Abstract
Background:
Automated insulin delivery (AID) systems are limited by the short wear time of insulin infusion sets, which typically need replacement every 2 to 3 days, significantly shorter than the 14-day lifespan of continuous glucose monitoring (CGM) sensors. Infusion set failure remains a major obstacle to AID reliability and patient adherence. This study examined the roles of insertion trauma and biomaterial composition in causing acute inflammatory responses using both swine and mouse models.
Methodology:
We evaluated three commercial CGM sensors (Abbott Libre 2, Dexcom G7, Medtronic Guardian 3) and two Teflon-based IIS catheters (Medtronic QuickSet and i-Port Advance). In swine, tissue was histologically analyzed one day after implantation to assess neutrophil extracellular trap (NET) formation. In a murine air pouch model, we isolated material-specific immune responses by reducing mechanical injury. Lavage fluids collected at 1 and 3 days postimplantation were examined for immune cell infiltration and cytokine expression using flow cytometry and MSD multiplex assays.
Results:
NETs were observed at all insertion sites, indicating that tissue trauma, rather than the material itself, is the primary trigger of early NET formation. However, Teflon catheters caused a more prolonged inflammatory response, with increased recruitment of macrophages and mast cells, and higher levels of TNF-α and KC/GRO. In contrast, polyurethane-based sensors induced minimal immune activation, suggesting greater biocompatibility. The findings were consistent across models, although some species-specific differences were noted.
Conclusion:
These findings underscore the importance of minimizing insertion trauma and selecting biocompatible materials to promote device-tissue integration, prolong wear time, and enhance AID system performance.
Keywords
Introduction
Advances in diabetes technology have significantly enhanced patients’ ability to maintain euglycemia, which in turn lowers the risk of long-term complications such as neuropathy, retinopathy, and cardiovascular disease.1-5 One of the most influential innovations is the development of automated insulin delivery (AID) systems. These systems combine data from continuous glucose monitoring (CGM) devices with insulin pumps to automate subcutaneous insulin delivery, resulting in more consistent glycemic control.6-8
However, a significant limitation of AID systems is the mismatch in the lifespan of their components. CGM sensors typically last up to 14 days, whereas standard insulin infusion sets are FDA-cleared for use up to two days (steel) or three days (Teflon). Extended-wear designs, such as the Medtronic Extended (EIS) and Tandem Diabetes Care SteadiSet infusion sets, can last up to 7 days.9-12 This discrepancy makes infusion sets the weakest link in the AID system.8,13 Unlike CGMs, infusion sets not only reside in subcutaneous tissue but also actively deliver insulin, introducing additional biological and material-related challenges that further complicate efforts to extend their wear duration.
One major obstacle to the longevity of implanted diabetes devices is the host immune response, which is triggered by both insertion-related tissue trauma and recognition of the device materials as foreign. 14 In this study, we investigated the early tissue response to both insertion injury and biomaterial composition in two types of diabetes devices: three commercially available CGM systems: Abbott Libre 2, Dexcom G7, and Medtronic MiniMed Guardian 3, and two insulin delivery systems, Medtronic i-Port Advance and MiniMed QuickSet. Sensors are primarily composed of polyurethane (PU),15,16 except for the Libre 2, while IIS are made of Teflon.
A key focus was the formation of neutrophil extracellular traps (NETs), as well as broader immune cell infiltration and cytokine expression profiles. Neutrophil extracellular traps are web-like structures made of DNA, histones, and antimicrobial proteins released by activated neutrophils. 17 Although NETs play a protective role in trapping pathogens, they are also implicated in chronic inflammation and fibrosis around implants, factors that can compromise device performance and reduce lifespan.18,19
To evaluate immune responses, we used both swine and mouse models. Swine are a particularly relevant model for studying diabetes devices because of their physiological and metabolic similarities to humans, including comparable skin structure, subcutaneous tissue composition, and insulin pharmacokinetics. 20 Along with assessing in vivo responses at implantation sites, we analyzed the immune profile prompted by the biomaterials themselves, independent of insertional trauma. This was done using an air pouch model, in which the sensors and insulin catheters were inserted. Lavage fluid was then collected for in vitro assays to evaluate the biomaterial-specific immune response.
Our findings show that NET formation occurs regardless of the material, highlighting mechanical impact as a key trigger of this immune response. However, Teflon induces significantly more inflammatory cell infiltration and higher cytokine levels than PU, suggesting it enhances local immune activation. These results may help explain the shorter lifespan of insulin infusion sets compared with CGM sensors when material-tissue interactions are considered in isolation.
Methodology
Swine Study
Yucatan pathogen-free mini pigs were used (Premier Bioresources/S&S Farms, Indiana), weighing between 25 and 89 kg. The animals were housed per the standards of the Division of Laboratory Animal Resources at Wayne State University. Before insertion, the animals received an intramuscular (i.m.) dose of anesthetics: midazolam (0.4 mg/kg, Hospira Inc., Lake Forest, IL, USA), xylazine (1.0 mg/kg), and butorphanol (0.2 mg/kg, Zoetis, Parsippany, NJ, USA). Anesthesia was maintained with an Isoflurane/oxygen mixture as needed. The belly area was shaved and cleaned with warm water to enhance device adherence, then coated with Skin Tac (Torbot Group Inc., Warwick, RI, USA). A 30-second drying period followed. Up to nine devices were inserted subcutaneously into the abdominal region for one day. The following devices were investigated: Abbott Libre 2, Dexcom G7, MiniMed Medtronic Guardian 3 sensors, Medtronic QuickSet catheter, and Medtronic i-Port Advance. Elastikon tape (Johnson & Johnson, New Brunswick, NJ, USA) was used to secure the individual devices, followed by a Tegaderm Transparent Film Dressing (3M, Maplewood, MN, USA). The site was then wrapped with self-adhesive bandages. The reversal agent Antisedan (Zoetis, Parsippany, NJ, USA) was administered to assist the animals’ recovery from anesthesia. Sensor and catheter tissues were obtained using an 8-mm SHARD premium + Disposable Biopsy Punch (AD Surgical, Sunnyvale, CA, USA). Wounds were closed with a single cruciate pattern suture, and biopsies were performed for histological analysis. This study was conducted with approval from the Institutional Animal Care and Use Committee at Wayne State University.
Histological Evaluations
After the biopsy, the tissue was immersed in Zinc Fixative (BD Pharmingen, San Diego, CA, USA) for 24 hours, followed by standard paraffin embedding and tissue sectioning (see Table 1, supplementary material for the number of biopsies). The tissue samples were stained with the standard hematoxylin and eosin (H&E) (Fisher Scientific, Waltham, MA, USA). To evaluate extracellular DNA, the slides were stained with DAPI (Electron Microscopy Sciences Fluoro-Gel II, Hatfield, PA, USA) and anti-Histone 3 (H3) (ab3594, Abcam, Cambridge, UK). This was followed by incubation with the secondary antibody 5/6-fluorescein isothiocyanate (FITC) (#ab7086, Abcam, Cambridge, UK). To evaluate whether the extracellular DNA originated from neutrophils, myeloperoxidase (MPO) staining was performed using AF3667 (R&D Systems Inc., Minneapolis, MN, USA) followed by incubation with secondary antibody Anti-goat IgG-NL637 (#NL002, R&D Systems Inc., Minneapolis, MN, USA). Imaging and analysis were conducted using a Nikon Eclipse Ni microscope and NIS Elements Imaging software. Slide scanning was performed on an Aperio CS2 (Leica Biosystems, Nussloch, Germany), and the scans were analyzed using Aperio ImageScope version 12.4.6.
Air Pouch Generation and Glucose Sensor Insertion
C57BL/6 mice, both males and females, aged eight to 12 weeks, were purchased from Jackson Laboratory (Bar Harbor, ME) and kept under temperature and light-controlled conditions (20°C-24°C, 12-hour light-dark cycle), receiving food and water ad libitum (see Table 2 animal numbers in supplementary material). Following an acclimation period, the animals were stratified by weight and then randomly assigned to each time point group. The dorsal surface of the animals was shaved, and an air pouch was created as described by Keserwan et al.21,22 Briefly, animals received a subcutaneous injection of 3 mL of sterile filtered air. A 23-gauge needle (23G) was used to pierce the caudal surface of the air pouch, and the sensing element, without its applicator and circuit board, was gently inserted. The QuickSet catheter (i.e., Teflon Catheter) was inserted using a 23G needle. No insulin or other substances were administered. For the sham control, a 23G needle was used for the Libre2 and G7 sensors, as well as for the QuickSet catheter. The Medtronic Inserter Needle (MNI) was used for the sham group with the Guardian 3 sensor instead of a 23G needle. This substitution was necessary because the Guardian 3 sensor is made of a softer material and has a larger size, making the 23G needle insufficient to pierce the mouse skin and enable proper insertion. All air pouch procedures were performed 24 hours before the implantation of the sensing device or catheter. Mice were sacrificed at one and three days post-sensor insertion to withdraw the sensor and lavage the air pouch. Cells were collected from the air pouch model through gentle lavage with PBS/EDTA (1 mM) using a 10 mL syringe. The cells were then centrifuged at 200 g for five minutes at room temperature (RT) for the flow cytometry assay. The cell-free lavage fluid was collected and stored frozen at −80°C until further use.
Flow Cytometry
Cells collected from each air pouch model were washed twice with FACS buffer (PBS/FBS 2%), resuspended in 100 µL of FACS Buffer, and subjected to flow cytometry as previously described. 23 In brief, cells were counted using the Automated Cell Counter TC 20 Bio Rad and kept on ice. After blocking with anti-mouse C16/32 (#101302, Biolegend, San Diego, CA, USA) for 10 minutes, cells were washed and incubated for 20 minutes on ice with the following antibodies: Pacific Blue anti-mouse-Ly-6C clone HK1.4 (#128013, Biolegend, San Diego, CA, USA), FITC anti-mouse-CD45 clone 30-F11 (#103107, Biolegend, San Diego, CA, USA), APC anti-mouse-CD11b clone M1/70 (#101211, Biolegend, San Diego, CA, USA), PE-Cy7 anti-mouse-Ly6G clone 1A8 (#127617, Biolegend, San Diego, CA, USA), Super Bright 600 anti-mouse-FceR1 alpha clone MAR-1 (#63-5898-82, Invitrogen, Waltham, MA, USA), PE anti-mouse-CD117 clone 288 (#12-1171-82, Invitrogen, Waltham, MA, USA). Cells were washed again with FACS buffer and resuspended in 300 µL of PBS. The viability dye DRAQ7 (#D15106; ThermoFisher Scientific, Waltham, MA, USA) was added at the time of acquisition using the Cytek Northern Lights (NL-3000). Single colors, FMOs, and unstained cells were used as controls.
Cytokine/Chemokine Analysis
Cytokines in the lavage fluids collected after one and three days of sensor and Teflon catheter implantation were measured using the Meso Scale Discovery (MSD) assay, according to the manufacturer’s instructions. The mouse custom V-Plex plate (K152AOH-2, MesoScale Discovery [MSD], Rockville, MD, USA) was designed for the multiplexed panel of cytokines/chemokines containing IL-1β, IL-6, KC/GRO, TNF-α, IL-4, IL-33, and MCP-1. The concentration of each analyte was determined via an electrochemiluminescent reaction detected using the Meso QuickPlex SQ 120MM microplate reader. The raw data were analyzed using MSD Discovery Workbench 4.0 software.
Statistical Analysis
Statistical analysis was performed using SigmaPlot version 15. To compare normally distributed data with equal variance, a Student’s t test was performed, whereas a Welch’s t test was performed on data with unequal variance. Nonnormally distributed data were evaluated using a Mann-Whitney Rank Sum test. Differences with a *P-value < 0.05 were considered statistically significant.
Results
Diabetes Technology Device Insertion Induces Neutrophil Extracellular TRAPs
Using a nondiabetic porcine model, we evaluated the tissue response toward the implantation of glucose sensors and insulin delivery devices, including infusion catheters and i-Port injection systems. Histological analysis with hematoxylin and eosin (H&E) staining revealed a dense infiltration of neutrophils, accompanied by deeply stained, web-like structures consistent with NETs, indicative of neutrophil activation and cell death.
To confirm the identity of these structures as NETs, we performed immunofluorescence staining on corresponding tissue sections to co-localize DNA (DAPI) with the biomarkers citrullinated histone H3 (CitH3) and myeloperoxidase (MPO). MPO, which is abundantly expressed in neutrophils,24,25 confirmed the presence of NETs across all tested devices (Figure 1a). Both glucose sensors and infusion catheters induced comparable NET formation at the insertion sites. Notably, NET accumulation was more prominent at the distal tips of the devices and along the insertion tracts (Figure 1b), suggesting a localized inflammatory response primarily driven by mechanical tissue disruption during device placement. These findings imply that the physical act of insertion may be a dominant factor in triggering acute inflammation, with the material composition of the devices playing a secondary role.
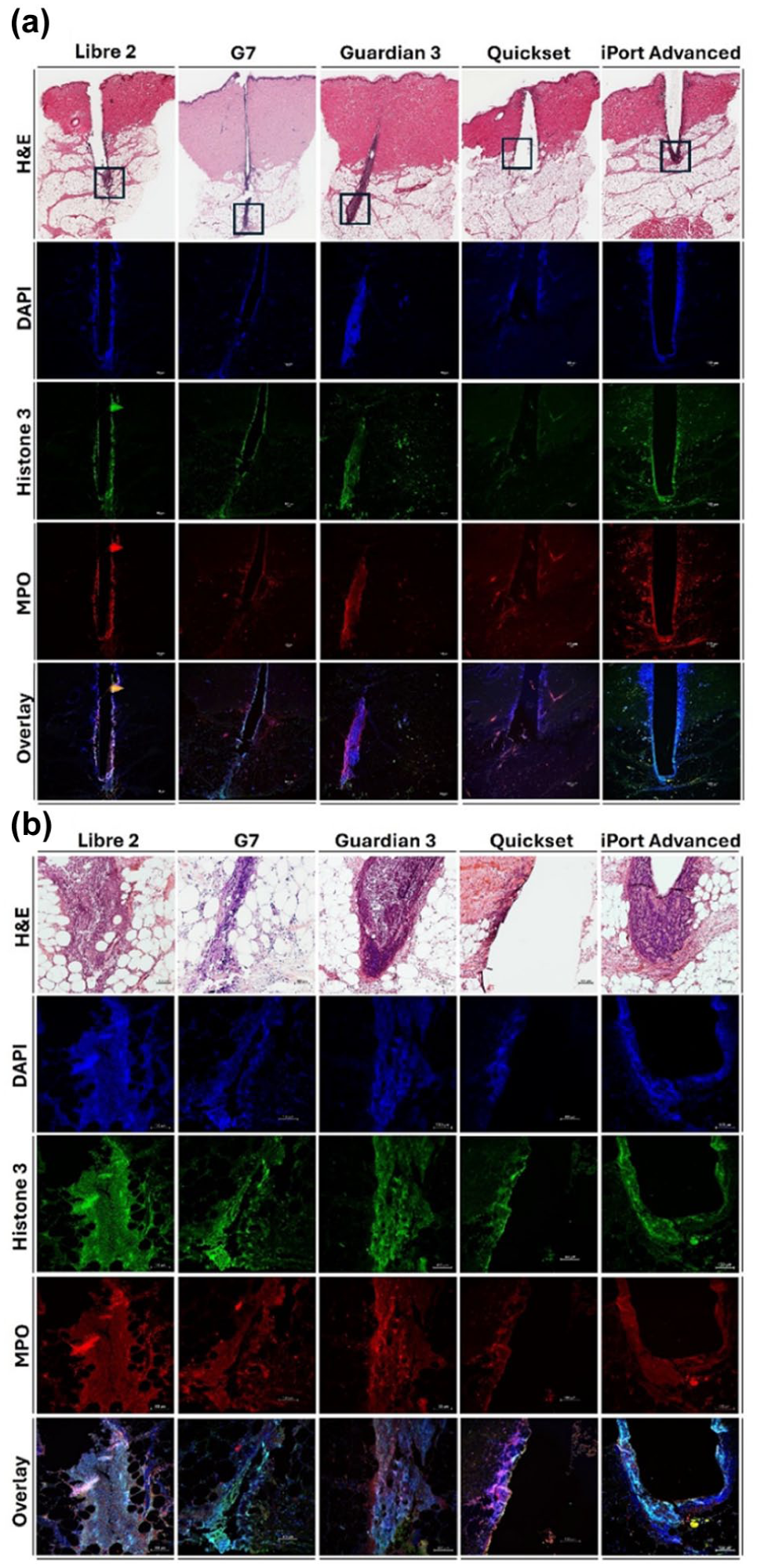
Detection of neutrophil extracellular traps (NET) by immunohistochemistry in swine skin at one day after sensors and catheter insertion. (a) Hematoxylin and eosin (H&E) staining reveals extracellular trap-like structures along the insertion pathway at low magnification (1×); scale bar = 1 mm. (b) Immunofluorescence staining at higher magnification (10×) highlights NET formation both along the insertion tract and at the distal tip of the device; scale bar = 100 μm. NETs were confirmed by the colocalization of DNA (DAPI, blue), Citrullinated histone H3 (CitH3, green), and myeloperoxidase (MPO, red), with merged images showing the overlap of all markers.
Teflon Triggers Material-Specific Inflammatory Responses in a Controlled Insertion Model
To determine whether device material contributes independently to inflammation, we employed a murine air pouch model that minimizes trauma during insertion. This model requires only a small needle puncture, allowing isolation of the immune response to the material itself. We compared the inflammatory cell influx induced by Teflon catheters and polyurethane-based CGM sensors (Libre 2, G7, and Medtronic Guardian 3) at one and three days postinsertion (Figure 2).
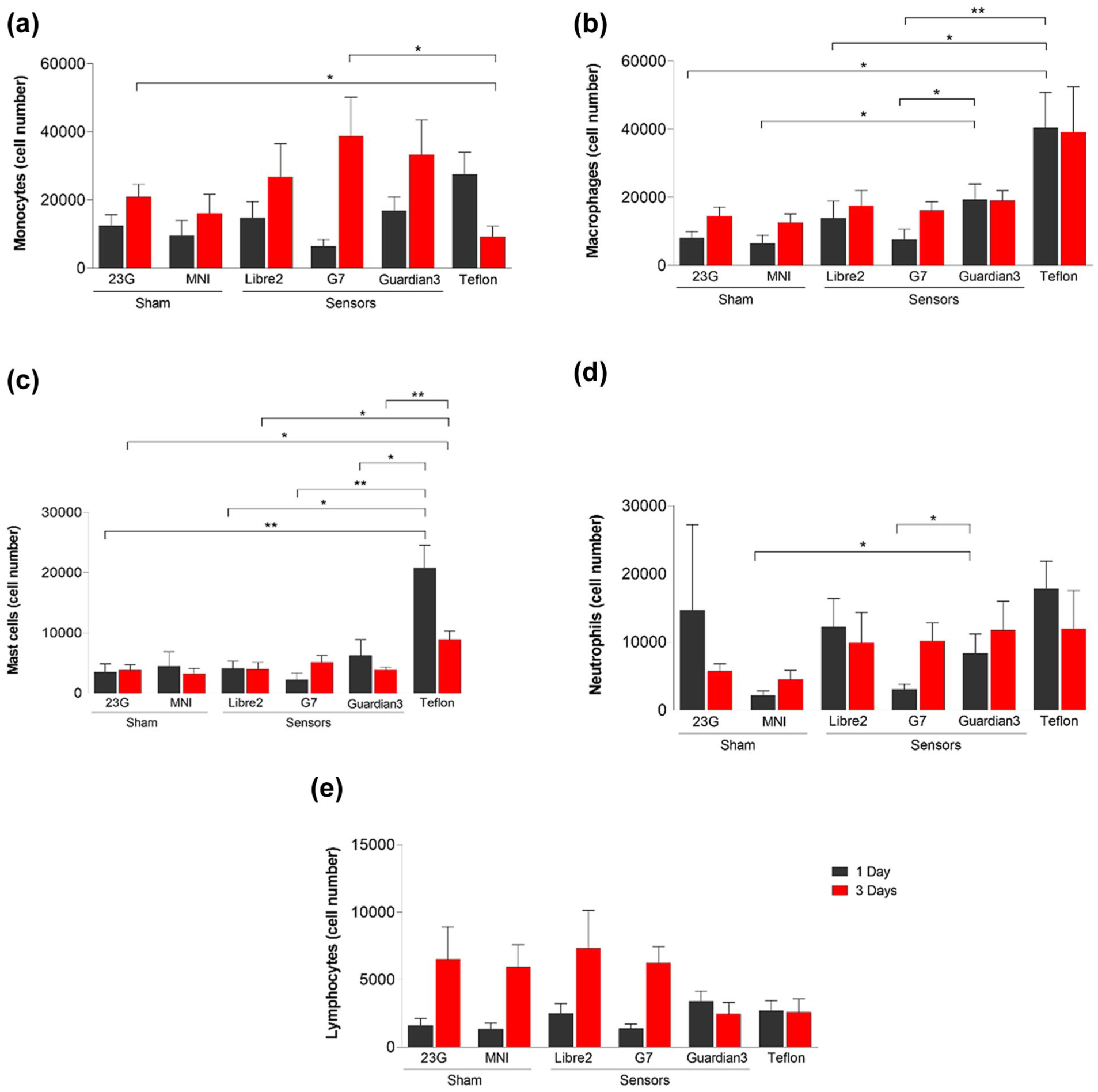
Immune cell population profile in response to Libre 2, G7, and Guardian 3 and Teflon catheter implantation in the murine air pouch model. Sham controls consisted of 23G needle insertions for Libre2, G7, and Teflon catheter groups, and the Medtronic Inserter Needle (MNI) for the Guardian 3 group. Immune cell infiltration was quantified at one and three days postimplantation. Graphs show the number of (a) monocytes, (b) macrophages, (c) mast cells, and (d) neutrophils. Data are presented as the mean ± SEM of at least six mice per group. Statistical comparisons were performed using Student’s t test. P < .05 (*); P < .005 (**).
On day 1, all devices and sham controls caused similar levels of monocyte infiltration. By day 3, however, the Teflon catheter showed significantly less monocyte infiltration than the sham and G7 sensor groups (Figure 2a). Macrophage infiltration at day 1 was similar across Libre 2, G7, and sham groups, but the Guardian 3 sensor induced a significantly higher macrophage response than both the sham and G7 groups (Figure 2b). Notably, the Teflon catheter produced a greater macrophage influx at day 1 than the sham, Libre 2, and G7 groups, indicating a unique material-specific immune profile.
Mast cell (MC) recruitment was also increased in response to the Teflon catheter. At one day post-insertion, the Teflon catheter induced higher MC counts than all sensors and the sham group. At three days, significant increases were observed compared with sham, Libre 2, and Guardian 3 (Figure 2c). Neutrophil levels were significantly higher in the Guardian 3 group compared with the sham and G7 groups at day 1 (Figure 2d). Lymphocyte levels remained consistent across all groups at both time points (Figure 2e).
The findings indicate that although the insertion process primarily causes inflammation, the material composition, especially Teflon, can affect the recruitment of specific immune cells, such as macrophages and MCs.
Material Composition Shapes Cytokine Responses to Implanted Devices in Murine Air Pouch Model
To assess the inflammatory cytokine profile induced by glucose sensors and Teflon catheter, we analyzed lavage fluid from the murine air pouch model at one and three days postimplantation (Figure 3). At day 1, the Guardian 3 sensor induced significantly elevated levels of monocyte chemoattractant protein-1 (MCP-1) compared with the sham group, G7, and Teflon catheter (Figure 3a). By day 3, MCP-1 levels in the Teflon catheter group were significantly lower than those in the sham and Libre two groups (Figure 3a).
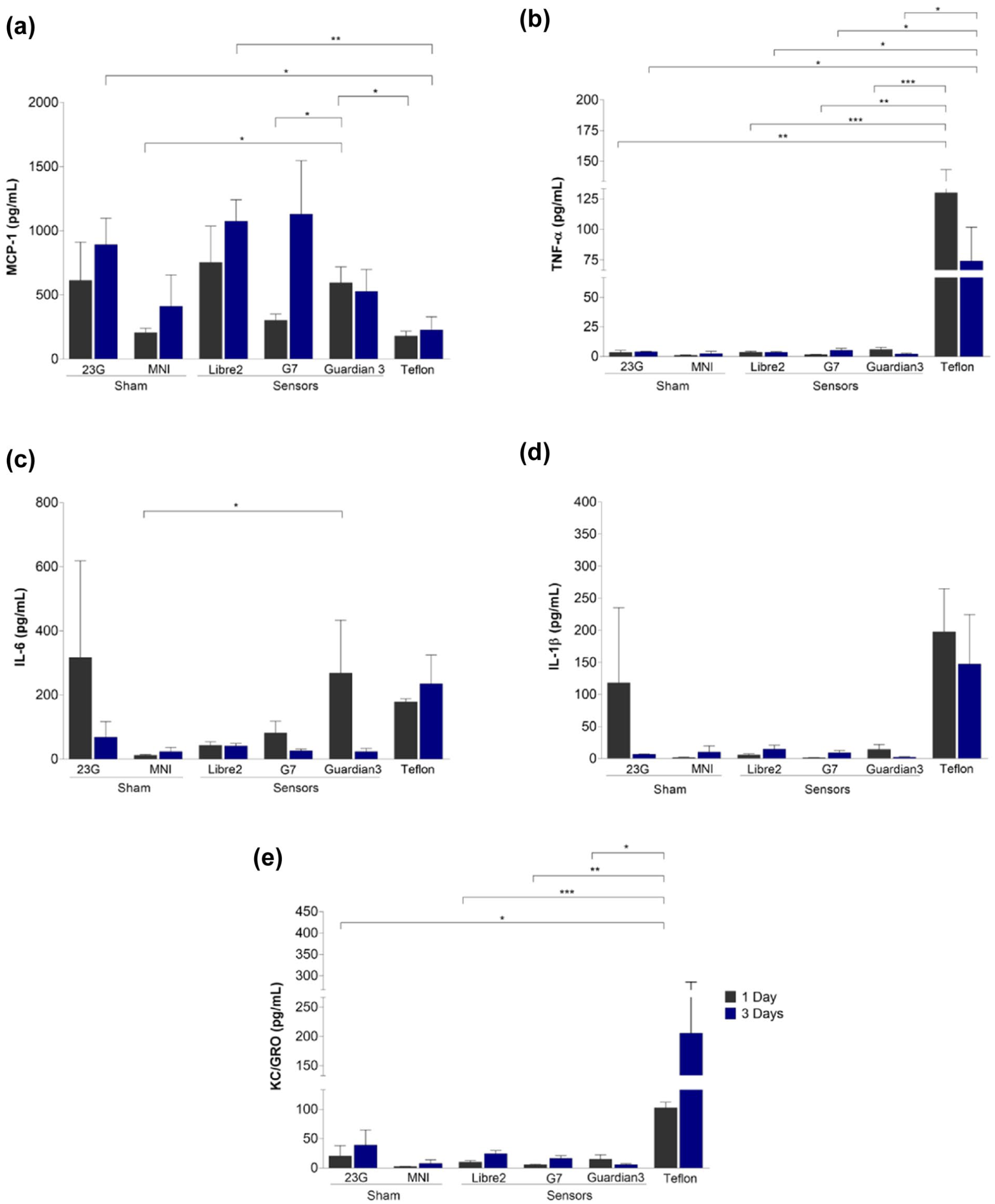
Release of proinflammatory cytokines following the implantation of Libre 2, G7, and Guardian 3 glucose sensors, as well as a Teflon catheter in a murine air pouch model. Sham controls included 23G needle insertions for the Libre 2, G7, and Teflon catheter groups, as well as the Medtronic Inserter Needle (MNI) for the Guardian 3 group. Cytokine concentrations in lavage fluid were measured at one and three days postimplantation. The graphs display levels (pg/mL) of (a) monocyte chemoattractant protein-1 (MCP-1), (b) tumor necrosis factor-alpha (TNF-α), (c) interleukin-6 (IL-6), (d) interleukin-1 beta (IL-1β), and (e) keratinocyte-derived chemokine/growth-regulated oncogene (KC/GRO). Data are presented as mean ± SEM from at least three mice per group.
Tumor necrosis factor-alpha (TNF-α) levels were comparable among all sensor groups and their respective sham groups. However, the Teflon catheter induced significantly higher TNF-α levels than both the sham and sensor groups at both time points (Figure 3b), indicating a sustained proinflammatory response.
For interleukin-6 (IL-6), only the Guardian 3 sensor showed a significant increase compared to its sham control on day 1 (Figure 3c). IL-1β levels remained consistent across all groups, including the sensors and the Teflon catheter (Figure 3d).
Notably, keratinocyte-derived chemokine/growth-regulated oncogene (KC/GRO) levels were significantly elevated in the Teflon catheter group compared with all sensor groups and their respective sham groups at day 1 (Figure 3e), further supporting the heightened inflammatory response associated with the Teflon material.
These results suggest that while glucose sensors induce mild cytokine responses, Teflon catheter material triggers a more significant and prolonged inflammatory cytokine profile.
Discussion
The American Diabetes Association and the European Association for the Study of Diabetes have identified infusion set failures as a major limitation in AID systems. 8 These failures highlight the urgent need for improved infusion set technologies that align with the extended wear duration of CGM systems, which can last up to 14 days, significantly longer than the typical two to three days for standard infusion sets. Although the Medtronic Extended Infusion Set (EIS) and Tandem Diabetes Care’s SteadiSet are FDA-cleared for up to seven days of use,26-29 many users do not reach the full duration, leading to disrupted insulin delivery, higher A1C levels, greater glycemic variability, and reduced adherence.26,30,31 Key factors contributing to this loss include infusion set occlusion, which may result from biochemical issues such as insulin aggregation or mechanical problems like cannula kinking, dislodgement, leakage, and local inflammation.32-36
Local inflammation at the site of the IIS cannula or CGM electrode is particularly concerning, as it can impair device performance.37-39 At CGM sites, inflammatory responses lead to the accumulation of metabolically active immune cells, such as neutrophils and macrophages, at the sensor-tissue interface. These cells consume interstitial glucose, causing the sensor to detect artificially low glucose levels. 40 This reduces CGM accuracy and may result in interstitial glucose readings that are lower than corresponding capillary blood glucose levels.
In contrast, inflammation at the IIS can increase insulin uptake and degradation by phagocytic immune cells, thereby reducing insulin availability and contributing to hyperglycemia.41,42 Additionally, if acute inflammation is not resolved, it can progress to chronic inflammation, resulting in tissue damage and potentially leading to fibrosis. This fibrotic response can have long-lasting effects on tissue integrity, further compromising both sensor accuracy and insulin absorption over time.
In this study, we examined the shared factor of skin insertion and the role of biomaterials in driving acute inflammatory responses, using both swine and mouse models. In swine, similar levels of NET formation were observed at insertion sites for CGM sensors and Teflon-based infusion catheters (QuickSet and i-Port), suggesting that mechanical trauma from insertion is a primary trigger of NET formation, regardless of the biomaterial. NETs, web-like structures composed of DNA, histones, and antimicrobial proteins, are released by activated neutrophils to trap pathogens.43,44 However, under sterile conditions, excessive NET formation can hinder tissue integration, interfere with wound-healing cytokine signaling, and lead to fibrosis.45,46 NETs may also break down implanted devices by releasing reactive oxidants, thereby sustaining chronic inflammation and the foreign body response.18,47,48
Although Teflon is known to promote NET formation in vitro due to its hydrophobicity, 49 we did not observe elevated NETs in vivo compared with PU-based glucose sensors. However, Teflon catheters did induce increased levels of the neutrophil-recruiting cytokine KC/GRO and significantly reduced levels of monocytes and monocyte chemoattractant protein-1 (MCP-1) three days postinsertion. MCP-1 signals through CCR2 to recruit monocytes and macrophages ,50,51 and its reduction may reflect a negative feedback mechanism after early monocyte influx .52,53 Given that monocytes can differentiate into macrophages at inflammatory sites, 54 this may explain the observed increase in macrophages near Teflon catheters, along with elevated TNF-α at one and three days postinsertion, both indicators of a sustained inflammatory response.
These findings are consistent with previous in vitro research by McKiel et al, 55 which showed Teflon-induced macrophage activation and cytokine production through adsorption of damage-associated molecular patterns and activation of the MyD88-dependent TLR2 signaling pathway. We also observed increased mast cell recruitment at Teflon catheter sites. Our earlier work indicated that mast cells are involved in inflammatory responses to phenolic preservatives in insulin formulations.21,22 However, their role in Teflon-mediated inflammation warrants further investigation.
In contrast, PU, a material widely used in medical devices, including CGM sensors, elicited a more favorable immune response. Polyurethane coatings enhance durability while maintaining permeability to glucose and oxygen. They also help minimize diffusion interference. 56 Unlike some polymers (e.g., polyethylene glycol, PEG) that can induce NETs, 57 PU did not significantly attract macrophages, mast cells, or inflammatory cytokines in our models. These data suggest that in CGM systems, acute inflammation is mainly caused by insertion trauma rather than the PU material itself.
Although these studies highlight differences in materials, mainly Teflon in IIS and PU in CGM devices, they do not separately assess factors, such as insertion force and angle, device size and shape, surface composition and texture, modulus relative to surrounding soft tissue, and both macro- and micro-motion. This narrow focus risks oversimplifying tissue responses. Importantly, as IIS devices serve as drug delivery systems, they also introduce triggers that are not relevant to CGMs. For example, preservatives and insulin-derived fibrils (IDFs), which form under mechanical and chemical stress, including thermomechanical energy from patient movement, can provoke immune responses and reduce insulin bioavailability.27,58-62 Both preservatives and IDFs are cytotoxic, promote inflammation, and contribute to fibrosis, damaging subcutaneous tissue and impairing insulin delivery.22,42,63,64 Thus, while some inflammatory mechanisms are shared between IIS and CGM devices, others are distinct to each, and multiple factors beyond material composition may play a more significant role in driving tissue responses at the device site.
Notably, the swine model showed minimal differences in inflammatory response among devices, while the mouse model exhibited more distinct material-specific responses. These findings suggest that the choice of animal model can significantly influence experimental results because of species-specific immune differences. Our findings provide insights that could inform future studies design and enhance human outcomes by elucidating the distinct roles of insertion trauma and biomaterial composition in the performance of diabetes devices.
Conclusion
This study shows that both insertion trauma and biomaterial composition influence the acute inflammatory response to subcutaneously implanted diabetes devices. Mechanical injury from insertion seems to be a key factor driving NET formation across different device types. However, differences specific to materials were also observed, particularly with Teflon catheters, which triggered sustained recruitment of macrophages and mast cells, along with increased production of proinflammatory cytokines. In comparison, polyurethane-based glucose sensors produced a milder immune response, indicating better biocompatibility during the early phase. While these findings highlight the importance of considering both mechanical and material factors in designing next-generation infusion sets and sensors, it is also crucial to recognize that other variables beyond material composition may have a more significant impact on tissue responses at the device site. These results also underscore the importance of selecting animal models that are suitable for the specific research question. For example, when devices are inserted into swine, the tissue response may be predominantly driven by insertion trauma, potentially masking the effects of biomaterial composition. In contrast, mouse models can help isolate the immune response to the material itself by minimizing mechanical injury. This distinction is critical for accurately assessing biocompatibility and immune responses to device materials.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251389945 – Supplemental material for Device Insertion Versus Material: Drivers of Inflammation in Diabetes Device Interfaces
Supplemental material, sj-docx-1-dst-10.1177_19322968251389945 for Device Insertion Versus Material: Drivers of Inflammation in Diabetes Device Interfaces by Priscila Silva Cunegundes, Kenneth Wood, Jean Gabriel de Souza, Anjul Bhangu, Li Mao and Ulrike Klueh in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
Carol Atkinson, Executive Director of Insulin for Life USA, provided the infusion set supplies.
Abbreviations
Automated Insulin Delivery (AID), Continuous Glucose Monitoring (CGM), Insulin Infusion Set (IIS), Foreign Body Response (FBR), Extended Infusion Set (EIS), Polyurethane (PU), Neutrophils Extracellular Traps (NETs), Myeloperoxidase (MPO), A 23-gauge needle (23G), Fluorophore Minus One (FMO), Room Temperature (RT), Medtronic Inserter Needle (MNI), Hematoxylin and Eosin (H&E), Meso Scale Discovery (MSD), Citrullinated histone H3 (CitH3), Monocyte Chemoattractant Protein-1 (MCP-1), Tumor Necrosis Factor- alpha (TNF-α), Interleukin-6 (IL-6), Interleukin-one beta (IL-1β), Interleukin-four (IL-4), Interleukin-thrirty three (IL-33), Mast cell (MC), Keratinocyte-derived Chemokine/Growth-Regulated Oncogene (KC/GRO), Damage-Associated Molecular Patterns (DAMPs), Polyethylene glycol (PEG), Insulin-Derived Fibrils (IDFs), Food and Drug Administration (FDA).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr UK is a co-founder of Cell and Molecular Tissue Engineering, LLC, and the founder of Alva Innovations, Inc., a small business developing diabetes technology. These affiliations represent potential financial conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the NIDDK Institute within the National Institute of Health (NIH) (grant number R01DK129681) and Digestive and Kidney Diseases (NIDDK) at the NIH (grant number 1R01DK133789).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
