Abstract
Arguably, diabetes mellitus is one of the best quantified human conditions. In the past 50 years, the metabolic monitoring technologies progressed from occasional assessment of average glycemia via HbA1c, through episodic blood glucose readings, to continuous glucose monitoring (CGM) producing data points every few minutes. The high-temporal resolution of CGM data enabled increasingly intensive treatments, from decision support assisting insulin injection or oral medication, to automated closed-loop control, known as the “artificial pancreas.” Throughout this progress, mathematical models and computer simulation of the human metabolic system became indispensable for the technological progress of diabetes treatment, enabling every step, from assessment of insulin sensitivity via the now classic Minimal Model of Glucose Kinetics, to in silico trials replacing animal experiments, to automated insulin delivery algorithms. In this review, we follow these developments, beginning with the Minimal Model, which evolved through the years to become large and comprehensive and trigger a paradigm change in the design of diabetes optimization strategies: in 2007, we introduced a sophisticated model of glucose-insulin dynamics and a computer simulator equipped with a “population” of N = 300 in silico “subjects” with type 1 diabetes. In January 2008, in an unprecedented decision, the Food and Drug Administration (FDA) accepted this simulator as a substitute to animal trials for the pre-clinical testing of insulin treatment strategies. This opened the field for rapid and cost-effective development and pre-clinical testing of new treatment approaches, which continues today. Meanwhile, animal experiments for the purpose of designing new insulin treatment algorithms have been abandoned.
Keywords
Introduction
In health, glucose metabolism is tightly controlled by a hormonal network including the gut, the liver, the pancreas, and the brain to ensure stable fasting blood glucose (BG) levels and brief transient postprandial glucose fluctuations. In diabetes, this network control is disrupted by deficiency or absence of insulin secretion, which have to be compensated by technological means and/or behavioral changes (eg, exercise, diet). Type 1 diabetes (T1D) is characterized by absolute deficiency of insulin secretion resulting from autoimmune response targeting the β-cells in the islets of Langerhans of the pancreas—the site of insulin secretion and synthesis. Affected individuals require insulin therapy to control hyperglycemia and sustain life.1-3 There is no cure; the only proven treatment for T1D is the tight optimal control of BG fluctuations via insulin delivery.4-8 Thus, T1D is a prime example of a significant health care problem, the only solution of which is advanced technologies aiming at the functional replacement of the failed beta cell. Major efforts target the optimization of diabetes control and large international organizations are dedicated to the treatment and, ultimately, the cure of diabetes and its complications, including American Diabetes Association (ADA), 9 the European Association for the Study of Diabetes (EASD,) 10 , the International Diabetes Federation (IDF,) 11 , and the Diabetes Technology Society (DTS). 12 These efforts are picking up speed and new diabetes technologies are introduced daily. To put this progress in perspective, for the 1900 years following the introduction of the term diabetes (Aretaeus the Cappadocian, 1st century AD), diet was the only treatment, albeit unsuccessful in T1D. In the 19th century, the nature of diabetes was understood and with the discovery of insulin in 1921 by Frederick Banting at the University of Toronto, T1D was no longer a death sentence. For this breakthrough, Banting and John Macleod were awarded the Nobel Prize in Physiology or Medicine in 1923. The field of Diabetes Technology was born.
The Glucose-Insulin Control Network
As presented in Figure 1a, BG levels are raised by food containing carbohydrates, and glucose is also produced by the body (mainly by the liver), after which it is distributed and utilized through both insulin-independent (eg, central nervous system and red blood cells) and insulin-dependent (muscle and adipose tissues) pathways. Insulin secreted by the pancreatic β-cell is the primary regulator of glucose homeostasis; it reaches the system circulation after liver degradation and is peripherally cleared primarily by the liver. The glucose and insulin systems interact by feedback control signals, eg, if a glucose perturbation occurs (eg, after a meal), β-cells secrete more insulin in response to increased plasma glucose concentration and in turn insulin signaling promotes glucose utilization and inhibits glucose production to bring rapidly and effectively plasma glucose to its pre-perturbation level. These control interactions are usually referred to as insulin sensitivity and β-cell responsivity. 13 In T1D, insulin secretion is virtually absent, while glucagon secretion from the α-cell is still preserved, which removes the insulin-dependent pathways lowering BG levels, and therefore, BG can only go up, leading to hyperglycemia (Figure 1b). Thus, insulin replacement is mandatory. A battery of counterregulatory hormones is also at work, including glucagon, epinephrine, cortisol, and growth hormone, which defend on different time scales the body from life-threatening hypoglycemia. Both hypoglycemia counterregulation and insulin control are neuromediated. However, with intensive insulin treatment, the counterregulatory defenses may fail, risking potentially severe hypoglycemia.13-17 Thus, people with T1D face a life-long optimization problem: to maintain strict glycemic control and reduce hyperglycemia, without increasing their risk for hypoglycemia. Blood glucose level is both the measurable result of this optimization and the principal feedback signal to the patient for his/her control of diabetes. This understanding of the glucose-insulin control network has been described in formal mathematical terms, which in turn led to the modeling, simulation, and control methods outlined in the following sections.
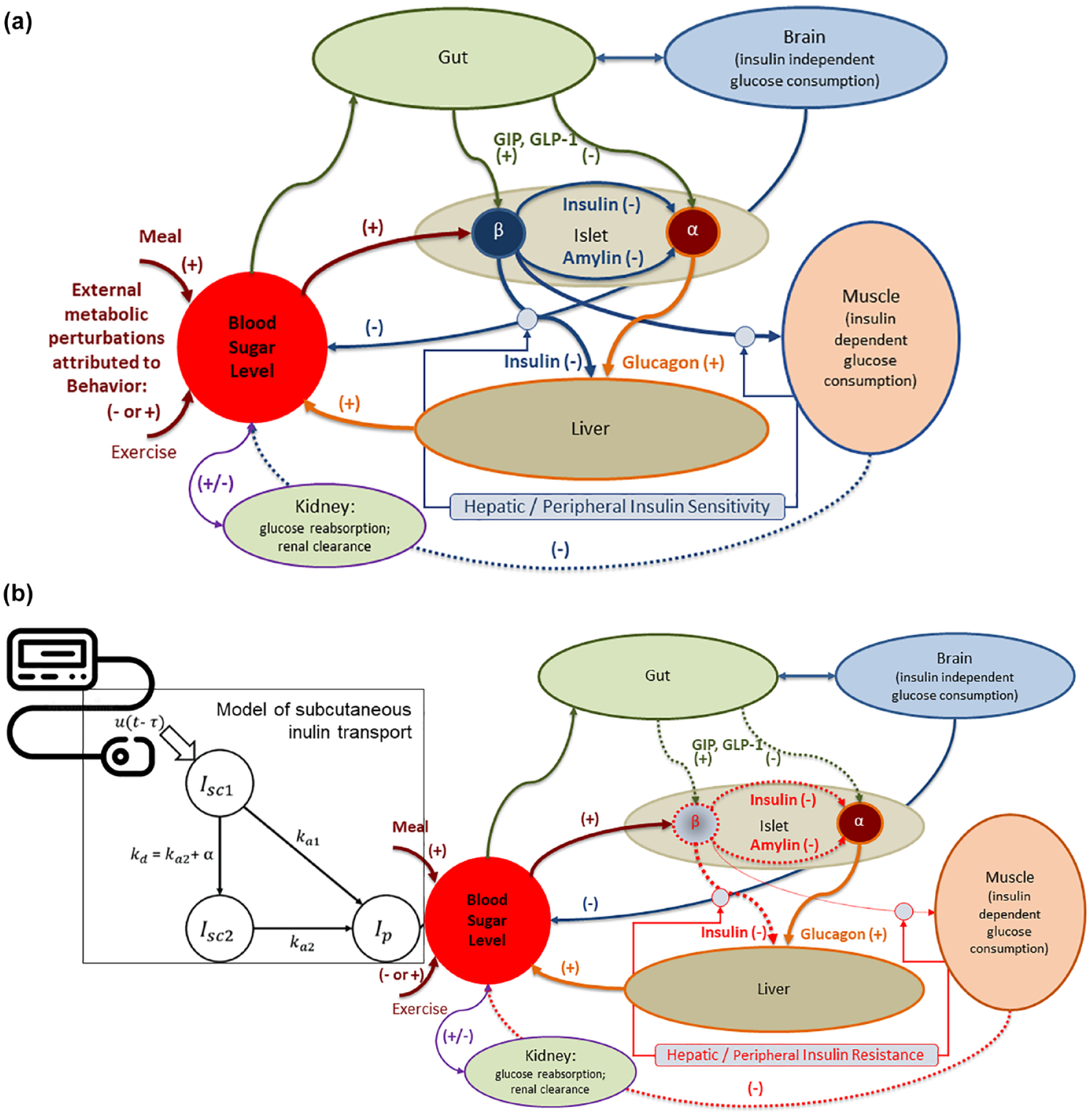
Hormonal response to metabolic disturbances in health (a) and disruption of the hormonal response to metabolic disturbances in type 1 diabetes, due to the absence of insulin secretion from the β-cell, and the need for external insulin replacement (b).
The Dawn of Diabetes Technology
Forty-two years after the discovery of insulin, an insulin pump delivering insulin and glucagon (to counteract hypoglycemia) was designed by Kadish. 18 In 1969, the first portable BG meter—the Ames Reflectance meter—was manufactured. The first commercial subcutaneous (SC) insulin pump—the Auto Syringe—was introduced by Dean Kamen in the 1970s, and by the end of the decade, the first trials of continuous subcutaneous insulin infusion (CSII) were reported by Pickup et al 19 in the United Kingdom and Tamborlane et al 20 in the United States, showing the feasibility of this minimally invasive mode of insulin replacement. The next logical step was automated insulin delivery (AID) controlled by a mathematical algorithm—a method that became known as closed-loop control or the “artificial pancreas (AP).” The AP idea can be traced back to the 1970s when the possibility for external BG regulation was established by studies using intravenous (i.v.) glucose measurement and i.v. infusion of glucose and insulin. Five teams reported i.v. closed-loop control results between 1974 and 1978: Albisser et al, 21 Pfeiffer et al, 22 Mirouze et al, 23 Kraegen et al, 24 and Shichiri et al. 25 In 1977, one of these designs 22 resulted in the first commercial device—the Biostator 26 —a large (refrigerator-sized) device that has been used extensively for glucose-control research. Systems such as the Biostator have been employed in hospital settings to maintain normoglycemia using negative (via insulin) and positive (via glucose or glucagon) control.27-29 A review of methods for i.v. glucose control can be found in Parker et al. 30 In 1979, another key element—the Minimal Model of Glucose Kinetics—was introduced by Bergman et al. 31 This, and subsequent mathematical models, serve as the “intelligent tool” behind the majority of control algorithms used in contemporary AP systems. Detailed description of the major early algorithm designs can be found in the literature.32-35 More work followed, spanning a range of control techniques powered by physiologic modeling and computer simulation.36-39 Between 1980 and 2000, the insulin pumps became smaller and portable, while the models of the glucose-insulin system became larger and more elaborate, allowing first computer simulation and then automated model-predictive glucose control of diabetes. In 1997 to 1998, the first elements of the risk analysis of BG data were introduced,40,41 which later became the base for closed-loop safety systems embedded in the AP design. The final critical technological leap enabling minimally invasive closed-loop was made at the turn of the 21st century with the introduction of continuous glucose monitoring (CGM)42-44—an event that started the ongoing quest for a wearable AP.
Modeling Diabetes Control
These technological developments created an environment that was very conducive to, and requiring, the use of mathematical models of the human metabolism. The Minimal Model of Glucose Kinetics (Figure 2) assumed that glucose kinetics can be described with one compartment and that remote (with respect to plasma) insulin controls both net hepatic glucose balance and peripheral glucose disposal. 31 A novel feature of the model was that insulin action did not emanate from plasma but from a compartment remote from plasma. This was a model ingredient requested by data and modeling methodology, in agreement with Sherwin et al. 45 Later, this remote compartment was experimentally proven in dog studies to be the interstitial fluid. 46 The minimal model has been widely employed—more than 1000 papers were published with its use since its introduction in 1979.
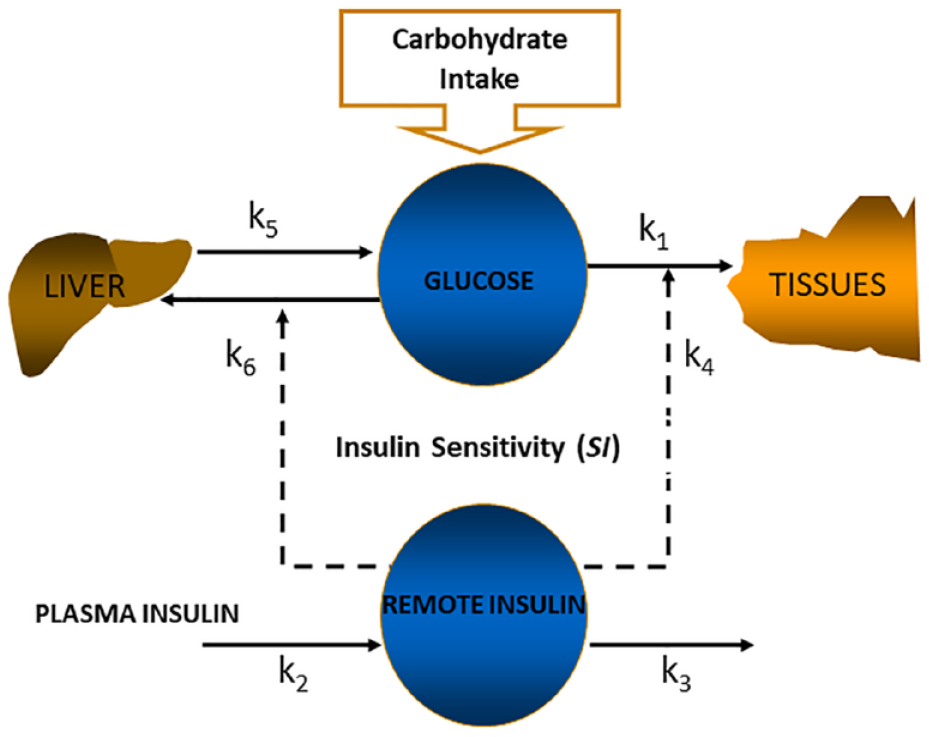
The minimal model of glucose kinetics.
Fast forward to 2006, the minimal model technology has evolved into elaborate quantitative understanding of the human metabolic system, which allowed its comprehensive description, fully implementing the body of knowledge about metabolic regulation into large, nonlinear models of high order, with a large number of parameters. 47 These sophisticated models became possible because of unique phenotypic data of carbohydrate metabolism obtained from studies conducted in >200 adults and children applying the triple tracer technique to derive key metabolic indices and parameters.48-54 The Triple Tracer Mixed Meal Test, 48 pioneered by Drs Rita and Andy Basu (now refined with the use of naturally labeled real-world food, eg, rice), and the stable-labeled oral glucose tolerance test have provided robust estimates of parameters of postprandial carbohydrate turnover in humans. These parameters include rates of endogenous glucose production (EGP), whole body glucose disappearance (Rd), and systemic appearance of meal carbohydrates (MRa). Furthermore, use of the meal minimal model in conjunction with the triple tracer method permits robust model assessments of indices of insulin action on the periphery (SI) and on the liver (SIL), glucose effectiveness (SG), insulin secretion, and β-cell function (disposition index).49-61 The triple tracer method has been tested in adolescents as well to estimate these parameters. 53 Since physical activity of any intensity modulates glucose control and postprandial glucose excursions,62,63 exercise effects on postprandial glucose turnover have been tested using the triple tracer mixed meal test in health and in T1D.62,64 The tracer-based “maximal” metabolic models enabled the next step of diabetes technology—the computer simulation of the human metabolic system.65,66 In order to do so, two steps were accomplished: step 1—the physiological understanding of the human metabolic system was converted into a compartmental model, where each compartment was a homogeneous entity in terms of glucose and insulin concentration, eg, plasma (Figure 3a) and step 2—the inter-compartment fluxes were described by a system of parameterized differential equations, including more than 30 parameters, which were the rate constants of fluxes from one compartment to another. Any physiologically realistic combination of these parameters became an in silico “subject”—complex entity of more than 30 individual parameters (Figure 3b).
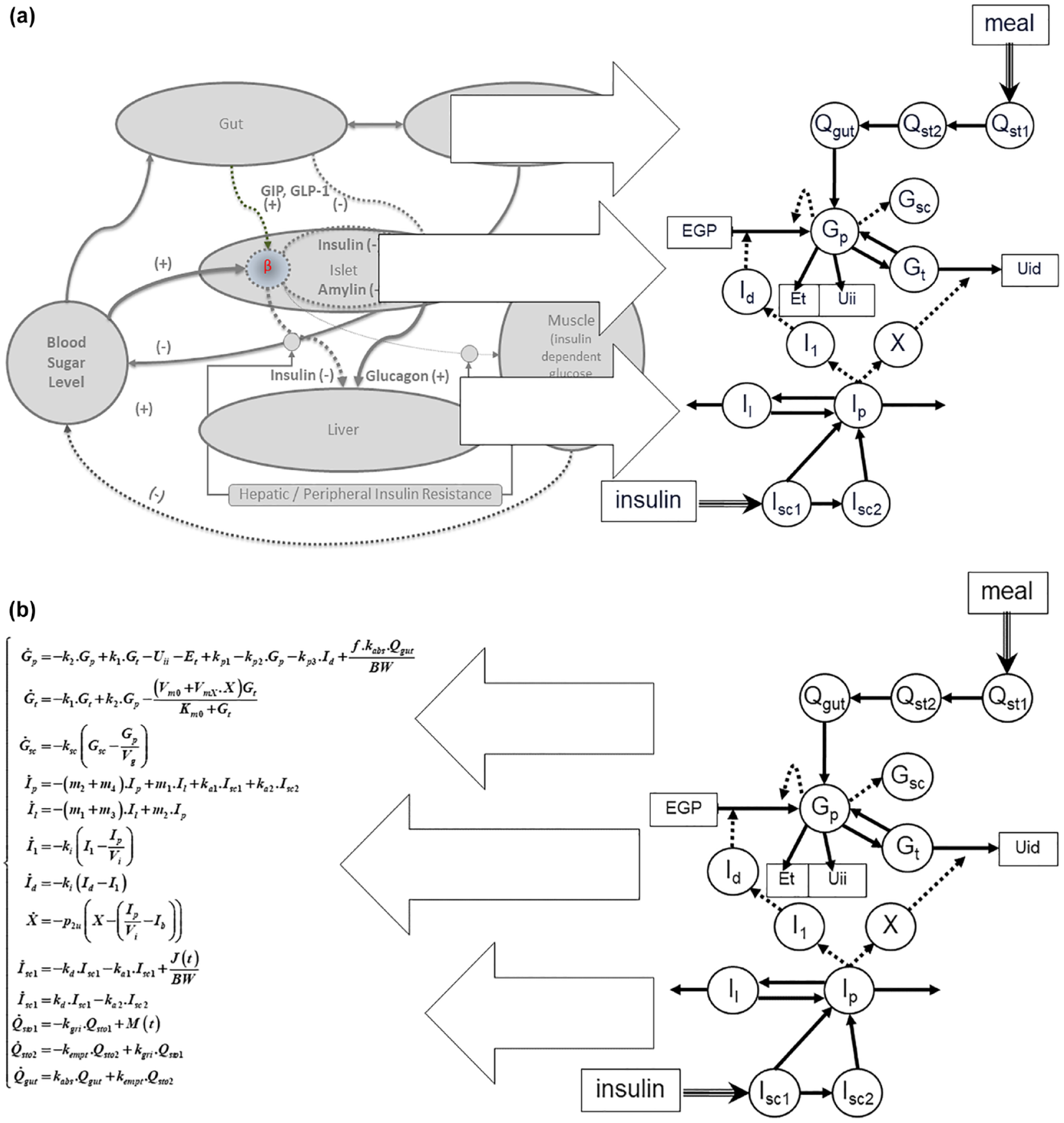
Step 1: converting the physiological understanding of the metabolic system into a compartmental model (a) and step 2: describing the inter-compartment fluxes by a system of parameterized differential equations. An in silico “subject” is a complex entity of more than 30 individual parameters (b).
Enabling In Silico Pre-clinical Trials
The regulatory validation of a metabolic simulation model was a critical step toward accelerating the progress of the AP development: simulation allowed rapid and cost-effective in silico testing of closed-loop control algorithms. The first simulation environment circa 2007 was based on the previously introduced Meal Model of glucose-insulin dynamics 47 and was equipped with a “population” of in silico images of N = 300 “subjects” with T1D, separated in three age groups: N = 100 simulated “children” below the age of 11; N = 100 “adolescents” 12 to 18 years old, and N = 100 “adults.” The characteristics of these “subjects” (eg, weight, daily insulin dose, carbohydrate ratio, etc.) were tailored to span a wide range of inter-subject variability approximating the variability observed in people in vivo. 66 Thus, the creation and validation of the in silico population involved proving that the parameters of the in silico population cover well key parameter distributions observed in vivo and provide comprehensive analysis of control performance.66,67 The observed distributions of key metabolic parameters are presented in Figure 4.
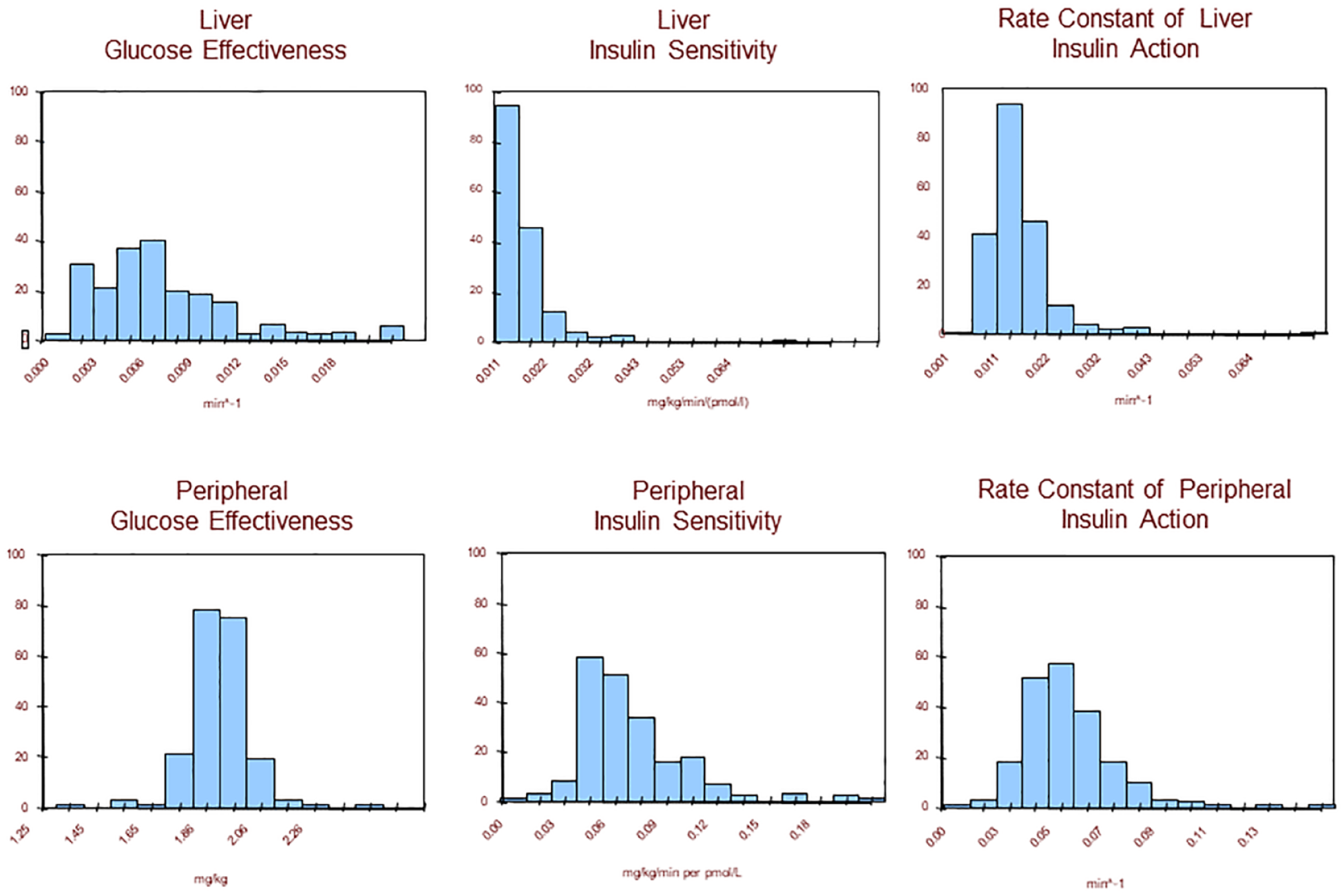
Creating an in silico population: the parameters of the in silico “population” must cover well key parameter distributions observed in vivo, thus providing comprehensive analysis of control performance.
Thereafter, simulation experiments allowed any CGM device, any insulin pump, and any control algorithm to be linked in a closed-loop system in silico, prior to their use in clinical trials. In particular, the critically important process of SC glucose transport was described by the so-called “triangular model,” as presented in Figure 1b. 68 With this technology, any meal and insulin delivery scenario could be pilot-tested very efficiently—a 24-hour period of closed-loop control is simulated in under 2 seconds. We need to emphasize, however, that good in silico performance of a control algorithm does not guarantee in vivo performance—it only helps test extreme situations and the stability of the algorithm and rule out inefficient scenarios. Thus, computer simulation is only a prerequisite to, but not a substitute for, clinical trials. In January 2008, in an unprecedented decision, the US Food and Drug Administration (FDA) accepted this computer simulator as a substitute to animal trials for the testing of closed-loop control strategies. For the first time in the diabetes field, a mathematical model equipped with 300 distinct sets of parameters (eg, 300 in silico “subjects”) became a medical device, deposited in FDA Device Master File 1521, February 2008, 67 with the following label: “Accepted for approximation of human glucose/insulin utilization, interstitial sensor performance, and SC insulin delivery, for the pre-clinical testing of insulin treatments and for the development of artificial pancreas (closed-loop control) algorithms.” This opened the field for efficient and cost-effective in silico experiments leading directly to human studies. Only three months later, in April 2008, the first human trials began at the Universities of Virginia, Montpellier (France), and Padova (Italy), using a control system designed entirely in silico. 69
Since then, the simulation environment had a number of updates and refinements that added functionality enabling increasingly sophisticated in silico treatment scenarios. These updates were based on emerging physiological data related to diurnal patterns of insulin sensitivity, dawn phenomenon, and glucagon effects amongst others.55,70-72 For example, a new model of glucagon-like peptide 1 (GLP-1) was introduced in response to meal ingestion 71 that could assist AID via mechanisms, such as suppression of glucagon secretion and delayed gastric emptying, without directly stimulating insulin secretion; the simulation environment was extended beyond its initial single-meal realm of application; 73 models of acetaminophen pharmacokinetic and long-acting insulin were introduced;74-76 the intraperitoneal (IP) absorption of insulin was modeled;77,78 and replay simulation and insulin sensitivity assessment techniques were developed.79,80 Extensive reviews of the models of the human metabolism and the simulation tools based on these models can be found in Cobelli et al.13,81
The Legacy of the Type 1 Diabetes Simulator: The Artificial Pancreas
In the years since its introduction and FDA acceptance, the simulation environment became known as the “UVA/Padova” simulator and was used in numerous in silico studies, primarily related to assessment of sensor characteristics and the development of new closed-loop control algorithms. It is safe to say that virtually all control algorithm developments done by the groups in Italy and several US sites (eg, Santa Barbara/Harvard, Illinois Tech, and the University of Virginia) were based on the UVA/Padova simulator. Many others used this tool as well. While it is not possible to review all uses of this simulation environment to date, we include a sample to 30 publications spanning 12 years, all of which used the UVA/Padova simulator as a central tool for their developments.82-111 Overall, a PubMed search (Figure 5) shows that since the introduction of the Minimal Model in 1979, about 555 papers were published on the topics of closed-loop control of diabetes or AID algorithms. Two notable dates mark the beginning of these developments: The 2005 National Institutes of Health (NIH)/Juvenile Diabetes Research Foundation (JDRF)/FDA workshop “Obstacles and Opportunities on the Road to Artificial Pancreas: Closing the Loop” and the 2008 acceptance of the UVA/Padova simulator as a substitute to animal trials for the development of closed-loop algorithms.
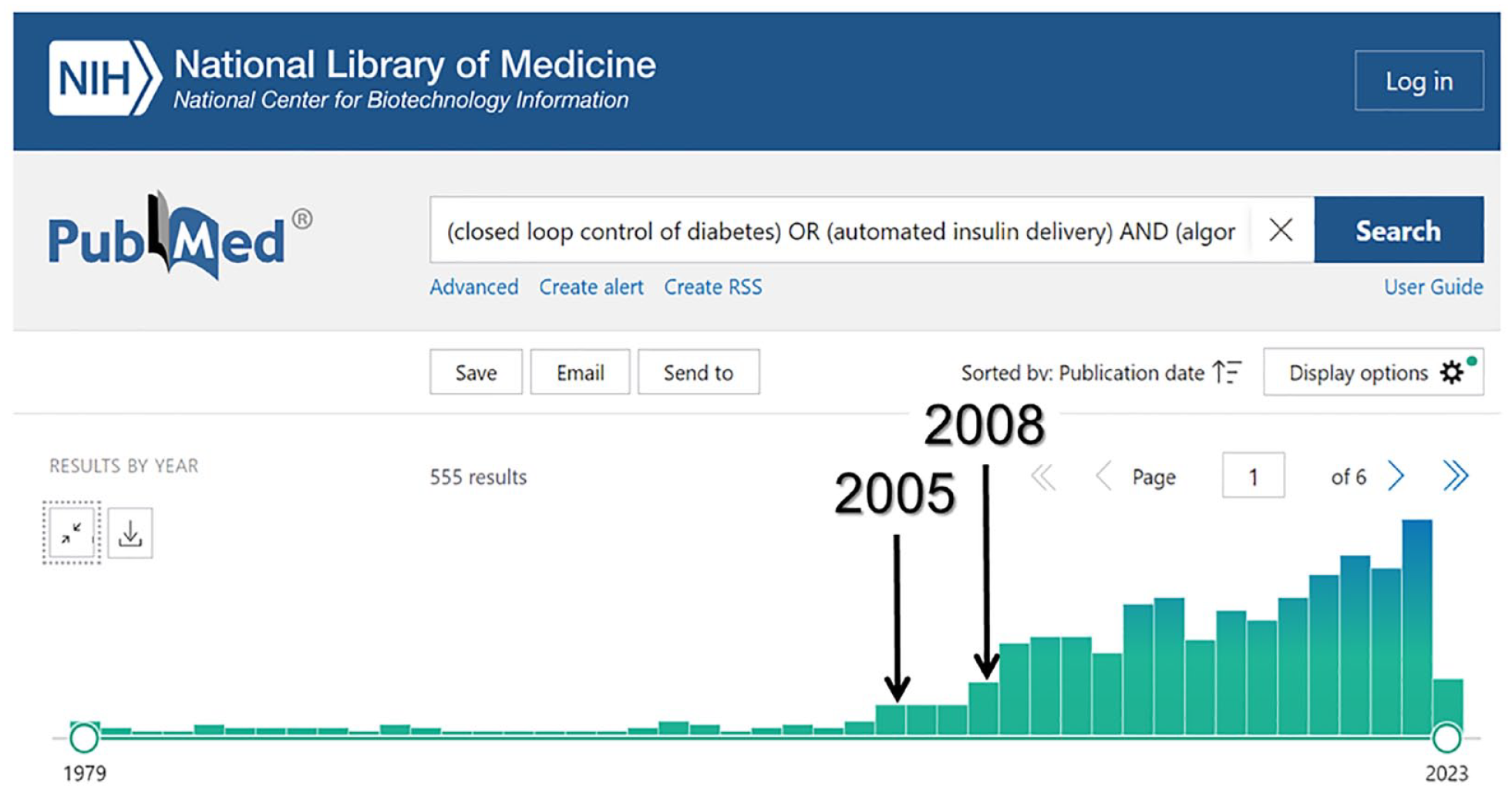
Since the introduction of the minimal model in 1979, approximately 555 papers were published on closed-loop control or automated insulin delivery algorithms. Two notable dates mark the beginning of these developments: The 2005 NIH/JDRF/FDA workshop “Obstacles and Opportunities on the Road to Artificial Pancreas: Closing the Loop” and the 2008 acceptance of the UVA/Padova simulator as a substitute to animal trials for the development of closed-loop algorithms. By 2023, six out of these proposed algorithms advanced to systems accepted in the clinical practice.
By 2023, six out of these proposed algorithms advanced to systems accepted in the clinical practice: Medtronic 670G/770G, Medtronic 780G, CamAPS FX developed in Cambridge, UK, the French system Diabeloop, Tandem’s Control-IQ, and Insulet’s Omnipod 5. 112 Two of these systems, Medtronic’s 670G/770G and Tandem’s Control-IQ, have FDA clearance for clinical use in the United States and CE mark for clinical use in Europe. Medtronic 780G,113,114 CamAPS FX,115,116 and Diabeloop 117 received CE mark for use in European countries; Omnipod 5 118 and, most recently Tidepool Loop, 119 were cleared by FDA as well. These systems are at different stages of their clinical use: while 670G/770G and Control-IQ already have hundreds of thousands of users around the world, Diabeloop and CamAPS FX are making their first strides in real-life application.
Of particular relevance to the subject of this article is the development and refinement, entirely in silico, of a series of control algorithms, which began in 2008 and continues today: The first wearable AID system was the Diabetes Assistant (DiAs) developed by our team at the University of Virginia in 2011. DiAs was equipped with a first-generation control algorithm designed using the UVA/Padova simulation and used in a number of clinical trials at ten clinical centers in the United States and Europe120-128 including (1) a long-term six-month outpatient pilot study; 126 (2) a successful use of AID in high-risk patients, ie, those who experienced frequent hypoglycemia or had hypoglycemia unawareness or history of severe hypoglycemia, 127 and (3) the first “stress test” of an AID system—the Artificial Pancreas Ski Trial—which involved children with T1D wearing DiAs during five-day winter-sport camps in Virginia, Colorado, and California to show that the use of AID under extreme conditions is safe and effective. 128 In July 2016, Diabetes Care published a symposium, 129 which featured several outpatient AID studies using DiAs running in two different in silico designed algorithm configurations.123-125
In 2015, NIH/National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) funded four major research efforts to test advanced AID systems. One of these studies became the International Diabetes Closed-Loop (iDCL) Trial, which tested two algorithms originally designed in silico.130,131 This large-scale study and its continuation with young children ages 2 to 6 years old completed several clinical protocols, three of which were published in the New England Journal of Medicine.132-134 These prestigious publications reported pivotal trials of the commercial descendant of DiAs—a new AID system (Control-IQ by Tandem Diabetes Care)—in different age groups: 14 years old and older, 132 6 to 13 years old, 133 and 2 to 6 years old. 134 As a result, in 2019, the FDA authorized the Control-IQ system as the “first interoperable, automated insulin dosing controller.” 135 Today, Control-IQ is cleared in over 25 countries and has over 400,000 users around the world. Two publications of real-life data, one reporting 9300 users for a year 136 and the other 21,000 users for four months, 137 confirmed that the system performance in real life is virtually identical to its performance in the pivotal clinical trials.132-134,136,137
A recent development of the simulator has been stimulated by the European Horizon 2020 project FORGETDIABETES 138 which aims to develop a fully implantable, fully automated, physiologic AP targeting IP glucose sensing and IP insulin delivery. In fact, the improvement of glycemic control through SC insulin delivery does not come without a cost: individuals with T1D are exposed to peripheral nonphysiologic hyperinsulinemia that, over time, is a major contributor to macrovascular complications, including heart failure and macrovascular disease that are still 3 times more frequent in those with T1D respect to their healthy peers (see Gregory 139 also for pertinent literature review). These diseases represent the first cause of mortality even for those who achieve optimal glycemic control. 140 SC insulin delivery creates a paradoxical peripheral hyperinsulinemia necessary to achieve minimal insulin concentration in the portal system able to inhibit hepatic glucose production, thus preventing fasting hyperglycemia. This observation has prompted, over the past decade, the development of dedicated pumps for IP insulin delivery, which provide a more physiological way to optimize glycemic control and prevent peripheral hyperinsulinemia (Figure 6). Inpatient 24-hour trials with three unannounced meals demonstrated that an AP with IP infusion can achieve significantly better glucose control compared to SC infusion and spare meal announcement, 141 due to the fast insulin delivery route represented by the peritoneum. This calls for novel control algorithms able to account for the specificities of IP insulin kinetics and IP glucose sensing.
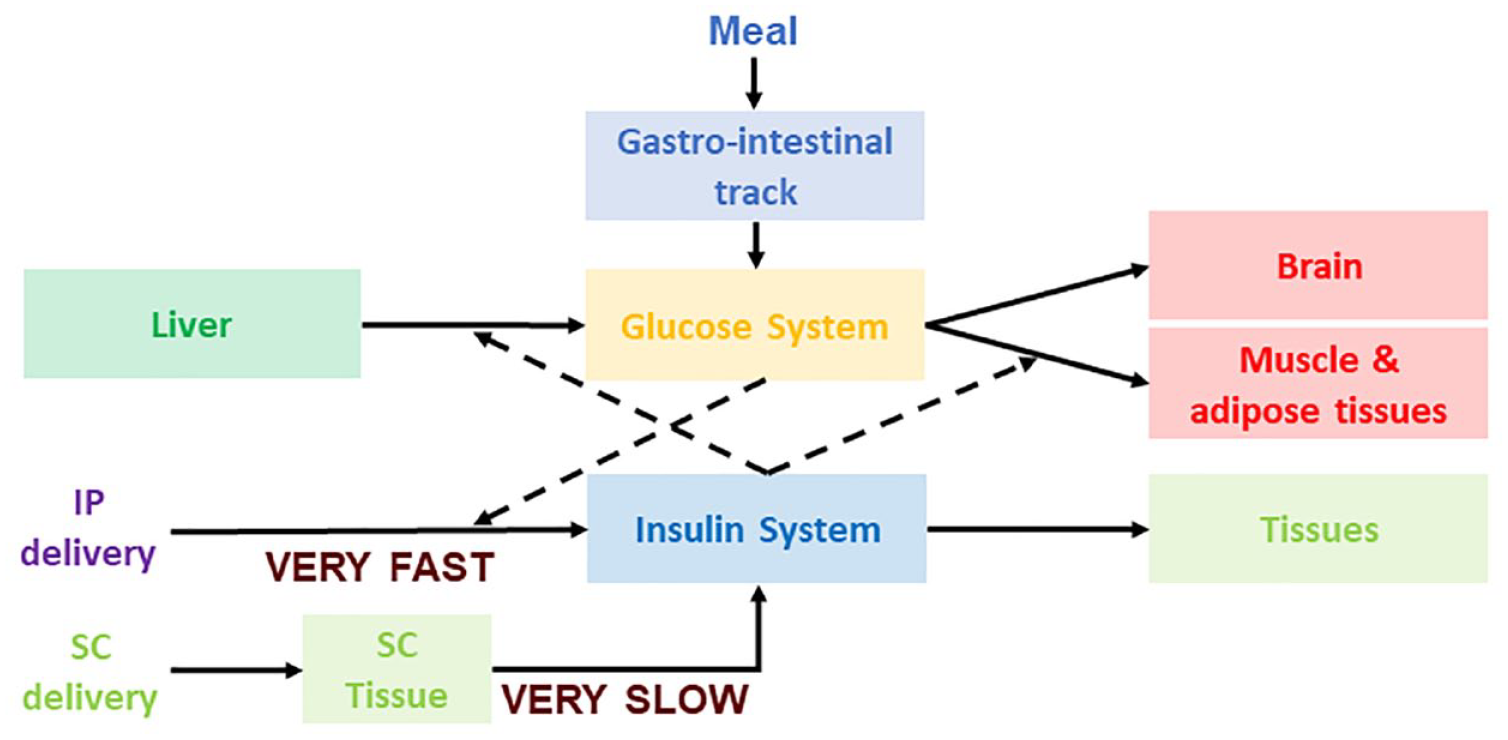
A schematic view of subcutaneous vs intraperitoneal insulin delivery.
The FDA-accepted UVA/Padova T1D simulator for SC insulin delivery and sensing 73 has been updated for IP insulin delivery and IP glucose sensing. In detail, we developed a two-compartment model of IP insulin kinetics, proving that the peritoneal space acts as a virtual compartment and IP insulin delivery is virtually an intraportal (intrahepatic) delivery, thus mimicking the physiology of insulin secretion. 78 As regards IP sensing, we simulated the model proposed by Burnett et al 142 which has an average time delay of 0.68 minutes and a time constant of 5.6 minutes. The inter- and intra-day insulin sensitivity variability of the FDA-accepted population 73 was retained. This IP simulator has been used in an in silico (100 adults) study of IP vs SC insulin delivery 143 and to design and validate a proportional-integral-derivative (PID) controller to guide IP insulin delivery in a fully closed-loop mode that does not require meal announcement. 144
Discussion
The 2008 FDA acceptance of the UVA/University of Padova computer simulator as a substitute to animal trials in the design and safety pre-clinical testing of AP controllers opened the field for rapid and cost-effective in silico experiments. Since then, virtually no animal experiments have been conducted for the purpose of designing AP algorithms. Thus, we can expect that in silico testing will permanently replace animal trials in the development of future diabetes technologies. Virtual environments will be created that will allow thorough testing of myriad control cycles per second in extreme physiological situations, or during low-probability system failures, that cannot be reproduced in real life. The AID algorithms designed with the assistance of in silico experiments, stood the test of time and are now in routine clinical use, alleviating the burden of T1D for hundreds of thousands of people.
Footnotes
Abbreviations
ADA, American Diabetes Association; AID, automated insulin delivery; AP, artificial pancreas; BG, blood glucose; CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; DiAs, Diabetes Assistant; DTS, Diabetes Technology Society; EASD, European Association for the Study of Diabetes; EGP, endogenous glucose production; FDA, Food and Drug Administration; GLP-1, glucagon-like peptide 1; iDCL, International Diabetes Closed-Loop; IDF, International Diabetes Federation; IP, intraperitoneal; JDRF, Juvenile Diabetes Research Foundation; MRa, meal carbohydrates rate of appearance; NIH, National Institutes of Health; Rd, glucose disappearance; SC, subcutaneous; SG, glucose effectiveness; SI, insulin action on the periphery; SIL, insulin action on the liver; T1D, type 1 diabetes; UVA, University of Virginia.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BK declares research support handled by the University of Virginia by Dexcom, Novo Nordisk, Tandem Diabetes Care; patent royalties handled by the University of Virginia by Dexcom, LifeScan, Novo Nordisk, and Sanofi. Honoraria: Tandem. CC declares consulting by Indigo and Ypsomed.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The writing of this review was supported by grants RO1 DK 133148 from the National Institute of Diabetes and Digestive Diseases and the Kidney, National Institutes of Health (to BK) and European Commission Horizon 2020, FORGETDIABETES-FET-EU951933 (to CC).
