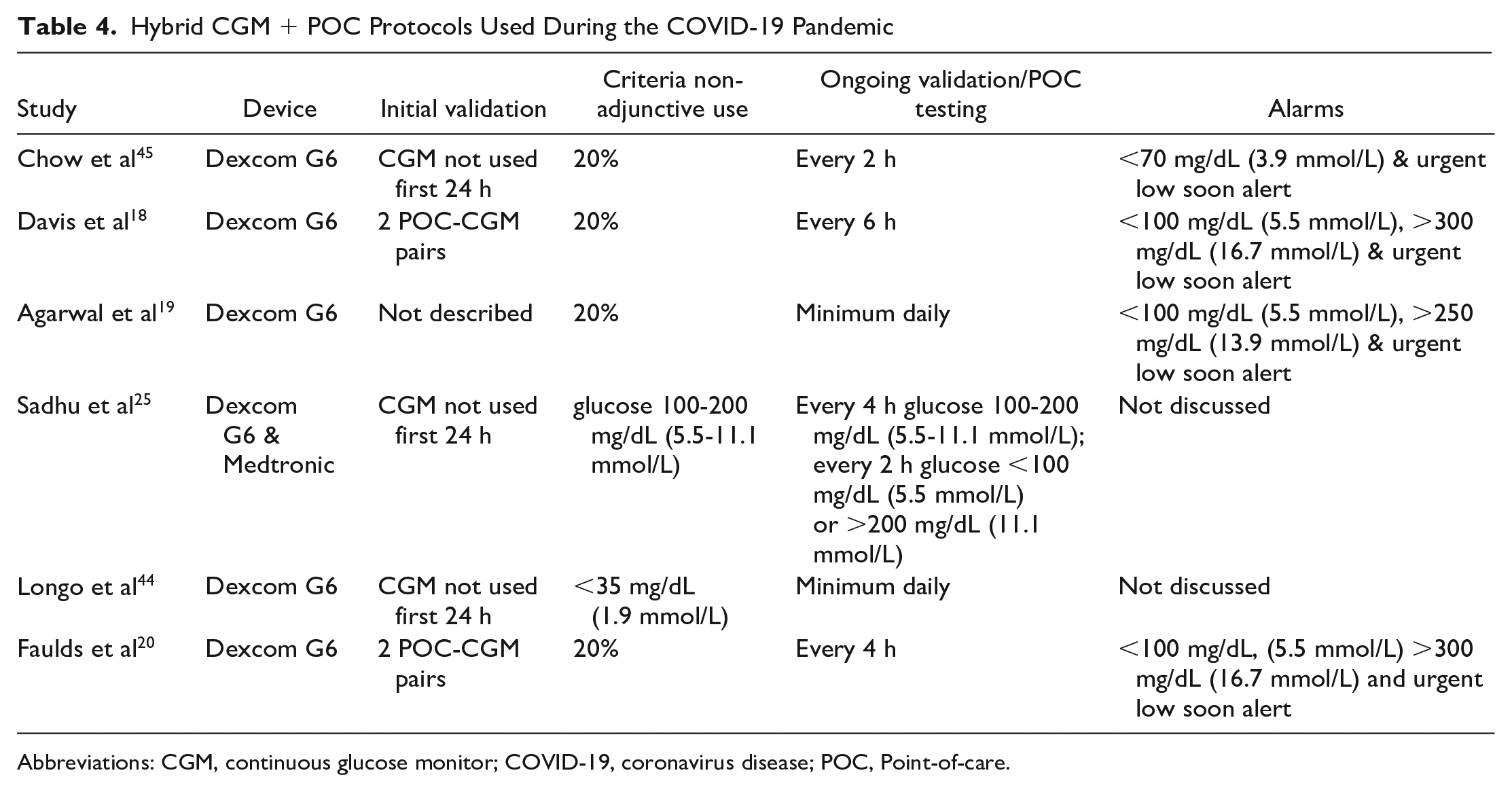
Abbreviations: CGM, continuous glucose monitor; COVID-19, coronavirus disease; POC, Point-of-care.
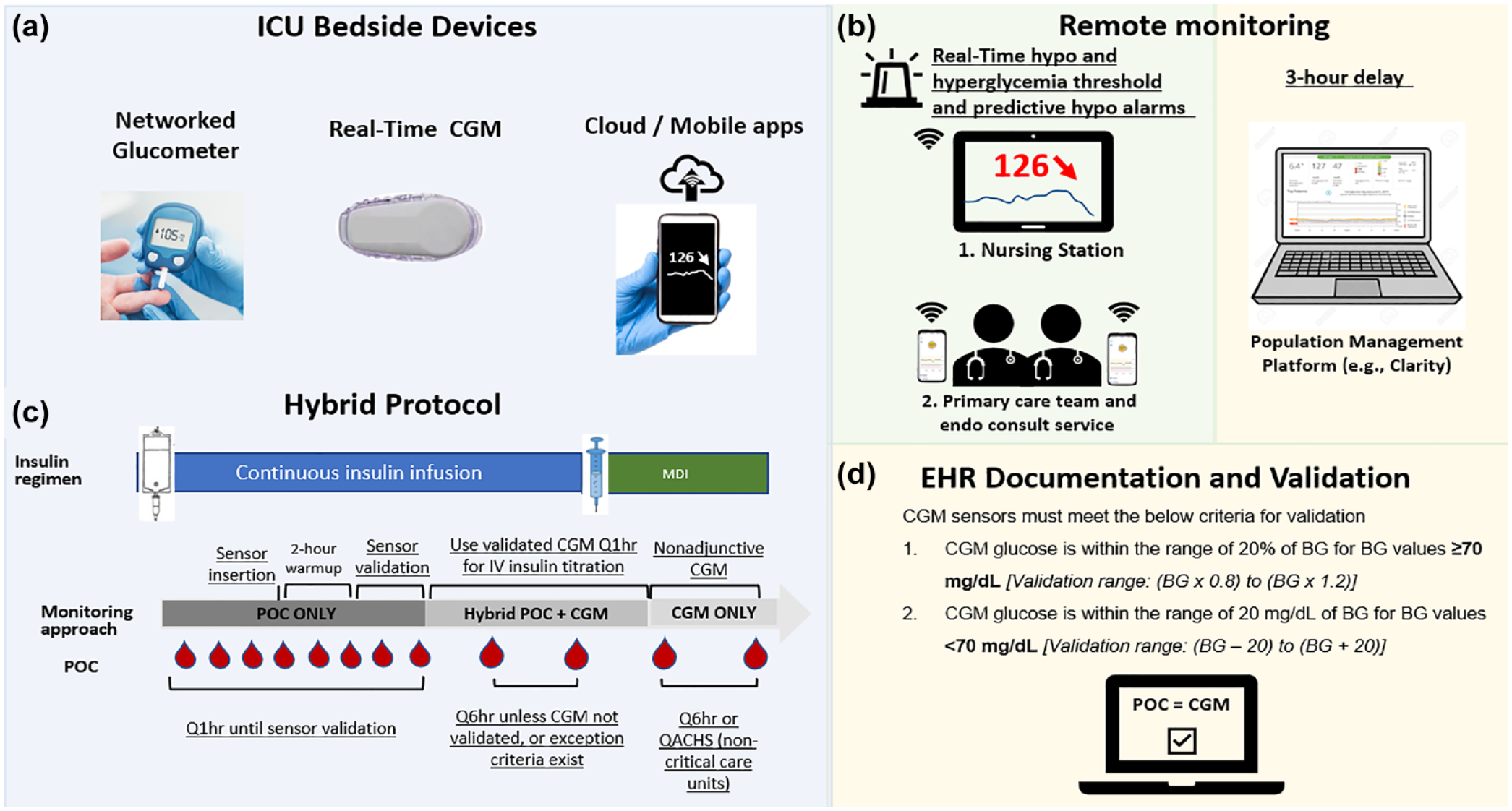
Implementation of CGM in the ICU Setting. Based on our recent experience, 4 main components are useful for implementation in the ICU including: (a) Devices for real-time CGM used in the ICU. (b) Protocol for hybrid monitoring: before sensors are validated POC test are conducted hourly to guide CII. Once initial sensor readings are available, they can be compared with a simultaneous POC test to determine accuracy and decision to initiate CGM guided insulin titration. A confirmatory validation can be performed 1 hour after the first POC-CGM comparison. Every 4 to 6 hours confirmation of sensor accuracy is recommended. After discontinuation of CII nonadjunctive CGM can be used for insulin management with multiple daily injections (with periodic validation, eg, QACHS or Q6h). (c) Data sharing for real-time remote monitoring (eg, nursing station, primary team) and download of summary data for interpretation (eg, population management platform). (d) EHR documentation and periodic validation. Abbreviations: CGM: Continuous glucose monitoring; CII: continuous intravenous insulin; EHR: Electronic Health Record; ICU, intensive care unit; MDI: multiple daily injections; POC: point of care; Q1hr: every hour; Q6hr: every 6 hours; QACHS: before meals and at bedtime.
Beyond Glycemic Control
The use of CGM certainly holds the potential to improve outcomes beyond glycemic control including reducing nursing burden, increasing nursing satisfaction, improving health system workflows, decreasing patient discomfort, and subsequently improving patient satisfaction. The current standard of care POC glucose testing can be a burdensome task for nurses and nursing support personnel. Boom conducted a time-in-motion analysis and found that each POC test took approximately 3 minutes to perform and the use of CGM significantly reduced nursing workload by a total of 19 minutes in 24 hours. This reduction in nursing personnel cost contributed to a significant reduction in hospital costs associated with CGM compared to traditional POC testing. 12 While there are few studies examining nursing perspectives on the implementation of CGM, Faulds et al found a high degree of acceptance and a high degree of perceived accuracy among nurses 20 and Chow et al reported that 63% of nurses believed CGM systems improved the care they provided their patients. 45 Even though frequency of POC was not significantly reduced overall, due to the nature of the protocol.
While improvement in glycemia associated with CGM could have huge impact on patient clinical outcomes, there are additional potential patient benefits for consideration. Point of care glucose monitoring is a painful procedure for patients and can disrupt a patient’s sleep and recovery especially when performed hourly in association with CII. The longer wear time of a CGM systems means patients may only need to have a sensor insertion performed once over the course of a hospitalization. Moreover, CGM results in minimal blood loss, particularly in comparison to use of arterial or venous samples. These and other outcomes beyond glycemia should be studied and incorporated into clinical trials whose primary emphasis is glycemic control.
Limitations and Barriers to Implementation
The use of CGM in the ICU may assist glycemic control by providing more information compared to intermittent POC. However, there are limitations to this technology that are important to consider, especially in the ICU. Critically ill patients frequently require vasopressors, present with hypoxia or hypothermia, are exposed to multiple medications and intravenous solutions, have variable parental/enteral nutrition support, or require unexpected procedures. Factors that cause a rapid rate of change in glucose may magnify any potential discrepancy between interstitial and blood glucose measurements. Limitations include (1) circumstances that affect accuracy, and (2) implementation barriers, resources, documentation, and costs.
Hypoperfusion may produce falsely low readings and the mechanical compression of the sensor can result in signal loss. 18 However, the accuracy of devices appears to be preserved among patients receiving vasopressors.11,47 Medications with substances interfering with sensor readings may produce falsely elevated or decreased glucose values (summary in Table 2). Patients admitted to the ICU require multiple radiology procedures, but information about their potential effects on sensor accuracy is limited. In a small observational study, the accuracy of the device after CT and X-rays was preserved. 27 According to device manuals specifications, devices need to be removed before magnetic resonance imaging (MRI). 48 However, Freestyle 2 and 3 maintain functionality and data integrity after MRI, as well as the G6. 49 During the perioperative period, a few studies have provided good accuracy and no interference with electrocautery/diathermia. 50 However, for patients undergoing Coronary Artery Bypass Graft (CABG), the CGM devices were associated with intermittent interference and potentially decreased accuracy after the procedure. 47 Patients’ safety and well-being are priorities. Therefore, further study of this potential limitation is needed.
There are also several barriers to implementation, including the need for approval and collaboration with multiple stakeholders, which may differ by institution. Buy-in from the end-users (nursing staff) is essential to ensure adoption of CGM into the clinical workflow in order to effectively reduce the burden associated with hourly glucose monitoring. 51 In addition, due to high nursing staff turnovers, identifying “nurse champions” that can help new staff with training/troubleshooting will promote continued use of the technology. Developing a training curriculum and additional tools (summary sheets, presentations, videos, remote support) may also facilitate the training process and sustained effective CGM use.
Due to potential known and unknown CGM interferences, documentation of sensor accuracy provides another layer of safety when using this technology in the hospital. It is critical to have procedures in place to track inaccuracy events, decisions for sensor replacement, or discontinuation of CGM therapy. Involvement of information technology services early during the implementation process may help facilitate the implementation of a system for proper tracking of CGM use in the hospital. 18 Recent efforts are focusing on the creation of data standards and workflow guidance for integration of CGM data in the electronic health record (EHR). 52
Future Directions
The use of CGM is changing the life of patients living with diabetes. In the outpatient setting, this tool helps to monitor glucose levels without the need for fingersticks, provides trends to predict changes in glucose, alarms for prevention, and detection of severe hypo or hyperglycemia, can promote behavior change, and is widely accepted by users. In the ICU, this technology thus far has been useful to reduce the burden of nurses taking care of ICU patients while allowing the continued use of the standard of care therapy during COVID-19. Studies conducted in ICU populations show this technology may significantly reduce the time spent in hypoglycemia. The use of alarms and visualization of trends can both help prevent the incidence or reduce the time spent in hypoglycemia that already occurred. 53 In addition, the information can help monitor the recovery after a hypoglycemic episode and prevent recurrence in high-risk patients.
With the expanding knowledge about the safe and helpful use of technology, we are hopeful the path for regulatory approval will become more efficient. These technologies continue to advance rapidly for outpatient use and translation of each new device is not a viable path for inpatient use of CGM.
The fact that the technology provides continuous data with visualization of trends as well as alarms for hypoglycemia prevention and early detection, suggests the criteria for accuracy should not be as stringent as it currently is for POC. This is also relevant when POC is used as reference, as single POC values may significantly differ from interstitial glucose levels during a rapid rate of glucose change.
Current approaches with hourly fingersticks are not just a significant burden to ICU staff but are suboptimal compared to the wealth of data and mitigation elements (ie, alarms, trends, digital sharing) provided by CGM devices. Once the accuracy of devices designed for the hospital is established for the ICU, electronic health records (EHR) integration and protocols for CGM use (ie, hybrid protocols) are proved to be effective, this technology has the potential to transform ICU glycemic care. The available and emerging data and experience gained with CGM in the ICU suggest this technology should at least be approved as adjunctive to POC in validated hybrid protocols until more definitive technology is available and ready to replace inconvenient hourly fingesticks.
Footnotes
Authors’ Contributions
L.G.A., E.F., and M.C.P.G. prepared the tables and wrote the first draft. G.M.D. and K.D. critically reviewed and edited the manuscript. E.F. and F.J.P. prepared the Figure. F.J.P. conceptualized the approach and wrote and edited different sections of the manuscript. All authors reviewed and approved the manuscript.
Abbreviations
ICU, intensive care unit; HCP, healthcare providers; POC, point-of-care; CGM, continuous glucose monitoring; CII, continuous intravenous insulin; FDA, Food and Drug Administration; MARD or MedARD, mean or median absolute relative difference; IV-CGM, intravascular CGM; SC-CGM, subcutaneous CGM; CABG, coronary artery bypass graft; EHR, electronic health record.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EF discloses research support with Dexcom Inc. KD discloses research support from Novo Nordisk, Sanofi, Viacyte, Abbott; consulting with Eli Lilly, Novo Nordisk, Boehringer-Ingelheim, and Tolerion; and honorarium from UptoDate, Medscape, and Elsevier. GD discloses research support from Insulet and honorarium from Medscape. FJP discloses unrestricted research support from Insulet, Dexcom and Ideal Medical Technologies and personal fees from Boehringer Ingelheim, Dexcom, and Medscape for consulting activities.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
