Abstract
The landmark Diabetes Control and Complications Trial (DCCT) showed that glucose control is critical to reducing the risk of diabetes-related complications. This chapter outlines a series of innovations and investigations that followed the DCCT, aimed at minimizing the risk of hypoglycemia while further improving glucose control. The chapter presents an example of innovations in wired enzyme technology that facilitated the movement from capillary glucose monitoring to continuous glucose monitoring (CGM) and ultimately, the first-factory calibrated CGM system. The next glycemic management innovation was to connect CGM data to an insulin pump containing an algorithm able to adjust insulin delivery based on the changing glucose levels and trends. The key features of automated insulin delivery (AID) systems, currently approved in the United States, are presented. The AID summary table includes type of pump, type and function of the insulin delivery algorithm, the data management system, and the indications for use. The next section explores the innovation of alternative routes of insulin delivery to move toward the goal of a fully automated insulin delivery system. The main trials in developing and implementing an implantable intraperitoneal programmable system are summarized. The last section explores if sensor input in addition to glucose levels such as continuous sensing of ketone, lactate, or insulin levels may provide valuable feedback to move us closer to a fully autonomous AID system. Much of this diabetes innovation and investigation work has been supported by the National Institute of Diabetes and Digestive and Kidney Diseases over that last 75 years.
Keywords
Introduction
This article is part of a collection of publications to commemorate the 75th Anniversary of the NIDDK.
The focus of this chapter is on the development of continuous glucose monitors (CGMs), the evolution of insulins, and the integration of CGMs, algorithms, and insulin delivery into artificial pancreas systems and the potential for implantable systems.
Prior to CGM development, urine glucose monitoring progressed to blood glucose monitoring which was chronicled in a symposium in Diabetes Care in 1980. 1 For the evolution of CGMs, one needs to go back to the development of the Clark electrode for monitoring oxygen tension in 1956, 2 and its evolution to an enzyme electrode for measuring glucose by Updike and Hicks in 1967. 3 The field was reviewed in an NIDDK (then NIAMDD)-sponsored meeting chronicled in a symposium in Diabetes Care in 1982, 4 which included contributions from both Clark and Updike, among others. An overview of early CGM development was published in Diabetes Technology and Therapeutics in 2009 5 and a roadmap to the effective use of continuous glucose monitoring was published in Diabetes Spectrum in 2023. 6
Meanwhile, insulin pumps were being developed, as was chronicled in a symposium in Diabetes Care in 1980. 7 AP systems were evolving, as reviewed in a paper in Diabetes in 2011, 8 with the development of algorithms to integrate insulin delivery as reviewed in 2014, 9 and the potential of a dual hormone artificial pancreas as reviewed in 2021. 10
As discussed in this chapter, CGM now includes use of a wired electrode, and multiple hybrid closed-loop systems have been launched. The chapter also highlights future potential developments for implantable systems and for newer insulins.
CGM-Targeting Research at the University of Texas
Continuous glucose monitors (CGMs) are a prerequisite for closed-loop insulin delivery and have become the standard of care for treatment of people with type 1 diabetes. A brief summary of some of the key work done at the University of Texas at Austin, funded in part by NIDDK, provides a glimpse into the evolution of several key discoveries linked together to create the most broadly implemented CGM system worldwide.
While animal studies between 1993 and 1998 at the University of Texas at Austin established the feasibility of CGM, in 1996 the focus shifted first to painless blood glucose monitoring (BGM) hoping to later build the CGM effort. Multiple hurdles were overcome, including eliminating the need for oxygen in a 0.25 mm diameter glucose concentration monitoring electrode. Then the effects of electrooxidizable interferants were sufficiently reduced to allow calibration with a single independent glucose assay. Following these developments, subcutaneous implantation in the rat and in a type 1 insulin-dependent chimpanzee suggested that a CGM system warranted further development.
In the 1990s NIH/NIDDK supported studies of implantable amperometric continuously glucose monitoring (CGM) sensors for diabetes management. These glucose monitoring electrodes utilized glucose oxidase,
11
an enzyme catalyzing the oxidation of glucose by dissolved oxygen, the reaction producing gluconolactone and hydrogen peroxide. The electrodes usually monitored the concentration of the hydrogen peroxide produced, though a few tracked the depletion of oxygen. Most electrodes were subcutaneously implanted; some were, however, intravascular, residing for example in the
Our studies differed from others in several ways, including the following:
Tests in rats suggested the feasibility of CGM with 0.25 mm diameter subcutaneously implanted electrodes.17 -23 Our notion of utility in managing T1D was reinforced by subcutaneously implanting the 0.25 mm diameter electrically wired glucose oxidase electrode in an unconstrained, naturally diabetic, brittle, type I, insulin-dependent chimpanzee. The glucose concentration measured with the electrode was consistent with the strip-measured blood glycemia (Figure 1). 24

Correlation of the subcutaneously implanted electrically wired sensor measured glucose concentration with strip measured blood glucose concentration on an unrestrained naturally type 1 insulin dependent diabetic chimpanzee. 24
Working with TheraSense this wired enzyme technology resulted in the first commercial version of the FreeStyle blood glucose meter (introduced in 2000) that was accurate, required a very small blood sample and the blood sampling technology was nearly painless. Abbott acquired TheraSense in 2004 and begun work on a CGM system. Key elements from the University of Texas, the TheraSense FreeStyle program and the Abbott team contributed to the development of the FreeStyle Navigator CGM (2008) and then the first factor-calibrated FreeStyle Libre Flash CGM (2010), culminating in Abbott’s launch of the FreeStyle Libre CGM in 2014.
Automated Delivery Systems
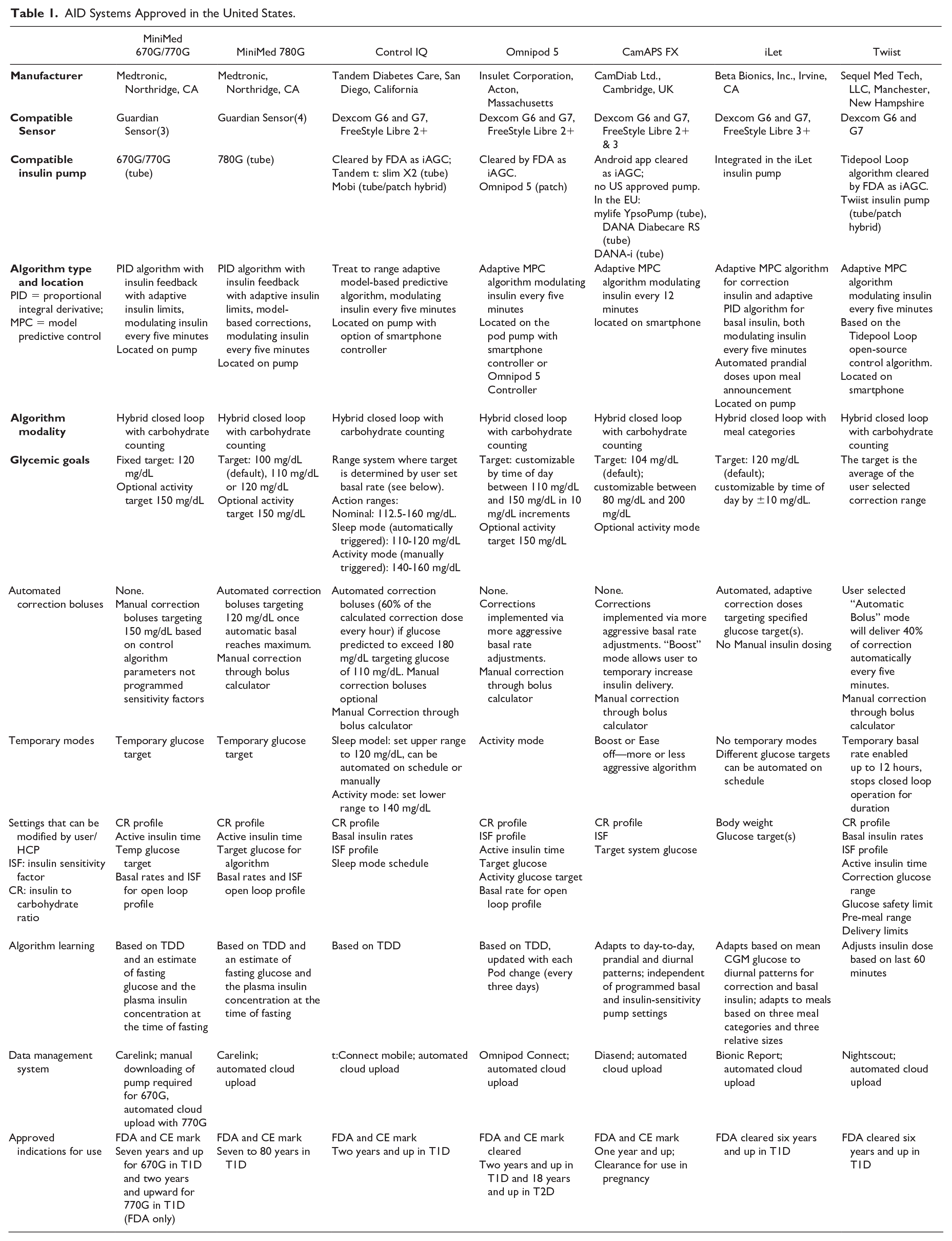
Automated insulin delivery (AID) is now established as the best-practice treatment for type 1 diabetes and is being rapidly adopted among people with type 2 diabetes who use insulin to manage their glucose levels. Several commercial AID systems are now approved in the United States, including Medtronic’s MiniMed 670G, 770G, and 780G25 -29; t:slim X2 and Mobi with Control IQ Technology by Tandem Diabetes Care,30 -34 OmniPod 5 system by Insulet Corporation,35 -37 the Tidepool Loop control algorithm that is now embedded in the twiist AID system by Sequel,38,39 and the iLet Bionic Pancreas system by Beta Bionics.40 -43 The CamAPS FX smart phone application (CamDiab Ltd., Cambridge, UK) is approved by the FDA as an interoperable Automated Glucose Controller (iAGC) but awaiting FDA clearance of the mylife YpsoPump as its interoperable pump to enable an integrated AID system.44 -48 In Europe, the United Kingdom, and Canada, other AID systems are available as well, for example, Diabeloop DBLG1 (Diabeloop, France),49,50 Inreda AP (Inreda Diabetic B.V., the Netherlands). 51 OpenAPS is an alternative to commercial systems providing advanced users with the opportunity to assemble their own AID system. 52 All contemporary AID systems perform significantly better than either multiple daily injections or sensor-augmented insulin pump therapy. Real-world AID use by thousands of people for extended periods of time have extended findings from clinical trials and demonstrate translation of this technology in clinical practice.53 -59
The key features of the most-used commercially available AID systems are summarized in Table 1, adapted and updated from the 2023 Consensus Recommendations for the Use of Automated Insulin Delivery Technologies in Clinical Practice. 60 Generally, the AID systems employ various continuous glucose monitoring (CGM) sensors from Medtronic, Dexcom, and Abbott and use a variety of control algorithms, with PID (Proportional Integral Derivative) and MPC (Model Predictive Control) being most common. Control-IQ was the first AID system designated by the FDA as an iAGC (not bound to a unique pump/sensor combination), followed by Omnipod 5, Tidepool Loop, the iLet, and then CamAPS. Most MPC algorithms are adaptive, adjusting their action to changes in user physiology. The systems are distinguished by their insulin pumps, expected user interactions, and adjustability of target glucose values and other parameters.
AID Systems Approved in the United States.
Implantable Insulin Delivery Systems Using Intraperitoneal Route
Subcutaneous (SC) insulin delivery is associated with several drawbacks, including delayed insulin absorption and action and peripheral hyperinsulinemia.61,62 Besides the cumbersome wear of external devices to use SC insulin in an automated insulin delivery system (AID), the delays of insulin action and glucose sensing when performed in the SC tissue require the need for meal and physical exercise announcements, resulting in so called “hybrid” AID.8,63 The goal of fully-automated insulin delivery necessitates alternative routes of insulin delivery.
From the 1990s, implantable intraperitoneal programmable systems (IIPS) embedded in a SC pocket of the abdominal wall that infuse insulin through the peritoneal route have been clinically used by several hundreds of people in Europe.64,65 Despite not being connected to continuous glucose monitors (CGMs), these systems have shown improvements of glucose control when compared with SC insulin pumps, including sustained lower HbA1c levels and reduction of severe hypoglycemic events.66 -68 Additional reported benefits include better diabetes-related quality of life, 69 improvements of lipids, 70 IGF-1 levels, 71 and restored glucagon secretion during hypoglycemia 72 and physical exercise. 73 Limitations came from the aggregation of insulin in the pumps, 74 catheter occlusions due to foreign body reactions,75,76 occasional pump-pocket infections 77 and low-affinity anti-insulin antibody production. 78
Pilot clinical trials have been performed to assess AID using these IIPS connected to intravenous (IV) or SC glucose sensors.79 -81 SC sensors wirelessly connected to the IIPS through an external platform managing the control algorithm (either of proportional-integral-derivative [PID] 82 or MPC 83 modes) have allowed better glucose control than manually-controlled IIPS 82 or CGM-connected SC infusion. 83 See Figure 2.
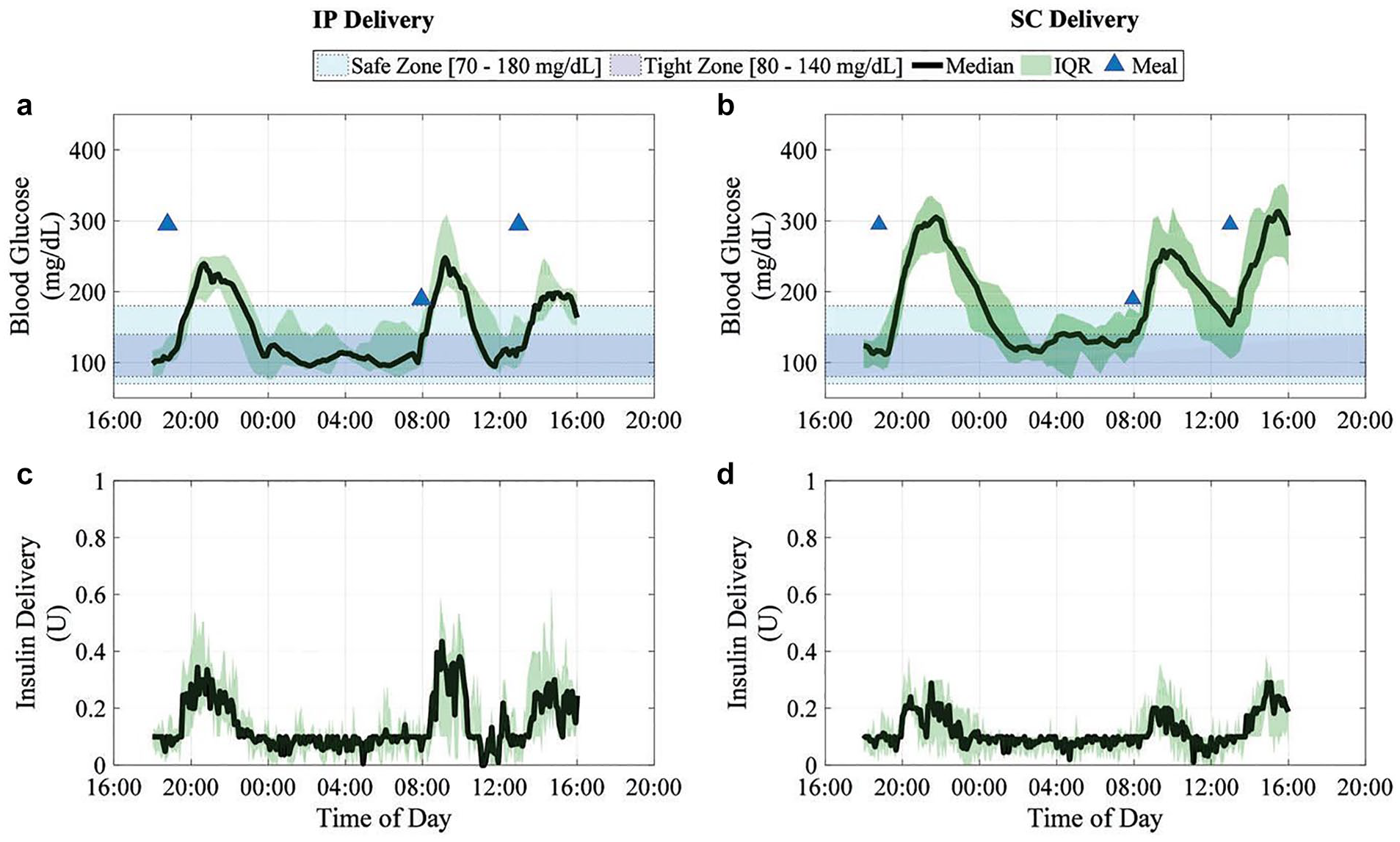
Intraperitoneal insulin delivery provides superior glycemic regulation to subcutaneous insulin delivery in model predictive control-based fully-automated artificial pancreas in patients with type 1 diabetes: a pilot study. Blood glucose results are summarized in (a) and (b) as median YSI-measured values (continuous thick line), interquartile range (shaded), for closed-loop trials with intraperitoneal (IP) insulin delivery and subcutaneous (SC) insulin delivery, respectively. The closed-loop duration is 22 hours from 6:00 PM. The control objective (80-140 mg/dL) is shown as the blue rectangular band and the safety region (70-180 mg/dL) as the cyan rectangular band. The median insulin delivery from insulin pump for all trials is shown in (c) and (d) for IP delivery and SC delivery, respectively, along with the interquartile range. 82
Based on these investigations, the concept of developing AID using the pharmacokinetics and –dynamics advantages of intraperitoneal insulin infusion are appealing. Research and development efforts are expected to focus on more physically stable highly concentrated insulin formulations, further miniaturized IIPS with coated catheters to prevent occlusions 84 and implantable glucose sensors eventually located in the peritoneal space. 85 In silico research has shown that PID control algorithms based on a two-compartment model of insulin kinetics would allow fully automated insulin delivery while using the peritoneal route. 86 Current EU-funded and industry-led projects are targeting this innovative approach of AID that would both allow optimized glucose control and to “forget” diabetes by the users.86 -89
New Insulin Formulations and Future Multiplexed Metabolic Sensors (Cortisol, Lactate, etc.)
New Insulin Formulations
In the six decades following insulin’s discovery in 1921, its utility was progressively enhanced through innovations in formulation. These enabled distinct pharmacokinetic-pharmacodynamic (PK-PD) classes 90 from “regular” solutions of zinc-stabilized insulin hexamers (rapid) to insulin-protamine microcrystalline suspensions (NPH; intermediate) and ultra-lente zinc-insulin crystals (basal). Recombinant DNA technology in the 1970s not only enabled a transition to human insulin but also stimulated design of analogs to optimize these PK-PD classes. 91 The underlying motivation was (and remains) to match subcutaneous absorption to the daily pattern of normal pancreatic insulin secretion. Prandial insulin analogs were designed, for example, to accelerate SQ absorption, thereby enhancing post-prandial glucose disposal and limiting late-post-prandial hypoglycemia. Conversely, basal analogs were sought with flat PK-PD profiles extending for 24 hours. In the past two decades prandial- and basal insulin analogs (rather than native human insulin) have assumed a predominant role in insulin replacement therapy. Remarkably, as insulin begins its second century, such molecular innovation continues as exemplified by once-a-week analogs.92,93 Furthermore, the threat of hypoglycemia is motivating efforts to insert glucose-responsive conformational “switches” into the insulin molecule94,95 toward self-regulation as a function of glycemic state. Additional molecular innovations are being sought to protect insulin from degradation at elevated temperatures96,97: such enhanced stability promises to circumvent the costly cold chain of insulin delivery and storage. In the coming decade we envision that combinations of such innovations may bring to fruition the “pursuit of a perfect insulin” 91 both in affluent societies and in the developing world.
Future Multiplexed Metabolic Sensors (Cortisol, Lactate, etc.)
Advanced automated insulin delivery (AID) systems have improved glucose control and reduced hypoglycemia, but self-management remains demanding for people with diabetes. Current AID systems rely on a single sensor source, CGM, to adjust insulin. A multi-metabolite sensor could measure glucose along with other data, advancing glucose management with limited to no user inputs.98,99 For example, ketone bodies signal when there's insufficient insulin, indicating system performance, while lactate levels rise with physical activity, helping artificial pancreas systems identify exercise without user input.
Researchers are exploring ways to integrate more comprehensive metabolic data into AID systems. The inclusion of ketone body sensing can provide critical insights into metabolic status, especially during periods of insulin deficiency, such as diabetic ketoacidosis (DKA). 99 Continuous monitoring of lactate levels offers another layer of understanding, particularly useful for detecting physical exertion that might otherwise result in unexpected drops in blood sugar levels.100 -103 The focus on multiplex biosensors like continuous ketone measurements (CKM), lactate, and continuous insulin measurements (CIM) has the potential to allow for near real-time feedback on insulin usage and efficacy and could enable more precise tailoring of insulin delivery, resulting in better overall glucose management and fewer incidences of hypo- or hyperglycemia.102,104
While AID systems have made significant strides, ongoing research and technological innovations combined with data-driven AI hold the promise of creating a fully autonomous system (Figure 3). These advancements could transform diabetes care, making it less burdensome and more effective, ultimately enhancing the well-being of millions worldwide. 100 Many of the advances in technology contributing to the transformation of diabetes care have been supported by the NIDDK over the last 75 years.
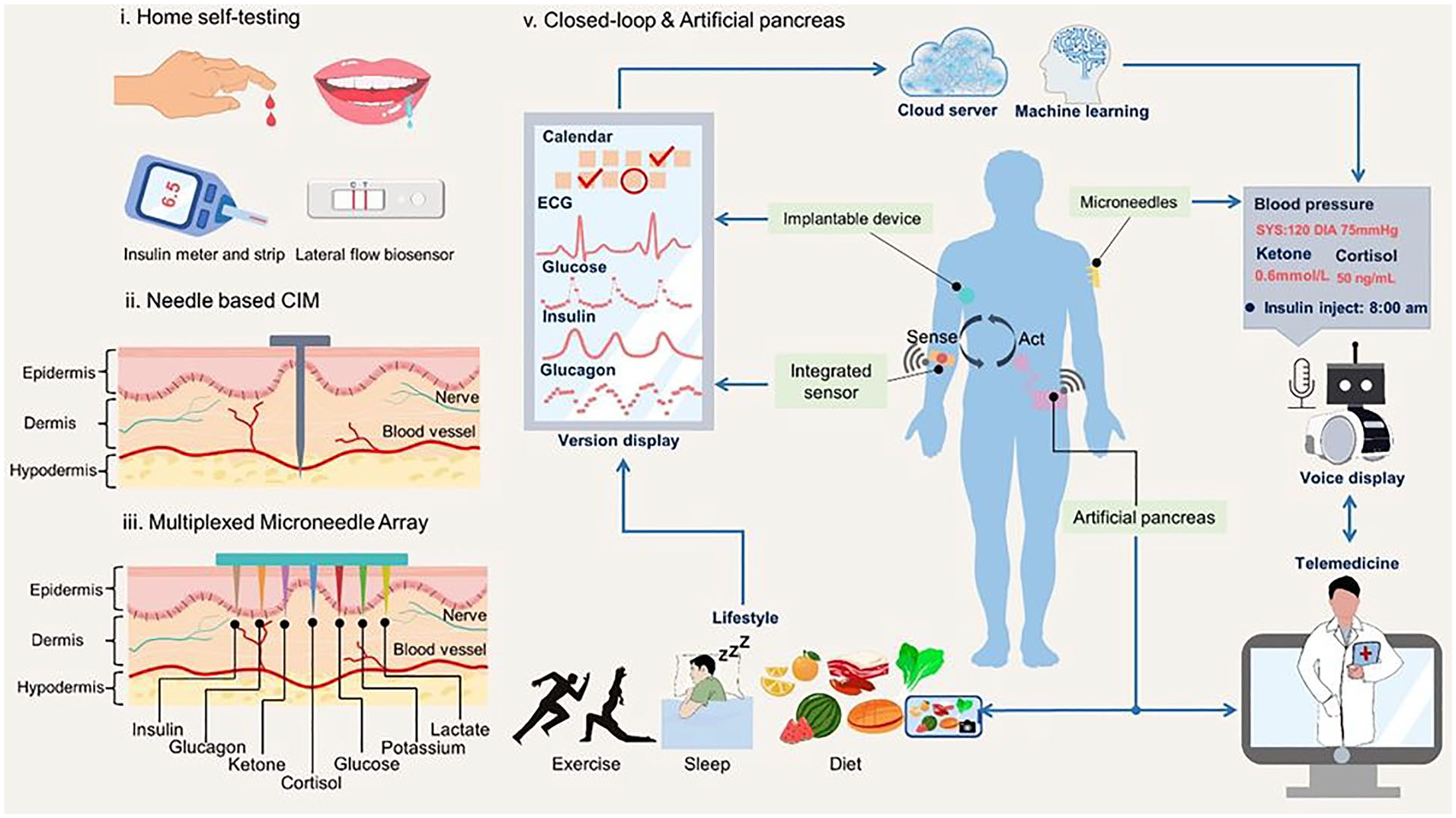
Future vision of diabetes management and personalized closed-loop autonomous artificial pancreas system integrating multi-modal sensing with AI. Adapted from Vargas. 103
Footnotes
Abbreviations
AID, automated insulin delivery; BGM, blood glucose monitoring; CGM, continuous glucose monitor; CIM, continuous insulin measurements; CKM, continuous ketone measurements; DKA, diabetic ketoacidosis; iAGC, interoperable automated glucose controller; IGF-1, insulin-like growth factor 1; IIPS, implantable intraperitoneal programmable systems; MPC, model predictive control; NIAMDD, National Institute of Arthritis, Metabolism and Digestive Diseases; NIDDK, National Institute of Diabetes and Digestive and Kidney Diseases; NIH, National Institutes of Health; NPH, Neutral Protamine Hagedorn Insulin; PID, proportional integral derivative; SC, subcutaneous.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RMB has no personal conflicts, but reports that his institution, HealthPartners Institute, has received support on his behalf for research grants, consulting, or scientific advisory board participation for Abbott Diabetes Care, Ascensia, CeQur, DexCom, Eli Lilly, Embecta, Hygieia, Insulet, Medscape, Medtronic, Novo Nordisk, Onduo, Roche Diabetes Care, Tandem Diabetes Care, Sanofi, United Healthcare, Vertex Pharmaceuticals, and Zealand Pharma. AH consults to Abbott Diabetes Care of Alameda, CA 94502. MDB receives research support through his institution from NovoNordisk, Dexcom, and Tandem; MDB received honoraria/consulting fees from Tandem, Sinocare, Boydsense, and Roche; MDB receives royalties through his institution from Dexcom, Lifescan, Sanofi, and Tandem. RV is a full-time employee of Medtronic Diabetes. SAB has received research funding to her institution from Dexcom, Insulet, Roche, Tandem, and Tolerion and participated on a data safety and monitoring board for MannKind. GPF conducts research sponsored by Medtronic, Dexcom, Abbott, Tandem, Insulet, and Beta Bionics and has been a speaker/consultant/ad board member for Medtronic, Dexcom, Abbott, Tandem, Insulet, Beta Bionics, Lilly, and Sequel. JLS has conducted clinical trials for Abbott Diabetes Care, Dexcom, JDRF/Breakthrough T1D, Insulet, Medtronic, and Provention Bio and has received in-kind support for research studies from Dexcom and Medtronic. She has consulted for Abbott Diabetes, Insulet, Medscape, Medtronic, Vertex, and Ypsomed. She has been a member of advisory boards for Cecelia Health, Insulet, Mannkind, Medtronic Diabetes, StartUp Health T1D Moonshot, and Vertex. RH reports having received speaker honoraria from Eli Lilly, Dexcom, and Novo Nordisk, receiving license fees from Braun; receiving consultancy fees from Abbott Diabetes Care, patents related to closed-loop, and being director at CamDiab. SJR reports being a Beta Bionics employee and stockholder, being an inventor on patents related to the bionic pancreas that are assigned to Massachusetts General Hospital and licensed to Beta Bionics, and payments for stock options related to the acquisition of Companion Medical by Medtronic. ERD holds equity in Beta Bionics, Inc. and is a named inventor on several issued patents, which are owned by Boston University and licensed to Beta Bionics, Inc. MSP has received research funding and honoraria from Vertex Pharmaceuticals, investigator-initiated research funding from Dexcom Inc and Samsung, and serves of the scientific advisory board of Anagram Therapeutics. MDB receives research support through his institution from NovoNordisk, Dexcom, and Tandem. RL has received consulting fees from Abbott Diabetes Care, Adaptyx Biosciences, Biolinq, Capillary Biomedical, Deep Valley Labs, Gluroo, PhysioLogic Devices, Portal Insulin, Sanofi, and Tidepool. He has served on advisory boards for Provention Bio and Lilly. He receives research support from his institutions from Insulet, Medtronic, Sinocare, and Tandem. CC has no disclosures relevant to this work. ED has received personal fees from Roche and Eli Lilly and Company; holds patents on artificial pancreas technology; and has received product support from Insulet Corporation, Tandem Diabetes Care, Roche, and Dexcom, Inc. The work presented in this article was performed as part of his academic appointment and is independent of his employment with Eli Lilly and Company. HKA reports research support and honorarium through the University of Colorado from Medtronic, Dexcom, Roche, and Tandem Diabetes Care. RWB reports no personal financial disclosures but reports that his institution has received funding on his behalf as follows: grant funding, study supplies, and consulting fees from Insulet, Tandem Diabetes Care, and Beta Bionics; grant funding and study supplies from Dexcom; grant funding from Bigfoot Biomedical; study supplies from Medtronic, Ascencia, and Roche; consulting fees and study supplies from Eli Lilly and Novo Nordisk; and consulting fees from embecta, Sequel Med Tech, Vertex, Hagar, Ypsomed, Sanofi, and Zucara. ER declares consultant/speaker fees from A. Menarini Diagnostics, Abbott, Air Liquide SI, Astra-Zeneca, Becton-Dickinson, Boehringer-Ingelheim, Cellnovo, Dexcom Inc., Eli-Lilly, Hillo, Insulet Inc., Johnson & Johnson (Animas, LifeScan), Medtronic, Medirio, Novo-Nordisk, Roche, and Sanofi-Aventis and research support by Abbott, Dexcom Inc., Insulet Inc., Roche, and Tandem Diabetes Care. MAW is an inventor of U.S. Patents issued to Case Western Reserve University pertaining to novel insulin analogs. JSS has equity in 4Immune, Abvance, AiTA, Applied Therapeutics, Avotres, Dexcom, Immunomolecular Therapeutics, Oramed, SAB Biotherapeutics, Signos, vTv Therapeutics, and WiNK.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
