Abstract
Background:
In critically ill patients, deviations in glucose levels may lead to significant harm to individuals with and without diabetes. Although subcutaneous continuous glucose monitoring (scCGM) has proven beneficial for patients in standard wards, its implementation in critical care settings has been limited due to multiple factors, potentially resulting in inadequate glycemic control and consequent complications; here, intravascular systems (ivCGM) have the potential to overcome these limitations.
Method:
This single-center, open-label study, aimed to assess accuracy and safety of a novel intravenous glucose monitoring system in patients with and without diabetes, admitted to a cardiothoracic surgery intensive care unit. Glucose levels were continuously monitored for up to 72 hours in the predefined glucose range of 20 to 400 mg/dL and compared with arterial glucose measurements (blood gas analyses [BGAs]).
Results:
Twenty-eight participants successfully completed the study, allowing the collection of 1224 ivCGM/BGA data pairs. Due to the exploratory nature of the trial in this vulnerable patient population, no data pairs <70 mg/dL and limited data pairs in level 2 hyperglycemia (>250 mg/dL) were observed. A mean absolute relative difference (MARD) of 8.7 ± 7.8% was found, whereas the mean absolute difference (MAD) for values <100 mg/dL was 3.3 ± 2.7 mg/dL. In participants with diabetes (N = 8,332 ivCGM/BGA data pairs), MARD was 9.6 ± 8.1%. Diabetes Technology Society Error Grid (DTSEG) analysis revealed that all data pairs fell within clinically acceptable zones A and B. Notably, no serious adverse events associated with the device were observed during the study.
Conclusion:
The present findings indicate that the investigated intravenous glucose monitoring system provides accurate glucose monitoring and demonstrates its safety in critical care settings. This technology offers promise for improved glycemic management in critically ill patients, particularly those with diabetes, potentially mitigating the associated risks and complications.
Background
In critical care, maintaining glycemic control in patients with or without pre-diagnosed diabetes mellitus is a considerable challenge for health care professionals (HCPs). Patients who require critical care are known to develop insulin resistance and are prone to increased glucose variability (GV) and hypoglycemia, all of which are independent risk factors for mortality.1,2 Even though the optimal glycemic range for critically ill patients remains unclear and seems to differ depending on preexisting diabetes, 3 guidelines recommend a narrow range of 140 to 180 mg/dL as the glycemic target range for insulin-treated patients. 4 Maintenance of this target is hampered by various factors, including systemic inflammation, endogenous stress, hormone excess leading to insulin resistance, impaired organ perfusion, impaired kidney function, fluid shifts, parenteral nutrition, concomitant medication (eg, glucocorticoids), intravenous glucose administration, fasting periods, and, eventually, the half-life of insulin. Especially, patients with diabetes undergoing cardiac surgery may benefit from achieving glycemic targets through continuous intravenous insulin infusion rather than subcutaneous therapy.4,5 This complex therapy requires regular glucose measurements to monitor and adjust insulin infusion rates. To date, bedside point of care measurements or venous/arterial blood gas analysis (BGA) represents the standard for providing information on glycemic control in critically ill patients. Point of care measurements are often criticized for low accuracy in critically ill patients. 6 Moreover, these methods fail to provide alarms or predict hypo- or hyperglycemic events due to the limited data on glucose excursions. Elevated glucose levels are often neglected due to other critical tasks that require health care professional (HCP) attention,6-8 resulting in unmet glycemic targets. 9 To optimize glycemic control, accurate bedside measurements providing more information than intermittent glucose readings are needed. 10 Continuous glucose monitoring may enhance glycemic control. Nevertheless, available systems for subcutaneous monitoring (scCGM) are not recommended in critically ill patients. Intravascular devices may overcome the issues related to scCGM, detecting changes in glucose levels faster and providing acceptable accuracy. Although regulatory authorities approved some invasive CGM systems, their use is limited. 11
The present study evaluated a novel intravenous CGM system. As ivCGM may represent a suitable option for optimizing glycemic control in critical care while reducing the workload for HCPs, accuracy and safety of the device in a postsurgery cardiothoracic intensive care unit (ICU) were investigated.
Methods
A monocentric, open-label, single-arm, pilot study investigating the performance of GlucoMen Day ivCGM (A. Menarini Diagnostics, Florence, Italy) was conducted in patients hospitalized in a cardiothoracic surgery ICU after surgical interventions at the Department for Cardiac Surgery at the Medical University Graz. The study was approved by the local institutional review board (33-615 ex20/21) and was performed according to the International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use—Good Clinical Practice (ICH-GCP) and the Declaration of Helsinki.
Primary objective of the study was to evaluate the accuracy of the ivCGM device in measuring blood glucose, compared with the standard laboratory reference measurements. Secondary objective was to evaluate the safety of the system.
Study Participants, Study Device, and Study Procedure
According to the inclusion criteria, patients were hospitalized for cardiac surgery, aged older than 18 years; 10 out of 35 enrolled patients were people living with diabetes. Exclusion criteria included, among others, pregnancy and breastfeeding, history of bleeding disorders, hypersensitivity to fondaparinux sodium, anemia, and absence of suitable veins for intravenous catheter insertion. A detailed list of inclusion and exclusion criteria can be found in Additional file 1.
After a screening visit conducted before surgery, including physical examination and laboratory testing, eligible patients were included in the study. Patients were admitted to the ICU after cardiac surgery, where the ivCGM system was applied.
The ivCGM system is an intravascular microdialysis-based device intended to be used to continuously monitor intravenous glucose levels for up to 72 hours over the predefined range of 20 to 400 mg/dL. It comprises single-use and reusable components, as described in Figure 1 and depicted in Figure 2.
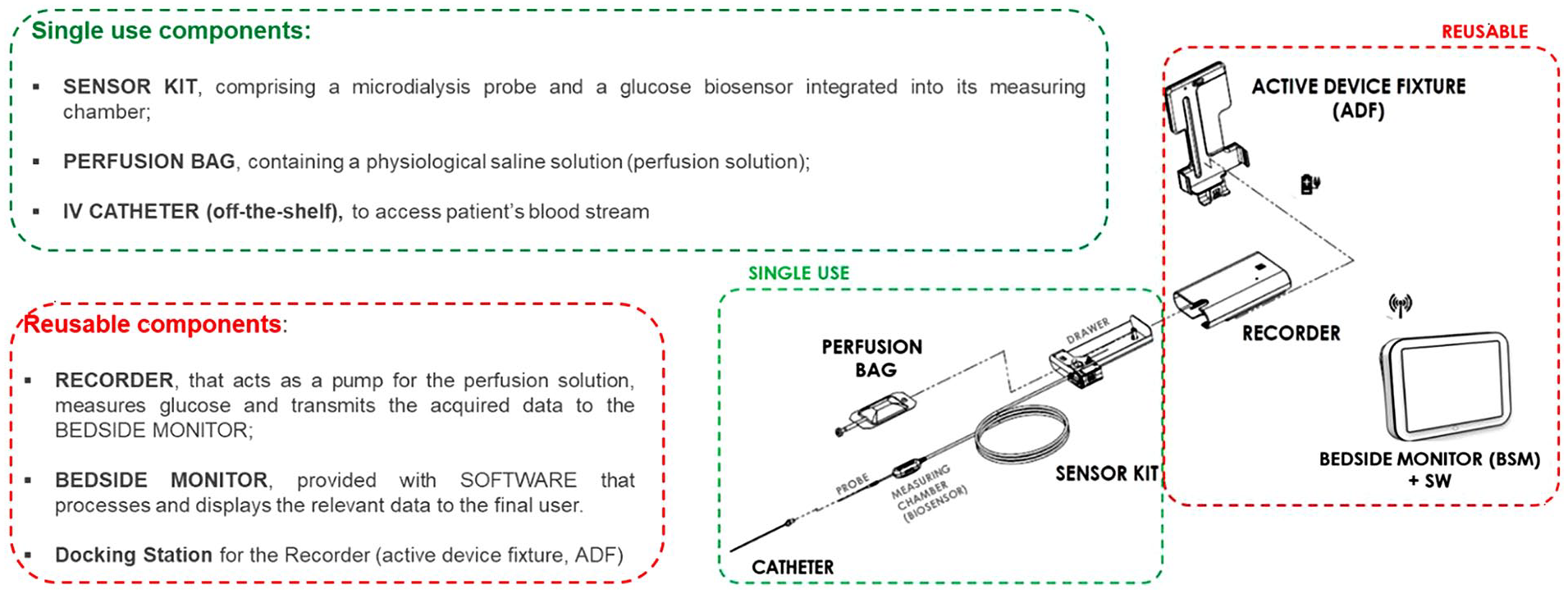
The investigated ivCGM system: device description and composition.
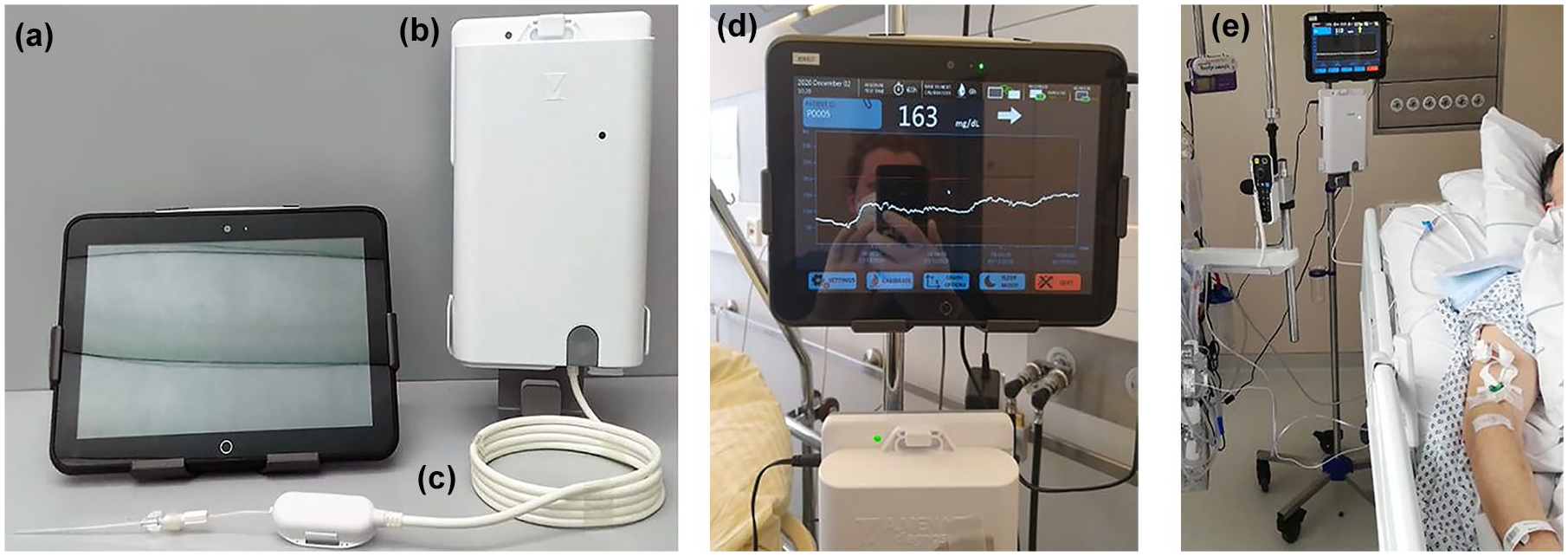
The investigated ivCGM system overview: bedside monitor (a), recorder (b), sensor kit (c), real-time glucose data display (d), ivCGM system arranged at patient bedside (e).
For all the eligible subjects, the following procedures were performed. An 18G intravenous catheter was inserted into a suitable peripheral vein of the arm as the access for the ivCGM microdialysis probe. The cephalic vein was primarily recommended, other veins were chosen at investigator’s discretion. The perfusion bag (containing a physiological saline solution injected with 20 mg of fondaparinux sodium to prevent blood clotting at the microdialysis probe) was plugged to the sensor kit drawer, which was then inserted into the recorder and the system initialized. Following the run-in phase, the probe was inserted and locked to the pre-inserted intravenous catheter and the monitoring session started. Based on the microdialysis principle, glucose molecules in the blood stream diffuse into the stream of the circulating perfusion solution to the measuring chamber where they are detected by a glucose oxidase (GOD)-based amperometric biosensor. Due to its low operating voltage, the ivCGM biosensor is not interfered by chemicals such as uric acid, adrenalin, dobutamine, levosimendan, L-DOPA, acetaminophen, and ascorbic acid; out of the tested endogenous and exogenous chemicals (45 species), only glutathione, at concentrations exceeding 1 mmol/L, slightly increased the glucose readings. The system required calibrations: at the start of the CGM session [t = 0], after the first four hours, and every eight hours thereafter. The study staff was thus periodically requested to input a reference glucose value into the bedside monitor, which then displayed the calibrated CGM profile. At the end of the monitoring session, which lasted for a maximum of 72 hours, microdialysis probe and intravenous catheter were removed from the blood vessel.
Reference measurements were obtained every 30 to 120 minutes using samples from the patients’ arterial line. Such samples were immediately processed by either ABL FLEX 90 or ABL Flex 800 Blood Gas Analyzers (Radiometer America Inc., Middleburg Heights, Ohio). Both systems were used for routine diagnostics in the cardiothoracic ICU, underwent regular routine maintenance, and were subjected to auto-calibration. The arterial line was placed according to local standards; there was no restriction on choosing a site.
During monitoring, treatment of hyperglycemia or hypoglycemia was performed as per routine ICU standards. Neither the ivCGM nor the additional BGA readings interfere with glycemic management provided by ICU staff.
A follow-up visit to investigate patients’ adverse events (AEs) related to study procedures was performed three to seven days after sensor removal.
Statistical Analysis
All statistical analyses were conducted according to the intention-to-treat principle. The primary endpoint of this study was point accuracy (ivCGM readings vs BGA arterial glucose reference measurements), evaluated as either mean absolute relative difference (MARD) (for reference measurements ≥100 mg/dL) or mean absolute difference (MAD) (for reference measurements <100 mg/dL). In addition, Diabetes Technology Society Error Grid (DTSEG) analysis was performed by pairing ivCGM and BGA results. 12 Additional statistics included the calculation of MedARD, mean amplitude of glycemic excursions (MAGE), total applicable monitoring duration, number of alarm episodes versus BGA values, time spent in the hypoglycemic range (<70 mg/dL), euglycemic range (70-180 mg/dL and 140-180 mg/dL), and hyperglycemic range (>180 mg/dL). Safety endpoints included the number of AEs and device-related adverse effects.
A sample size calculation for the primary endpoint with α = .05, a power of 90% and an effect size of 0.05 resulted in 359 test data pairs. To allow for possible dropouts and sensor failures, 35 subjects were recruited aiming for 30 complete subjects. Subjects who dropped out before completing 24 hours of monitoring were replaced. The sample size was consistent with the available literature on microdialysis-based CGM in critically ill patients or patients undergoing cardiac surgery.13,14
The time lag between ivCGM signals and reference glucose measurements, due to both perfusion solution flow rate and biosensor response time, was automatically considered by the system during calibration and is estimated to be two to three minutes, depending on the perfusion solution flow rate. During accuracy assessment, ivCGM readings were directly compared with BGA results with no time lag taken into account, reflecting the real-time CGM values displayed by the system that may be used for decisions in clinical practice.
Results
Study Population
Thirty-five patients were enrolled in the study, 29 of whom completed the study. Subject 001 was withdrawn from the analysis due to a lower default perfusion rate (ca. 12.8 µL/min) set in the device as compared to other participants, leading to inaccurate sensor readings at high glucose concentrations. From the second enrolled participant onward, the default perfusion rate was increased to 17.6 µL/min. Therefore, 28 participants were considered homogeneously treated and eligible for accuracy analysis. The patients included in the study predominantly had overt cardiovascular disease (N = 23, 82.1%) or a high risk for arteriosclerotic cardiovascular events (N = 27, 96.4%). Eight patients (28.6%) had pre-diagnosed diabetes mellitus. Seven of these received coronary artery bypass graft partly in combination with aortic valve replacement. One had transfemoral aortic valve implantation. All participants with diabetes received insulin therapy and in six intravenous dextrose was administered throughout the study. Baseline characteristics are displayed in Table 1.
Baseline Characteristics.

Collected Data
Up to 1711 reference data points, covering a test period of 1592 hours, could have been collected for the 28 patients during the prospective analysis period (Table S1, Additional file 1). Out of these theoretical values, 1658 reference data points, covering an actual test period of 1534 hours were recorded (occasionally, due to necessary medical procedures and arterial line malfunction, blood sampling for BGA measurement was not possible). Reasons for early termination of the monitoring session included patient transfer to a different ward, patient refusal to continue the study, catheter dislocation, procedural issues, or sensor kit failure. The median sensor run time was 60.2 hours (min 22.9 hours, max 72.1 hours); in participants with diabetes, the median did not differ from the overall population (60.2 hours; 22.9 hours, 72.1 hours). Out of the recorded CGM data (1525 hours), 89.3% (1361 hours) were included in the statistical analyses, representing useful ivCGM data; the difference was excluded because of system self-detection of nonphysiological variability of the signal (in which case, no CGM values were displayed to the user) and recovery periods from device-related or procedure-related system malfunctions. Out of the collected BGA reference measurements (n = 1658), 95.0% of the data (n = 1575) were included in the statistical analyses, with 1224 BGA results used for assessing the accuracy of the ivCGM system. The difference accounts for the values used for ivCGM system calibration or collected while the system malfunctioned.
Accuracy Assessment
For accuracy assessment, 1224 useful ivCGM-BGA data pairs were available.
%15/15 accuracy. 82.3% of the ivCGM results (n = 1007) fell within ±15 mg/dL of the BGA reference values at glucose levels <100 mg/dL and within ±15% of the BGA reference values at glucose levels ≥100 mg/dL.
%20/20 accuracy. 90.4% of the ivCGM results (n = 1107) fell within ±20 mg/dL of the BGA reference at glucose levels <100 mg/dL and within ±20% of the BGA references at glucose levels ≥100 mg/dL.
The aggregated overall MARD for all obtained values was 8.7 ± 7.8%. The aggregated MAD, calculated for values <100 mg/dL, was 3.3 ± 2.7 mg/dL; the aggregated MARD calculated for values ≥100 mg/dL was 8.8 ± 7.8%. When looking at elevated glucose levels, MARD was 10.3% between 180 and 250 mg/dL (N = 122), and 9.2% above 250 mg/dL (N = 7). Mean absolute relative difference and numbers of data pairs falling in the different glycemic ranges can be found in Table S2 (Additional file 1). The patient-specific MAD (<100 mg/dL), MARD (≥100 mg/dL), and overall MARD are shown in Table S3 (Additional file 1).
Similar to scCGM in patients after major cardiac surgery, the ivCGM accuracy improved throughout the 72-hour life cycle, with a trend consistent with gradual stabilization of the recovery performance by the microdialysis probe (Figure 3). 15
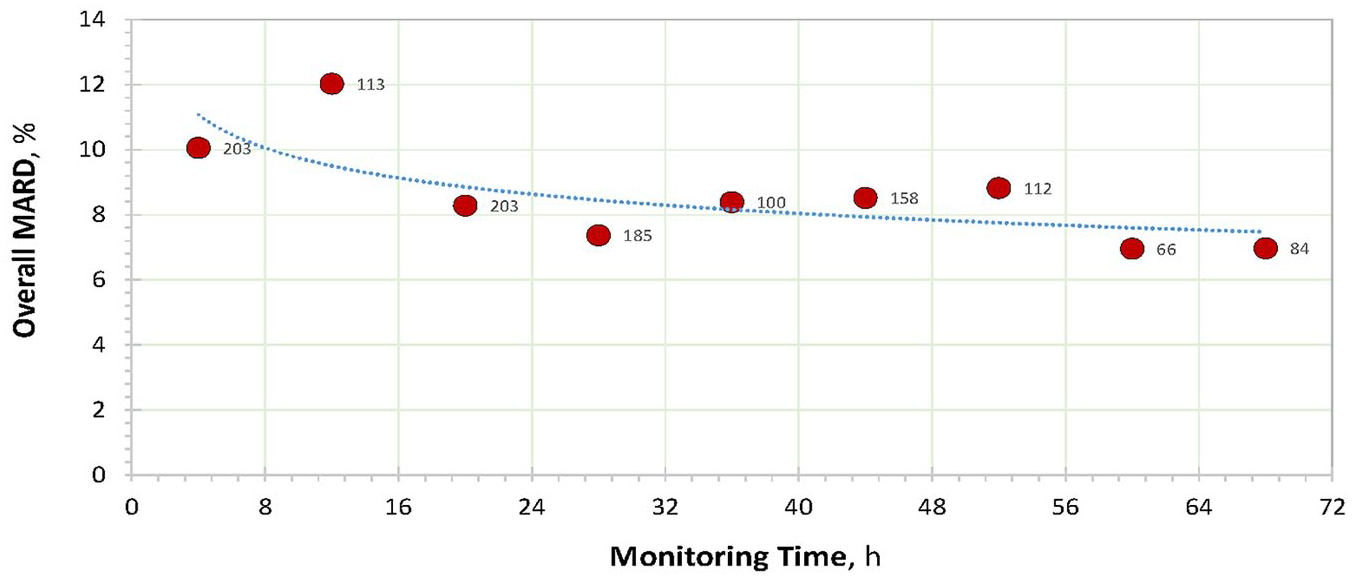
Evolution of overall MARD throughout the 72-hour monitoring period (values next to the red dots: number of data pairs collected within each 8-hour timeframe and used for MARD calculations).
The aggregated MedARD [interquartile range] for all obtained values was 6.9 [2.8-12.0]% when looking at the entire patient population (N = 28, 1224 data pairs); 7.9 [3.9-13.1]% when looking at patients with diabetes (N = 8, 332 data pairs); 6.5 [2.6-11.3]% for patients without diabetes (N = 20,892 data pairs).
According to the DTSEG analysis (Figure 4), all data pairs (n = 1224, 100%) fell within the clinically useful zones A and B, with 1105 (90.3%) in zone A and 119 (9.7%) in zone B, respectively. Figure 5 displays an example of glycemic profile (patient #5), including ivCGM readings and reference BGA measurements. All glycemic profiles are displayed in Figures S1-28 (Additional file 1).
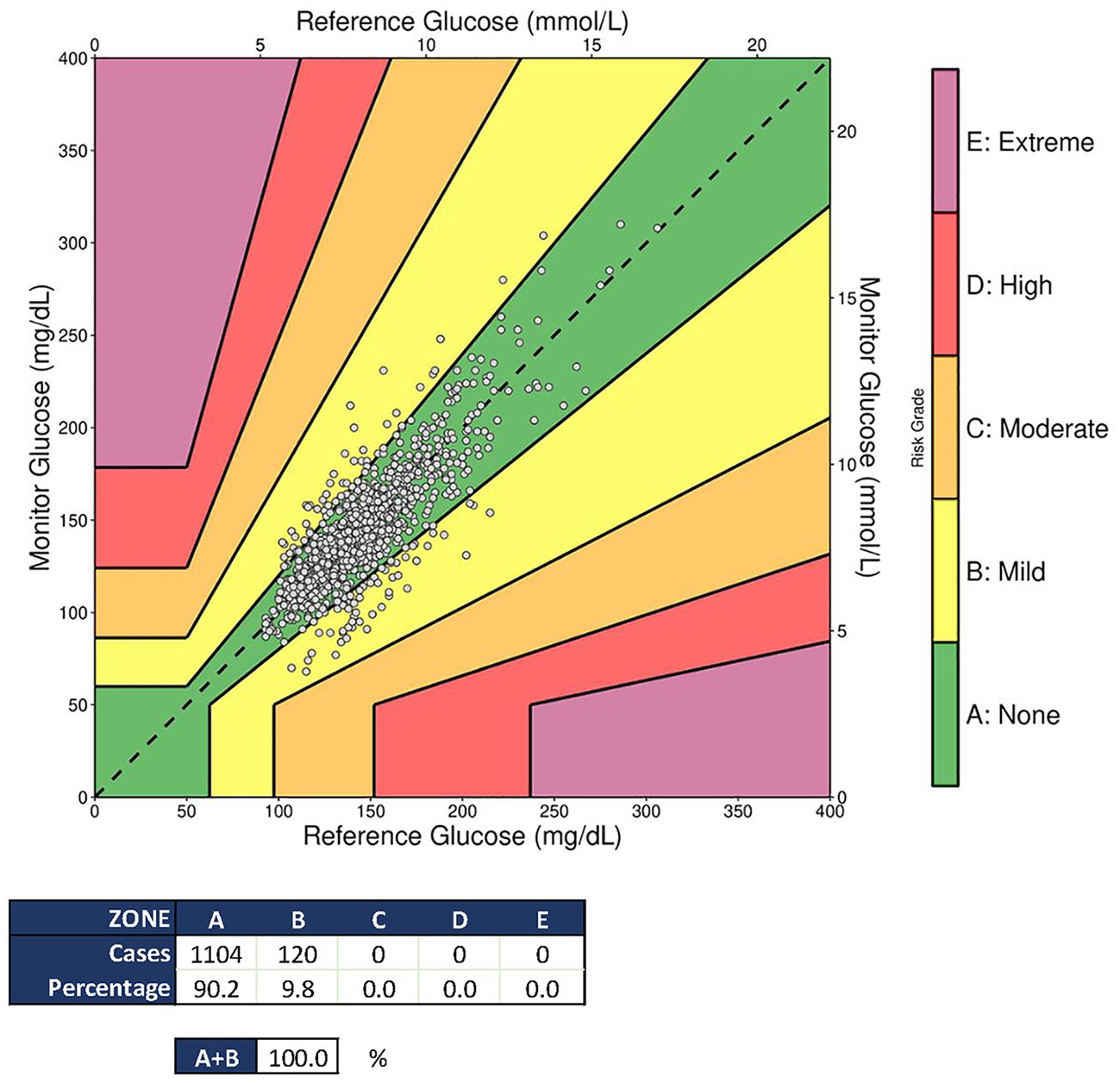
DTSEG plot of the ivCGM-BGA data pairs.
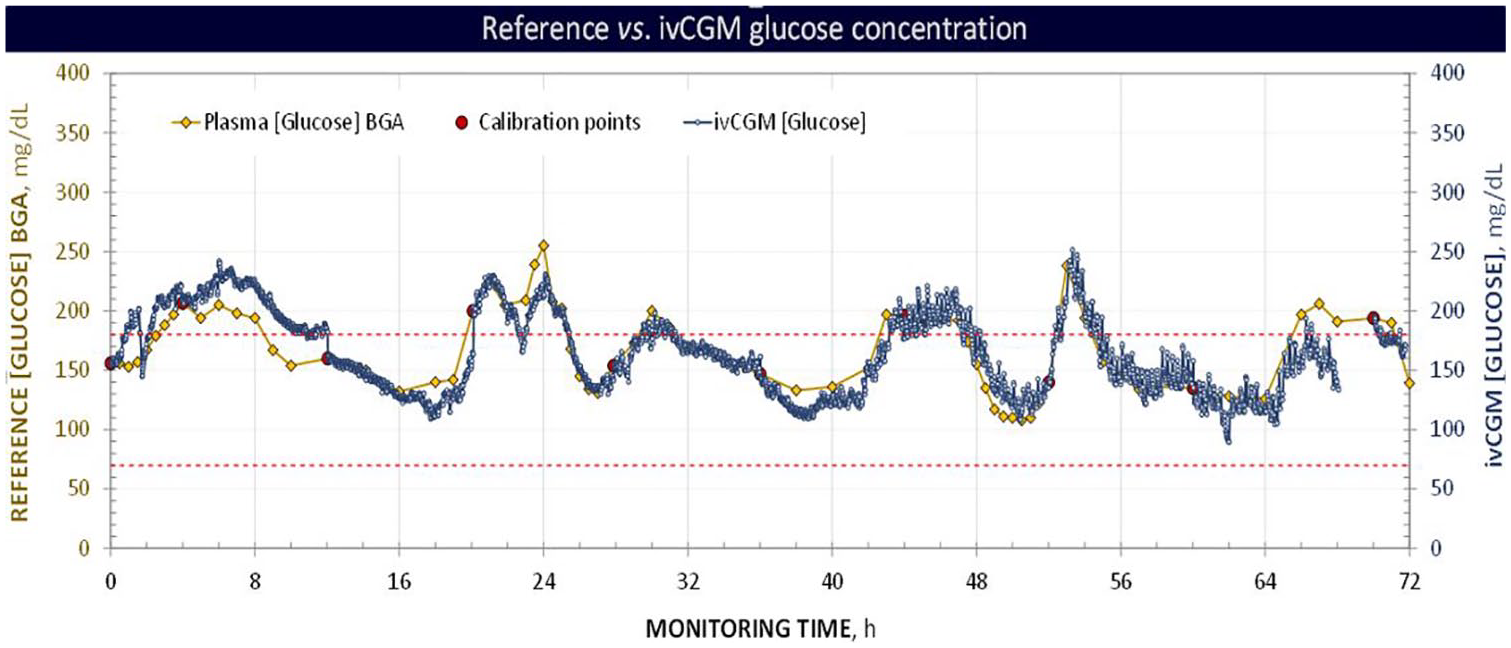
Example of glycemic profile.
Glycemic Control
Even though the ivCGM readings were not used for therapeutic decisions, understanding the glycemic excursions occurred during the monitoring period was crucial for evaluating MAD and MARD.
Glycemic control endpoints included the number of hypoglycemic (<70 mg/dL) and hyperglycemic (>180 mg/dL) events detected by either reference instrument (BGA) or ivCGM (allowed bias vs BGA measurement: ±10%). Reflecting the high number of patients with no diabetes (20 out of 28), no hypoglycemic events were observed during the study, whereas 53 hyperglycemic events were confirmed by BGA. These hyperglycemic events were correctly detected by ivCGM in 90.6% of the cases (n = 48); the remaining five events were not tracked by the system (false negatives); two false-positive hyperglycemic readings were tracked by the system.
Time in range (TIR) (70-180 mg/dL) as reported by the ivCGM was 90.6% (1234 hours), whereas TIR (140-180 mg/dL) was 34.5% (469 hours), which both reflect a population where patients without diabetes mellitus were the majority (Table S4, Additional file 1). The time spent below 70 mg/dL was 0.1% (1.5 hours), as detected by ivCGM, and time spent above 180 mg/dL was 9.3% (126 hours).
A median MAGE of 49 mg/dL (min 27 mg/dL, max 152 mg/dL) was observed throughout the study.
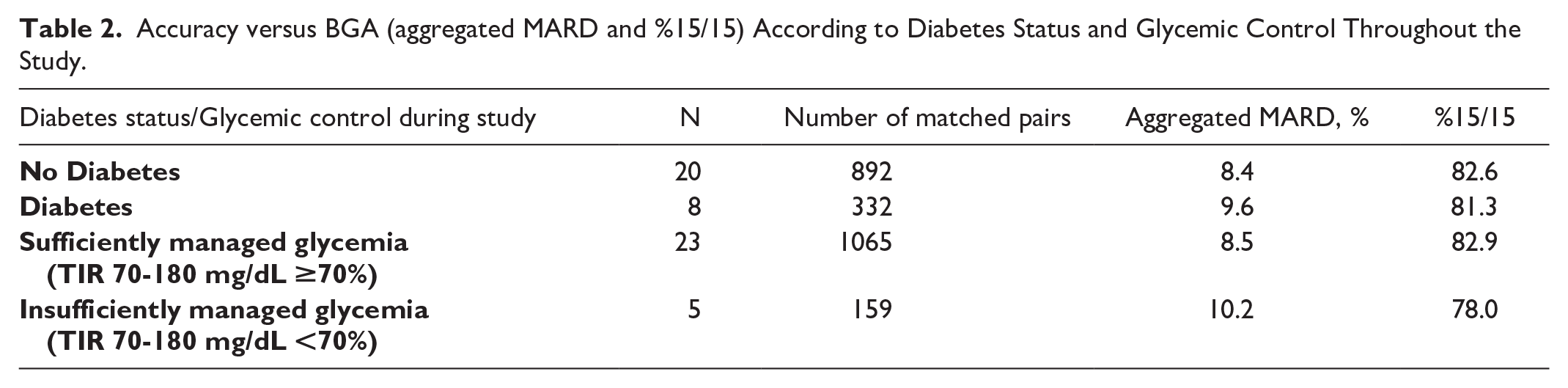
The group of participants with diabetes had a total of 374 hours of useful recordings. In total, 75.4% (282 hours) was spent in the target range of 70 to 180 mg/dL, 0.1% (0.3 hours) was spent below 70 mg/dL, and 24.5% (91.6 hours) was spent above 180 mg/dL. These patients had a MARD of 9.6 (6.5; 13.3)%. When looking at all insufficiently managed patients (ie, those having an individual TIR <70%, according to the TIR consensus), 16 five individuals were found (median TIR: 59.1 [41.5; 65.3]%). These patients had a MARD of 10.2 (6.3; 13.3)%. An overview of system accuracy in relation to glycemic control and diabetes status is shown in Table 2.
Accuracy versus BGA (aggregated MARD and %15/15) According to Diabetes Status and Glycemic Control Throughout the Study.
Adverse Events
When assessing safety endpoints, nine AEs were documented. Four were classified as serious adverse events (SAEs), primarily attributable to the high-risk profile of the study population and the postcardiac surgery environment. These SAEs included secondary hemorrhage and postoperative hemodynamic failure. None of the SAEs were considered to be related to the device employed in the study.
Furthermore, five of the observed AEs were categorized as adverse device effects (ADEs). These included instances of accidental catheter dislocation, localized hematoma formation, extravasation, swelling, and pain at the sensor insertion site. All ADEs were successfully resolved within the study’s designated timeframe and, in nature and impact, could be compared with AEs commonly associated with routine peripheral intravenous catheter usage.
Notably, no unexpected adverse reactions, serious adverse device effects, or unexpected adverse device reactions occurred throughout the study.
Discussion
The present pilot trial assessed accuracy and safety of a novel ivCGM system in cardiothoracic surgery patients with and without diabetes admitted postoperatively in the ICU. The cardiovascular high-risk patient population was selected to rigorously evaluate the system’s safety in this vulnerable population, who often requires urgent management of hyperglycemia and hypoglycemia events. 9 The ivCGM system provided reliable glucose measurements with adequate accuracy compared with BGA measurements over the 72-hour monitoring period, even in patients with insufficiently managed glycemia. Notably, the aggregated overall MARD was well below the 10% threshold recommended by Danne et al. 17
Overall, there is an urgent need for accurate and reliable CGM technologies in ICU settings. Intermittent glucose measurements, representing the present standard of care, inadequately capture glycemic trends. In critically ill patients, quarter-hourly BG measurements improve glycemic control. 18 This frequency is only achievable in clinical practice using CGM systems. Hypoglycemia is associated with poor outcomes, especially in critically ill patients. Although clinically challenging due to possibly masked neuroglycopenia or sympathoadrenal symptoms, 19 CGM has shown a reduction in hypoglycemic events in critically ill patients compared with intermittent BGA measurements. 20 CGM may also help to maintain glycemic target ranges, even though the optimal glycemic range for critically ill patients is still subject of ongoing debate.21-28 As available scientific research often does not include CGM data and does not address the definition of a meaningful TIR, CGM use in critically ill patients may add further knowledge on glycemic targets. So far, observational studies showed an association with increased survival in insulin-treated patients who achieved TIR (70-139 mg/dL) of 80%. 29 Krinsley and Preiser 30 obtained similar results in patients without diabetes. Notably, in the HALF-PINT study, therapy guidance in critically ill children after cardiac surgery was performed by scCGM. 31 Thus, maintaining a safe glycemic range (140-180 mg/dL, as recommended by current guidelines) 4 and avoiding both hypoglycemia and high GV are crucial for managing glycemia in ICU patients. In the present observational study, TIR (140-180 mg/dL) was 43.8% for patients with preexisting diabetes, showing significant deviations from this target range despite all efforts by the treating physicians to maintain glycemic control by standard of care treatment in this vulnerable population.
For CGM use in vulnerable ICU patients, high levels of accuracy and reliability are required. However, accuracy assessment of CGM has not been standardized yet. Recently, clear recommendations on reporting scCGM accuracy have been published; in part, these may also be applied to ivCGMs. Notably, common CGM accuracy metrics are point accuracy (eg, MARD), clinical accuracy (eg, DTSEG), and trend accuracy. 32 Continuous glucose monitoring accuracy assessment in ICU patients seems especially challenging due to multiple variables and requires high standards. Available scCGM systems may not meet these requirements due to a general lack of accuracy and time lag in glucose measurements during rapid glycemic changes. Factors such as alterations in tissue perfusion, pH, electrolyte, and fluid shifts caused by hypovolemic, distributive, cardiogenic, or obstructive shock, frequently present in critical illness, impact accuracy. 33 Even if preliminary data show no association between impaired microcirculation or vasopressor use and sensor accuracy in critically ill patients admitted to medical and surgical ICUs, accuracy of scCGM compared with BGA is impaired.34-37 On the other hand, ivCGM can provide blood glucose readings without relevant delay, is not affected by pressure artifacts when positioning the patient (semi prone/supine position), and is not dependent on fluid shifts between bloodstream and subcutaneous tissue. Disadvantages of ivCGM compared with sc devices such as higher implementation resources, care burden, thrombosis, and infection risk are mentioned in literature. 11 Such device-related AEs did not occur in the present study, and the mentioned disadvantages may be mitigated by further developments or using existing central or peripheral intravenous lines. Despite the complexities of critically ill patients, ivCGM may address the clinical needs of ICU settings, allowing accurate glycemic monitoring with minimal time lag and alarm functions availability. It is important to note that the present study has limitations. Due to the pilot nature of a trial in critically ill patients and ethical concerns, no active glucose challenges were performed to induce high rates of glucose change. Furthermore, patients were treated according to internal guidelines for maintaining glycemic control by the ICU staff, who acted independently from study conduction. Therefore, the prespecified measurement range of 20 to 400 mg/dL was not fully covered in this study, none of the patients experienced hypoglycemia (<70 mg/dL), and only a few data pairs fell above 250 mg/dL. Accuracy in hypoglycemia and level 2 hyperglycemia (>250 mg/dL) is important for assessing the performance of a CGM system and was not completely covered in this study.
Current consensus guidelines on CGM and metrics for clinical trials 38 do not give specific recommendations on how to deal with devices at this stage of investigations. Awaiting for future trials where accuracy at extreme glucose levels and during fast glucose changes are assessed, the ivCGM system may be used in a adjunctive setting by confirming hypoglycemia and level 2 hyperglycemia by BGA measurements. It should be noted that the clinical accuracy of the ivCGM system should remain substantially unaffected at high rates of glucose change due to an instrumental time lag as short as 2 to 3 minutes.
The study population showed a low median MAGE (49 mg/dL) compared with CGM trials with participants living with type 1 diabetes 39 and type 2 diabetes. 40 This primarily because, in the present observational study, participants living with diabetes were in the minority, and all patients were managed so as to minimize the occurrence of wide glycemic excursion. Nevertheless, due to preexisting diabetes, perioperative stress, and the use of intravenous glucose administration as a solution for amiodarone or levosimendan, in some patients, significant glycemic fluctuations were recorded throughout the study period. The MARD observed in patients with either diabetes or insufficiently managed glycemia indicates acceptable accuracy even during phases of high rates of glucose change.
Conclusion
In this pilot study, the ivCGM system was accurate compared with BGA in a population of high-risk cardiovascular patients admitted to post-surgery cardiothoracic ICU. Notably, point accuracy by aggregated overall MARD was 8.7%. Clinical accuracy was high according to DTSEG, with 100% of the values in zones A and B. No hypoglycemic events were observed throughout the study. Yet, the ivCGM system provided accurate readings in euglycemia and hyperglycemia over the whole 72-hour monitoring period and demonstrated good tolerability and safety profiles when applied to this vulnerable patient population. Further assessment of ivCGM accuracy in more extensive studies with significant coverage of hypoglycemia, level 2 hyperglycemia, and high rates of glucose change, including more patients with diabetes in surgical and medical ICU wards, is needed. Subsequently, randomized controlled trials should evaluate the clinical benefits of ivCGM over standard glucose monitoring practices in critically ill patients, particularly when coupled with validated insulin infusion protocols. These efforts may solidify the position of the ivCGM system as a valuable tool in the pursuit of optimal glycemic control and improved outcomes within the critical care domain.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251342598 – Supplemental material for Accuracy and Safety of a Novel Intravenous Continuous Glucose Monitoring System in Patients Admitted to a Cardiothoracic ICU: A Pilot Trial
Supplemental material, sj-docx-1-dst-10.1177_19322968251342598 for Accuracy and Safety of a Novel Intravenous Continuous Glucose Monitoring System in Patients Admitted to a Cardiothoracic ICU: A Pilot Trial by Daniel Hochfellner, Tina Poettler, Michael Schoerghuber, Edita Lukic, Ameli Yates, Ingeborg Keeling, Daniel Zimpfer, Felix Aberer, Francesca Berti, Fausto Lucarelli, Francesco Valgimigli and Julia K. Mader in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
We would like to acknowledge Laura Roubik who helped to perform the study.
Abbreviations
AE, adverse events; ADE, adverse device effect; ASCV, atherosclerotic cardiovascular event; BGA, blood gas analysis; BSM, bedside monitor; CABG, coronary artery bypass graft; DTSEG, Diabetes Technology Society Error Grid; CGM, continuous glucose monitoring; GOD, glucose oxidase; GV, glycemic variability; HCP, health care professional; ICU, intensive care unit; ICH-GCP, International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use—Good Clinical Practice; iv, intravenous; MAD, mean absolute difference; MARD, mean absolute relative difference; MedARD, median absolute relative difference; MAGE, mean amplitude of glycemic excursions; POC, point of care; SADE, serious adverse device effect; SAE, serious adverse events; scCGM, subcutaneous CGM; TIR, time in range; UADR, unexpected adverse device reaction; UAR: unexpected adverse reaction.
Author Contributions
DH, JKM, FB, FL, and FV were involved in conceptualization of the trial and designed the protocol; Project administration was conducted by TP; DH, JKM, MS, FA, EL, IK performed the study; JKM, AY, and DZ supervised the study; FL performed the statistical analyses and developed the figures and visualizations; DH wrote the original manuscript draft. All authors critically revised and edited the article and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DH received travel grants from Eli Lilly, MSD, Novo Nordisk, Novartis, and Sanofi as well as speaker’s honoraria from Novo Nordisk. FV, FB, and FL are full-time employees at A. Menarini Diagnostics. FA received speaker honoraria from Sanofi Aventis, Novo Nordisk, AMGEN, Eli Lilly and participated in advisory boards of Eli Lilly and Novo Nordisk. JKM is a member in the advisory boards of Abbott Diabetes Care, Becton-Dickinson/Embecta, Biomea, Eli Lilly, Medtronic, Novo Nordisk, Pharmasens, Roche Diabetes Care, Sanofi, and Viatris; received speaker honoraria from Abbott Diabetes Care, A. Menarini Diagnostics, Becton-Dickinson/Embecta, Boehringer-Ingelheim, Eli Lilly, MedTrust, Novo Nordisk, Roche Diabetes Care, Sanofi, Servier, and Ypsomed; and is shareholder of decide Clinical Software GmbH and elyte diagnostics. The other authors have no conflict of interest to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by A. Menarini Diagnostics, Florence, Italy.
Ethics Approval and Consent to Participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use—Good Clinical Practice (ICH-GCP) and with the Declaration of Helsinki. Approval of the local institutional review board: (EC #33-615 ex20/21) was obtained. Informed consent was obtained in written form before any study related procedures were performed.
Consent for Publication
Not applicable
Trial Registration
Availability of Data and Materials
The authors provided as much detailed information on data and materials as possible within the manuscript and in the Additional file 1: theoretical and collected BGA and ivCGM data for all patients eligible for data analysis as well as Results of accuracy evaluation versus BGA reference for each patient. Furthermore, subject plots and complete data on TIR calculations are provided in Additional file 1.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
