Abstract
The introduction of automated insulin delivery (AID) systems has enabled increasing numbers of individuals with type 1 diabetes (T1D) to improve their glycemic control largely. However, use of AID systems is limited due to their complexity and costs associated. The user must wear both a continuously monitoring glucose system and an insulin infusion pump. The glucose sensor and the insulin catheter must be inserted at two different body sites using different insertion devices. In addition, the user must pair and manage the different systems. These communicate with the AID software implemented on the pump or on a third device such as a dedicated display device or smart phone application. These components might be developed and commercialized by different manufacturers, which in turn can cause difficulties for patients seeking technical support. A possible solution to these challenges would be to integrate the glucose sensor and insulin catheter into a single device. This would allow the glucose sensor and insulin catheter to be inserted simultaneously, eliminating the need for pairing, and simplifying system management. In recent years, different technologies have been developed and evaluated in clinical investigations that combine the glucose sensor and the insulin catheter in one platform. The consistent finding of all these studies is that integration has no adverse effect on insulin infusion and glucose measurements provided that certain conditions are met. In this review, we discuss the perceived challenges of such an approach and discuss possible solutions that have been proposed.
Keywords
Introduction
Ongoing innovation in continuous glucose monitoring, insulin treatment technologies and automated insulin delivery (AID) algorithms in the last 15 years has led to the development of AID systems that enable users with type 1 diabetes (T1D) to significantly optimize their glucose control in daily life. Clinical studies have shown significant improvements in glycemic outcome across all age groups, gender, diabetes duration, prior insulin delivery modality, or baseline HbA1c.1-5 Respective analysis have also suggested cost-effectiveness of AID systems.6-9
An AID system automatically adjusts insulin infusion rates of an insulin pump between meals and during the night in response to current and predicted glucose levels as measured by a continuous glucose monitoring (CGM) system. CGM systems provide immediate and continuous feedback to users, displaying current glucose levels, recent trends, and rate-of-change trend arrows that indicate the direction and magnitude of glucose changes. As an added safeguard, programmable alerts warn users of immediate and impending acute glycemic events.
Nevertheless, use of commercially available AID systems requires users to carry around a number of devices at all times and insert the glucose sensor of the CGM system and the catheter of the insulin infusion set at different body sites.10,11 The insertion of the sensor and the insulin catheter is supported by different insertion devices. This imposes an added burden on patients, causing unnecessary pain, increase risk of infection or skin problems, and may result in diminished freedom of movement.12,13 If the user needs technical support, additional complexity arises when different manufacturers supply the components to a given AID system. If the system or one of these components fails, the question arises about whom to contact (also from a liability point of view). Other disadvantages of combining products from different manufacturers into one AID system are increased plastic waste and costs. All these factors can contribute to suboptimal adherence in using AID systems.
Such considerations have prompted developers in the academic and commercial sector to explore the feasibility of combining glucose sensing and insulin infusion at the same body site in a single platform (Table 1). In earlier studies, microdialysis or microperfusion catheters were inserted into the subcutaneous tissue to enable collection of interstitial fluid (ISF) samples for extracorporeal glucose measurement.14-16,18 In some studies, the same catheter was used to infuse the insulin into the tissue, so that a separate infusion catheter was not needed. In a different approach, the glucose sensor was either inserted into the lumen of the insulin catheter or the sensor was applied to the outer walls of the insulin catheter.19-23,27-29,31 Both approaches have in common that only one device had to be inserted (“single-port device”). In another approach, the glucose sensor and insulin catheter were inserted as separate components at a defined horizontal distance from each other (“dual-port device”) (Figure 1).17,24-26,30
Summary of Studies Investigating Single Glucose Sensing/Insulin Infusion Platform.
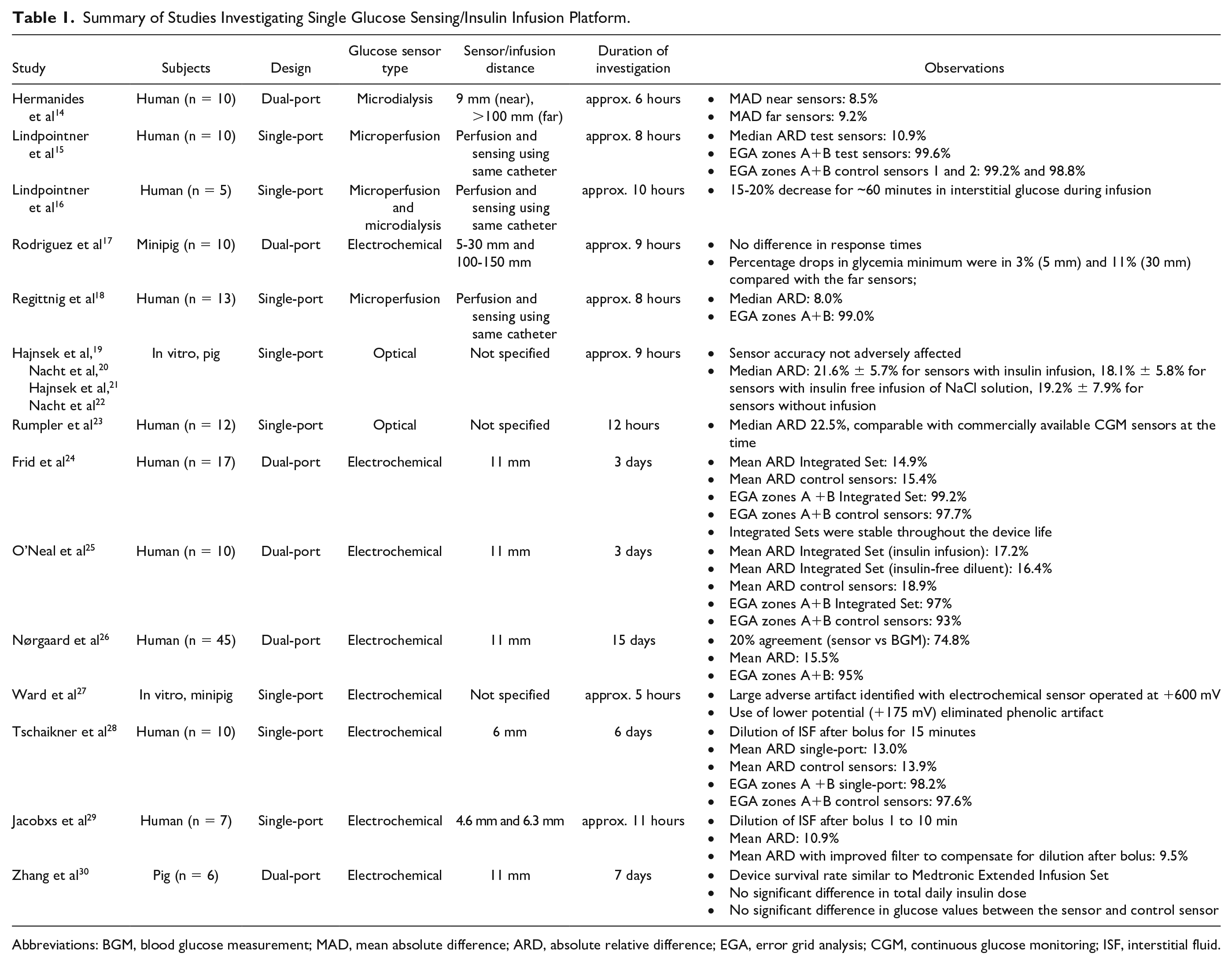
Abbreviations: BGM, blood glucose measurement; MAD, mean absolute difference; ARD, absolute relative difference; EGA, error grid analysis; CGM, continuous glucose monitoring; ISF, interstitial fluid.
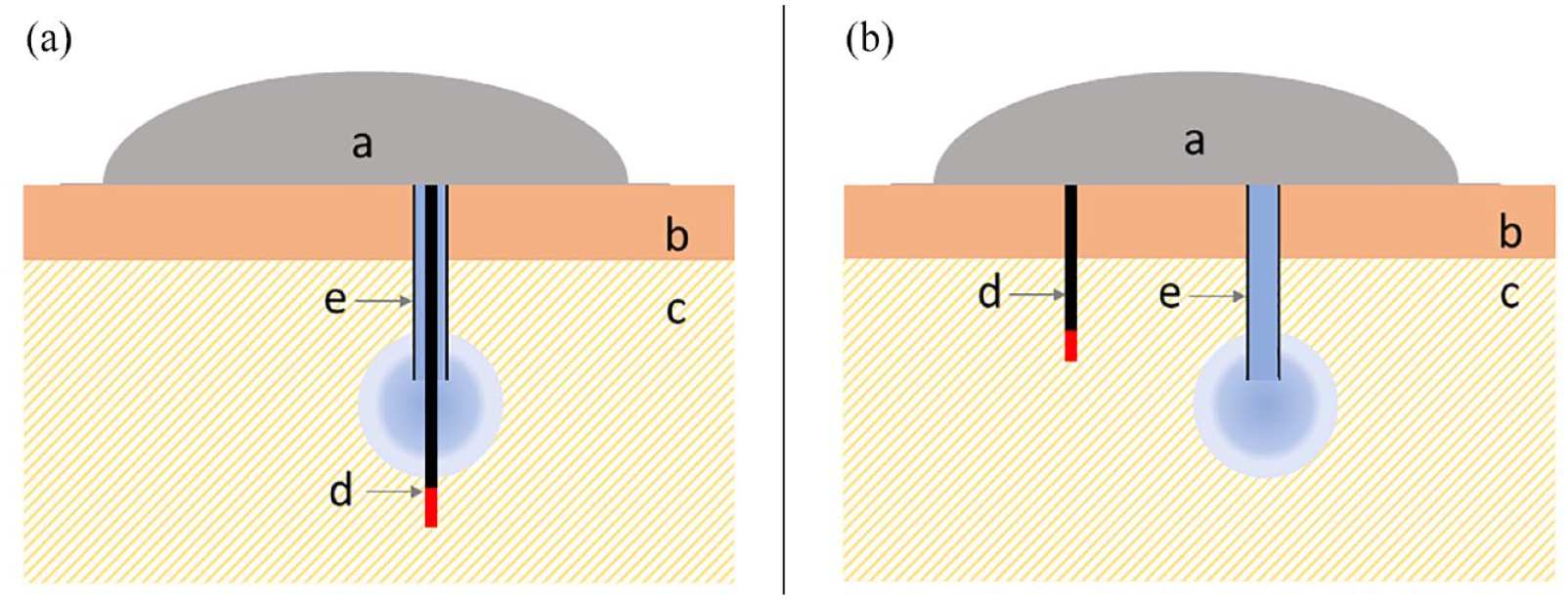
Schematic drawing of single- and dual-port integrated devices. (a) single-port device; (b) dual-port device; a, integrated body-worn device; b, dermis; c, subcutaneous tissue; d, glucose sensor with glucose sensitive tip (red); e, insulin catheter.
However, combining glucose monitoring and insulin infusion into a single device brings together two different activities in close proximity. There is a risk of interactions that may lead to incorrect glucose measurement results. Essentially, three different types of interactions between insulin infusion and glucose measurement are possible:
High local insulin concentrations at the infusion site may lead to an increased uptake of glucose by adipocytes nearby, leading to a local decrease in glucose concentration (resulting in erroneously low glucose readings)
Effects of infused insulin solution in diluting glucose levels at the infusion site (resulting in erroneously low glucose readings); and
Glucose measurement interference caused by insulin and/or excipients present in commercial insulin preparations (resulting in erroneously high or low glucose readings).
To assess these challenges and discuss proposed solutions, we searched PubMed from January 1, 2000, to June 1, 2023, defining three categories with specific keywords. The categories were combined with “and,” and the keywords within each category were combined with “or.” The categories (keywords) were CGM (glucose monitoring, glucose sens*, glucose measurement, glucose sampling), insulin delivery (insulin infusion, insulin delivery, infusion cannula), integration (combin*, adjacent, conjoined, simultaneous*, single-port, single-site, integrat*, proximity, near). Of the 352 publications found, 333 were excluded because of unrelated content or language. The remaining 19 publications included in this review presented results of in vitro and preclinical studies as well as early clinical feasibility studies or discussed general aspects of the integration of glucose sensors and insulin catheters in one body-wearable platform. One publication and two poster abstracts were identified in addition through a non-systematic search.
Background
In an early study by Hermanides et al, 14 investigators inserted two microdialysis based glucose sensors (GlucoDay S; A. Menarini Diagnostics, Firenze, Italy) in the periumbilical region of 10 individuals with T1D. The mean insertion distance between the test sensor microfiber and tip of the infusion set catheter was 0.9 ± 0.2 cm. A control sensor microfiber was inserted at least 10-cm away from the test glucose sensor. Following a standardized breakfast, investigators administered an augmented insulin bolus while maintaining current basal insulin rates. An average delay of 10.4 ± 4.0 minutes was observed in the glucose values measured with the test sensor compared to the reference venous blood measurements, while the average delay between the control sensor measurements and the venous blood measurement was not significantly lower at 5.7±4.0 minutes. The difference in glucose concentration between the test and control sensors was not different as well (Figure 2). The authors concluded that microdialysis glucose monitoring can be accurately performed in the proximity of insulin infusion site.
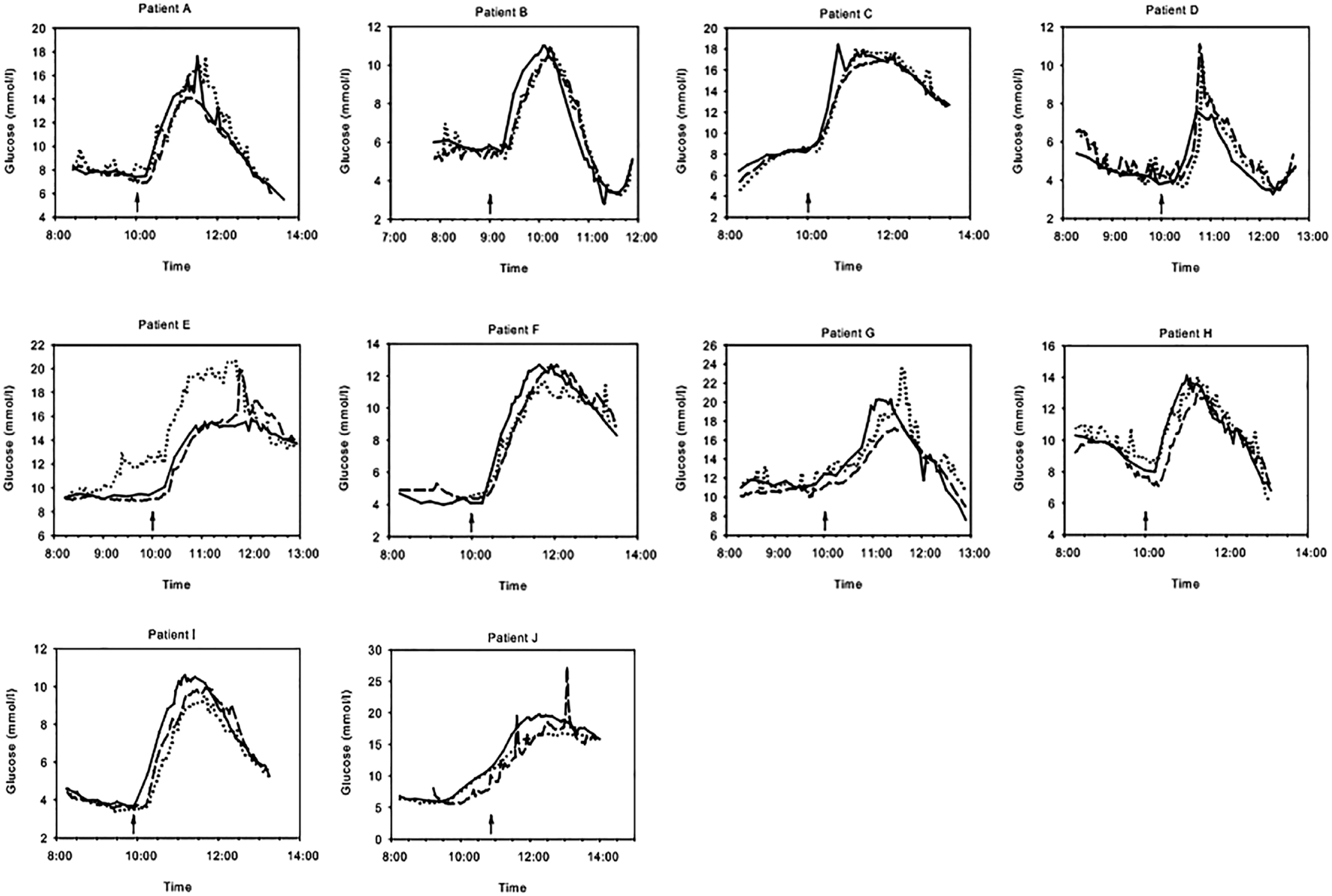
The results of all 10 subjects are displayed. The continuous line represents the blood glucose values, the dotted line represents the control sensor, and the long dashed line represents the test sensor. The time of breakfast is indicated by the arrow.
In a study done by Lindpointner et al, 15 three microperfusion catheters were inserted in 10 subjects with T1D. One test catheter was used for insulin infusion into the subcutaneous tissue and ISF sampling. As controls, the other two microperfusion catheters were inserted at a distance of >3.5 cm. They were perfused with either with insulin-free mannitol solution or used for glucose sampling only. Perfusate was sampled in 30-minute fractions for glucose analysis. Twenty minutes before participants ingested 75 g of glucose, investigators administered an insulin bolus. This was followed by continuous infusion of insulin at basal rates over the 8-hour observation period. Interstitial fluid glucose levels obtained from test catheters were compared with control catheters and with plasma glucose measurement results. A strong concordance of glucose levels measured with microperfusion catheters with plasma glucose levels was observed (Figure 3). A high percentage of paired glucose values was within the clinically acceptable accuracy error grid zones A + B, which were comparable with the two insulin-free infusion sites. Based on these results, the authors concluded that a reliable estimation of plasma glucose levels at the site of subcutaneous insulin infusion is feasible.
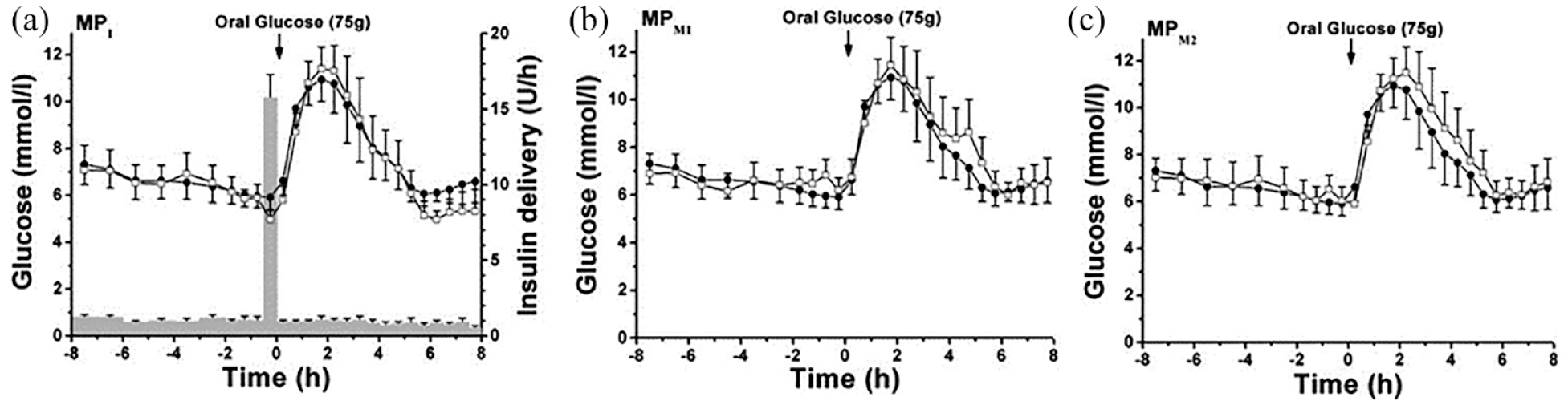
Comparison of plasma and ISF-derived glucose concentrations observed during an overnight fast and oral gucose tolerance test (OGTT) in subjects with diabetes. (a) Average time course (n = 10, means ± SE) of plasma glucose concentration (•) and the tissue glucose concentration obtained with the microperfusion (MP) catheter used for insulin delivery and simultaneous glucose sampling (MPI, □). A also shows the average time course (n = 10, means ± SE) of the insulin delivery rate (bars) used to control glucose concentration during experiments. (b and c) Average time course (n = 10, means ± SE) plasma glucose (•) and the tissue glucose obtained with the mannitol-perfused microperfusion catheters (MPM1 and MPM2, □). Abbreviation: ISF, interstitial fluid.
Another study was performed by the same research group using microdialysis and microperfusion catheters for simultaneous insulin infusion and ISF glucose sampling in five subjects without diabetes. 16 Catheter effluents were continuously collected in 30-minute fractions. Before and after the 6-hour insulin delivery period both catheters were perfused with an insulin-free solution for 2 hours and used for glucose sampling only. After the start of insulin delivery, a slight decrease in tissue glucose concentration was observed, which stabilized after ~60 minutes regardless of the infusion rate. After switching to the insulin-free solution at the end of the 6-hour insulin period, glucose concentration increased slowly and re-attained before insulin delivery levels. The authors attributed this small decline to the local effect of insulin on the tissue which becomes saturated and attained steady state values after ~60 minutes and to the experimental setup. The authors concluded that because of this saturation insulin delivery and glucose sensing may be performed simultaneously at the same site.
A subsequent study by the same group investigated the feasibility of glucose measurements at the insulin infusion site during periodic short-term interruptions of the continuous insulin infusion using microperfusion catheters in 13 subjects with T1D. 18 The results, median absolute relative difference (median ARD) 8.0 and 99% of data within Error Grid Analysis (EGA) zones A+B, showed that tissue glucose concentration at the infusion site closely reflect blood glucose levels and that this technology could be used to simplify and improve glucose management in diabetes.
Hajnsek et al19,21 developed a luminescence-based optical glucose sensor that was applied to the outer walls of an insulin catheter and first tested and optimized in vitro before being evaluated in a preclinical in vivo study in pigs.20,22 Glucose levels were clamped from 40 to 250 mg/dL by intravenous glucose infusion and subcutaneous insulin infusion via the glucose sensor. Sensor glucose values correlated well with reference blood glucose values, despite infusion at the site of glucose measurement. The average median ARD value was 21.6% ± 5.7% for sensors used for insulin infusion, 18.1% ± 5.8 % for sensors used for infusion of an insulin free NaCl solution and 19.2% ± 7.9 % for sensors without infusion. 21 After further optimization and miniaturization, two single-port systems were tested in each of 12 subjects with T1D in a clinical study. 23 Without additional data processing, the median ARD was 22.5%. The authors concluded that the results were comparable with commercially available CGM systems at the time and that the single-port system had great potential to become the central element of an artificial pancreas system.
In another study, Rodriguez et al 17 administered a bolus of diluted short-acting insulin to minipigs in the flank, where three to five glucose sensors (Freestyle Navigator; Abbott Diabetes Care, Alameda, CA) measured the subcutaneous glucose concentrations. The glucose sensors were placed 5, 10, 20, and 30 mm near the injection site and 100 or 150 mm distant from the site. No significant differences were observed between the response times to glucose changes measured by the near and far sensors. However, mixed evidence for differences in the measured glucose levels was observed. Sensors placed 5 and 30 mm from the injection site showed a percentage decrease in the glycemic minimum of 3% and 11%, respectively, compared to those placed far away (p<0.05). The sensors placed 10 and 20 mm from the infusion site did not show a decrease. No explanation for these results was provided. The limitations of this study include failure to provide calibrated glucose data and the use of highly diluted insulin (10 U/mL), the purpose of which was to allow more accurate dosing. Due to the dilution of the insulin, it is difficult to extrapolate results from this study to standard insulin concentrations (100 U/mL).
Frid et al 24 used a prototype of a dual-port device with the glucose sensor and infusion cannula 11 mm apart (MiniMed Duo; Medtronic, Inc., Northridge, CA) to assess sensor performance in subjects requiring relatively large insulin boluses (average meal bolus 13.1 ± 5.7U). The study included 17 subjects who were followed over the 3-day observation period, which included high carbohydrate meals. Investigators reported no significant difference in mean absolute relative difference (mean ARD) between the integrated sets and control glucose sensors (Enlite; Medtronic Inc., Northridge, CA) which were inserted on the other site of the abdomen (14.9% and 15.4%, respectively). The investigators concluded that glucose sensor performance of the integrated set is not affected even when recurrent large boluses are delivered.
O’Neal et al 25 evaluated the performance of the same dual-port device set as Frid et al 24 in 10 adult subjects with T1D over a 3-day observational period. A control glucose sensor (Sof-Sensor; Medtronic Inc., Northridge, CA, USA) was inserted on the other side of the abdomen. The integrated set delivered insulin-free diluent except during meal tests on days 1 and 3 when insulin was delivered as a meal bolus and post-meal basal infusion. Analyses revealed no evidence of spike artifacts during insulin or diluent infusion. The mean ARD values from the integrated platform were not significantly different from the control glucose sensor results (17.0% and 18.9%, respectively) (Figure 4). The authors concluded that a device providing for simultaneous adjacent placement of an insulin infusion catheter and a CGM sensor is feasible and functions within acceptable limits.
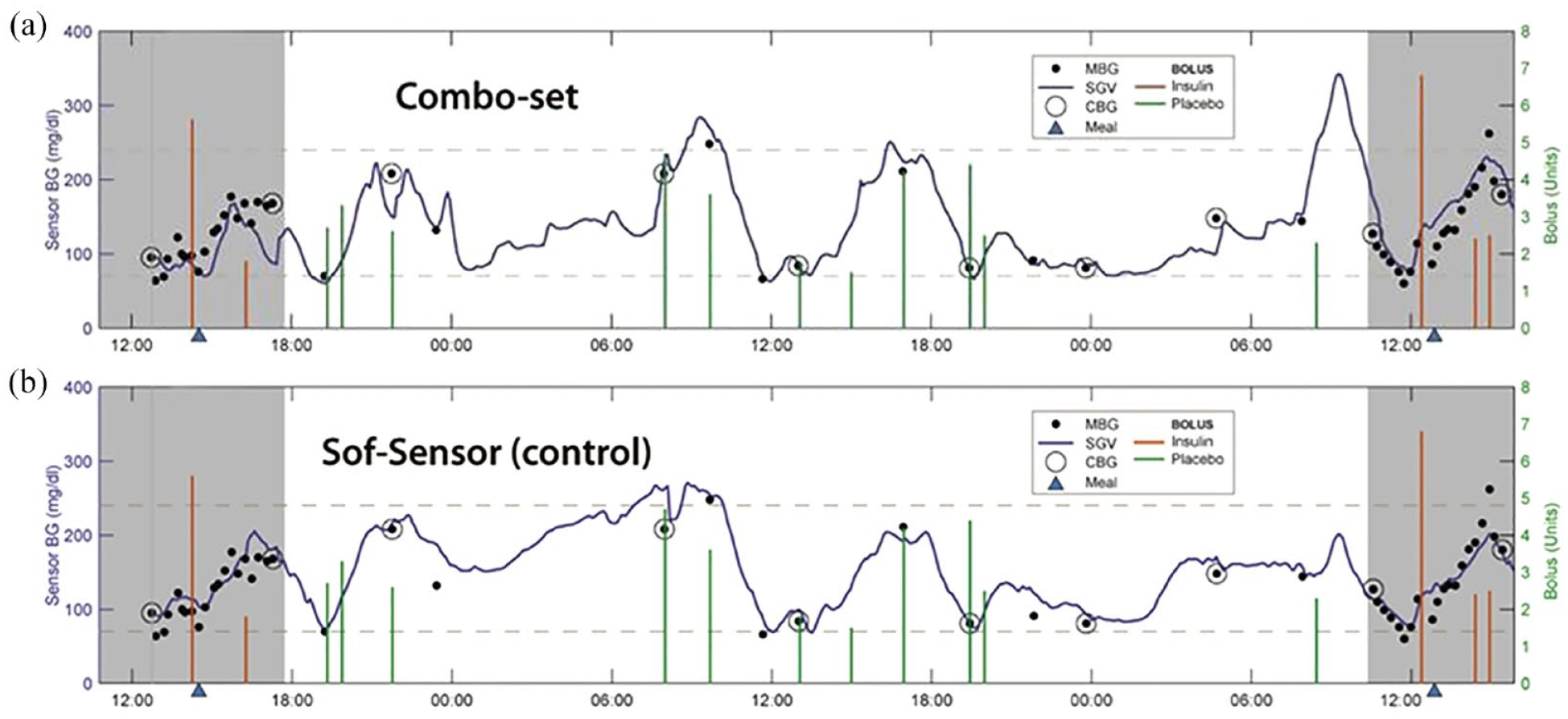
(a) Combo-Set sensor versus (b) control Sof-Sensor glucose tracings.
Nørgaard et al 26 reported findings from a study that investigated the performance and acceptability of the same integrated set as Frid et al 24 and O’Neal et al 25 Forty-five participants with T1D measured their blood glucose levels 7 times a day and the integrated set was changed every three days for 15 days. The majority (74.8%) of the paired sensor glucose—blood glucose values were within 20%, meeting the predefined primary endpoint accuracy limits. Mean ARD was 15.5% ± 17.1%, which was the typical performance of glucose sensors of CGM systems at that time when this study was performed. The authors concluded that the integrated set provided accurate glucose readings during routine clinical use, was safe to wear and was acceptable to most patients.
It has been suggested that insulin or excipients like phenol/cresol preservatives in the commercial insulin formulations interfere with the glucose measurement by direct reactions at the electrodes of electrochemical glucose sensors.25,27,29 In their investigations, Tschaikner et al 31 assessed the effects of inserting a glucose sensor into an insulin infusion set (Sof-Set; Medtronic Inc, Northridge, CA). Investigators evaluated the feasibility using glucose sensors in three different commercial CGM systems (Dexcom, Abbott, and Medtronic). No artifacts or interferences in the in vitro studies performed were observed. Based on the in vitro evaluation and design considerations, investigators selected one glucose sensor (G4 Platinum; Dexcom Inc., San Diego, CA) for a subsequent evaluation, resulting in a 6-mm distance between catheter tip and sensor tip. During a first in vivo study with this single-port device, investigators found that when insulin is administered at a high infusion rate (bolus delivery), it may dilute the ISF surrounding the glucose sensitive probe tip. Usually, it took only about 15 minutes until the insulin fluid has been absorbed and the ISF is again undiluted. 31
The single-port device was then used by the same group in a clinical study with 10 subjects with T1D. 28 Subjects were using the device for up to six days in their home/work environment for open-loop insulin delivery and glucose sensing. On one additional day, the device was used in combination with an algorithm to perform automated glucose control under hospital settings. To mitigate the effect of ISF dilution after bolus delivery, the insulin solution was spiked with glucose to obtain a glucose concentration of 200 mg/dL. The average mean ARD from capillary blood glucose concentrations did not differ from that of the additionally worn control CGM system (Dexcom G4 platinum) which was inserted >100 mm from the insertion site of the single-port device (13.0% and 13.9%, respectively). In the error grid analysis, 98.2% of the data from the single-port system fell into the clinically acceptable zones A+B, which again was comparable with the results obtained with the control system (97.6%). The authors concluded that insulin delivery and glucose sensing with the single-port device was reliable and safe in the open-loop and closed-loop setting.
Ward et al 27 investigated the interference effects of using a single-port device, with the glucose electrode laminated to the outer wall of the insulin delivery cannula. Using a glucose sensor operated at +600 mV, they identified a large sensor artifact in the presence of insulin resulting in subsequent irreversible electrode poisoning in vitro. Subsequent work revealed that this effect was due to the oxidation of phenolic preservatives at the electrodes. Use of a lower potential (+175 mV), made possible by a change in sensor chemistry, effectively eliminated the phenolic artifact.
In a more recent study with seven subjects with T1D, the same group assessed the accuracy of the single-port glucose sensing/insulin infusion cannula compared with a control sensor to evaluate whether there was an artifact caused by the delivery of either insulin or insulin-free solution (phosphate buffered saline [PBS]). 29 Two different experimental glucose sensing/insulin infusion cannula designs were tested. In the first design, the sensor was 4.6 mm from the infusion tip (distal sensor design) and in the second design the sensor was laminated 6.3 mm from the tip (proximal sensor design). Investigators observed a small and transient artifact in both conditions that increased with larger volumes of fluid; however, there was no difference between the artifacts in the sensing cannula delivering insulin compared with the sensing cannula delivering insulin-free PBS. The authors concluded that this artifact is due to the dilution of glucose in the ISF. The time for the sensor to recover from the artifact was longer for larger fluid amounts compared with smaller fluid amounts (10.3 ± 8.5 minutes vs 41.2 ± 78.3 seconds). Sensor accuracy was improved using a smart-sampling Kalman filtering smoothing algorithm from mean ARD 10.9% to 9.5 %. The authors concluded that despite this small and transient artifact after bolus, which is very likely due to dilution by fluid delivery, it is possible to continuously measure glucose in a cannula that simultaneously applies insulin.
Discussion
The aim of this review is to critically question whether it is possible to integrate the glucose sensor and insulin infusion components required for AID systems into a single body-worn device. Surprisingly, all studies conducted so far indicate that integration is possible and previously held concerns could not be confirmed.
Given the high local insulin concentration in the subcutaneous tissue at the infusion site, one might expect a local effect of insulin to markedly lower the local concentration of glucose. Interestingly, only Lindpointner et al 16 identified a slight reversible decrease of 17% to 23% in tissue glucose concentration after start of insulin infusion in five subjects without diabetes. The investigators attributed this decrease to the biological action of insulin. However, this effect stabilized after approximately 60 minutes and remained stable even with changing insulin infusion rates. The researchers hypothesized that after this equilibration period, the local effect of insulin on tissue glucose concentration saturates and reaches steady-state values.
Possible explanations for the fact that a significant local reduction in glucose concentration due to insulin-induced glucose uptake by adipocytes has not yet been clearly demonstrated and that this effect is therefore much smaller than originally expected were discussed by Ward et al 32 :
1. Slow glucose uptake into adipocytes
As reported in early studies, glucose uptake by muscle tissue is approximately 75 times greater than uptake in adipose tissue,33,34 whereas adipose tissue is responsible for <1% of glucose uptake in hyperinsulinemic people without diabetes. 35
2. Competition between insulin absorption into circulating blood and local action
Because of the quite low glucose uptake in adipose tissue and the large capillary blood flow (3-5 mL per 100 g of tissue per minute, which is 2- to 3-fold higher than in muscle tissue), 36 it is likely that rapid absorption of injected insulin into capillaries successfully competes with the local action of insulin in adipocytes.
3. The reduced effects of insulin at high concentrations
In earlier studies, it was discovered that when insulin-responsive cells were exposed to high concentrations of insulin, their response per unit of insulin immediately fell to low levels.37-39 This effect was called negative cooperativity and interestingly is never operative during normal physiology, only at pharmaceutical insulin concentrations such as injection into fat tissue.
Jacobs et al 29 and Tschaikner et al 31 demonstrated a transient drop of 1 to 15 minutes in glucose level because of ISF dilution after delivery of an insulin bolus. Jacobs et al 29 were able to reduce this effect and improve the accuracy of the system using improved data filter algorithms. Other solutions to avoid transient erroneous glucose sensor readings were discussed by Tschaikner at al. 31 and included administering an extended insulin bolus instead of a normal bolus, using predictive algorithms to compensate for the transient drop, adding glucose to insulin, or simply ignoring glucose sensor readings for about 15 minutes after the insulin bolus. If the sensor needs to be user calibrated a calibration immediately after bolus delivery must be avoided to prevent erroneous sensor readings after the dilution effect has disappeared. An effect of the transient dilution on the calibration of a factory calibrated sensor is unlikely but remains to be investigated.
In vitro studies done by Ward et al 27 gave strong evidence that phenolic components, such as those added as preservatives to common insulin preparations, can oxidize and subsequently may poison the electrodes of the glucose sensor. There was no evidence that insulin itself or other excipients of the insulin formulation caused the interference. However, Tschaikner et al 31 and Ward et al 27 have clearly shown that the design of the electrochemical sensor can effectively prevent interference and subsequent poisoning of the electrodes by phenolic preservatives. An appropriately designed membrane system can prevent phenolic components from diffusing to the electrode surface, and the use of redox mediators allows the glucose sensor to be operated at low electrode potentials, which are not sufficient for the oxidation of the phenolic components.
All clinical studies performed so far have certain limitations. The effects of glucose monitoring adjacent to the infusion site on insulin infusion have not been studied specifically. However, in a preclinical study Zhang et al 30 observed no significant difference in total daily insulin dose between the integrated MiniMed Duo set and the Extended Wear Infusion Set (Medtronic Inc, Northridge, CA). Regardless of study duration, which ranged from a few hours in a clinic setting to 15 days in a home setting (Table 1), no abnormal failure rates were reported for either insulin infusion or glucose monitoring. However, a glucose sensor integrated within the insulin catheter as investigated by Tschaikner et al28,31 may lead to increased back pressure and higher probability of leakage, or occlusion of the insulin catheter. To proof robust and safe systems worn on the body 24/7, longer-term studies are required. A further limitation is that most studies analyzed only the calibrated sensor signal. Only Rodriguez et al 17 investigated the impact on the raw signal of the sensor during insulin infusion which could give additional, more detailed insights. All studies used the periumbilical region as the insertion site. CGM sites are expanding to include the back of the arm, where there may be less adipose tissue, raising the question of whether the results can be extrapolated to other areas of the body. Another general weakness is that in practice it is difficult to maintain the exact intended distance between the insulin catheter and the glucose sensor in the tissue. A soft catheter, in particular, can bend after insertion. The intended distance to the glucose sensor may increase or decrease unnoticed.
The integration of a CGM sensor into an insulin infusion set or patch pump may result in a larger form factor of the body-worn device. However, given the already high degree of miniaturization of newer CGM systems, it can be expected that the size of an integrated device will not be significantly larger than that of previous CGM patches or insulin patch pumps.
Although the current evidence is both conclusive and compelling, questions remain regarding the most effective design approaches. Microdialysis and microperfusion catheters, as used in the early studies, have the advantage that they can be used simultaneously for insulin infusion and ISF sampling followed by glucose analysis.15,16,18 However, such catheters are relatively large compared to current CGM sensors and insulin infusion catheters and require a complex fluidic management (eg, precision pumps, tubes, valves, reservoir- and waste-containers). As a result, these systems would be quite difficult to miniaturize and expensive to manufacture. This makes it unlikely that these technologies will be used to design an integrated AID platform. Rumpler et al 23 and Jacobs et al 29 have shown that novel optical and electrochemical glucose sensors can be applied directly to the walls of the insulin infusion catheter. This allows for the most minimal invasive approach for an integrated AID platform. As promising as the published data are, history shows how long it takes to develop glucose sensors that are reliable and accurate enough for an AID system. Thus, despite the advantage of miniaturization and minimal invasiveness, it will be some time before these novel highly integrated systems will be approved as part of an AID system. Tschaikner et al28,31 used commercially available glucose sensors and insulin infusion catheters to design an integrated single-port system. A disadvantage of this design is the length of the integrated glucose sensor/insulin catheter required to provide adequate distance between the glucose sensor and insulin infusion tip, which may cause pain and discomfort, especially in lean T1D users. The most promising approach for the realization of a first-generation product is therefore the dual-port design, in which a separate, already approved glucose sensor and an insulin catheter are integrated into a single body-worn platform and inserted with a common insertion mechanism. The body-worn platform can be based on an insulin infusion set in combination with a durable insulin pump as used in Fried et al, 24 O’Neal et al, 25 and Nørgaard et al 26 or an insulin patch pump.
It is an interesting question why, despite the promising evidence presented before, there are still no AID systems commercially available that integrate the glucose sensor and the insulin catheter in one body-worn device. One possible reason why early technologies based on microdialysis or microperfusion did not catch on may be that these systems are too expensive to manufacture due to the complex fluidics required. Another reason may be that the duration of use of glucose sensors and insulin infusion sets, from seven to 14 days and until recently three days respectively, was too different to make integration cost-effective. 40 Replacing a combined platform every three days would also affect sensor accuracy, as all commercially available CGM sensors have limited accuracy on the first day. 40 The recent approval of novel infusion sets that last up to seven days and the fact that manufacturing costs can be saved by integrating CGM and insulin pump technology together with the AID algorithms into a single body-worn device may change the situation in the future.
Conclusion
Several clinical studies have demonstrated the feasibility of combining a glucose sensor and an insulin catheter into a single body-worn device. Challenges associated with integrating glucose sensing and insulin infusion have been successfully addressed by designing devices that maintain a distance of 5 to 11 mm between the glucose sensing electrodes and the insulin infusion catheter, whether it is a single-port or dual-port device. A well-designed system should also incorporate predictive algorithms that compensate for the temporary dilution of glucose in the ISF after bolus insulin delivery. In addition, electrochemical sensor technology should be used that does not interfere with the substances contained in the insulin formulations. Based on these considerations, the development of a fully integrated glucose sensing/insulin infusion device appears to be feasible. A device like this will have less components and be easier to use than existing AID systems and therefore significantly improve user acceptance and ultimately improve clinical outcomes of a broader base of users.
Footnotes
Acknowledgements
We thank Chris Parkin, CGParkin Communications, Inc. for his assistance in writing this publication and Stephanie Wehrstedt, Institut für Diabetes Technologie Forschungs-und Entwicklungsgesellschaft mbH an der Universität Ulm, for conducting the literature search.
Abbreviations
AID, automated insulin delivery; ARD, absolute relative difference; BGM, blood glucose measurement; CGM, continuous glucose monitoring; EGA, error grid analysis; ISF, interstitial fluid; MAD, mean absolute difference; MARD, mean absolute relative difference; PBS, phosphate buffered saline; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MS and AM are full-time employees of PharmaSens AG; JKM is a member of advisory boards of Abbott Diabetes Care, Becton-Dickinson, Boehringer Ingelheim, Eli Lilly, Embecta, Medtronic, NovoNordisk A/S, Roche Diabetes Care, Sanofi-Aventis, Viatris and received speaker honoraria from A. Menarini Diagnostics, Abbott Diabetes Care, AstraZeneca, Boehringer Ingelheim, Dexcom, Eli Lilly, Medtrust, MSD, NovoNordisk A/S, Roche Diabetes Care, Sanofi, Servier, and Ypsomed; she is shareholder of decide Clinical Software GmbH and elyte Diagnostics where she also serves as CMO; GF has received speakers honoraria or consulting fees from Abbott, Ascensia, Berlin Chemie, Boydsense, Dexcom, Lilly, Metronom, MySugr, Novo Nordisk, PharmaSens, Roche, Sanofi, and Terumo; P-YB has received speaker honoraria from Abbott, Eli Lilly, Novo Nordisk, and Sanofi, is chief medical officer for Diabeloop, and served on advisory board panels for Abbott, Dexcom, Insulet, LifeScan, Eli Lilly, Pharmasens, Novo Nordisk, and Sanofi; Peter Diem is member of the PharmaSens AG Board of Directors; Lutz Heinemann is a consultant to several diagnostic and therapeutic companies in the development of new products, he is a shareholder in the Profil Institute for Metabolic Research in Neuss, Germany.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was funded by PharmaSens AG, Biel, Switzerland.
