Abstract
Introduction:
The Virtual Diabetes Specialty Clinic (VDiSC) study demonstrated the feasibility of providing comprehensive diabetes care entirely virtually by combining virtual visits with continuous glucose monitoring support and remote patient monitoring (RPM). However, the financial sustainability of this model remains uncertain.
Methods:
We developed a financial model to estimate the variable costs and revenues of virtual diabetes care, using visit data from the 234 VDiSC participants with type 1 or type 2 diabetes. Data included virtual visits with certified diabetes care and education specialists (CDCES), endocrinologists, and behavioral health services (BHS). The model estimated care utilization, variable costs, reimbursement revenue, gross profit, and gross profit margin per member, per month (PMPM) for privately insured, publicly insured, and overall clinic populations (75% privately insured). We performed two-way sensitivity analyses on key parameters.
Results:
Gross profit and gross profit margin PMPM (95% confidence interval) were estimated at $−4 ($−14.00 to $5.68) and −4% (−3% to −6%) for publicly insured patients; $267.26 ($256.59-$277.93) and 73% (58%-88%) for privately insured patients; and $199.41 ($58.43-$340.39) and 67% (32%-102%) for the overall clinic. Profits were primarily driven by CDCES visits and RPM. Results were sensitive to insurance mix, cost-to-charge ratio, and commercial-to-Medicare price ratio.
Conclusions:
Virtual diabetes care can be financially viable, although profitability relies on privately insured patients. The analysis excluded fixed costs of clinic infrastructure, and securing reimbursement may be challenging in practice. The financial model is adaptable to various care settings and can serve as a planning tool for virtual diabetes clinics.
Introduction
Continuous glucose monitoring (CGM) is recommended as standard of care by the American Diabetes Association (ADA) for individuals with type 1 diabetes (T1D) and insulin-treated type 2 diabetes (T2D). 1 Although significant research has demonstrated that CGM is associated with improved glycemic outcomes and quality of life,2-8 this technology has not yet been widely disseminated into routine patient care.9-11 Limited resources, technology readiness, insurance coverage, and a lack of CGM training and education are often cited as factors that limit the uptake of CGM. 12 Moreover, accessing subspecialty care in the United States remains a pernicious problem. Although the burden of diabetes already accounts for one in four health care dollars in the United States 13 and is projected to grow substantially in the coming decades, 14 most patients are treated in the primary care setting.15,16
Telemedicine presents a unique opportunity to expand access to underserved populations who do not have local access to endocrinologists, measure patient outcomes in real time, and deliver timely, on-demand interventions. Models of care that incorporate CGM initiation and support and remote patient monitoring (RPM) into routine care have been associated with improved time in range (TIR) and hemoglobin A1c (HbA1c), as well as reduced provider review time per patient.17-20 Moreover, a growing body of evidence suggests that virtual models of care, whereby patients are initiated on CGM and evaluated remotely, may also be feasible and effective.21,22 In particular, the Virtual Diabetes Specialty Clinic (VDiSC) 23 enrolled 234 patients with T1D or T2D into a fully virtual clinic providing comprehensive diabetes care, CGM support, behavioral health services (BHS), and algorithm-enabled decision support technology. The VDiSC study reported clinical benefits: For T1D patients, average HbA1c decreased from 7.8% at baseline to 7.1% at both three and six months, and average TIR increased by 11% over six months. For T2D patients, average HbA1c decreased from 8.1% at baseline to 7.1% at both three and six months, and average TIR increased by 18% over six months. 23 Moreover, patients also reported less diabetes-related distress and greater satisfaction with managing their condition. 24 Among patients who participated in an additional six-month extension study, clinical benefits were sustained at the 12-month mark.
While previous studies have suggested that there exist financially sustainable pathways for diabetes clinics to adopt telemedicine as an adjunct to routine, in-person care,25,26 it is unclear whether a fully virtual clinic is feasible from an economic point of view. In this article, we created a financial model to estimate the expected monthly utilization of virtual visits and RPM services for the patient population enrolled in the VDiSC study. Then, we projected the variable costs, reimbursement revenue, and gross profit margin of the virtual care model on a per member, per month (PMPM) basis.
Methods
VDiSC Study Protocol
The protocol for the VDiSC study has been previously described. 23 Briefly, 234 eligible participants with T1D or T2D (characteristics described in Table 1) were assigned a certified diabetes care and education specialist (CDCES) who provided CGM training and diabetes management education. After completing a minimum of three protocol-scheduled remote training sessions, additional contacts with the CDCES to review CGM data in an ongoing manner were scheduled as needed. A study endocrinologist was also available for patient referral as needed. Patients completed behavioral health questionnaires and were referred to a nonphysician BHS coach if they met thresholds for a positive screen. Finally, ongoing RPM included device support with a clinician, a patient-facing mobile app with a bolus calculator to calculate insulin dose, and a platform with onboarding, monitoring, and algorithm-enabled recommendations for insulin dosing (endo-digital; DreaMed Diabetes, St. Louis Park, MN). 27 Additional details on the study protocol can be found in the Supplemental Methods.
Patient Characteristics From the VDiSC Study (n = 234).
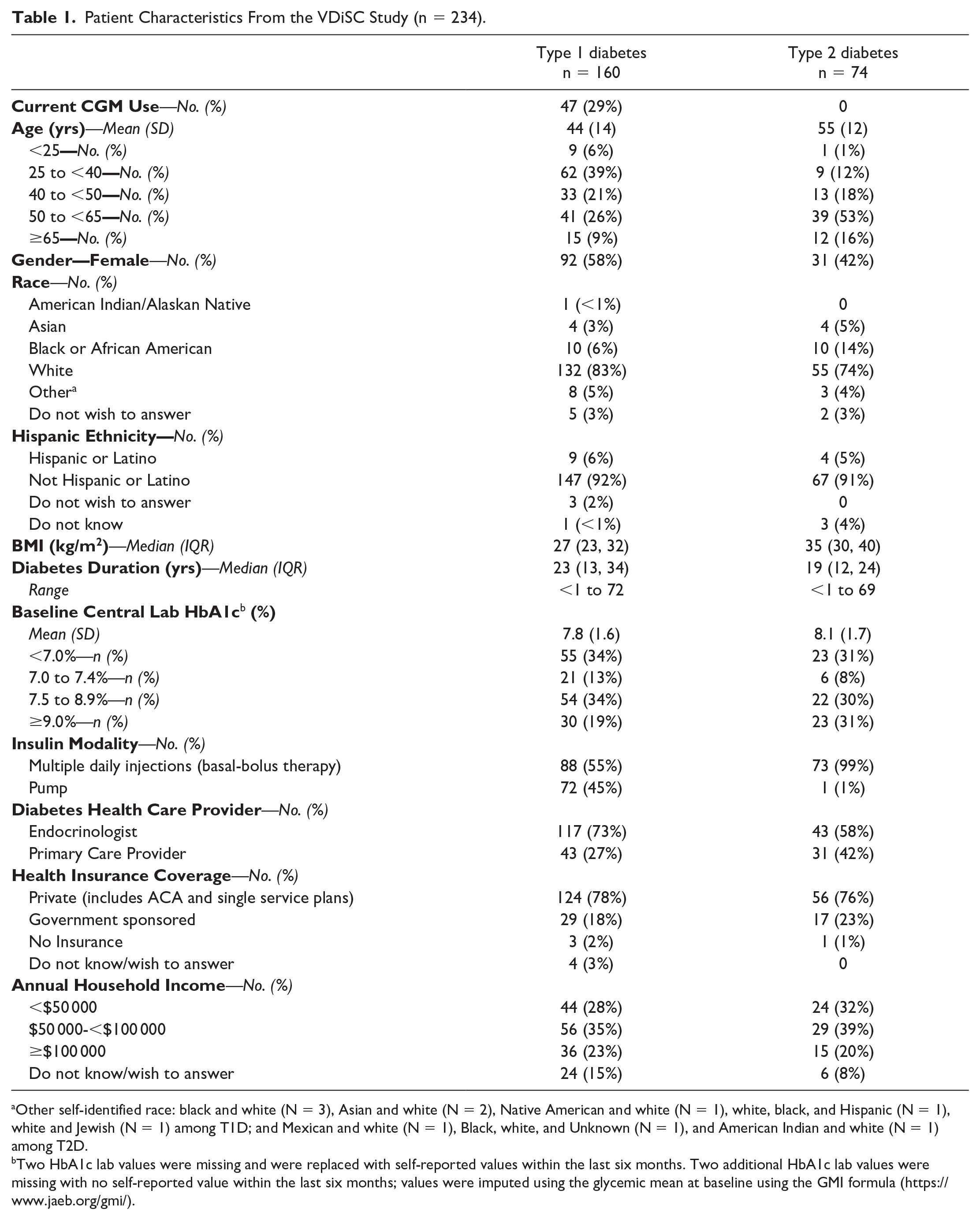
Other self-identified race: black and white (N = 3), Asian and white (N = 2), Native American and white (N = 1), white, black, and Hispanic (N = 1), white and Jewish (N = 1) among T1D; and Mexican and white (N = 1), Black, white, and Unknown (N = 1), and American Indian and white (N = 1) among T2D.
Two HbA1c lab values were missing and were replaced with self-reported values within the last six months. Two additional HbA1c lab values were missing with no self-reported value within the last six months; values were imputed using the glycemic mean at baseline using the GMI formula (https://www.jaeb.org/gmi/).
Overview of Financial Model
We created a financial model to estimate the average variable costs and revenues of providing virtual diabetes care to the patient population enrolled in the VDiSC study on a PMPM basis. First, we estimated the average monthly utilization of CDCES, BHS, and endocrinologist visits from the patient visit schedule data, which recorded the duration and date of appointments for all patients during the entirety of their participation in the study. Second, we estimated the monthly costs of these services by converting the charges to costs using a nationally representative cost-to-charge ratio (CCR). Third, we calculated the monthly revenue using Medicare Current Procedural Terminology (CPT) codes which are currently approved for virtual visits and RPM. Finally, we calculated the gross profit and gross profit margin PMPM of the clinic. We conducted these analyses for both privately and publicly insured patients.
Average Utilization of Services
Because CDCES, BHS, and endocrinologist visits in the VDiSC study were allocated based on patient need, rather than scheduled at a regular cadence, we modeled the average monthly utilization per patient. We assumed that CDCES and BHS appointments would be charged in 30-minute increments (Table 2). For example, a 45-minute visit would be charged at the same rate as a 60-minute visit (ie, the rate for two 30-minute visits). For each patient, we summed the number of 30-minute visits incurred over the duration of their participation in the study and calculated the average number of visits incurred per month. We visualized the distribution of 30-minute visits per month, stratified by insurance type (public vs private) and by diabetes type (T1D vs T2D). Next, we calculated the mean utilization of CDCES and BHS 30-minute visits PMPM by averaging over all patients and calculated a corresponding 95% confidence interval (CI). Our estimate of CDCES utilization included the initial three training sessions scheduled for all patients.
Charge and Cost Structure for Virtual Visits and Remote Patient Monitoring (RPM) Services, and Associated Sources of Reimbursement. Certified Diabetes Care and Education Specialist (CDCES), Behavioral Health Services (BHS), and Endocrinologist Virtual Visits Are Dynamically Allocated and Charged per Visit, While RPM Is Charged per Member per Month (PMPM).
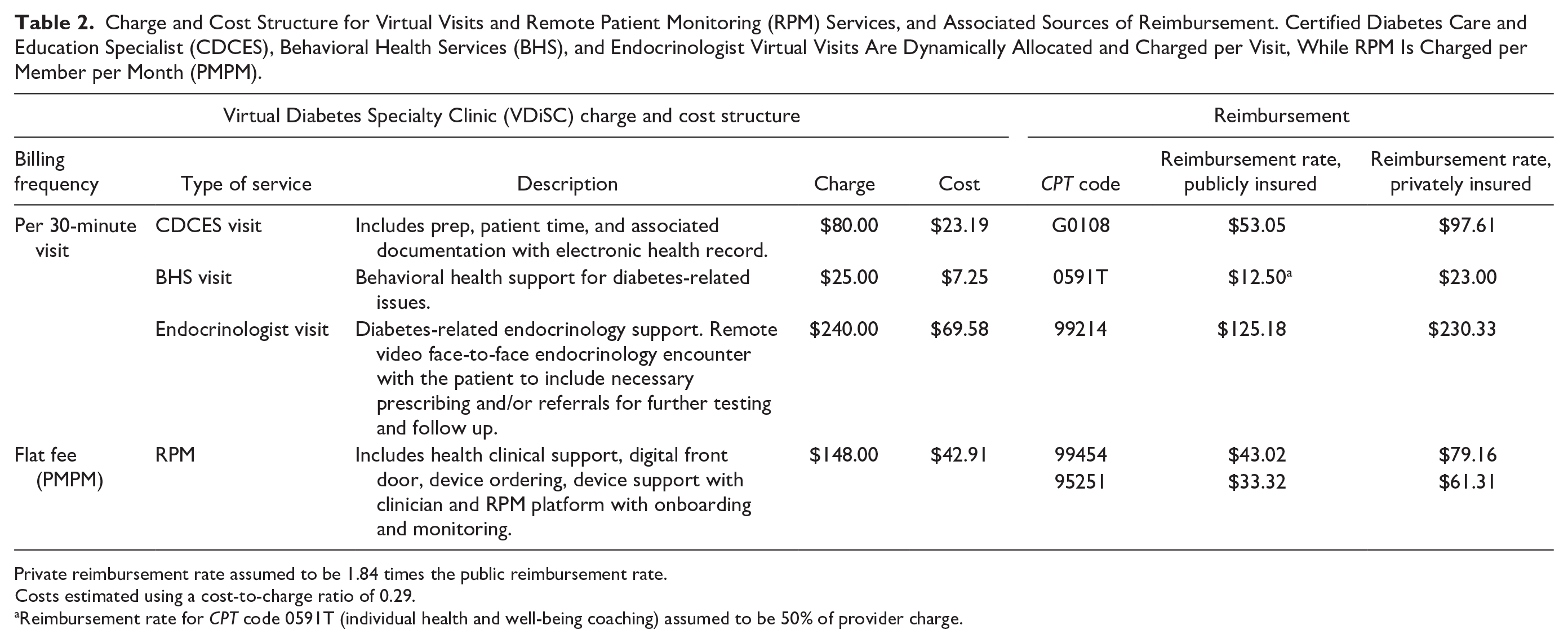
Private reimbursement rate assumed to be 1.84 times the public reimbursement rate.
Costs estimated using a cost-to-charge ratio of 0.29.
Reimbursement rate for CPT code 0591T (individual health and well-being coaching) assumed to be 50% of provider charge.
For the endocrinologist visits, the dataset only included the total number of 30-minute visits utilized in the study and the specific patients who required escalation to a specialist. We assumed that these visits were evenly distributed across the specific patients who utilized this service and calculated the mean utilization of 30-minute visits PMPM using the same methodology as above. Although we could not assess potential variability in the distribution of visits among these specific patients, this simplifying assumption did not bias the estimate of mean utilization.
Costs and Potential Reimbursement Revenue
Because the VDiSC study was conducted in the context of a clinical trial, participants were not billed for any services. The amount that the study provider could have charged for each service outside of a clinical trial setting is shown in Table 2. Visits were charged in increments of 30 minutes on a per-visit basis. Ongoing RPM services incurred a monthly, flat charge for each patient. To estimate the variable costs of these services, we multiplied the charge amount by a nationally representative CCR of 0.29, which is the median for hospitals in the United States, reported in the 2022 Hospital Provider Cost Report published by the Centers for Medicare and Medicaid Services. 28 All costs are in US dollars.
To estimate the potential reimbursement revenue for these services, we identified CPT codes currently approved for the treatment of diabetes, and used the national average amount reported in the 2025 Medicare National Fee Schedule. 29 The CDCES visits were reimbursed by CPT code G0108, which covers diabetes self-management training. Moreover, we assumed that publicly insured patients could only be reimbursed for a maximum of two hours of CDCES visits per year, as per Medicare reimbursement guidelines. 30 Endocrinologist visits were reimbursed by CPT code 99214, which covers outpatient evaluation and management of an established patient. We assumed that BHS virtual visits may be eligible for CPT code 0591T, a temporary category III CPT code that covers health and well-being coaching by certified nonphysician health care professionals. For RPM services, CGM interpretation was reimbursed by CPT code 95251, while data transmission and device servicing were reimbursed by CPT code 99454. We assumed that 96% of patients would meet the CGM wear time requirements for CPT code 99454, based on the median CGM usage of patients in the VDiSC study. 23 The CPT codes and reimbursement amounts are shown in Table 2. Detailed assumptions about eligibility for reimbursement can be found in the Supplemental Methods.
Public and private payors have different rates of reimbursement. Thus, we assumed that the expected reimbursement rate for privately insured patients was 1.84 times greater than the rate for publicly insured patients, which is the average commercial-to-Medicare price ratio for professional services in the United States in 2022. 31 For our base case analysis, we assumed that 75% of patients were privately insured and that the remaining 25% were publicly insured, which reflects the insurance makeup of the VDiSC study population (Table 1). To estimate the overall revenue for the clinic, we calculated a weighted average reimbursement rate for each service.
Profitability of Virtual Care
To estimate the expected monthly variable costs and revenues PMPM, we multiplied the average monthly utilization of each service by its cost and by the reimbursement rate, respectively. Subtracting the cost from the revenue yielded the monthly gross profit per patient. Finally, we divided the gross profit by the revenue to obtain a gross profit margin PMPM, both for each individual service and for the overall clinic.
Sensitivity Analysis
To account for the significant hospital-specific variability in CCR and price ratio, we conducted a two-way sensitivity analysis of these parameters on the gross profit margin. We varied the CCR between 0.1 and 1 (with 1 representing parity between charge and cost), as 92% of hospital CCRs fall within this range, with a majority falling between 0.2 and 0.5. 28 We varied the price ratio between 1 and 2.5 (with 1 representing parity between prices paid by public payors and commercial health plans), as previous work has found that the ratio varies between 1.41 and 2.47. 32 We visualized the gross profit margin as a function of both parameters. Moreover, we conducted two scenario analyses, where we varied the proportion of privately insured patients to 50% and 25% (compared with 75% in the base case).
Results
Average Utilization of Services
Of the 234 patients in the VDiSC study, 97 patients (41%) required a BHS appointment at some point in their enrollment, while the other 137 patients (59%) did not utilize this service at all. Among the patients that utilized a BHS visit, a majority (78%) were privately insured (Figure 1). There were 104 thirty-minute endocrinologist visits across 33 participants (14%). Supplemental Figure 1 illustrates the distribution of CDCES visits by diabetes type.
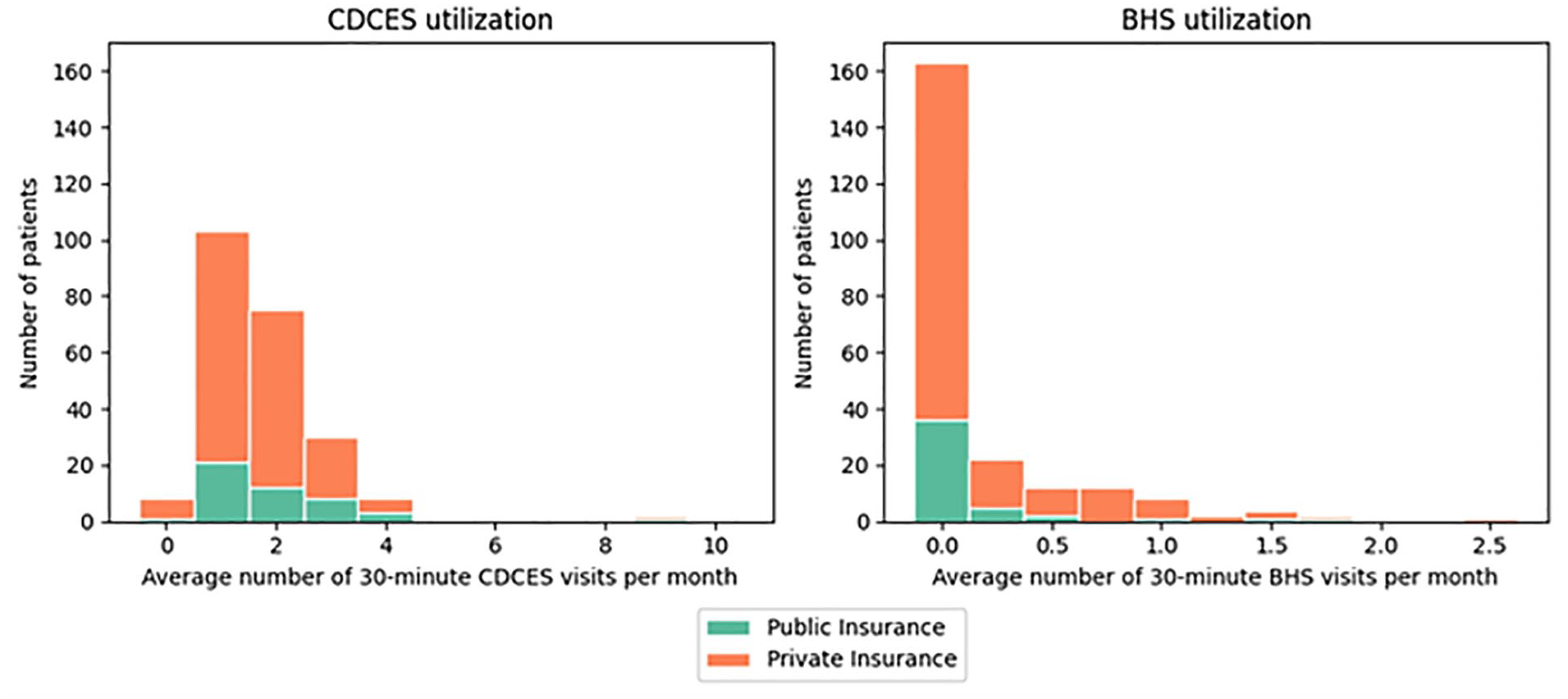
The average utilization of 30-minute certified diabetes care and education specialist (CDCES) visits (left) and 30-minute behavioral health services (BHS) visits (right) per month. The colors represent insurance types (public vs private).
The average utilization of 30-minute visits PMPM and the economics of care are shown in Table 3. For publicly insured patients, the utilization of CDCES, BHS, and endocrinologist visits was 2.43 thirty-minute visits PMPM (95% CI: 2.00-2.85), 0.20 thirty-minute visits PMPM (95% CI: 0.078-0.32), and 0.030 thirty-minute visits PMPM, respectively. For privately insured patients, it was 2.18 thirty-minute visits PMPM (95% CI: 2.04-2.33), 0.20 thirty-minute visits PMPM (95% CI: 0.078-0.32), and 0.030 thirty-minute visits PMPM, respectively. For the overall clinic, it was 2.24 thirty-minute visits PMPM (95% CI: 1.67-2.82), 0.24 thirty-minute visits PMPM (95% CI: 0.02-0.46), and 0.04 thirty-minute visits PMPM, respectively.
Expected Utilization of Services and Revenues, Costs, and Gross Profit of Care Model per Member, per Month (PMPM).
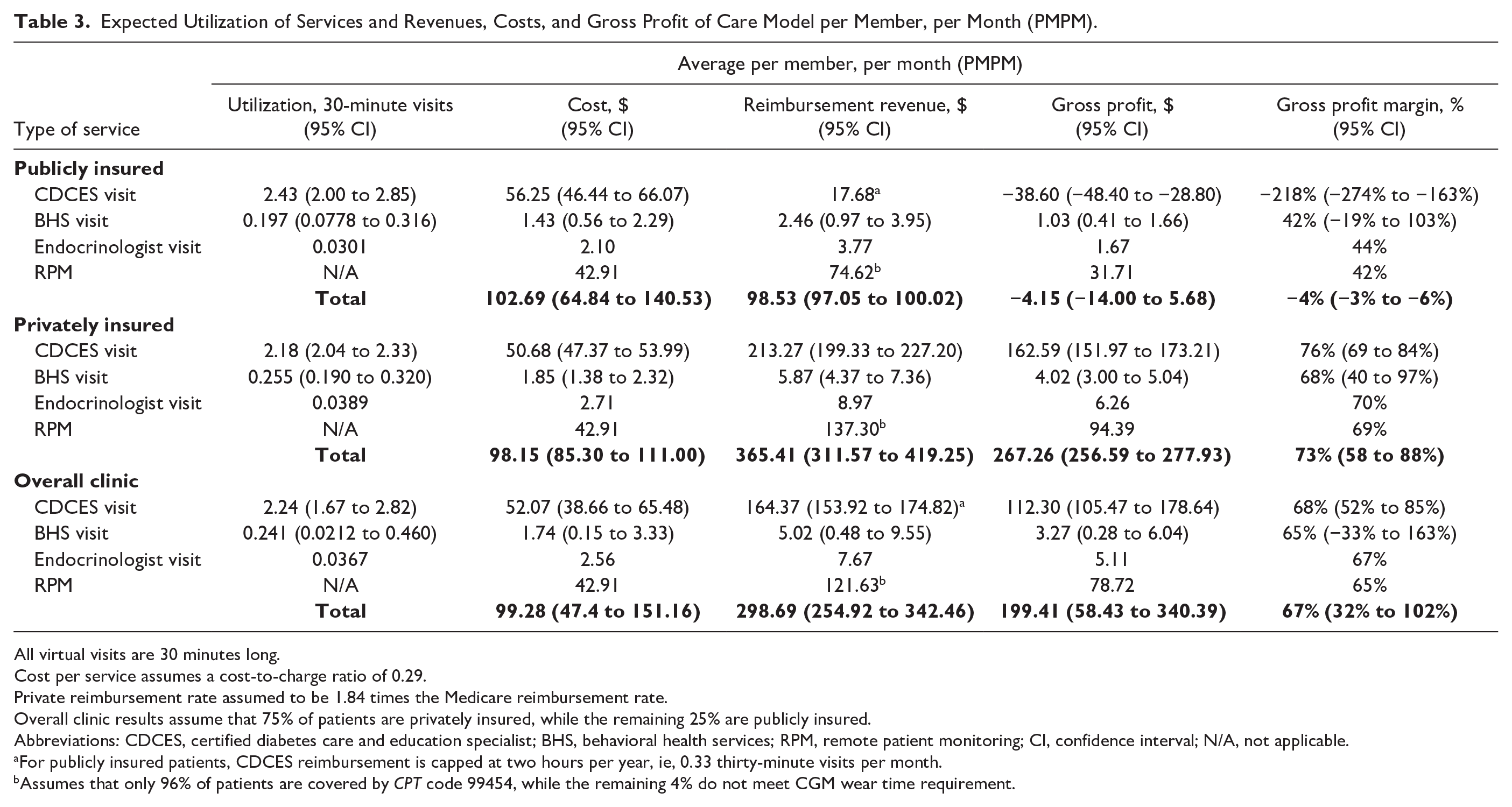
All virtual visits are 30 minutes long.
Cost per service assumes a cost-to-charge ratio of 0.29.
Private reimbursement rate assumed to be 1.84 times the Medicare reimbursement rate.
Overall clinic results assume that 75% of patients are privately insured, while the remaining 25% are publicly insured.
Abbreviations: CDCES, certified diabetes care and education specialist; BHS, behavioral health services; RPM, remote patient monitoring; CI, confidence interval; N/A, not applicable.
For publicly insured patients, CDCES reimbursement is capped at two hours per year, ie, 0.33 thirty-minute visits per month.
Assumes that only 96% of patients are covered by CPT code 99454, while the remaining 4% do not meet CGM wear time requirement.
Profitability of Virtual Care
For publicly insured patients, the gross profit PMPM and gross profit margin PMPM were projected to be $−4.15 (95% CI: $−14.00 to $5.68) and −4% (95% CI: −3% to −6%), respectively. For privately insured patients, these were projected to be $267.26 (95% CI: $256.59-$277.93) and 73% (95% CI: 58%-88%), respectively. For the overall clinic, these were projected to be $199.41 (95% CI: $58.43-$340.39) and 67% (95% CI: 32%-102%), respectively. In all cases, the reimbursement revenue and gross profit were primarily driven by CDCES visits and RPM, due to their outsized utilization compared with BHS and endocrinologist visits (Table 3).
Sensitivity Analysis
In two-way sensitivity analysis, the gross profit margin increased as the price ratio increased and the CCR decreased (Figure 2). Within a range that is representative of most health care providers (CCR between 0.2 and 0.5 and price ratio between 1.4 and 2.5), the gross profit margin varied between 27% and 83%. Within the same representative range, gross profit margins were less favorable in the scenario analyses (Supplemental Figures 2 and 3). When 50% of patients were publicly insured, the gross profit margin varied between 9% and 77%. When 75% of patients were publicly insured, the gross profit margin varied between −22% and 65%.
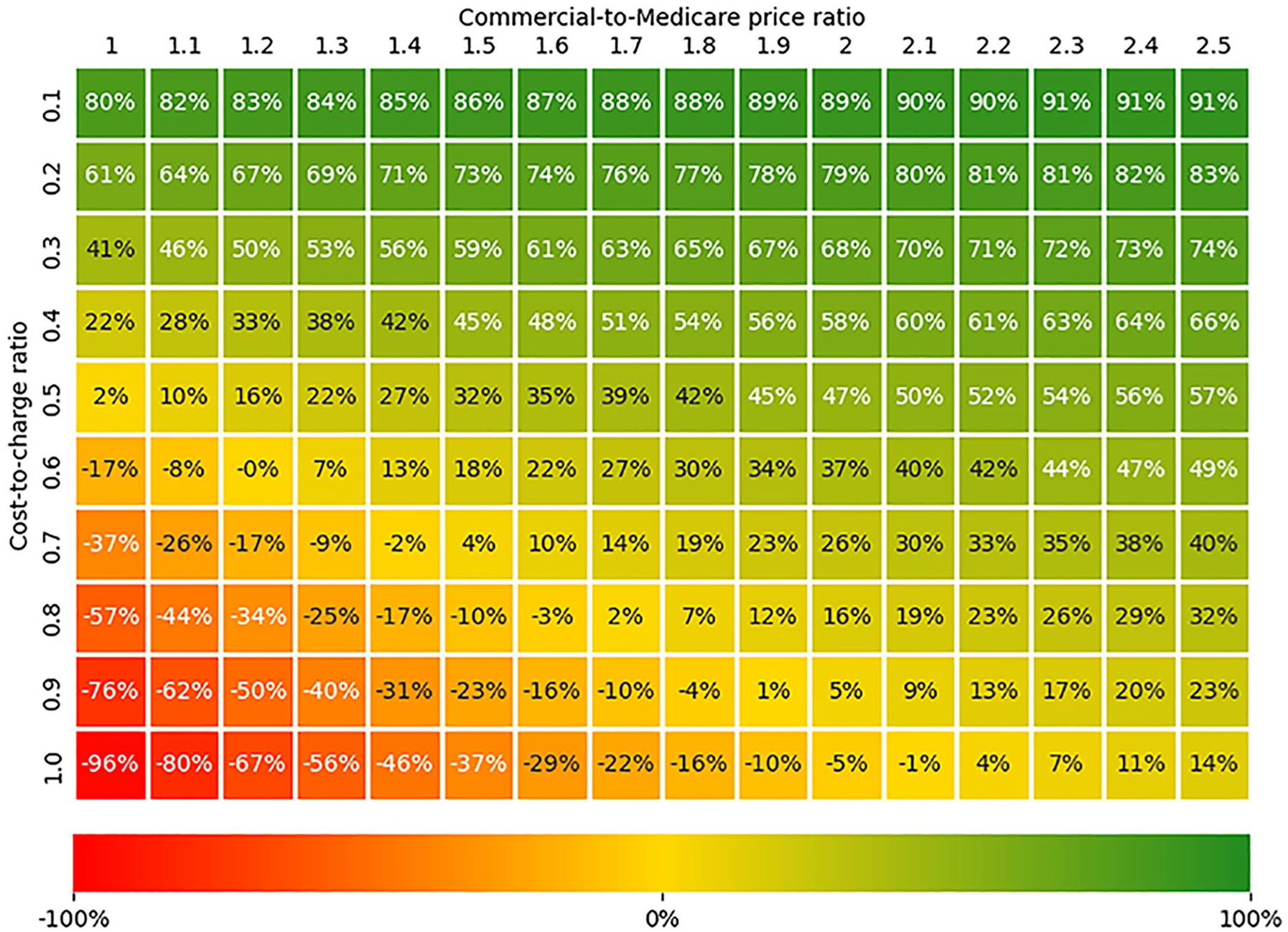
Two-way sensitivity analysis: expected gross profit margin of virtual clinic per member, per month, as a function of cost-to-charge ratio and commercial-to-Medicare price ratio.
Discussion
Virtual models of diabetes care are clinically effective and operationally feasible.17-23 In particular, the VDiSC study exemplified the clinical viability of virtual care delivery, as both T1D and T2D patients demonstrated improvements in HbA1c and TIR. 23 In addition to improving glycemic outcomes, virtual care may also reduce provider review time, increase clinic capacity, and improve patient quality of life. Virtual care may also address health care disparities by expanding access to subspecialty care for underserved populations with limited access to endocrinologists. Although virtual diabetes care is extremely promising, its financial sustainability is unclear. Thus, we developed a financial model to evaluate its economic viability, given current reimbursement guidelines. In a patient population representative of the VDiSC study, we estimated a gross profit margin of 67% PMPM, with expected variation between 27% and 83% depending on CCRs and price ratios typical of most healthcare providers.
This margin was driven by CDCES visits and RPM (Table 3), which were utilized at much higher rates than BHS and endocrinologist visits. In addition, 75% of the patients in the VDiSC study were privately insured and achieved a higher gross profit margin than publicly insured patients (73% vs −4%), who were projected to not be profitable. This profitability gap is due to different rates of reimbursement, and the fact that publicly insured patients can only be reimbursed for a maximum of two hours of CDCES time per year. If this reimbursement restriction was lifted, the gross profit margin for publicly insured patients would be 51%. Therefore, under current reimbursement guidelines, it may not be possible to provide virtual diabetes care to Medicare beneficiaries in a financially sustainable way, especially given that older adults require more CGM training and support than younger adults to achieve similar glycemic benefits. 33 One potential strategy to address this limitation is to incorporate CGM onboarding during the initial 10 hours of diabetes self-management training that Medicare covers as a once-in-a-lifetime benefit. Nonetheless, this would be insufficient to provide adequate coverage even in the first year of treatment, nor would it address the fundamental long-term financial viability concerns of serving this patient population.
The estimated gross profit margin represents an optimistic projection of the reimbursement potential. In practice, reimbursement may be challenging to secure. Reimbursement varies substantially across payors and states,34,35 while some commercial plans may not reimburse RPM or may have limits on the billing frequency of certain CPT codes. Some plans may require bundling diabetes care with other services as a condition for reimbursement. Implementing billing workflows for RPM also presents significant operational challenges, and requires close collaboration with numerous health care administration and compliance stakeholders. 26
Limitations
One limitation of our analysis is that it did not account for the one-time fixed costs of developing and deploying the necessary hardware and software, nor did it account for other fixed costs related to the establishment of the clinic infrastructure (eg, legal, licensing, and technology fees). These implementation costs vary substantially, from tens of thousands to hundreds of thousands of dollars per year. 25 Although including fixed costs would not affect the estimated gross profit margin, it would be a central component in deciding how much to invest in establishing a virtual clinic. Future work could explicitly account for fixed costs and for other downstream benefits of virtual care such as decreased provider review time.
Next, we did not account for one-time CPT codes that could be billed for (eg, CPT code 95250) or other ongoing RPM codes that could be stacked (e.g., CPT codes 99457 and 99458). We estimated the number of patients meeting the monthly data submission requirements for CPT code 99454 using median CGM usage as a proxy. However, this may not accurately reflect the actual proportion of patients who fulfill this reimbursement criterion. Moreover, it was unclear whether the BHS coaches were eligible for CPT code 0591T, nor was the exact reimbursement amount available. Nonetheless, given that the expected gross profit stemming from BHS visits only accounted for roughly 2% of the total gross profit (Table 3), it was unlikely to have any material impact on our conclusions, even in cases with little to no reimbursement from BHS visits.
Another limitation is that the number of visits may be mis-estimated based on the study dataset. On one hand, our estimate of the utilization of CDCES visits may be inflated by the study protocol, which mandated a minimum of three initial training sessions. On the other hand, patients may have consulted other providers (eg, their primary care physician) regarding their diabetes care plan and treatment outside of the VDiSC study, which was not captured in the dataset. As a result, our estimate of the demand for visits could be overestimated or underestimated. In addition, this virtual care model was deployed in a study population and may not be generalizable to a wider patient population. For instance, the population of the VDiSC study was predominantly white with private insurance, which may not be representative of other groups with comparatively worse outcomes due to existing health disparities. 36
Conclusions
Multiple future directions exist for this work. Although we used charge amounts from the VDiSC study, only slight modifications would be necessary to extrapolate the results to other healthcare systems. In addition, we developed our model in a traditional fee-for-service setting. However, virtual clinics that provide whole-population care and targeted interventions may be more suited to value-based payment models that reward providers for improving aggregate glycemic outcomes.
In conclusion, our findings suggest that comprehensive diabetes care can be financially sustainable, but primarily when serving privately insured populations. Significant changes to existing reimbursement guidelines would be necessary to extend economically viable care to Medicare beneficiaries. Our analysis provides a customizable financial planning tool that is broadly applicable to any clinic delivering or transiting to virtual diabetes care—a model of care that can improve glycemic outcomes and expand access to specialized care for underserved communities.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251340664 – Supplemental material for Assessing the Financial Sustainability of a Virtual Clinic Providing Comprehensive Diabetes Care
Supplemental material, sj-docx-1-dst-10.1177_19322968251340664 for Assessing the Financial Sustainability of a Virtual Clinic Providing Comprehensive Diabetes Care by Paul Dupenloup, Grace Guan, Grazia Aleppo, Richard M. Bergenstal, Korey Hood, Davida Kruger, Teresa McArthur, Beth Olson, Sean Oser, Tamara Oser, Ruth S. Weinstock, Robin L. Gal, Craig Kollman and David Scheinker in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
None.
Abbreviations
ADA, American Diabetes Association; BHS, behavioral health services; CDCES, Certified Diabetes Care and Education Specialist; CI, confidence interval; CGM, continuous glucose monitor; CCR, cost-to-charge ratio; CPT, current procedural terminology; HbA1c, hemoglobin A1c; PMPM, per member per month; RPM, remote patient monitoring; TIR, time in range; T1D, type 1 diabetes; T2D, type 2 diabetes; VDiSC, virtual diabetes specialty clinic.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.A. reported grants from The Leona M. and Harry B. Helmsley Charitable Trust during the conduct of the study and personal fees from Bayer, Dexcom, and Insulet; nonfinancial support from Eli Lilly; and grants from Dexcom, Eli Lilly, Fractyl Health, Emmes, Mannkind, Tandem Diabetes Care, and Welldoc outside the submitted work. R.M.B. reported grants from Abbott Diabetes Care, Eli Lilly, Hygieia, Dexcom, Sanofi, Tandem Diabetes Care, Insulet, and Medtronic and consulting and/or advisory board fees paid to his institution from Abbott Diabetes Care, Dexcom, Onduo, Sanofi, Roche, Embecta, and Medtronic during the conduct of the study. R.L.G. reported grants from The Leona M. and Harry B. Helmsley Charitable Trust during the conduct of the study. K.H. reported consulting fees from Sanofi, Havas Health, and MannKind; investigator-initiated grant from Embecta. C.K. reported grants from The Leona M. and Harry B. Helmsley Charitable Trust and nonfinancial support from Dexcom during the conduct of the study; grants from JDRF, Diasome, and MannKind outside the submitted work; and nonfinancial support from Tandem, Dexcom, and Insulet outside the submitted work. D.K. reported grants from Abbott and advisor and/or speaking fees from Abbott, Dexcom, and Jaeb Center for Health Research outside the submitted work. B.O. reported personal fees from Lagoon Health during the conduct of the study and stock ownership in Abbott Laboratories. S.O. reported grants from The Leona M. and Harry B. Helmsley Charitable Trust during the conduct of the study and consulting fees from Cecelia Health and advisory board fees from Dexcom outside the submitted work. T.O. reported grants from The Leona M. and Harry B. Helmsley Charitable Trust during the conduct of the study and consulting fees from Cecelia Health, advisory board fees from Dexcom, and an investigator-initiated grant from Abbott Diabetes Care outside the submitted work. R.S.W. reported personal fees from The Leona M. and Harry B. Helmsley Charitable Trust and Jaeb Center for Health Research during the conduct of the study; grants for participation in multicenter clinical trial through her institution from Eli Lilly, Novo Nordisk, Insulet, Tandem, Amgen, and MannKind outside the submitted work; and discounted devices for clinical research from DexCom outside the submitted work. D.S. has received support from Stanford MCHRI, Stanford HAI, the NSF, and the Helmsley Charitable Trust, and is an advisor to Carta Health. P.D. has received support from Stanford HAI and Stanford SIGF. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by The Leona M. and Harry B. Helmsley Charitable Trust. Dexcom provided devices used in the VDiSC study. D.S. and P.D. are supported in part by the Stanford Institute for Human-Centered Artificial Intelligence (HAI). G.G. is supported in part by the National Science Foundation Graduate Research Fellowship under Grant No. 1656518 and the Stanford Data Science Scholars Program. Any opinion, findings, and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Science Foundation.
Trial Registration
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
