Abstract
The use of continuous glucose monitoring (CGM) and automated insulin delivery (AID) technologies can improve patient outcomes and overall quality of life while helping to reduce the long-term costs. However, current eligibility criteria imposed by many major commercial insurers limit access to these technologies among a large portion of the diabetes population. This narrative review and commentary highlights the evidence supporting the use of CGM and AID in the various diabetes populations, discuss the current eligibility criteria that make these technologies inaccessible to individuals who would benefit, and present recommendations for modifying these criteria.
Introduction
The growing prevalence of diabetes in the United States continues to be a significant public health concern. An estimated 38.4 million Americans are living with diabetes; approximately 36.1 million have type 2 diabetes (T2D). 1 The majority of these individuals, but certainly not all, are also overweight or obese. 1
Landmark studies have shown that persistent hyperglycemia leads to a constellation of microvascular and macrovascular complications.2,3 However, almost half of Americans with T2D are not achieving the established glycemic goal of HbA1c <7%. 4 Approximately 13% have HbA1c levels >9%. 1 Individuals who are overweight or living with obesity are at even higher risk for developing complications of diabetes.
Innovations in glucose monitoring and insulin delivery technologies have led to the development of new tools such as continuous glucose monitoring (CGM) and automated insulin delivery (AID) systems that enable individuals with diabetes to better manage their disease.5,6 Importantly, the use of these tools has been shown to lower the number and severity of diabetes-related events and hospitalization rates and reduce the overall cost of diabetes, 7 which, in the United States, now exceeds $412.9 billion. 8
Based on this large and growing body of evidence, the American Association of Clinical Endocrinology (AACE) expanded its CGM recommendation to include all individuals with problematic hypoglycemia (frequent/severe hypoglycemia, nocturnal hypoglycemia, hypoglycemia unawareness) regardless of therapy. 9 The American Diabetes Association (ADA) has also adopted CGM use as a standard care for all individuals treated with intensive and nonintensive insulin therapy. 10 Moreover, the ADA guidelines state that individuals using CGM and/or AID for diabetes management should have continued access across third-party payers, regardless of age or HbA1c levels. 10 This is particularly important in light of results from a recent study that showed significant deterioration in glycemic control among individuals treated with basal insulin therapy who switched from CGM back to the use of blood glucose monitoring (BGM). 11 In alignment with the current evidence and modern glucose monitoring capabilities, Recommendation 7.9 was added to emphasize that reports for all CGM devices, connected insulin devices, and continuous subcutaneous insulin infusion and AID systems should be standardized with at a minimum the ambulatory glucose profile and weekly summary.
Unfortunately, many individuals with T2D cannot utilize these technologies due to restrictive eligibility criteria imposed by government and commercial health plans. In this narrative review, we review the clinical and economic evidence supporting the use of current diabetes technologies in various patient groups, highlight the inherent dangers of coverage denial, and propose changes to current eligibility criteria that are evidence-based, which would expand access to these innovative tools. This narrative review highlights the evidence supporting the use of CGM and AID in the various diabetes populations, discusses the current eligibility criteria that make these technologies inaccessible to individuals who would benefit, and presents recommendations for modifying these criteria.
Advent of Innovative Diabetes Technologies
Continuous Glucose Monitoring
The availability of CGM has shifted the paradigm of diabetes management from reactive to prospective. Unlike traditional fingerstick BGM systems, which provide only a point-in-time “snapshot” of an individual’s current glucose level, CGM sensors transmit a continuous stream of real-time glucose data to patients’ smartphones or handheld readers (Figure 1). The data are displayed in numerical and graphical formats, indicating the current glucose level, and historical trends. Trend arrows show the direction and velocity of changing glucose levels. Importantly, current CGM devices feature programmable alerts that warn patients of immediate and impending hypoglycemia and hyperglycemia.
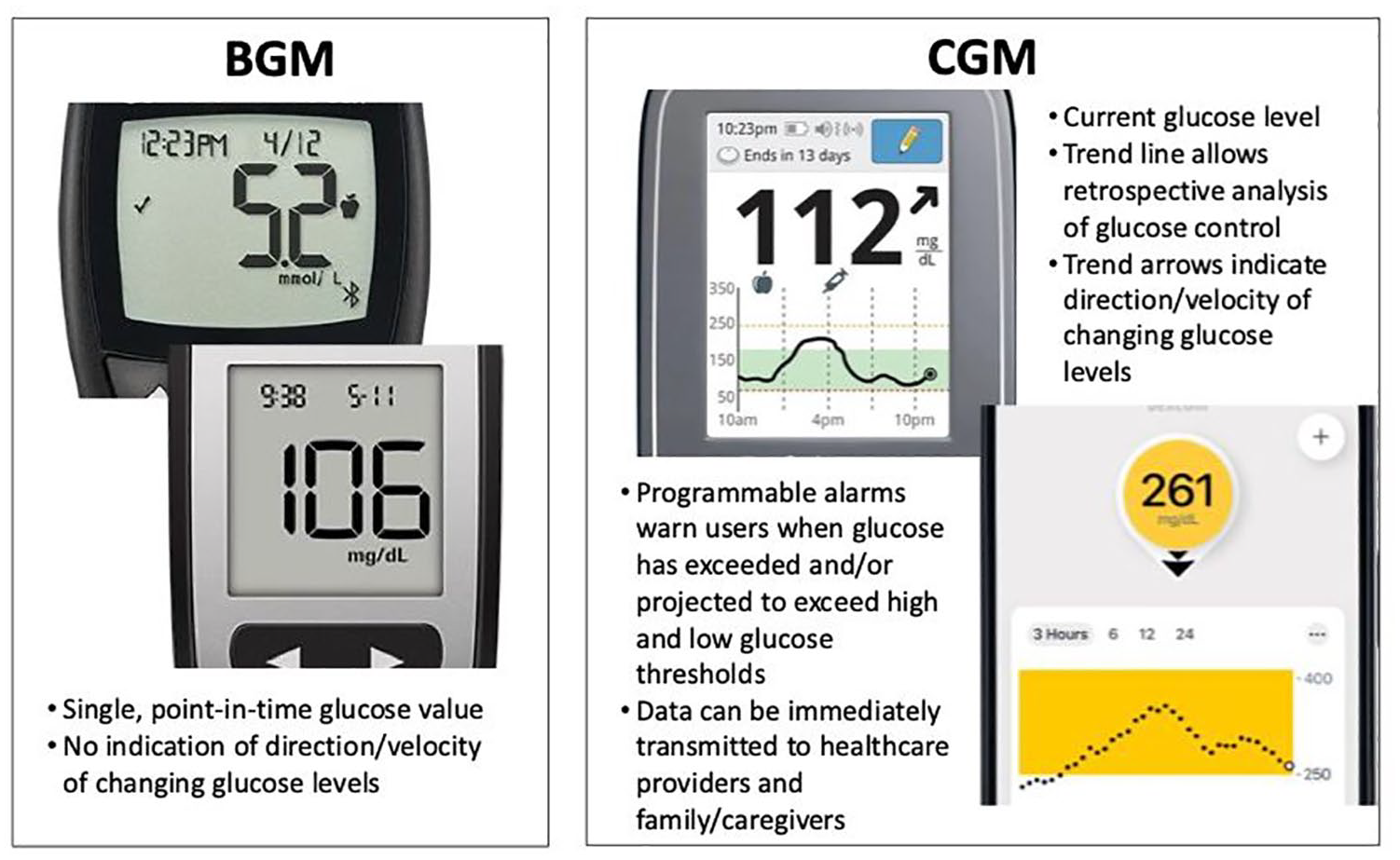
Glucose information displayed with BGM and CGM.
While the real-time data generated by CGM facilitates immediate and more informed decisions in daily diabetes self-management, the full value of this technology is realized through retrospective analysis of the data. When an international consensus panel published its list of 10 core CGM metrics for use in clinical practice, the panel also recommended the use of a one-page report, the ambulatory glucose profile (AGP), 12 which now aligns with the recent ADA recommendations. The AGP aggregates data from a CGM device via cloud-based transmission or download from a reader/receiver and displays the data in an easy-to-interpret report based on established CGM metrics. Retrospective analysis of the AGP report enables clinicians to quickly identify problematic glucose patterns and make appropriate adjustments in therapy when needed. It also supports more effective clinician-patient collaboration and provides patients with a better understanding of their diabetes and the impact of medications, food, and other health behaviors on glycemic control.
Improvements in CGM accuracy and reliability have led to the development of AID. Current AID systems utilize a sophisticated controller algorithm that continuously increases or decreases insulin delivery in response to changing glucose levels obtained from an integrated CGM, residual insulin action (eg, insulin-on-board), and other inputs, such as meal intake and exercise announcement. Advanced systems are also becoming available with the added capability to deliver automated correction boluses.
Acute Clinical Consequences of Poorly Controlled Diabetes
Hyperglycemia
All individuals with poorly controlled diabetes are at elevated risk for developing severe, chronic complications of diabetes, including cardiovascular disease (CVD), nephropathy, neuropathy, and retinopathy. 13 Diabetic ketoacidosis (DKA), a life-threatening acute complication, can also occur in persons with uncontrolled T2D. In addition, the risk of hyperglycemic hyperosmolar state (HHS) is often overlooked in T2D.
The HHS is characterized by hyperglycemia, hyperosmolarity, and dehydration in the absence of significant ketosis or acidosis. 14 The HHS can occur in type 1 diabetes (T1D), T2D, and other diabetes types but is more frequently found in older adults with diabetes. 14 Common causes include infection and omission of antihyperglycemic medications such as insulin and sulfonylureas. While the rate of hospital admissions for HHS in T2D is <1% of all diabetic-related admissions, 15 mortality can reach up to 20%. 16
Hypoglycemia
Hypoglycemia is the major limiting factor in achieving optimal glycemic control among individuals with diabetes. 17 Associated with increased mortality and debilitating morbidities, 18 hypoglycemia occurs when an individual’s blood glucose level drops below <70 mg/dL. Hypoglycemic episodes are categorized as level 1 (glucose 54-70 mg/dL), level 2 (glucose <54 mg/dL), and level 3 (requiring assistance from another person or medical intervention). 19 While more frequent in T1D, the number of hypoglycemic events in T2D is much higher due to the large and increasing number of individuals with T2D. 20
Severe hypoglycemia is a significant burden for all individuals with T2D who are treated with insulin and other antihyperglycemic medications. 21 However, it is particularly dangerous for older individuals with underlying heart disease. 22 Other factors that put elderly individuals with diabetes at significant risk are hypoglycemia unawareness, nocturnal hypoglycemia, cognitive impairment, and dementia. 22 One early study reported that insulin-treated patients with diabetes who were >80 years of age were more than twice as likely to require emergency department services and nearly five times as likely to be subsequently hospitalized for hypoglycemic events than those aged 45 to 64 years. 23
Use of Continuous Glucose Monitoring Reduces Acute and Long-term Complications
Numerous studies have demonstrated the clinical efficacy of CGM in individuals with T1D and those with T2D who are treated with intensive insulin therapy regardless of insulin delivery method. 5 The benefits of CGM use in this population include reductions in HbA1c, fewer severe hypoglycemia events, less hypoglycemia fear, increased time within target glucose range (TIR), and reductions in time below range. 5 Large observational database studies have also shown an association between CGM use and significant reductions in hospitalizations for severe hypoglycemia and DKA. 24 The clinical benefits of CGM have also been reported in individuals treated with nonintensive insulin and noninsulin regimens.5,25 Several recent studies have demonstrated improvements in glycemic control, acute diabetes-related events (ADEs), and costs in this population. 5 In addition to T2D, a growing body of evidence also supports the efficacy and safety of CGM in gestational diabetes (GDM). 26
Recent large database analyses have shown an association between CGM use and reductions in all-cause hospitalizations (ACHs), 27 ADEs, overall health care resource utilization, and associated costs in diabetes populations treated with intensive insulin therapies. 24 Recent studies have also demonstrated that daily CGM use reduces diabetes-related events and is cost-effective in individuals with T2D regardless of their therapy. 5
A detailed summary of the design and outcomes of the studies cited here was recently published, highlighting the need for a more evidence-based approach to policy-making within the Medicare and Medicaid systems. 5
Enhanced learning, patient engagement, and increased treatment satisfaction are additional benefits of CGM use in noninsulin-treated populations. In a study of 594 adults with T2D (insulin users, n = 217; noninsulin users, n = 377) who used CGM intermittently, Bergenstal et al 28 found that most respondents reported improved understanding of the impact of eating (97.0%), increased diabetes knowledge (95.7%), and improvements in diabetes control even when not wearing the sensor (79.4%). Importantly, HbA1c decreased from 7.7% to 7.1% (P < .001) over an average of 10.2 months. As recently reported by Kicaj et al, 29 the value of CGM in noninsulin-treated populations cannot be overestimated, given the effects of diabetes self-management education, particularly in those who are newly diagnosed.
Revisiting Current Continuous Glucose Monitoring Eligibility Policy-Making
Recent changes in the Centers for Medicare & Medicaid Services (CMS) eligibility criteria have expanded access to CGM technology for all Medicare beneficiaries with diabetes who are treated with insulin or have a history of significant hypoglycemia. Once limited to beneficiaries treated with intensive insulin regimens, CGM coverage is now available to beneficiaries who are treated with insulin therapy (intensive and nonintensive) or experience problematic hypoglycemia regardless of their therapy. 30 Importantly, CMS has dropped the requirement for documentation of ≥4 fingerstick BGM tests per day. While we applaud CMS for making these changes in its Medicare program, we urge CMS to put pressure on the various state Medicaid programs. Many programs do not provide coverage, while others continue to impose restrictive eligibility criteria.
In our review of CGM eligibility criteria from eight major commercial health insurance providers, we found notable differences in eligibility criteria for coverage (Table 1). While many plans have modified their eligibility requirements, the differences between plans could adversely impact individuals who change their insurance providers.
Differing Eligibility Criteria (EC) for CGM Coverage Eligibility.

Despite their differences, many of these relatively new requirements, such as coverage for nonintensive insulin therapy (EC-6, EC-7, EC-8), serve to expand access to CGM. However, some criteria are more troubling. For example, insurers with an eligibility requirement for frequent BGM (EC-1) are both unduly restrictive and medically unfounded, as demonstrated in large CGM trials that included patients with a baseline BGM frequency of <4 times per day who achieved the same glycemic benefits as those with higher testing frequency. 24 Similarly, the requirement for multiple (≥3) daily doses (EC-1, EC-2, EC-4, EC-5) can be easily disputed by the growing number of studies demonstrating the clinical benefits of CGM use in individuals treated with nonintensive insulin and noninsulin therapies. 5 Equally concerning is the caveat in which coverage is denied to individuals who are meeting glycemic goals and/or have no problematic hypoglycemia (EC-2, EC-3, EC-5, EC-7). In essence, patients are forced to forego CGM use until their problematic hypoglycemia returns or if they have poor overall glycemic control.
Most disappointing is the lack of CGM coverage for individuals treated with noninsulin therapies. Despite the evidence of efficacy reported in several recent studies, the majority of individuals with T2D are unable to afford this technology due to lack of coverage, particularly for those living in low socioeconomic and racial/ethnic minority communities where the prevalence of diabetes and severity of diabetes complications are disproportionately higher. 39 Moreover, CGM use is already disproportionally low among individuals in racial/ethnic minority and low socioeconomic populations.40,41 In a retrospective cohort of 300 young adults with T1D, Agarwal et al 41 reported significantly lower use of CGM among Black patients (28%) compared with White patients (71%). Results from multivariate regression to assess the impact of socioeconomic status, health care factors, and diabetes self-management behaviors, investigators could not fully explain these disparities, suggesting the potential for implicit bias. Similar disparities were observed in a retrospective review of 227 adult T1D patients who were seen in an urban, safety-net endocrinology clinic. 42 Investigators reported that only 85 patients (37%) used any diabetes technology, and a significantly lower percentage of Black (4%) and Hispanic (9%) patients used CGM and/or insulin pumps compared with White (35%) patients. Given that all of these individuals were eligible for CGM use according to virtually all commercial and government coverage policies, one can safely assume that use of CGM by T2D patients within lower socioeconomic and racial/ethnic communities is close to nil.
Rational for Continuous Glucose Monitoring in All Individuals With Type 2 Diabetes
Learning From Mistakes in the Past
The ADA/EASD algorithm for the management of T2D that was first introduced in 2009 outlined a step-wise approach to managing glycemia. 43 Some criticized this approach as too restrictive, limiting clinicians’ ability to individualize diabetes care. 44 However, the net effect was the propagation of what has been termed “treat to failure,” which is characterized by adding or titrating medications when HbA1c levels are not achieved or maintained. Although the algorithm has evolved over the years to include new medication classes, thereby mitigating some of the original criticisms, the unintended consequence of this approach has been therapeutic inertia, the delay in treatment intensification, which is often due to clinician and patient fear of hypoglycemia and lack of timely, accurate, and complete glucose information. 45 Therapeutic inertia is recognized by the ADA as the underlying cause of suboptimal glycemic control in a large proportion of individuals with T2D 46 most of whom are treated with noninsulin therapies. 1
A key learning from the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) 2 and the United Kingdom Prospective Diabetes Study was the importance of early intensification of therapy to achieve optimal glycemic control. 47 Early and effective intervention is particularly important today, given the increasing number of individuals who are developing T2D at earlier ages.48,49 Therefore, early interventions are essential to help these individuals achieve their glycemic targets and live longer without the debilitating and costly complications of diabetes.
Recommendations
Addressing the current and future need for optimal glycemic control and improved outcomes can be achieved through expanding access to CGM for all individuals with diabetes. Therefore, we recommend that CGM be offered to all individuals with T2D at the time of diagnosis regardless of the initial therapy (insulin or noninsulin), and that their use of CGM is continued without interruption due to lack of insurance coverage throughout the course of their disease. A substantial and growing body of evidence demonstrates the value of CGM use among noninsulin-using T2D patients. 5 These studies consistently show clinically meaningful reductions in HbA1c and increases in time in range. Given that 68.1% of Americans ages 18 to 64 years are covered by commercial health plans, 50 efforts to expand access to CGM must involve greater enlightenment within the commercial payer community.
Current eligibility criteria are based mostly on findings from large randomized controlled trials (RCTs), which are often out of date due to rapidly evolving CGM technologies. In one set of eligibility criteria, 29 we found that the evidence used to formulate coverage policies included only three RCTs, two of which were published in 2011 51 and 2017. 52 There were no studies involving patients treated with nonintensive insulin or noninsulin therapies. Importantly, the supportive evidence base included no real-world studies. Real-world evidence (RWE) is now required by the US Food and Drug Administration (FDA) for product clearance. 53 Although RCTs are considered the highest level of scientific evidence when investigating the efficacy of a given intervention, they do not provide critical information about the effectiveness of an intervention when used in uncontrolled clinical settings. This is particularly relevant to assessing the efficacy of diabetes technology, which is heavily dependent on user behavior. In our view, all relevant evidence should be considered in policy decisions.
Summary
The increasing prevalence of diabetes, diabetes complications, and associated costs threatens to overwhelm the US health care system. Although innovative and effective diabetes technologies have the demonstrated potential to reduce the burden of diabetes among all people with diabetes, these technologies remain inaccessible to those individuals who would benefit most due to restrictive and often medically unfounded eligibility criteria. The future impact of diabetes can no longer be ignored. The diabetes epidemic is the 50-foot tsunami, 50 yards from shore. Changes in eligibility criteria for all diabetes technologies are needed.
Footnotes
Abbreviations
AACE, American Association of Clinical Endocrinology; ACH, all-cause hospitalizations; ADE, acute diabetes event; AGP, Ambulatory Glucose Profile; AID, automated insulin delivery; ADA, American Diabetes Association; BGM, blood glucose monitoring; CGM, continuous glucose monitoring; CMS, Centers for Medicare & Medicare Services; DCCT/EDIC, Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications; DKA, diabetic ketoacidosis; EC, eligibility criteria; FDA, U.S. Food & Drug Administration; HbA1c, glycated hemoglobin; HHS, hyperosmolar state; RCTs, randomized controlled trials; T1D, type 1 diabetes; T2D, type 2 diabetes; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DFK institution has received research support from NIH, Helmsley Foundation, Novo Nordisk, Abbott Diabetes, and Dexcom. Consulting fees from Novo Nordisk, Abbott Diabetes, Mannkind, CeQur, Sanofi, Medical Module, Provention Bio, and Pendulum. She has been a speaker for Dexcom, Novo Nordisk, Sanofi-Aventis, Xeris, and CeQur. Stock options not redeemed Pendulum. CGP has received consulting fees from Abbott Diabetes Care, CCS, CeQur, Dexcom, Embecta, LifeScan, Mannkind, Roche Diabetes Care, and Tandem. IBH serves as an advisory board member for Abbott Diabetes Care, Roche, Bigfoot, and GWave and receives research grant support from Dexcom. JBM has received research support from the NIH, Helmsley Foundation, JDRF, Novo Nordisk, and Beta Bionics. GA has received research support from Northwestern University from AstraZeneca, Dexcom, Eli-Lilly, Emmes, Fractyl Health, Insulet, Novo Nordisk, and WellDoc. GA has received consulting fees from Eli-Lilly, Dexcom, and Insulet outside of this work. JBM has received research support from the NIH, Helmsley Foundation, JDRF, Novo Nordisk, and Beta Bionics. JBM has received consulting fees from Bayer, Boehringer Ingelheim Lilly, Mannkind, Novo Nordisk, and Thermo Fisher outside of this work. RJG is supported by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) of the National Institutes of Health (NIH) under Award Numbers 2P30DK1110246 and K23DK123384-3. RJG received research support from Emory University for investigator-initiated studies from Novo Nordisk, Dexcom, and Eli-Lilly and consulting fees from Sanofi, Eli-Lilly, Boehringer, Pfizer, and Weight Watchers, outside of this work. CJL has received research support from the NIDDK and Helmsley Foundation and industry support paid to the Icahn School of Medicine at Mount Sinai from Abbott Diabetes, Dexcom, Insulet, Novo Nordisk, Senseonics, and Tandem. CJL has received consulting fees from Eli-Lilly and Dexcom outside of this work. GEU is partly supported by research grants from the National Institutes of Health (NIH/NATS UL 3UL1TR002378-05S2) from the Clinical and Translational Science Award program and from the National Institutes of Health and National Center for Research Resources (NIH/NIDDK 2P30DK111024-06). GEU has received research support (to Emory University) from AstraZeneca, Bayer, Abbott, and Dexcom. GG reports receiving fees from Eli-Lilly, Novo Nordisk (speakers’ bureau) and Lifescan, and Nevro (advisory boards). GG reports receiving fees from Eli-Lilly, Novo Nordisk (speakers’ bureau), Lifescan, and Nevro (advisory boards). RMB has received research support, consulted, or has been on a scientific advisory board for Abbott Diabetes Care, Ascensia, CeQur Corporation, Dexcom, Hygieia, Insulet, Johnson & Johnson, Lilly, Medtronic, Novo Nordisk, Onduo, Roche, Sanofi, United Healthcare, and Zealand. His technology research is funded in part by NIH/NIDDK. RMB’s employer, nonprofit HealthPartners Institute, contracts for his services, and no personal income goes to RMB.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
