Abstract
Background:
Youth with type 1 diabetes (T1D) and public insurance have lower diabetes technology use. This pilot study assessed the feasibility of a program to support continuous glucose monitor (CGM) use with remote patient monitoring (RPM) to improve glycemia for youth with established T1D and public insurance.
Methods:
From August 2020 to June 2023, we provided CGM with RPM support via patient portal messaging for youth with established T1D on public insurance with challenges obtaining consistent CGM supplies. We prospectively collected hemoglobin A1c (HbA1c), standard CGM metrics, and diabetes technology use over 12 months.
Results:
The cohort included 91 youths with median age at enrollment 14.7 years, duration of diabetes 4.4 years, 33% non-English speakers, and 44% Hispanic. Continuous glucose monitor data were consistently available (≥70%) in 23% of the participants. For the 64% of participants with paired HbA1c values at enrollment and study end, the median HbA1c decreased from 9.8% to 9.0% (P < .001). Insulin pump users increased from 31 to 48 and automated insulin delivery users increased from 11 to 38.
Conclusions:
We established a program to support CGM use in youth with T1D and barriers to consistent CGM supplies, offering lessons for other clinics to address disparities with team-based, algorithm-enabled, remote T1D care. This real-world pilot and feasibility study noted challenges with low levels of protocol adherence and obtaining complete data in this cohort. Future iterations of the program should explore RPM communication methods that better align with this population’s preferences to increase participant engagement.
Keywords
Introduction
Sustained use of diabetes technology, including continuous glucose monitors (CGMs), insulin pumps, and automated insulin delivery (AID) systems, improves glycemic outcomes in youth with type 1 diabetes (T1D) thereby reducing the risk for complications.1-4 In the United States, diabetes technology use is lower in youth with public insurance, which is a marker of lower socioeconomic status (SES) and minoritized backgrounds. The technology use gap has widened as technology advanced rapidly.5-8 In California at time of study initiation, public insurance only covered CGM for youth with documented glucose checks four or more times daily for more than 30 days, translating to limited CGM access for a population who are struggling the most with diabetes management. 9 Youth with public insurance who met criteria for CGM coverage had high use of CGM, but gaps in CGM access led to discontinuations of CGM use and worsening of clinical outcomes.3,10 Therefore, timely recognition and interventions to address supply gaps will likely increase device use and reduce disparity.
Despite increasing awareness about diabetes care disparities, only a few interventional trials targeting racial-ethnic disparities in diabetes technology use have been published. Key studies demonstrated that supporting CGM use in youth and adults with T1D from marginalized backgrounds is associated with improved glycemic outcomes and thereby reduced disparities in diabetes care.11-13 We aimed to develop a clinical program that can support consistent CGM use and improve glycemia for youth with T1D from marginalized backgrounds, with consideration of program sustainability without research funding.
The Teamwork, Targets, Technology, and Tight Control (4T) Study is a pragmatic research study designed to improve clinical care for youth with newly diagnosed T1D. A key feature of this study is a CGM-based asynchronous remote patient monitoring (RPM) facilitated by the Timely Interventions in Diabetes Excellence (TIDE) platform.14,15 This is an asynchronous telehealth workflow that includes electronic transmission of patient CGM data to the Certified Diabetes Care and Education Specialists (CDCESs). Certified Diabetes Care and Education Specialists use the interactive TIDE dashboard to prioritize patients for review based on adapted consensus guidelines, allowing for increased opportunities for identifying and intervening on patients not meeting targets while minimizing provider time demands. The 4T Study demonstrated that early initiation of CGM and support with RPM was associated with hemoglobin A1c ( HbA1c) reduction 12 months after T1D diagnosis.14,16 As an extension to the 4T Study, we developed the CGM Time in Range Program at Stanford (CGM TIPS) to translate this improved care model to youth with existing T1D who therefore did not qualify for the 4T Study.
The primary purpose of the study was to assess the feasibility of a technology-enabled clinical workflow to support CGM use with RPM for a population with high levels of barriers to consistent CGM use and achieving glycemic targets. Additional exploratory hypotheses were that participants would have improvements in glycemic metrics. Since CGM is a required component for AID, we also hypothesized that participants would have increased uptake of AID.
Methods
Participants and Study Design
From August 2020 to June 2023, we approached youth who met eligibility criteria for enrollment: ages 2 to 23 years, existing T1D, public insurance, and challenges obtaining consistent CGM supplies. Eligible participants who declined participation may be approached again based on clinical team’s discretion; this commonly occurred when the youth was not reaching glycemic targets, and the clinician felt strongly that CGM and RPM would significantly benefit the youth. Patient characteristics were obtained from electronic health record (EHR) review. Diabetes technology use history was documented, inclusive of those who had appropriate prescriptions and education but were not using the devices at enrollment.
Continuous glucose monitor Time in Range Program at Stanford is a single-arm, prospective pilot and feasibility study modeled after the 4T Study, 14 which supports CGM use with RPM for youth with T1D. Due to the strong evidence that consistent CGM use is beneficial for management of T1D in youth, we felt it was unethical to use a randomized controlled trial design as it necessitates withholding CGM supply support for some participants. The study was designed to be sustainable without research funding. Participants received no financial compensation for participation and RPM was conducted with existing clinic infrastructure and staff. Consents and assents were obtained from the guardian and participant ages seven to 18 years, respectively. The Stanford Institutional Review Board approved this study protocol.
At enrollment, participants were initiated on Dexcom G6 CGM (Dexcom, San Diego, California) with a CDCES and received two months of CGM supplies. Certified Diabetes Care and Education Specialists used first month of CGM data to demonstrate consistent glucose monitoring and applied for insurance coverage for ongoing CGM supplies, which typically became available during the second month. Continuous glucose monitor data sharing with clinic was set up using participants’ compatible smart phone or a clinic-supplied iPod Touch. Participants could use alternate receivers (eg, t:slim ×2 insulin pump [Tandem Diabetes Care, San Diego, California] or Dexcom receiver) if they preferred, although these devices did not allow automatic data sharing to the cloud for RPM. Standard clinical care included a CGM teaching session one week after device initiation and quarterly clinic or telehealth visits. In addition, CGM TIPS provided 12 monthly RPM by a CDCES facilitated by the TIDE platform.14,15 The TIDE platform triggered CDCES review for patients who met the following criteria adapted from consensus guidelines: CGM data availability < 50%, time in range (TIR, 70-180 mg/dL) < 65%, week-over-week decrease in TIR > 15%, > 4% time below range (TBR, < 70 mg/dL), or > 1% time in clinically significant hypoglycemia (< 54 mg/dL). 17 For the many participants with persistently low TIR, CDCESs preferentially used hypoglycemia flags and decrease in TIR > 15% alerts to prioritize RPM contacts. Remote patient monitoring communication occurred as a templated message in English or Spanish via the secure patient portal MyChart (Epic Systems, Verona, Wisconsin), which is our clinic’s first line for communicating with patients. As per provider’s discretion, RPM communication may also occur via phone call. Monthly CDCES RPM allowed more frequent dose adjustments, troubleshooting CGM supply gaps, and education. After the 12-month program, participants may reconsent or exit the program to continue standard care.
Feasibility Assessment
The primary study outcome was program feasibility, which focused on participant engagement with standard clinical care and study interventions. Our clinic standard included quarterly clinic or telehealth visits with HbA1c measurements. Starting in March 2020, due to the COVID-19 pandemic, many clinic visits were transitioned to telehealth. Thus, many participants did not have quarterly HbA1c. As routine clinical care, glucose management indicator (GMI) was used as substitute for HbA1c.18,19 The rates of meeting clinic targets and the rates of having no-show to scheduled appointments were used as markers for engagement with standard care. Continuous glucose monitor use metrics included the rates of CGM adoption and rates of consistent CGM data availability as defined by ≥ 70% data availability for successive 14-day periods over the study duration. 20 Remote patient monitoring engagement was defined as participant reading provider messages within seven days, as tracked by the MyChart “read” timestamp in the EHR. Exploratory assessment of barriers contributing to low patient engagement was obtained based on retrospective chart review of Social Work notes to identify psychosocial risk factors and unstructured interviews with the program CDCESs.
Glycemic Assessment
Hemoglobin A1c was obtained using either point-of-care or laboratory testing as routine care. Hemoglobin A1c from enrollment ± 1 month was compared with HbA1c at 12 ± 2 months using paired t-test. The rate of participants achieving HbA1c 7% or lower as recommended by the American Diabetes Association (ADA) and the International Society for Pediatric and Adolescent Diabetes (ISPAD) guidelines20,21 and the rate of participants achieving glycemic target of HbA1c 9% or lower as set by the Healthcare Effectiveness Data and Information Set 22 were used as glycemic metrics. Continuous glucose monitor data were collected from Dexcom Clarity web portal (Dexcom) or from the t:Connect web portal (Tandem Diabetes Care). Continuous glucose monitor data from participants on other systems (eg, Omnipod and Medtronic) who did not use devices compatible with RPM data transmission were not accessible by the clinical team nor research team. Continuous glucose monitor metrics were calculated at two-week intervals over study duration, including GMI, CGM TIR, TBR, and time in clinically significant hypoglycemia.14,18
Statistical Analysis
Data from all enrolled participants were analyzed under the intention-to-treat principle, using mixed effects methods to utilize all available data. Sub-analysis compared participants with and without consistent CGM data as defined by ≥ 70% CGM data available. Hemoglobin A1c and GMI were visualized using locally estimated scatter plot smoothing (LOESS). 14 Time in range was visualized as stacked bar plots over time. Linear mixed-effect models with a subject random effect were used to compare glycemic metrics, including HbA1c, GMI, and TIR at enrollment versus study end for participants with sufficient data.
Results
Participant Characteristics
Ninety-one youth enrolled with median age 14.7 years and median duration of diabetes 4.4 years at enrollment (Table 1). The cohort included all public insurance users, 47.3% Hispanic, and 31.9% non-English speakers. Mean HbA1c was 9.7% ± 2.1% at enrollment for the 86/91 (95%) participants with HbA1c available ±1 month of enrollment. About half of CGM TIPS participants attended the recommended quarterly clinic/telehealth appointments during the year prior to enrollment (48.4%) and during study period (46.2%). More than half of participants had at least one no-show to scheduled appointments. Prior to enrollment, 45/91 (49.5%) of participants had history of CGM use, 31/91 (34.1%) insulin pump use, and 11/91 (12.1%) AID use. Supplement S1 lists the timing of initiating CGM, insulin pump, and AID system prior to study versus during study and the device types.
Characteristics of the CGM TIPS Cohort.

Data are presented as n (%), mean ± standard deviation, or median (interquartile range).
Other languages include Arabic, Tongan, and Turkish (n = 1 each) for parents of the participants, although in each case the participants spoke English.
Diabetes technology use prior to enrollment was characterized based on having a prescription and appropriate teaching for these devices. However, patients enrolled in CGM TIPS were primarily individuals who had inconsistent technology use due to various barriers.
Program Engagement
All but three participants initiated CGM and most started within one week after enrollment (Supplement S2). Three participants exited the study due to transferring care or switching to private insurance. During the initial two weeks after enrollment, 52.1% of the cohort had CGM data consistently (Figure 1). The rate of consistent CGM use down-trended with time, but this was not significantly different by study end. Despite provision of CGM supplies, only 21 participants (23%) had consistent CGM data available throughout the 12-month period. Some differences in consistent data availability were noted by race/ethnicity and language but not statistically significantly (Supplement S3). The 91 participants received 626 RPM MyChart messages during the study and 57.2% of the messages were read by participants within seven days.

Participants with consistent CGM use as defined as ≥ 70% CGM data available during 12-month study period. Time point zero included analysis of CGM data for the initial two weeks after enrollment and subsequent time points examined data availability over four weeks. Cohort with consistent CGM data was highest during the initial two weeks of enrollment, 37/71 (52.1%). Rate of consistent CGM data during the last month of study was 31/71 (43.7%), which does not significantly differ from that at initiation (P = .40).
Glycemic Control
Hemoglobin A1c was available for 86/91 (95%) participants at ±1 month of enrollment and 58/91 (64%) participants at 12 ± 2 months after enrollment. Participants with paired HbA1c values at study start and end had mean HbA1c reduction from 9.8% to 9.0% (n = 58, P < .001) (Figure 2a). Glucose management indicator showed no difference over study period (Figure 2b). Cohort mean CGM TIR ranged from 32% to 48% and was not different over study period (Figure 2c). Mean TBR was 2.3% ± 3.1% and time in clinically significant hypoglycemia was 0.48% ± 1.0% for the whole cohort (n = 88). Certified Diabetes Care and Education Specialist frequently attempted to communicate hypoglycemia recommendations via MyChart for individuals who had > 4% TBR or > 1% time in severe hypoglycemia, but many messages were unread. Many patients had follow-up phone calls from CDCESs if hypoglycemia persisted.
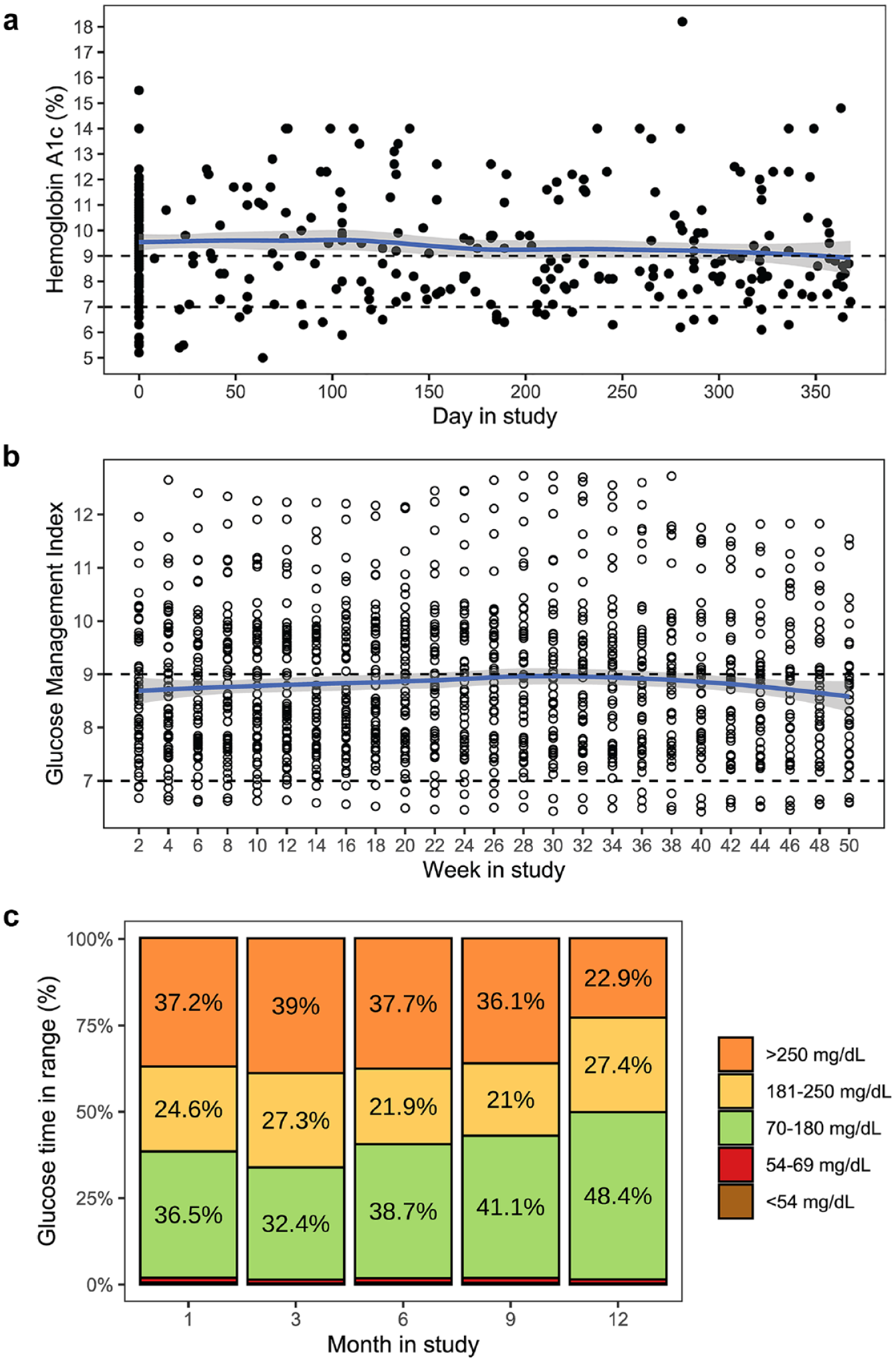
Glycemic control over study period as measured by hemoglobin A1c (HbA1c) (a), glucose management indicator (GMI) (b), and glucose distribution from continuous glucose monitor (CGM) readings (c). Scatter plot of HbA1c levels (a) over time with locally estimated scatter plot smoothing (LOESS) show significant reduction over 12 month study period (P < .001). Glucose management indicator was not different at enrollment versus at 1 year (8.6 ± 1.2 vs 8.5 ± 1.3, P = .08) (b). Continuous glucose monitor time in range was not statistically different at enrollment versus at one year (36.5% vs 48.4%, P = .8) (c).
For the sub-analysis, we characterized the 21 youth with consistent CGM data ≥ 70% throughout the study, which is needed to provide clinically meaningful CGM metrics. Locally estimated scatter plot smoothing and linear models showed consistent CGM data availability was associated with lower HbA1c (HbA1c 8.7 vs 9.7, P = .02) (Figure 3a) and lower GMI (GMI 8.2 vs 8.9, P = .02) (Figure 3b). Consistent CGM data availability was not associated with any changes with CGM TIR distribution over time (Figure 3c).
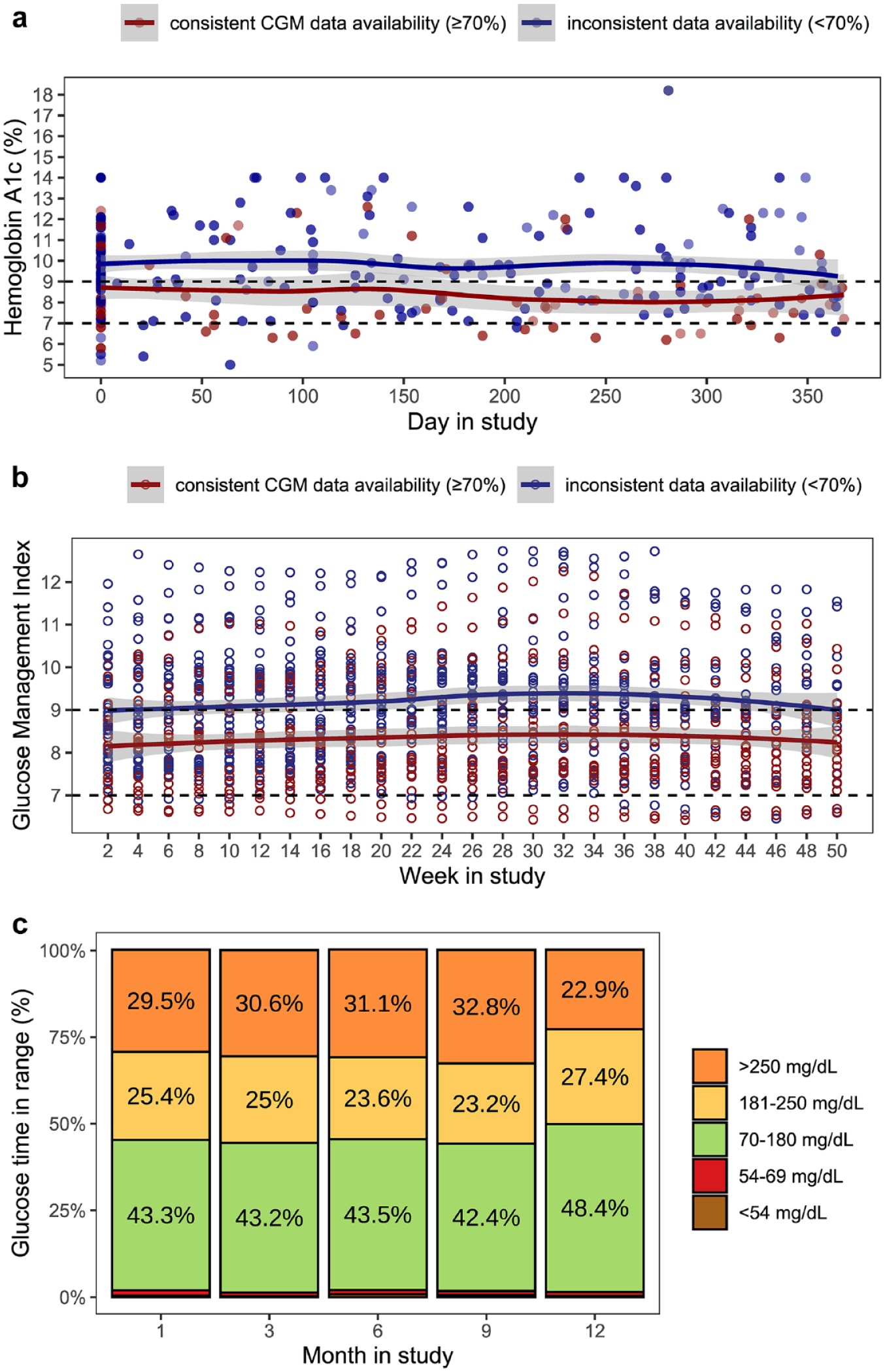
Glycemic metrics for sub-cohorts with consistent continuous glucose monitor (CGM) data availability (≥ 70%) in red vs inconsistent data availability (< 70%) in blue, including hemoglobin A1c (HbA1c) (a) and glucose management indicator (GMI) (b). Sub-cohort with consistent CGM data had lower HbA1c compared with the sub-cohort without consistent CGM data (HbA1c 8.7 vs 9.7, P = .02). Similarly, sub-cohort with consistent CGM data had lower GMI compared with the sub-cohort without consistent CGM data (GMI 8.2 vs 8.9, P = .02). Glucose distribution from CGM readings for the sub-cohort with consistent CGM data availability (n = 21) shows no difference at enrollment versus study end (c).
Rates of Insulin Pump and Automated Insulin Delivery System Use
During the study period, insulin pump users increased from 31 to 48 and AID users increased from 11 to 38. Additional description of device types and initiation timing are given in Supplement S1. Time in range up-trended for participants who initiated insulin pump (n = 17, TIR = 30.8%-63.2%) and AID (n = 27, TIR 34.3%-59.8%), but trends in glycemic effect were not statistically significant (Supplement S4, S5).
Discussion
Implementation of a pilot program using asynchronous RPM and timely CDCES support is an important step toward improving outcomes and identifying barriers to consistent CGM use for youth with established T1D and public insurance. Our program was successful in capturing a population of marginalized youth with T1D who are challenging to engage, have low technology use, and at high risk for diabetes associated complications. All participants used public insurance, a third of the cohort were non-English speakers, more than half were from ethnic minority backgrounds, majority had identified psychosocial risk factors that may be barriers to diabetes care, and nearly half did not attend recommended quarterly clinic visits. Notably, the CGM TIPS cohort included 47.3% Hispanic participants, which was much higher than the 4T Pilot Study (21.5%), the 4T Study 1 (36.8%), and other diabetes technologies studies ranging from 2% to 21%.16,23 Importantly, despite provision of CGM supplies and RPM, participant engagement was low—only 23% had sufficient CGM data and 64% had paired HbA1c at study initiation and end—which raises concern about attrition bias. Although the study did not meet typical feasibility threshold of > 70% data availability, this level of participation is still notable considering the baseline challenges engaging this population and the intervention designed for sustainability without ongoing research funding. W built upon prior studies to explore care models better suited to support youth with T1D from marginalized backgrounds through a team-based, algorithm-enabled approach. Lessons from this study provide insights for iterative improvements at the levels of RPM modality and data connectivity to better support this population.
Low participant engagement and data missingness were major limitations identified in this feasibility study. We noted important barriers, including delays in identifying CGM supply gaps, remote connectivity for CGM data sharing, logistical challenges with HbA1c collection in setting of fewer in-person visits during the COVID-19 pandemic, and participant psychosocial risk factors. Future iterations of CGM TIPS will be focused on further understanding and addressing these barriers.
Throughout the program, participants were encouraged to reach out to CDCES for help regarding supply gaps. However, many participants did not proactively reach out, so that, supply gaps were only identified up to four weeks later at scheduled RPM time points. Adjusting RPM frequency to flag gaps in CGM data earlier may be helpful as will identifying barriers to proactive outreach by participants.
Logistical factors contributed to data loss as well. Low rates of baseline clinic attendance and conversion to telehealth visits during the COVID-19 pandemic both contributed to missed opportunities for CGM data download and HbA1c testing. For patients using Dexcom receiver or t:slim ×2 pump as the receiver, any CGM data that were not transferred to the web portal within 30 or 90 days, respectively, were lost. Implementing alternate workflow to increase clinic attendance or support remote collection of glycemic data are additional areas of improvement for this program.
Despite provision of CGM supplies and monthly CDCES RPM support, some participants were still unable to consistently use CGM or provide the care team with CGM data to guide management. This suggests that significant barriers to diabetes technology use remain in place for this population. Anecdotal experience from clinicians involved in these patients’ care suggested that psychosocial risk factors including housing insecurity and food insecurity contributed to barriers for some families in the program. 9 We observed some interesting differences in the race/ethnicity and language of participants with and without consistent CGM data. Additional work is needed to assess these initial findings in order to inform interventions targeting these potential barriers.
Remote patient monitoring has potential to revolutionize management of T1D by enabling more frequent opportunities to encourage patient engagement.15,24,25 While the CGM TIPS and 4T studies used similar RPM support for youth with T1D, CGM TIPS participants with long-standing out-of-target glycemic control had much lower rates of achieving the glycemic target of HbA1c ≤ 7% at study end (16%) compared with the new-onset 4T Pilot cohort (53%), 4T Pilot Hispanic subgroup (47%), 4T Pilot public insurance subgroup (47%), and the 4T Study 1 (64%).14,16,26 We speculate that early initiation of CGM and building healthier habits for diabetes management in the new-onset period contributes to this difference. We further speculate that the CGM TIPS cohort requires different types of RPM support. Current clinic standard workflow includes CDCES sending RPM communication via patient portal messaging. Our finding of low RPM message read rate aligns with prior studies showing that patient portal messaging is not effective for communicating with families from minoritized background.27,28 We hypothesize that more personalized communication can improve engagement with RPM. Future studies will assess patient preferences for RPM modality, frequency, timing, and type of language. Since many participants in CGM TIPS are adolescents with years of diabetes self-management experience, it may be reasonable to target the adolescent participant vs parent participant for RPM communication.
Automated insulin delivery system adoption increased during this study, and AID adopters had a non-statistically significant but increasing TIR throughout the study. We hypothesize that AID uptake is related to participants having sustained CGM supplies, which is a necessary component of AID. However, we cannot rule out confounding factors, such as general trends of increasing AID uptake in our community as we offer insulin pump and AID education classes widely as standard care. Additional studies are needed to evaluate whether AID use improves glycemia in this population in the real-world setting.
Implementation of this technology-enabled RPM program to support CGM use in marginalized youth with T1D is an important step forward in our mission of provide equitable care. Though this program was faced with feasibility challenges to engage participants using existing standard communication practices, we are optimistic that lessons from this study can inform iterative improvements to better serve this patient population. Future directions include systematically identifying barriers to diabetes care and using psychosocial context to inform more effective RPM communication strategies to engage this population.
Supplemental Material
sj-docx-1-dst-10.1177_19322968241305612 – Supplemental material for Addressing Disparities Using Continuous Glucose Monitors and Remote Patient Monitoring for Youth With Type 1 Diabetes
Supplemental material, sj-docx-1-dst-10.1177_19322968241305612 for Addressing Disparities Using Continuous Glucose Monitors and Remote Patient Monitoring for Youth With Type 1 Diabetes by Ming Yeh Lee, Victor Ritter, Blake Shaw, Johannes O. Ferstad, Ramesh Johari, David Scheinker, Franziska Bishop, Manisha Desai, David M. Maahs and Priya Prahalad in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors would like to acknowledge all youth who participated in the CGM TIPS Study. They would also like to thank the other members of the 4T Study research team, including research coordinators, clinical staff, students in the Systems Utilization for Stanford Medicine (SURF) group, the Quantitative Sciences Unit, the T1D Working Group in Statistics, and Informatics at Stanford Medicine Children’s Health.
Abbreviations
ADA, American Diabetes Association; AID, automated insulin delivery; CDCES, Certified Diabetes Care and Education Specialist; CGM, continuous glucose monitor; CGM TIPS, CGM Time in Range Program at Stanford; EHR, electronic health record; GMI, glucose management indicator; HbA1c, hemoglobin A1c; ISPAD, International Society for Pediatric and Adolescent Diabetes; IQR, interquartile range; LOESS, locally estimated scatter plot smoothing; RPM, remote patient monitoring; SES, socioeconomic status; 4T, Teamwork; Targets; Technology; and Tight Control; TIDE, Timely Interventions in Diabetes Excellence; TIR, time in range; TBR, time below range; T1D, type 1 diabetes.
Author Contributions
MYL, PP, and DMM designed the study, researched data, conducted statistical analysis, wrote, and edited the manuscript. VR and BS conducted statistical analysis and wrote the manuscript. JOF, RJ, DS, FB, and MD contributed to the study design and editing of the manuscript. MYL and PP are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Maahs has had research support from the NIH his institution has had research support from Dexcom. Dr Maahs has consulted for Abbott, the Helmsley Charitable Trust, Lifescan, Sanofi, Medtronic, Provention Bio, Kriya, Biospex, and Bayer. Dr Prahalad has consulted for Sanofi. The remaining authors do not report any relevant disclosures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MYL is the Elizabeth and Russell Siegelman Postdoctoral Fellow of the Stanford Maternal and Child Health Research Institute (MCHRI) and is supported by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) of the National Institutes of Health (NIH) under Awards T32 DK007217-47 and 5K12DK122550-05. VR, BS, and MD are partially supported by the NIH through Stanford’s Center for Clinical and Translational Education and Research award, under the Biostatistics, Epidemiology and Research Design (BERD) Shared Resource (UL1TR003142). DMM, PP, JOF, and RJ have all received support from Stanford MCHRI, Stanford Human-Centered Artificial Intelligence, and the National Science Foundation (NSF) under grant number 2205084. JF has received support from the Stanford Data Science Scholars Program. DMM, DS, and RJ have received funding from the Helmsley Charitable Trust.
Funding for iOS devices and some CGM supplies were provided through philanthropic funding by the Lucile Packard Children’s Hospital Auxiliaries Endowment and by a grant through the Lucile Packard Foundation for Children’s Health—Moca Foundation. Additional CGM supplies were provided by Dexcom. No other research funding was used to support CGM TIPS. The funders had no role in the design, data collection, data analysis, and reporting of this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
