Abstract
Background:
Since the discovery of the life-saving hormone insulin in 1921 by Dr Frederick Banting in 1921, there have been many critical discoveries and technical breakthroughs that have enabled people living with type 1 diabetes (T1D) to live longer, healthier lives. The development of insulin pumps, continuous glucose monitoring (CGM) systems, and automated insulin delivery (AID) systems have enabled people living with T1D to safely manage their glucose, reduce their HbA1c, and improve their overall health and quality of life. Nevertheless, AID systems are not yet designed for all people with T1D, and they perform best during the overnight period when meals and exercise are not occurring. AID systems are not fully automated in that they require the person using the system to announce meals and exercise to the system to avoid dangerous hyper- or hypoglycemia, respectively.
Methods:
In this review, which is one of a collection of manuscripts to commemorate the 75th anniversary of the National Institute for Diabetes and Digestive and Kidney Diseases, we celebrate the commercialization of the AID and discuss the major challenges and research gaps that remain to be solved to enable single- and multi-hormone AID systems to more fully support glucose management in people living with T1D.
Results:
More research is required to design and evaluate more intelligent AID systems that do not require accurate carbohydrate estimations or announcements for meals and exercise. Current AID systems are also not designed to be used by older adults or pregnant people. Results are presented on new AID systems that can automatically respond to meals and exercise. Results are also presented on evaluations of AID systems in older adults and pregnant people.
Conclusions:
Next-generation AID systems will need to support all people, including older adults, people during pregnancy, athletes, and people who may be too busy to announce carbohydrates or exercise to the system. Solutions are now becoming available that will enable AID systems to support a broader range of people living with T1D by leveraging the latest technologies in artificial intelligence and adaptive control.
Keywords
Introduction
This article is part of a collection of publications to commemorate the 75th Anniversary of the NIDDK.
The National Institute for Diabetes and Digestive and Kidney Diseases (NIDDK), which was founded 75 years ago, has a mission to conduct and support medical research and to disseminate science-based information on diabetes and other endocrine and metabolic diseases to improve peoples’ health and quality of life. In this manuscript, we celebrate the 75 years of progress by the NIDDK by showcasing a major accomplishment that the NIDDK has enabled for saving lives and improving quality of life for people living with type 1 diabetes. Throughout its 75 years, NIDDK has supported its mission through support of basic, preclinical, and clinical research to develop novel treatments and technologies to improve the lives of people with diabetes. The commercialization of automated insulin delivery (AID) was only possible through substantial support through grants from the NIDDK. This investment has served the mission of NIDDK as the AID has enabled improved human health by enabling significant improvements in glucose outcomes for people living with type 1 diabetes (T1D). However, there is more work to do and NIDDK is leading the way in this effort. Current AID systems are not fully automated in that they still require meal carbohydrates to be announced to the systems. Exercise can still be a challenge for current AID systems, which do not automatically detect physical activity or adjust dosing to prevent exercise-induced hypoglycemia. Furthermore, AID has not yet been fully evaluated and specified for all populations of people with T1D including women during pregnancy or older adults. In this NIDDK 75th year anniversary review, we explore the current gaps and challenges with AID while suggesting new areas that offer opportunities for future improvement.
Meals and Hybrid Closed Loop
Current automated AID systems are hybrid, requiring carbohydrate content to be “announced” for insulin dose calculation before a meal starts. Although AID systems significantly improve glycemic outcomes, managing postprandial glucose excursions is still challenging due to several factors, including the timing of premeal bolus, accurate carbohydrate counting, missed meal boluses, and meal fat and protein content. This need for user input also increases diabetes burden and negatively impacts the quality of life.
Over the years, several strategies have been developed to overcome these challenges. One approach eliminates an accurate carbohydrate count requirement. Instead, it utilizes a qualitative estimate of carbohydrate content (“usual” “more,” or “less”) as compared with a typical meal for the individual.1,2 Other dual-hormonal (insulin and glucagon) algorithms 3 have explored a similar method. In a small study by Petrovsky et al, 4 70% of 34 adolescents with T1D reached the American Diabetes Association HbA1C goal using an AID system and simplified meal announcements with three preset personalized fixed carbohydrate amounts.
A smartwatch application capable of detecting eating behavior and alerting the user improved glycemic results by reducing the rate of late/missed meal boluses. It increased (time in range, 70-180 mg/dL) TIR and decreased HbA1C in a small cohort of individuals with T1D. 5 Using this “detecting gesture app,” an AID showed similar TIR while the system converted the gestures to carbohydrate content without requiring a manual bolus. 6 A retrospective analysis showed that the smartphone bolus feature increased the number of boluses by the user, 7 which may contribute to improved outcomes.
Automated insulin delivery systems rely mainly on the entered carbohydrate content for bolus dose calculations, along with minor adjustments for deviations from a pre-set target glucose range. However, it is well known that meal protein content has a dose-dependent effect, and fat content can increase insulin resistance, cause early hypoglycemia, and late hyperglycemia. 8 Most systems do not incorporate meal composition in their insulin dosing models. However, a few studies assessed the impact of nutrient variables on post-prandial excursions in individuals who use AID systems.9,10 More advanced algorithms help with small, missed meal boluses. In a small outpatient study of adults with T1D by Shalit et al 11 using AID, missed announcements of up to 20 g of carbohydrates did not decrease TIR significantly.
Current insulin formulations have a slower onset of action than endogenous insulin. A few studies have compared a more rapid-acting insulin’s impact on postprandial glucose trends in the setting of AID, but the findings are inconsistent.12-18 An alternative strategy is to use an adjunct treatment, such as pramlintide (an amylin analog), to delay gastric emptying and reduce glucagon secretion in a closed-loop algorithm to reduce the need for carbohydrate counting. In this study of 30 adolescents and adults with T1D, the insulin plus pramlintide system with simple meal announcement achieved the non-inferiority margin with only mild side effects. 19 Another study explored sodium-glucose co-transporter-2 (SGLT2) inhibitors in conjunction with simple meal announcements, which was also found to be non-inferior to the carbohydrate counting approach. 20
In summary, future work should combine the mentioned strategies, including simplifying carbohydrate entry, developing ultra rapid-acting insulin formulation, and creating meal detection algorithms integrated with meal composition models and adjunctive therapies. This approach will lead to more personalized and effective diabetes outcomes, particularly in achieving optimal postprandial glucose management.
Physical Activity and Multi-Signal Closed-Loop
Pioneering work on AID systems and exercise management has shown that sophisticated algorithms could limit activity-induced hypoglycemia.21-30 Because of this early work, most current commercially available AID systems have an activity/exercise mode, whereby the target glucose level is elevated above the usual level and insulin delivery is reduced during exercise. This can be effective at reducing exercise-related hypoglycemia across various forms of exercise.31-36 However, these systems all require user initiation well before exercise. Current clinical guidelines on exercise and AID systems recommend the use of higher temporary targets in advance of exercise. 37 Unfortunately, these and other hypoglycemia mitigation strategies are seldom used by most individuals with T1D on AID who exercise regularly. 38
Studies that model the complexity of glucose and insulin dynamics during physical activity in T1D have recently been conducted that should improve on current AID systems.39-44 Exercise “events” can now be detected using commercial products like smartwatches and activity bands or rings, which can be incorporated into proof-of-concept AIDs for exercise management.29,45,46 However, these signals and various activity-informed algorithms are not yet fully integrated into commercial AID systems for various technical and feasibility reasons and only a few AID systems with advanced exercise settings have been tested.26,27,29,30,47
Activity monitors typically use heart rate photoplethysmography and accelerometry data to detect exercise, sleep and stress events. Some activity trackers can estimate energy expenditures during daily physical activity, typically reported as metabolic energy equivalents, to estimate exercise intensity. This information may be particularly valuable in emerging AID systems as the requirement for insulin changes nonlinearly with increasing exercise intensities. 48 Technical and complex physiological challenges remain in the development of an AID system that automatically responds to physical activity. The known “hurdles” for this include the rapid changes in glucose turnover during exercise, the effect of exercise on insulin pharmacokinetics and pharmacodynamics, and the abnormal glucagon responses to exercise and/or hypoglycemia in T1D. 49
With prolonged aerobic exercise, subcutaneous insulin absorption increases in persons with T1D on pump therapy and systemic insulin levels tend to rise, which increases glucose disposal and limits hepatic glucose production. 50 Vasodilation of the skin and skeletal muscles caused by aerobic exercise increases skeletal muscle glucose oxidation rate while also increasing subcutaneously delivered insulin absorption rate, which contributes to a relative hyperinsulinemia that limits hepatic glucose production thereby causing a drop in glycemia unless carbohydrates are consumed. 51 The blunted glucagon response to exercise in T1D likely increases hypoglycemia risk.52,53 These pharmacokinetic and pharmacodynamic actions of insulin and glucagon during exercise may be overcome by using innovative dual hormone AID systems typically with model predictive controller (MPC) algorithms that can reduce or suspend insulin and infuse glucagon automatically.54-56
Activity-induced hyperglycemia can result from intensive exercise, resistance training or competition stress, typically caused by the rapid increase in counterregulatory hormones and lactate. 57 Some insulin-only AID systems already may manage post-exercise hyperglycemia well.32,58 Proof-of-concept AID systems can now accurately and automatically distinguish between various forms of exercise and alter insulin delivery accordingly. 59 Improving glucose time-in-range (TIR) outcomes during and following exercise may be supported in the future through use of faster insulins and AI-enabled precision-medicine systems that use pattern recognition and digital twins to provide automated dosing adjustments and decision support around exercise. 60
The Artificial Pancreas in Pregnancy
Pregnancies complicated by pre-existing diabetes have an increased risk of adverse pregnancy outcomes (APOs), such as fetal/neonatal deaths, congenital anomalies, preeclampsia, preterm delivery, abnormal fetal growth, and neonatal morbidity. Tighter glucose targets are required compared to those outside pregnancy (63-140 mg/dL pregnant vs 70-180 mg/dL non-pregnant) to reduce APOs. While AID use has demonstrated improvements in glycemic outcomes and reductions in the burdens of self-care for people with diabetes, less research has focused on the unique needs of pregnancy in individuals with T1D, 61 with even fewer data in those with type 2 diabetes (T2D).
Changing levels of maternal insulin resistance throughout gestation pose glycemic challenges.62,63 The insulin resistance of pregnancy is multifaceted and is influenced by hormonal, placental, genetic, and epigenetic contributions, unique to each pregnant person. Maternal glycemia is also influenced by maternal physical activity levels, diet/microbiome, and weight. 64 Together these commonly lead to changing insulin doses: 20% less insulin in early gestation, 200-300% more insulin by delivery, and up to 50% less insulin post-partum compared to pre-pregnancy.65,66
Implementing AID use during pregnancy requires unique configurations including: (1) ability to safely achieve the tighter pregnancy time in range prior to conception and throughout gestation, (2) algorithm adaptability to changes in insulin sensitivity throughout gestation, (3) assertive post-prandial insulin delivery including a mechanism to respond to changes in glycemia postprandially, (4) more aggressive glucose targets without an increased risk of maternal hypoglycemia, and (5) ability to rapidly adapt to changes in insulin requirements post-partum (through setting adjustments and algorithm adaptation). 67 Additionally, a pregnancy-specific system should be user-friendly, regardless of an individual’s health care status or literacy, to maximize benefits and reduce risks throughout gestation.
Use of continuous glucose monitoring (CGM) during pregnancy is associated with improvements in fetal outcomes for T1D pregnancies. While a few trials have shown improved maternal glycemia with AID therapy, none were adequately powered to assess maternal and/or neonatal health outcomes 68 (Table 1). To date, only one AID algorithm has an indication for use during pregnancy, and only two algorithms offer customization to reach the pregnancy glycemic targets. When these systems are unavailable, many people struggle with whether to use or discontinue an AID system which met preconception needs. Many pregnant users of off-label systems, without access to pregnancy-specific systems, use assistive bolusing techniques to increase insulin delivery with variable results. Others revert back to CGM with multiple daily injections or insulin pump delivery with inconsistent outcomes. 78 However, many pregnant women with T1D generally prefer using AID therapy, whether or not it is specifically designed to meet pregnancy goals.79-81
Selected Studies Evaluating AID Use During Pregnancies in Women With T1D.
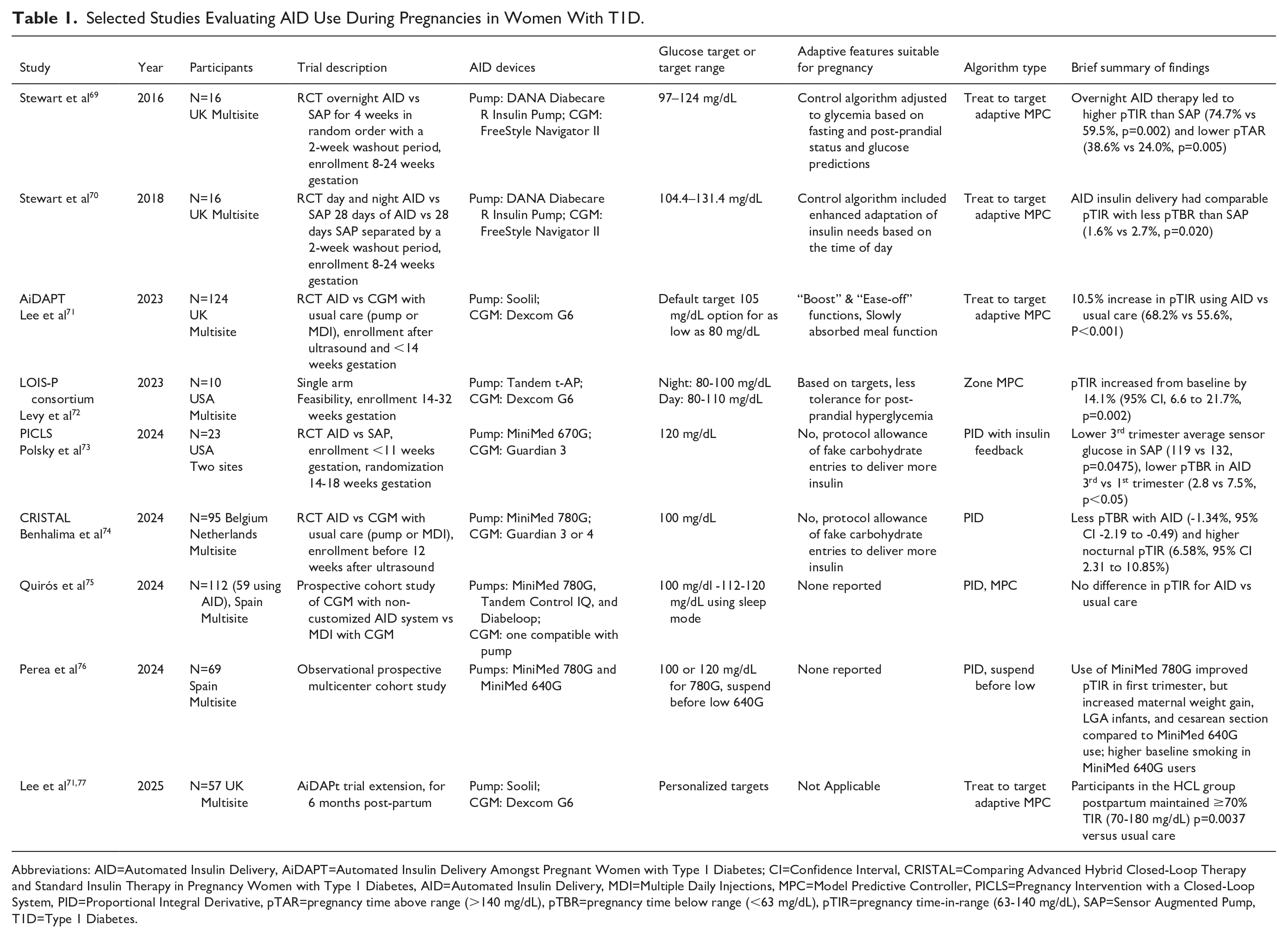
Abbreviations: AID=Automated Insulin Delivery, AiDAPT=Automated Insulin Delivery Amongst Pregnant Women with Type 1 Diabetes; CI=Confidence Interval, CRISTAL=Comparing Advanced Hybrid Closed-Loop Therapy and Standard Insulin Therapy in Pregnancy Women with Type 1 Diabetes, AID=Automated Insulin Delivery, MDI=Multiple Daily Injections, MPC=Model Predictive Controller, PICLS=Pregnancy Intervention with a Closed-Loop System, PID=Proportional Integral Derivative, pTAR=pregnancy time above range (>140 mg/dL), pTBR=pregnancy time below range (<63 mg/dL), pTIR=pregnancy time-in-range (63-140 mg/dL), SAP=Sensor Augmented Pump, T1D=Type 1 Diabetes.
More research is necessary to further develop and refine therapeutic options to maximize glycemic control, reduce self-care burden, and improve health outcomes prior to conception, throughout gestation, and into the post-partum period. Further evaluation and understanding of pregnancy-specific CGM metrics, strategies to achieve higher pregnancy-specific time in range to reduce large-for-gestational age babies and other APOs, along with automated approaches to support pregnancies with T1D and T2D, requires contributions from patients, academia, industry partners, and continued support from the NIH and other funding agencies.
The Artificial Pancreas in Older Adults
Older adults with T1D represent a growing population and despite advancements in treatment options, limited research accounts for the unique characteristics of aging with T1D. 82 Challenges in frailty, dexterity, vision, cognition/executive functioning among other complications need to be considered when implementing treatment plans. 83 These co-morbid conditions may affect the uptake and maintenance of diabetes-related technology which has shown superior glycemic outcomes in other age ranges and yet only emerging data exist in older adults with T1D.84,85 In older adults, concerns have been raised regarding increased risks of severe hypoglycemia (SH) and its attendant consequences including falls and fractures, particularly in the setting of impaired hypoglycemia awareness with increasing duration of diabetes.86-89 The WISDM study of CGM use in older adults with T1D notably identified unrecognized higher rates of hypoglycemia overnight and reported beneficial glycemic outcomes with CGM use alone.90,91 Recently, a randomized controlled study using CGM enhanced with geriatric principles consisting of setting appropriate glycemic goals and implementing simplification strategies based on overall health of older adults with T1D, has shown reduced hypoglycemia without worsening glycemic control in a cost-effective fashion. 84 To date, there have been at least three randomized controlled trials in AID use focused in older adults, two of which were sponsored by the NIDDK/NIH92,93 with encouraging glycemic results (Table 2).
Selected Randomized Trials in Older Adults With T1D on AID Systems.
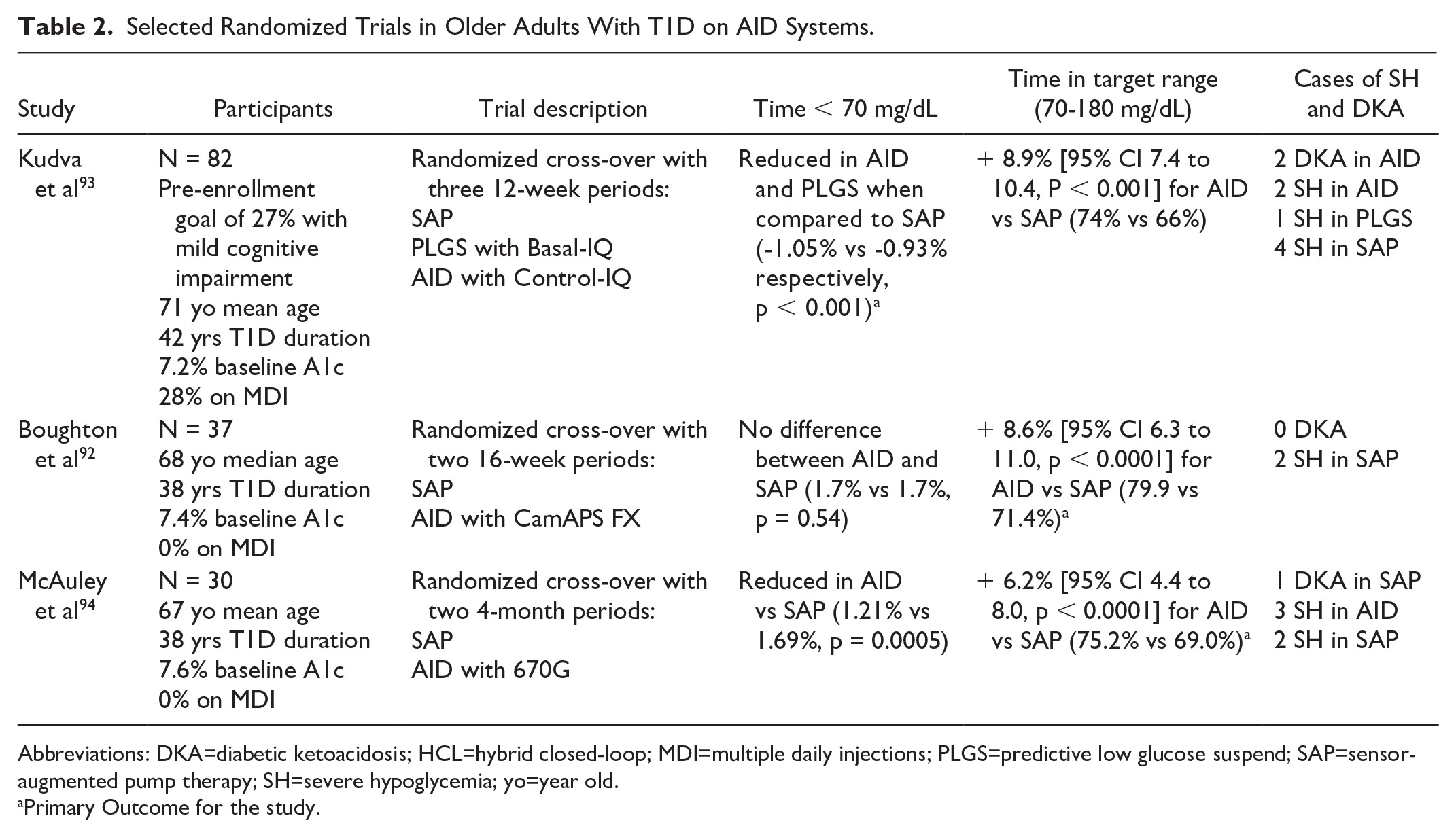
Abbreviations: DKA=diabetic ketoacidosis; HCL=hybrid closed-loop; MDI=multiple daily injections; PLGS=predictive low glucose suspend; SAP=sensor-augmented pump therapy; SH=severe hypoglycemia; yo=year old.
Primary Outcome for the study.
The largest randomized trial in older adults reported a reduction in hypoglycemia in AID and predictive low glucose suspend (PLGS) compared to sensor augmented pump (SAP) for 12 weeks. 95 In addition, AID improved time in range, reduced hyperglycemia, glycemic variability, and HbA1c to the levels below those recommended for older adults. An extension phase found that participants preferred the AID system over PLGS or SAP when given a choice suggesting that the AID treatment option was well received. 96 A second trial in older adults reported increased time in range using AID compared to SAP for 16 weeks without an increase in hypoglycemia. 92 Follow-up studies indicated favorable patient reported outcomes including less worry about diabetes. 97 An earlier randomized cross-over study of a first-generation system that required fingersticks reported improvements in time in range for AID compared to SAP for four months in older adults. 94 There have been subsequent reports in real-world use of these systems in older adults that are encouraging98-100 and report favorable outcomes of time in target range without increasing hypoglycemia risk,101-103 while there remains mixed outcomes in quality of life such as sleep.104,105
These studies collectively support the use of AID in older populations with T1D but are limited by relatively small number of participants with little demographic diversity (eg, ethnicity or educational attainment) and overall good health, making it more difficult to generalize. However, these findings begin to challenge the notion of whether guidelines in overall healthy and functionally independent older adults should in fact include relaxed glycemic targets to avoid hypoglycemia given that AID devices could potentially achieve less hyperglycemia without a concomitant increased risk of hypoglycemia. Future research should stratify individuals by frailty scores and include human factor studies with geriatric principles as well as non-glycemic outcomes such as falls, fractures, worry indices and sleep metrics to capture the characteristics of aging with T1D.
The Artificial Pancreas in Type 2 Diabetes
The use of AID systems in people with type 2 diabetes (T2D) is growing, but high-quality evidence from randomized controlled trials in this population is just starting to become available. In a 13-week randomized multicenter trial of 319 people with T2D, HbA1c decreased by 0.9 percentage points in the AID group compared with 0.3 percentage points in the control group. 106 In two single-center crossover trials, the CE marked CamAPS HX fully closed-loop system (no meal announcements) was safe and associated with increased time in target range compared with standard insulin therapy without increasing time in hypoglycemia.107,108 In a multicentre crossover trial involving adults with T2D using insulin pumps in France, hybrid closed-loop improved time in range compared to insulin pump and sensor without increasing hypoglycemia. 109
Data from non-randomized before and after studies of hybrid closed-loop systems including Control-IQ, Omnipod 5, and Medtronic MiniMed 780G in adults with T2D have recently been reported, but efficacy cannot be determined due to the lack of a control group.110-112 Real-world observational data is also available for Control-IQ, which recently received FDA approval for use in adults with T2D.113,114 Significant challenges around the resources required for clinical implementation in this population exist.
Fully-Automated Closed Loop: Clinical and Engineering Perspectives
The ultimate goal of insulin delivery automation for individuals with T1D is to reduce or eliminate user burden and need to interact with their diabetes devices. The challenging task of managing postprandial hyperglycemia can further be complicated by late hypoglycemia, mainly due to the delayed onset of insulin action. Various algorithms, mostly MPC, have been explored using different meal anticipation and detection approaches based on glucose and insulin data. A zone MPC showed promising results in short-term supervised studies facing unannounced meals and exercise115-121; a step toward fully closed loop (FCL) therapy was the use of adaptive layer to the Zone-MPC hybrid AID, resulting in a significant reduction in HbA1C following 12 weeks. 122 In addition, a multiple model probabilistic predictive control (MMPPC) that anticipated meals only when the patient was awake 123 demonstrated a mean CGM value of 157 mg/dL during a hotel study that included meal and exercise challenges.123,124 Artificial intelligence approaches have also been used to automatically detect and dose for meals. In a small study, Mosquera-Lopez and colleagues developed a neural network that could automatically detect meals, estimate the meal size, and dose insulin in response to the detected glucose, showing a 10.8% reduction in postprandial time in high glucose (<180 mg/dL). 125
The team at the University of Virginia Center for Diabetes Technology has developed an adaptative MPC schema designed to function in FCL, showing significant improvement over a current AID system in a supervised study of 18 adolescents. Further improvements were shown with automatic priming bolus triggering when meal-like CGM profiles were detected. 126 Furthermore, the MPC can be modified to anticipate repeating eating behavior while ensuring safety when user habits change; though while shown in-silico, postprandial TIR improvements were not apparent in a recent feasibility study.127,128 Home studies are currently under way (NCT06041971, NCT06633965).
The CamAPS algorithm from Cambridge was tested in adults with T1D and suboptimal glucose management. This FCL significantly improved glucose control without increasing the risk of hypoglycemia. 129 In addition, the system has been shown to improve glycemic control in complex medical and surgical inpatients with T2D with challenging glycemic management. 130
Fuzzy-logic control algorithm DreaMed GlucoSitter system with Faster Aspart was evaluated in FCL during a double-blind, randomized, crossover trial with 20 participants. There was no significant difference in glycemic outcomes between standard and faster Aspart arms with exercise and meal challenges. 131
Evidence shows adjunctive treatments like glucose-like peptide-1 receptor agonists (GLP 1 RAs), GLP-1/glucose-dependent insulinotropic polypeptide (GIP) dual receptor, and SGLT2 inhibitors can improve glycemic outcomes and decrease body weight and insulin requirement.132-134 In addition to glucose management, these drugs decrease cardiovascular and kidney disease risks. These adjunctive therapies may ultimately be utilized in combination with FCL systems, however, the algorithms may need to be informed of the adjunctive therapy and adjusted based on the treatment. Pramlintide, which is an analog of the hormone amylin, delays gastric emptying and when delivered along with insulin at the time of a meal, can significantly reduce the postprandial glucose spike by enabling the insulin kinetics to more closely match carbohydrate absorption rates. 135 In a study conducted by Tsoukas et al, 136 the combination of Fiasp and pramlintide in multi-hormone FCL was not found to be non-inferior to a hybrid closed loop system with Fiasp in adults with T1D with suboptimal glucose control at baseline.
The Inreda AP bihormonal (insulin and glucagon) FCL system (Netherlands) was tested in a trial with 78 adults with T1D. After 1 year of FCL treatment time in range was increased from 55.5% at baseline to 80.3% and median time below range was just 1.36%. 137
Conclusions
While the critical support of the NIDDK has enabled the commercialization of the AID, there is still a need to continue to improve AID technologies so that they can support all populations of people living with T1D. Next generation AID and multi-hormone automated hormone delivery systems will likely be able to automatically handle meals without user input and automatically detect and adjust dosing during exercise. New AID systems are expected to be more highly personalized so that they can adapt to each person’s physiology and the physiology of different groups of people living with T1D such as pregnancy and older adults. Continuing research is ongoing to identify the best metric, or group of metrics, for evaluation of AID systems including TIR, time in tight range (70-140 mg/dL), and how best to implement these to improve outcomes.138-141 Future studies should evaluate the cost-effectiveness and impact on quality-of-life of usage of AID by people with T1D. A narrative review by Mathieu et al 142 of 18 studies evaluating cost effectiveness and quality of life showed that nearly all studies demonstrated that AID systems were cost effective and improved quality of life. Artificial intelligence, adaptive controls, faster insulin, adjunctive medications, and additional studies in more heterogeneous cohorts will be critical for enabling innovations that can improve performance and glucose outcomes in people with T1D. We expect that the NIDDK will continue to fulfill its mission to improve human health by enabling innovative research and development efforts that will address these challenges to provide the next generation of therapeutics to improve health in people living with diabetes.
Footnotes
Abbreviations
ADA, American Diabetes Association; AID, automatic insulin delivery; CGM, continuous glucose monitoring; CSII, continuous sub-cutaneous insulin infusion; DKA, diabetic ketoacidosis; EASD, European Association for the Study of Diabetes; FCL, fully closed-loop therapy; GIP, glucose-dependent insulinotropic polypeptide; GLP 1 RA, glucagon-like peptide-1 receptor agonists; HbA1c, glycosylated hemoglobin (%); HCL, hybrid closed-loop system; MPC, model predictive control; NIH, National Institute of Health; NIDDK, National Institute of Diabetes and Digestive and Kidney Diseases; PLGS, predictive low glucose suspend; SAP, sensor-augmented pump therapy; SGLT-2i, sodium-glucose transport protein 2 inhibitor; T1D, type 1 diabetes; T2D, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PGJ receives research support from Dexcom and Eli Lilly and is a co-founder and shareholder in Pacific Diabetes Technologies. CJL has received research support from Tandem Diabetes, Dexcom, Abbott, Mannkind, NovoNordisk, and Insulet paid to her institution and has served as a consultant for Tandem Diabetes and Dexcom. SP was a contributing writer for diaTribe, was on the Medical Advisory Board for Medtronic MiniMed, Inc, has received research funding from Dexcom, Inc., Eli Lilly, JDRF, Leona & Harry Helmsley Charitable Trust, NIDDK, and Sanofi US Services, has received research support from Diasome Pharmaceuticals, Medtronic MiniMed, Inc., and Sanofi US Services, and has received honoraria from the Children’s Diabetes Foundation and the American Diabetes Association. MSP has received research funding and honoraria from Vertex Pharmaceuticals, investigator-initiated research funding from Dexcom Inc and Samsung, and serves of the scientific advisory board of Anagram Therapeutics. MDB receives research support through his institution from NovoNordisk, Dexcom, and Tandem; MDB received honoraria/consulting fees from Tandem, Sinocare, Boydsense, and Roche; MDB receives royalties through his institution from Dexcom, Lifescan, Sanofi, and Tandem. ED has received personal fees from Roche and Eli Lilly and Company; holds patents on artificial pancreas technology; and has received product support from Insulet Corporation, Tandem Diabetes Care, Roche, and Dexcom, Inc. The work presented in this article was performed as part of his academic appointment and is independent of his employment with Eli Lilly and Company. RH reports having received speaker honoraria from Eli Lilly, Dexcom and Novo Nordisk, receiving license fees from Braun; receiving consultancy fees from Abbott Diabetes Care, patents related to closed-loop, and being director at CamDiab. LMW receives research support from Dexcom and Eli Lilly. VNS’ institution has received research grant from Eli Lilly, Dexcom, Enable Bioscience, Zucara Therapeutics, Cystic Fibrosis Foundation, Breakthrough T1D, and NIH. VNS has received personal fees from Sanofi, NovoNordisk, Eli Lilly, Dexcom, Insulet, Tandem Diabetes Care, Ascensia Diabetes Care, Embecta, Sequel Med Tech, Biomea Fusion, Genomelink, and Lumosfit for speaking, consulting, or serving on advisory board. RP has received speaker fees from Lilly and Novo Nordisk; stock options from Altanine, Inc.; consulting fees from AbbVie Inc., Altanine Inc., Amgen Inc., AstraZeneca Pharmaceuticals LP, Bayer AG, Bayer HealthCare Pharmaceuticals, Inc., Boehringer Ingelheim Pharmaceuticals, Inc., Corcept Therapeutics Incorporated, Eli Lilly and Company, Gasherbrum Bio, Inc., Getz Pharma, Hanmi Pharmaceutical Co., Lexicon Pharmaceuticals, Lilly USA, Novo Nordisk, Regeneron, Scholar Rock Inc., and Sun Pharmaceutical Industries; and grants (directed to his institution) from Biomea Fusion, Carmot Therapeutics, Dompe, Endogenex, Inc., Fractyl, Lilly, Novo Nordisk, and Sanofi. AC has received research funding from NIDDK and JDRF, and receives research support from Dexcom. RJH has received research funding from the NIDDK. SAB has received research funding to her institution from Dexcom, Insulet, Roche, Tandem, Tolerion and participated on a data safety and monitoring board for MannKind. DMM has consulted for Abbott, Sanofi, Medtronic, Biospex, and Enable Biosciences. LE receives salary support from NIDDK. LE’s institution has received research support from Breakthrough T1D, Medtronic, Mannkind, and Abbot, and she has served on the advisory board of Abbot, Diabetes Center Berne, Sequel, and Medtronic. She has received consulting fees from Jaeb, and Tandem Diabetes Care, and has received honorarium fees from Med Learning Group (Sanofi-sponsored grant), Tandem Diabetes Care, Medtronic, and Insulet. CKB has received consultancy fees from CamDiab, speaker honoraria from Ypsomed and research support from Abbott Diabetes Care, Dexcom and Ypsomed. MCR reports receiving consulting fees from the Jaeb Center for Health Research, Eli Lilly, embecta, Zealand Pharma, and Zucara Therapeutics; speaker fees from Sanofi Diabetes, Eli Lilly, Dexcom Canada, and Novo Nordisk; and stock options from Zucara Therapeutics.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
