Abstract
Diabetes is the leading cause of chronic kidney disease (CKD) and end-stage kidney disease in the world. It is known that maintaining optimal glycemic control can slow the progression of CKD. However, the failing kidney impacts glucose and insulin metabolism and contributes to increased glucose variability. Conventional methods of insulin delivery are not well equipped to adapt to this increased glycemic lability. Automated insulin delivery (AID) has been established as an effective treatment in patients with type 1 diabetes mellitus, and there is emerging evidence for their use in type 2 diabetes mellitus. However, few studies have examined their role in diabetes with concurrent advanced CKD. We discuss the potential benefits and challenges of AID use in patients with diabetes and advanced CKD, including those on dialysis.
Keywords
Introduction
Diabetes is the leading cause of end-stage kidney disease (ESKD) throughout the world, responsible for 47% of all ESKD in the United States. 1 It is a major health epidemic that occupies considerable human and financial resources.2 -4 Maintaining tight glucose control within a healthy range is an established strategy that can prevent the development and progression of albuminuria as well as progression to ESKD.5,6 Furthermore, poor glycemic control both at initiation of renal replacement therapy and during dialysis treatment has been shown to be associated with decreased survival.7 -10 The presence of diabetic kidney disease has a significant impact on patient outcomes, associated with an increased morbidity and mortality, 11 particularly an increased risk of developing cardiovascular disease.
However, patients with concurrent diabetes and advanced chronic kidney disease (CKD) (defined as CKD stage G3b and onward) commonly display suboptimal glycemic control and, in particular, are prone to wide glycemic excursions. 12 This is multifactorial in nature associated with changes to insulin and glucose metabolism that occur with advanced CKD, as well as behavioral and cognitive factors.
The goal of insulin therapy in people with diabetes mellitus is to match as closely as possible normal physiologic insulin secretion to maintain healthy plasma glucose levels. 13 Recent major advances in automated insulin delivery (AID) systems have improved our ability to match exogenous insulin delivery dosing with the changing insulin requirements of day-to-day life. This has brought about the possibility of increasing time in glucose range for people with diabetes and CKD.
Changes in Glucose and Insulin Metabolism in Renal Impairment
Glucose and insulin metabolism in patients with diabetes are profoundly altered by the coexistence of advanced CKD. The risk of hypoglycemia is increased by a range of factors. With the kidneys usually responsible for 40% of systemic gluconeogenesis, 14 advanced CKD as a consequence results in impaired gluconeogenesis, thereby increasing the risk of hypoglycemia. 15 In addition, CKD is associated with impaired counterregulatory hormone responses (cortisol, growth hormone), predisposing to hypoglycemic events. 12 The above derangements are most likely driven by a state of uremia as discussed below. Finally, there is also increased erythrocyte glucose uptake during dialysis, which further contributes to the risk of developing hypoglycemia. 12
It is also known that CKD results in reduced insulin clearance by the kidney. 16 Up to 80% of endogenous insulin is degraded by the liver due to first-pass metabolism, whereas exogenous insulin is primarily eliminated in the kidney 12 (Figure 1). In normally functioning kidneys, insulin is readily filtered by the glomerulus and reabsorbed and degraded by the proximal tubular cells such that less than 1% of filtered insulin appears in the urine. 17 Ordinarily, the renal clearance of insulin far exceeds the glomerular filtration rate (GFR) due to the contribution of peritubular uptake, and as such, there is little change in the metabolic clearance of insulin until the GFR has decreased to 40 mL/min/1.73m2. 12 Peritubular insulin uptake increases as renal function declines to maintain normal insulin clearance until the GFR has fallen to less than 15 to 20 mL/min/1.73m2. At this point, the metabolic clearance rate of insulin falls significantly. This is most likely primarily responsible for the progressive reduction in insulin requirement for people with diabetes who develop progressive CKD. 14
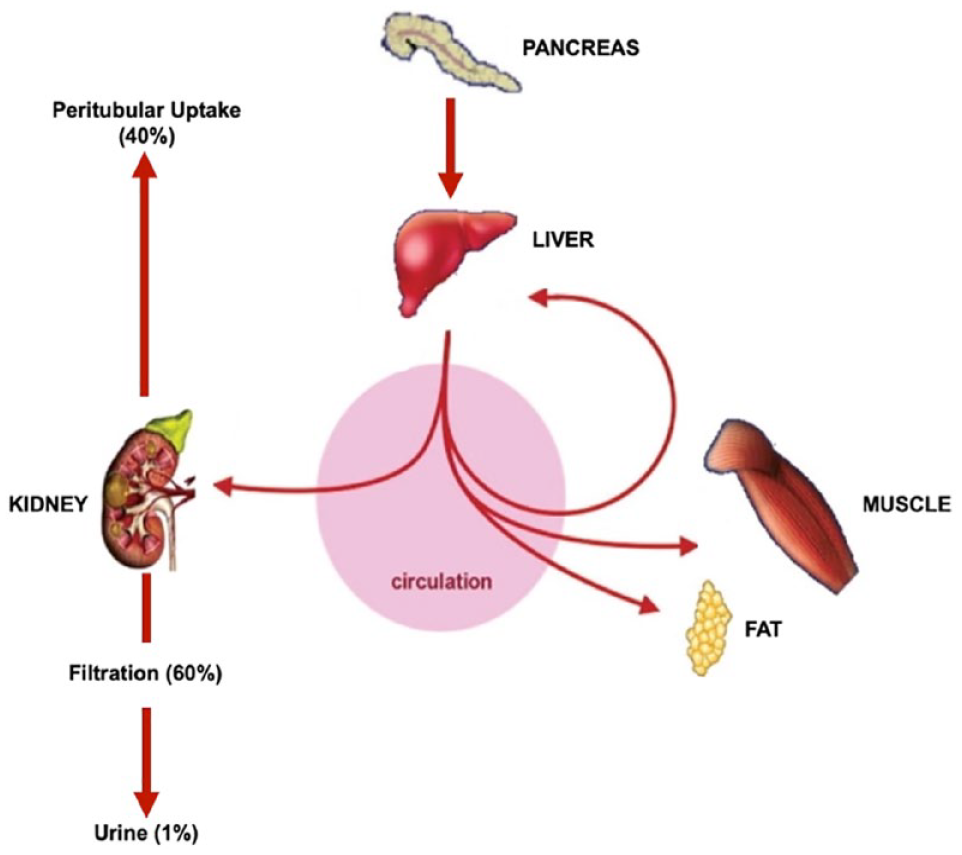
Metabolism of insulin. Insulin is filtered at the glomerulus and then extensively reabsorbed by the proximal tubule. Of the total renal insulin clearance, 60% occurs by glomerular filtration and 40% by extraction from peritubular vessels.
In addition, uremia has systemic effects, which results in impaired degradation of insulin in nonrenal tissues such as liver and muscle. 18 The overall effect is prolonging the half-life of insulin, which further contributes to the risk of hypoglycemia. This impaired metabolic clearance of insulin can be normalized by dialysis. 19 Conversely, uremia may also contribute to hyperglycemia by decreasing tissue insulin sensitivity and as a result, leading to increased insulin resistance. Finally, in those individuals with residual β-cell function, the direct toxic effects of urea on pancreatic β-cells result in impaired insulin secretion19,20 (Figure 2).
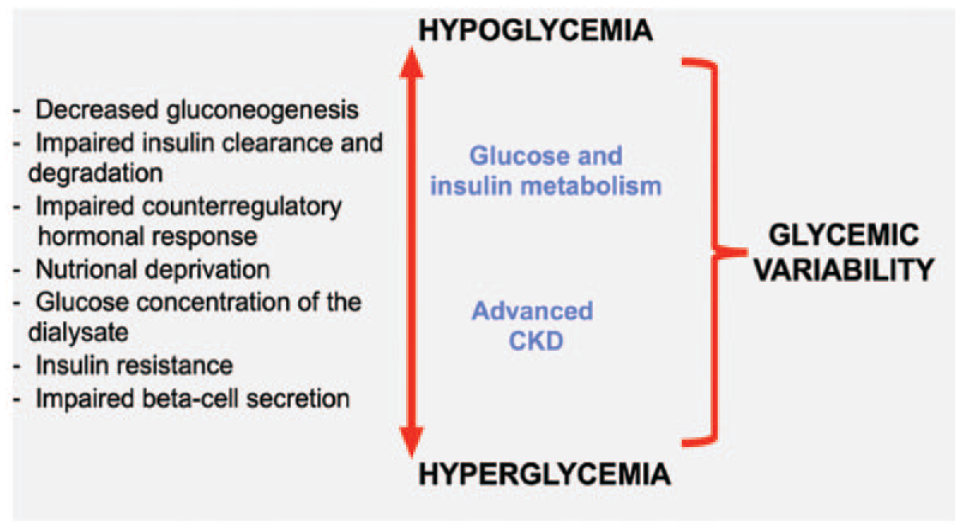
Changes to insulin kinetics and glucose metabolism with advanced CKD.
It is also worth noting that in individuals who receive a kidney transplant, immunosuppressive medications taken thereafter (eg, cyclosporine, tacrolimus, prednisolone) are known to impair glucose metabolism and lead to worsening glycemic control of pretransplant diabetes mellitus. New onset of diabetes after kidney transplant is also a well-described complication of kidney transplantation due to immunosuppressive medications.21,22 However, recurring advanced CKD and graft failure after kidney transplantation is a complex area prompting clinician-patient discussions regarding immunosuppression tapering or cessation, reinitiating dialysis and transplantectomy. 23 There are no data regarding the use of AID technology in this patient group, and while we will not be discussing this population in this review, we recognize a need for urgent future research.
The Effect of Dialysis on Glucose Control
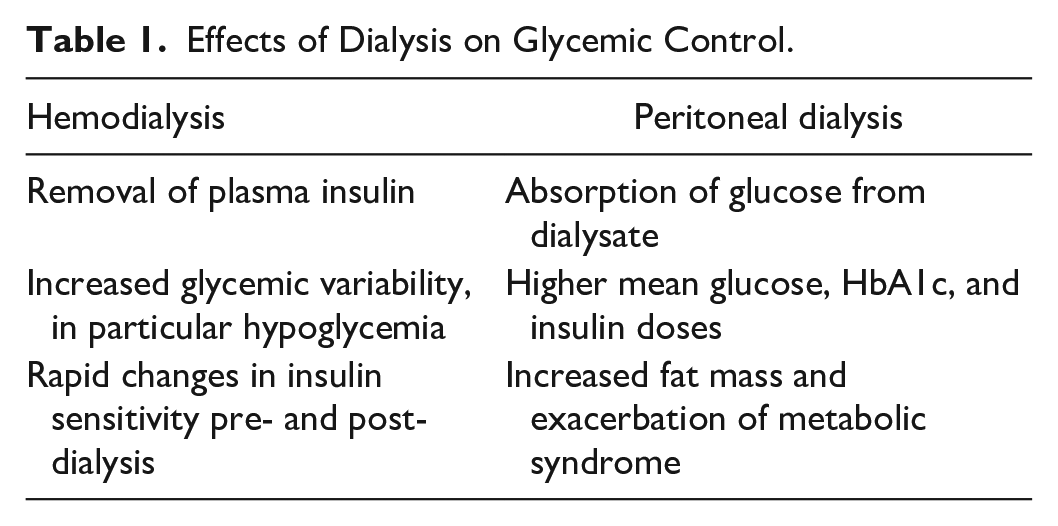
There are different glycemic challenges associated with peritoneal dialysis (PD) and hemodialysis (HD) (Table 1). 24 In PD, mean blood glucose concentrations, HbA1c, and insulin dose have been shown to be higher than in HD 24 with 50% incidence of hyperglycemia compared with 20% in HD-treated patients. 25 Conversely, one study has shown that the frequency of hypoglycemia was higher during HD than PD (13.2% vs 2.8%, respectively), and importantly, severe hypoglycemia was more common with HD. 26
Effects of Dialysis on Glycemic Control.
Peritoneal Dialysis
To achieve osmotic removal of the retained fluid, PD fluids contain supraphysiological concentrations of glucose. Sixty to eighty percent of the glucose in glucose-containing PD solutions instilled into the peritoneal cavity is absorbed, which corresponds to a daily intake of 100 to 300 g of glucose. This may lead to aggravation of hyperglycemia, obesity, and hyperlipidemia. All these factors are known to trigger the production of reactive oxygen species (ROS) and induce an inflammatory cascade that results in increased insulin resistance and impaired lipid metabolism.27 -30 It is well established that PD patients frequently gain weight (predominantly an increase in fat mass), especially during the first year of PD therapy and particularly if associated with the presence of diabetes or obesity at PD initiation. 31 Biocompatible PD solutions using glucose polymers or amino acids as the alternative osmotic agent have been developed, but the evidence supporting their use has been variable. 32 Furthermore, glucose polymers such as icodextrin can also interfere with some glucose testing strips and continuous glucose monitors (CGMs). 33
Hemodialysis
This presents several complexities in diabetes management. First, during HD, plasma insulin can be removed by diffusion and/or convection mechanisms owing to its small size (molecular weight, 6.2 kDa) and protein-binding rate of 1%. 34 This may affect circulating insulin concentrations for a patient in the pre-HD as opposed to the post-HD time frames. Furthermore, total daily insulin requirements are altered by dialysis. Euglycemic clamp studies conducted 24 hours pre, during, and post HD in type 2 diabetes mellitus (T2DM) patients showed that there is a significant 25% reduction in basal insulin requirements the day after dialysis compared with the day before. No significant change in bolus insulin requirements was observed. 35
Hemodialysis-induced hypoglycemia is also common with contributory factors being glucose loss into dialysate, diffusion of glucose into erythrocytes, and use of glucose-free dialysate. These factors can all contribute to inducing a catabolic state similar to that observed during fasting. When a glucose-free dialysate is used, asymptomatic hypoglycemia (defined as serum glucose <4 mmol/L) occurs in approximately 40% of patients regardless of having diabetes or not.36,37 Hypoglycemia can still occur, although less commonly, with the more commonly used 5.55 mmol/L glucose-containing dialysate. 34 Several hours after completion of HD, a paradoxical rebound in hyperglycemia may occur. A similar mechanism as to that of the Somogyi effect, whereby counterregulatory hormones of cortisol, catecholamines, and growth hormone are released, together with the underlying state of insulin resistance observed in ESKD patients is most likely responsible for this phenomenon. 34 It is worth noting that the above describes the glycemic challenges posed by conventional center-based HD. In recent times, home HD treatments, offering greater convenience, are gaining popularity. While home HD, similar to center-based HD, is still administered on a schedule, so that one would expect similar principals to apply, there are very little data on the specific glycemic challenges of home HD as distinct from center-based HD.
Patients with ESKD on dialysis have particularly poor glycemic control due to the aforementioned factors, and confounding this, treatment targets are difficult to determine in this population. Hemoglobin A1c is unreliable and significantly underestimates mean blood glucose due to decreased erythrocyte life-span in ESKD.38,39 The latest guidelines from Kidney Disease Improving Global Outcomes (KDIGO) emphasize the uncertainty of HbA1c and suggest using CGM as an alternative. 40 Furthermore, CGM may be of particular relevance as recent studies have indicated that for elderly and high-risk populations with type 1 diabetes mellitus (T1DM), changes in CGM time-in-range (TIR) correlate inversely with hyperglycemia but do not correlate with time-below-range (TBR). Therefore, hypoglycemia risk should be addressed independently of achieving TIR targets. 41 However, the clinical use of CGM in patients receiving dialysis has not been well studied. As a result, the international guidelines for CGM treatment goals for the elderly and at risk may not necessarily be applicable to the ESKD population.42,43
Current AID Systems
The AID systems have revolutionized the management of diabetes, particularly for people with T1DM with clinical trials and real-word studies demonstrating marked improvements in TIR (79-180 mg/dL) and HbA1c while at the same time minimizing hypoglycemia and improving quality of life (QoL).44 -46 Most AID systems used in an ambulatory setting have taken on a hybrid closed loop (HCL) format where users are still required to manually initiate food boluses. Currently, globally, there are five insulin-only HCL systems commercially available (Figure 3). In general, they use three main types of algorithms: model predictive control, proportional-integral-derivative, and fuzzy logic.
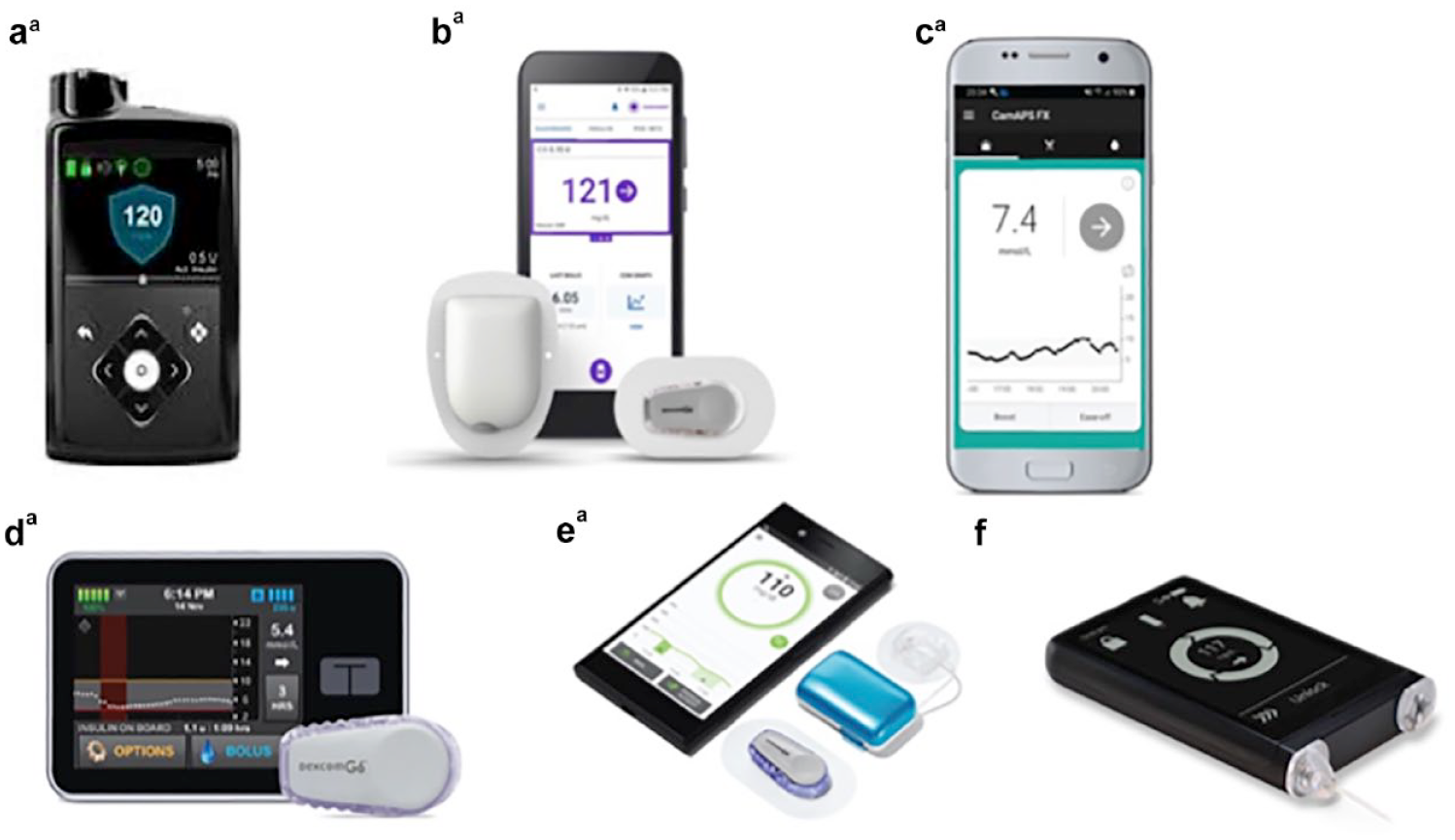
Commercially available and in-development hybrid closed-loop systems. (a) MiniMed 7800G with Guardian Link 3 sensor/transmitter. (b) Omnipod 5 patch-pump system. (c) CamAPS FX algorithm hosted on Android. (d) Tandem t: slim X2 pump paired with Dexcom G6 sensor. (e) Diabeloop DLBG1 algorithm with Kaleido patch-pump and Dexcom G6 sensor. (f) iLet insulin pump.
Potential Benefit of Aid Systems in Renal Disease
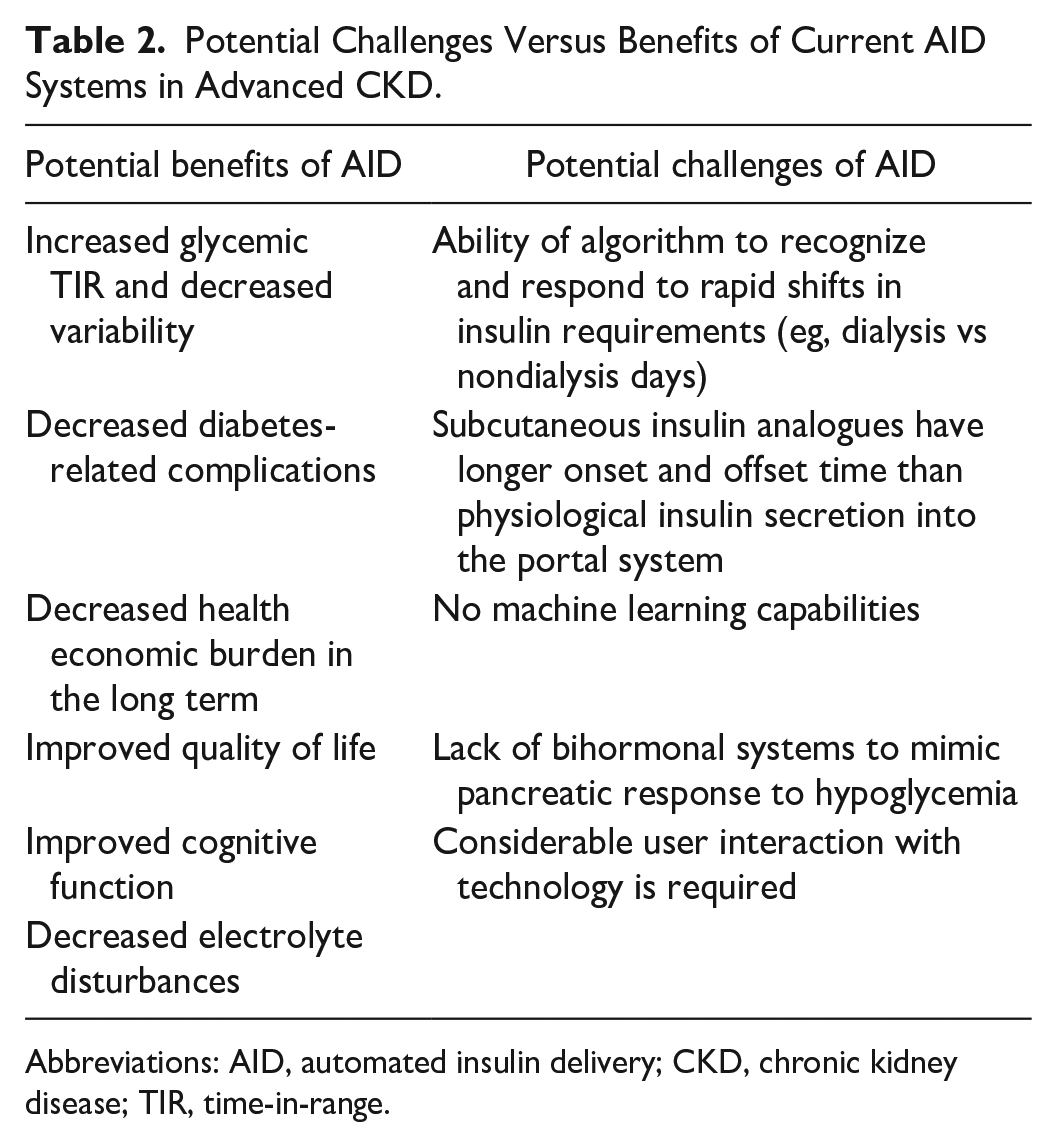
Limited research regarding the application of AID systems in the setting of CKD has generally reported benefits in glycemic control (Table 2). Baseline TIR for patients with advanced CKD generally is in the range of 30% to 40% and increases to 50% to 70% with the application of closed-loop systems.47,48 Of note, to date, these studies have been limited to the T2DM population using HD and using fully closed-loop systems without boluses for meals. In comparison, TIR for patients without CKD has been reported to increase from 60%-67% to 70%-79% using AID systems with an HCL format. 46 As summarized previously, advanced CKD and dialysis create a state of increased glycemic variability. Therefore, it follows that an insulin delivery system that is more dynamic and flexible compared with conventional modalities would be better suited to deal with these fluxes. It is anticipated that the benefits of stringent physiologic glycemic control with AID systems will translate to the decreased emergence and/or progression of diabetes-related complications with the concurrent health economics advantages.
Potential Challenges Versus Benefits of Current AID Systems in Advanced CKD.
Abbreviations: AID, automated insulin delivery; CKD, chronic kidney disease; TIR, time-in-range.
A reduction in glucose fluctuations may also contribute to improved cognition and QOL for advanced CKD patients.49,50 It is estimated that 70% of HD patients aged above 55 years exhibit moderate to severe cognitive impairment 51 with a similar prevalence for PD patients.52,53 Simultaneously, it is also known that T2DM has been associated with increased mild-to-moderate cognitive impairment and a 50% increased risk of all-cause dementia.33,54 This cognitive impairment is likely to be exacerbated by the effects of uremia in people with CKD. 55 Importantly, in elderly people with T2DM, cognitive dysfunction is associated with poor diabetes self-management, a resultant increase in assistance with personal care and increased risk of hospitalization. 56 Intensive glucose control has been shown to slow down cognitive decline in patients with T2DM, and to a lesser degree in people with T1DM.57 -59 There is some evidence to indicate that improving glycemia can even result in improved cognition.60 -62 However, there are no studies specifically assessing improving glycemic control in the advanced CKD population and the effect on cognitive dysfunction, and therefore, further research is required.
In addition, patients with diabetes frequently develop a constellation of electrolyte disorders,63,64 especially in the setting of poor glycemic control. 65 Apart from disturbances that occur in acute decompensated diabetic states such as diabetic ketoacidosis (DKA) or hyperglycemic hyperosmolar syndrome (HHS), chronic hyperkalemia is common in patients with diabetes via a hyporeninemic hypoaldosteronism state.63,66,67 This may be further exacerbated with the coexistence of advanced CKD and dialysis, 64 and therefore may benefit from improved glycemic control.
Potential Challenges to the use of Aid Systems in Advanced CKD
There are many potential challenges when considering the use of AID systems in people with diabetes and coexistent advanced CKD (Table 2). Hemodialysis may be a scenario that poses a unique set of challenges with potentially opposing changes to insulin and glucose metabolism occurring over a short space of time. First, the clearance of insulin from the circulation via the dialysis process would not be predicted by current AID systems and as such could impact the accuracy of subsequent insulin delivery calculations by the algorithm. Second, insulin sensitivity is low prior to dialysis and improves after dialysis with the clearance of uremic toxins. 68 Given that AID system algorithms use recent glucose history (particularly the past 48 hours) to calculate ongoing correction boluses and basal delivery, this acute reduction in insulin resistance may predispose the individual to hypoglycemia. Third, HD-induced hypoglycemia and subsequent rebound hyperglycemia are known phenomena that occur frequently and may further challenge the responsiveness of current CL systems.
The CGM performance has also not been well studied in the advanced CKD population. 42 It is unknown whether the rapid fluid shifts from the intravascular space to interstitial fluid which occurs with dialysis may affect the performance of current CGM systems which sample from the interstitium. Studies are underway to investigate the performance of CGM in the setting of uremia and metabolic acidosis associated with ESKD as these are factors that could possibly interfere with sensor accuracy.
The slower pharmacokinetics of subcutaneous insulin delivery also limits the responsiveness of AID systems to rapid changes in blood glucose levels. In people without diabetes, endogenous insulin is secreted directly into the portal circulation and has a half-life of four to six minutes allowing for rapid but self-limiting responses to blood glucose.69,70 The limitation of subcutaneous insulin delivery is the time taken for absorption into the intravascular space. Rapid-acting insulin analogues, such as aspart and lispro, have a half-life of ~1-2 hours, significantly longer than endogenous insulin. 71 Even the ultra-rapid-acting insulins faster aspart and ultra-rapid-lispro have an onset of action time of ~20 minutes and half-lives of 57 and 44 minutes, respectively, 72 which may be further lengthened in the setting of advanced CKD. Development of even faster insulin analogues with quicker onset and offset duration would be required to more accurately mimic the normal physiological pancreatic release of insulin and adapt to the many dynamic changes in glucose-insulin homeostasis that occur in dialysis.
Also on the horizon are bihormonal systems incorporating glucagon and insulin. These have been investigated in free-living randomized controlled trials and results have been promising with improved glycemic TIR together with reduced time spent in hypoglycemia.73,74 The utilization of glucagon in a pump may mitigate the longer half-life of insulin analogues and the subsequent risk of hypoglycemia.
Machine learning also represents an important potential approach in the optimization of insulin delivery, which may be of relevance to those with advanced CKD. It is a form of artificial intelligence that allows a system to develop self-learning capabilities to predict glucose fluctuations based on past behavior and lifestyle patterns.75,76 This technology has the potential to alleviate some of the decision-making burden for those living with diabetes to adapt to day-to-day challenges such as exercise or stress. The integration of machine learning may be particularly relevant in addressing changes to insulin requirements associated with predictably recurring events such as that which occurs with dialysis. Typically, patients on center-based HD will have three sessions of dialysis a week on fixed days (eg, Monday, Wednesday and Friday) which typically last three to five hours, 77 home-based dialysis can be performed anywhere from three times a week up to a daily basis, 78 whereas PD is usually undertaken daily at home. 79 A system that could learn the dialysis schedule as well as the glycemic response of an individual during dialysis could therefore pre-empt and mitigate glucose fluctuations prior to them occurring. Current commercially available systems do not have machine learning capabilities, but they are under development and may be of benefit in the near future. 80
Finally, as outlined above, cognitive impairment is common in both patients with advanced CKD and diabetes. Current AID systems still require a considerable amount of user interaction with troubleshooting of alarms, line, and set changes every three days, sensor changes every 7 to 10 days, and general carbohydrate counting proficiency and a minimum requirement regarding understanding of the technology. This complexity of interaction undoubtedly poses a difficulty for patients with cognitive impairment. In addition, limited manual dexterity and hand grip strength which is particularly prevalent in dialysis patients 81 may represent a further barrier to patients on renal replacement therapy from using AID systems unless there is a carer available to assist with tasks listed above.
Conclusions
Commercially available AID systems have been shown to improve glucose control and QoL in people living with diabetes. However, there is a paucity of data in those who also have advanced CKD including those on dialysis. The failing kidney creates a state of increased glucose variability. The AID systems represent the most adaptive form of exogenous insulin delivery we have available at the present time, and therefore have the potential to improve glucose control in this population. This in turn could lead to decreased diabetes-related complications, less hospitalizations, improved QOL, and an overall health economic benefit. However, it remains to be seen how the algorithms of the current AID systems will respond to the challenges that dialysis poses on glucose variability. There are identifiable limitations to current AID systems including the pharmacokinetics of subcutaneous insulin and complexity of operating the system, which may limit their use in people with impaired cognition. Some of these challenges could be addressed with a bihormonal AID system incorporating glucagon, as well as developing machine learning algorithms to alleviate some of the decision-making burden. Ultimately, good-quality AID trial data are required in the setting of advanced CKD to help optimize the application of this technology. Further advancements in insulin delivery are also required to ensure CL systems more closely mimic pancreatic function.
Footnotes
Abbreviations
AID, automated insulin delivery; CGM, continuous glucose monitor; CKD, chronic kidney disease; DKA, diabetic ketoacidosis; ESKD, end-stage kidney disease; GFR, glomerular filtration rate; HCL, hybrid closed loop; HD, hemodialysis; HHS, hyperglycemic hyperosmolar syndrome; KDIGO, Kidney Disease Improving Global Outcomes; PD, peritoneal dialysis; QoL, quality of life; ROS, reactive oxygen species; T1DM, type 1 diabetes mellitus; T2DM, type 2 diabetes mellitus; TIR, time-in-range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jean C Lu, Petrova Lee, and Francesco Ierino have no disclosures. Richard J MacIsaac has received research grants from Novo Nordisk, Servier, Medtronic, The Rebecca Cooper Medical Research Foundation, St Vincent’s Research Foundation, The Juvenile Diabetes Research Foundation, Grey Innovations, The Diabetes Australia Research Trust/Program, and The National Health and Medical Research Council of Australia. He has also received honoraria for lectures from Eli Lilly, Novo Nordisk, Sanofi Aventis, Astra Zeneca, Merck Sharp & Dohme, Novartis, and Boehringer Ingelheim and has been or is on the advisory boards for Novo Nordisk, Boehringer Ingelheim-Eli Lilly Diabetes Alliance, Astra Zeneca, and Merck Shape & Dohme. Travel support has been supplied by Novo Nordisk, Sanofi, Boehringer Ingelheim, and Astra Zeneca. He has been a principal investigator for industry-sponsored clinical trials run by Novo Nordisk, Sanofi, Bayer, Johnson-Cilag, and Abbive. The author has no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed. Elif Ekinci is on advisory boards for Eli Lilly, Sanofi, Ypsomed, and Pfizer and have provided lectures for Eli Lilly, Boehringer, and Sanofi. These monies are directly donated toward diabetes research at EIE’s institution. EIE’s institution gets research funding from Eli Lilly, Boehringer, Sanofi, Novo Nordisk, Medtronic, Applied Therapeutics, Endogenex, Pharmasol, and Arrowhead. David O’Neal has received research support from Medtronic, Insulet, Dexcom, Roche, GlySens, BioCapillary, and Endogenex. He is on the advisory boards for Medtronic, Insulet, Abbott, Ypsomed, Novo Nordisk, and Sanofi. He has also received honoraria for lectures from Medtronic, Insulet, Abbott, Novo Nordisk, and Sanofi.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
