Abstract
Background:
Lipohypertrophy is a common skin complication in people with insulin-treated diabetes. Despite its high prevalence and potential impact on diabetes management and outcomes, published data regarding the risk factors for the development of this complication are contradictory. The study aimed to determine risk factors for lipohypertrophy related to patient characteristics and insulin therapy.
Method:
Medical databases (MEDLINE/PubMed, Embase, CENTRAL) were searched from 1990 to August 21, 2023. All relevant studies describing potential risk factors for lipohypertrophy in people with insulin-treated diabetes (eg, sex, age, body mass index [BMI], type of diabetes, and injection technique) were included. Data enabling calculations of prevalence odds ratios (pOR) and mean differences (MD) with 95% confidence intervals [95% CI] were extracted and pooled in meta-analyses.
Results:
Fifty-one studies of risk factors for lipohypertrophy were identified. Performed meta-analyses indicate that the strongest contributor to lipohypertrophy was incorrect injection site rotation (pOR = 8.85 [95% CI: 5.10-15.33]), followed by needle reuse (3.20 [1.99-5.13]), duration of insulin therapy >5 years (2.62 [1.70-4.04]) and >2 daily injections per day (2.27 [1.58-3.25]). Those with type 1 diabetes and obese/overweight individuals also had significantly higher odds of developing lipohypertrophy. Sex, age, and insulin device (pen, syringes) were not significant risk factors for lipohypertrophy.
Conclusions:
This systematic review with meta-analysis revealed that incorrect injection site rotation and needle reuse are the most substantial factors in developing lipohypertrophy. Notably, both factors are modifiable through patient education, emphasizing the importance of teaching proper injection techniques for better diabetes management.
Keywords
Introduction
Lipohypertrophy, a localized change in the structure of subcutaneous fatty tissue, is a prevalent side effect of insulin therapy among people with diabetes. 1 Previous studies have shown that people with lipohypertrophy more often experience episodes of unexplained hypoglycemia2-9 and have significantly higher glycated hemoglobin (HbA1c)6,10-14 than those without this skin complication.
Despite its high prevalence among individuals with diabetes, based on a recent meta-analysis (41·8% [95% CI: 35.9-47.6]), 15 the precise etiology of lipohypertrophy remains elusive. Numerous risk factors for developing lipohypertrophy among insulin-injecting patients have been considered, including demographic variables,11,13,16-20 injection technique,3,10,16,19,21,22 and disease course,2,3,13,23 creating a complex web of potential contributors. For example, Vardar and Kizilci 24 identified that longer insulin therapy duration, incorrect injection site rotation, and needle reuse are independent risk factors for lipohypertrophy. Other researchers, including Blanco et al 3 and Hajheydari et al, 13 proposed that diabetes type also may contribute to lipohypertrophy development, with a higher prevalence observed in patients with type 1 diabetes compared with type 2 diabetes. At the same time, Gentile et al 25 and Lin et al 26 observed that the number of daily insulin injections is higher in people with lipohypertrophy compared with those without this condition. On the contrary, other authors found no associations between lipohypertrophy and site rotation,20,27,28 needle reuse,9,23,29,30 type of diabetes,30-32 and the daily number of insulin injections.7,20,27,33 These conflicting results leave important questions unanswered regarding what precisely predisposes people with diabetes to this condition.
This study aimed to address these contradictory data by conducting a systematic review and meta-analysis. The primary objective was to identify the risk factors associated with the development of lipohypertrophy in people with insulin-treated diabetes.
Materials and Methods
Selection Criteria and Search Strategy
This review focused on studies that included people with diabetes on subcutaneous insulin or glucagon-like peptide-1 (GLP-1) therapy administered via pens or syringes, excluding those using devices for continuous subcutaneous infusion of such drugs. The decision to include GLP-1 therapies was based on evidence indicating that subcutaneous nodules, resembling lipohypertrophy in imaging studies, have also been reported with GLP-1 analogs such as exenatide.34,35 Due to substantial differences in insulin delivery methods using syringes/pens versus pumps, we chose not to mix results for these populations and we are working on a separate research extensively discussing the issue of lipohypertrophy in pump users. Of primary interest were studies that compared the frequency of lipohypertrophy in groups with and without potential risk factors, including variables related to patient characteristics (sex, diabetes type, body mass index [BMI] category, duration of diabetes, and subcutaneous therapy >5 years) and injection technique (needle reuse, site rotation, insulin type, needle size, number of daily injections >2). In addition, studies reporting mean BMI, duration of diabetes and subcutaneous therapy, insulin dose, number of injections per day, and insulin dose in individuals with and without lipohypertrophy studies were considered.
No restrictions regarding study design were made, allowing the inclusion of experimental and observational studies. Only studies presenting data unsuitable for cumulation in meta-analysis, published in languages other than English, and conference proceedings were excluded from our review. Furthermore, only research that was conducted and published after 1990 was included to ensure the applicability of study results to current clinical practice. Earlier work was considered outdated because of the widespread use of animal-derived insulin, which no longer represents the standard of care in diabetes management.
The systematic search was conducted on August 21, 2023, in MEDLINE (via PubMed), Embase, and CENTRAL (via The Cochrane Library) using keywords and queries related to diabetes and lipohypertrophy. Detailed search strategies for each database were shown in the supplementary appendix (Supplementary Tables 1-3). To retrieve other relevant publications not indexed in searched medical databases, a hand-search of the references of the included studies was also performed. The review process was conducted by two independent analysts (AST, MM) following the PRISMA (Preferred Reporting Items for Systematic Review and Meta-Analyses) 2020 guidelines 36 and registered PROSPERO protocol (CRD42023459726). Any disagreements between reviewers on the full-text stage of the selection process were resolved by consensus.
Data Analysis
Two reviewers performed data extraction independently (AST, MM). Any identified discrepancies in extraction between reviewers were discussed and resolved by consensus. In cases of concerns about the correctness of the reported data in the included studies, such data were not extracted. Extracted items related to the methodology of the included studies, characteristics of patients with emphasis on details of anti-hyperglycemic therapy, and risk factors of lipohypertrophy. Regarding risk factors, if available, we extracted results of comparisons and P values made by authors of the individual studies. We contacted the corresponding authors of studies conducted by the same research groups to ensure that published articles do not duplicate results for the same patient cohorts. The quality of the included studies was evaluated by Joanna Briggs Institute tools (JBI) for cross-sectional 37 and quasi-experimental 38 studies.
Meta-analyses were conducted using a random-effects model (DerSimonian & Laird) as significant between-study heterogeneity of results (P value for Cochrane Q test < 0.10 and I 2 > 50%) was observed for all comparisons. Outcomes were presented either as prevalence odds ratios (pOR) for the percentage of patients with lipohypertrophy among risk factors groups or as mean differences (MD) for variables expressed by mean values and standard deviations (SDs). All results were given with 95% confidence intervals [95% CI]. Funnel plots were also drawn for the assessment of the publication bias.
Subgroup analyses and meta-regression were performed to address heterogeneity in meta-analyses and explore the impact of various confounding variables on the obtained results. The considered factors for subgroup analyses included the geographic region of the study, the number of participating centers, diabetes type, the age of the study population, the definition of lipohypertrophy, publication date, and the population size. In addition, for meta-regression, the impact of continuous variables related to injection technique and duration of anti-diabetes therapy was investigated, among others. Meta-regression calculations were conducted using the random-effects model (REML, restricted maximum likelihood estimation). Subgroup analyses and meta-regression were performed exclusively for meta-analyses with statistically significant results that included at least 10 studies. OpenMEE software 39 was used for all calculations.
Results
Of the 4131 records identified during databases and references search, 273 full-text articles were selected for further analysis. Among these, 51 studies met the criteria for inclusion in this meta-analysis (Table 1), and 222 were excluded. Detailed reasons for exclusion and the PRISMA flow diagram describing the selection process are provided in the supplementary appendix (Supplementary Figure 1).
Summary of the Included Studies.
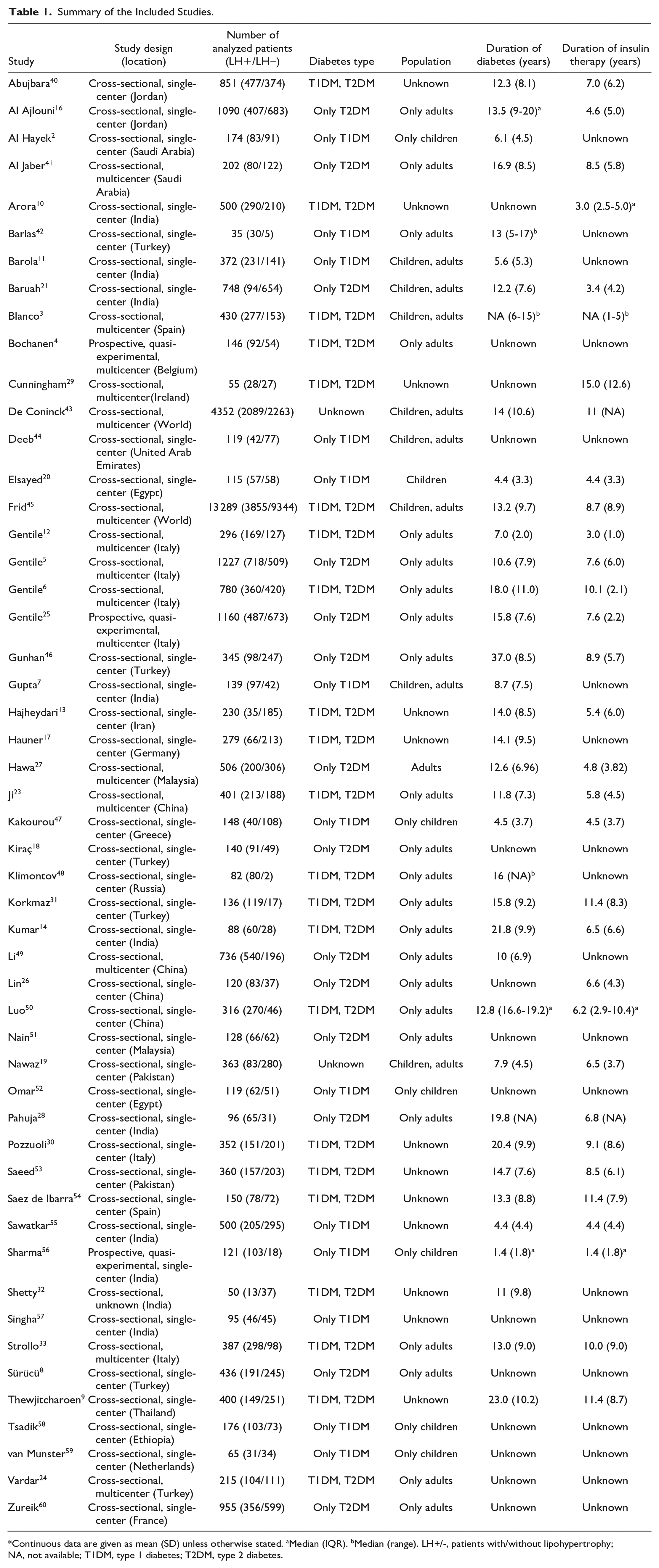
Continuous data are given as mean (SD) unless otherwise stated. aMedian (IQR). bMedian (range). LH+/-, patients with/without lipohypertrophy; NA, not available; T1DM, type 1 diabetes; T2DM, type 2 diabetes.
All eligible studies—except three prospective studies (Bochanen et al, 4 Gentile et al, 25 Sharma et al 56 )—were cross-sectional. Most studies were conducted in East Asia (19 studies), primarily in India (10 studies); 15 originated from Europe, while 14 described patients from Western Asia or Arab countries. In addition, two studies included global data (multinational studies) and one was conducted in Ethiopia. Of the 51 included studies, 69% were single-center studies.
The sample size in the included studies varied from 35 (Barlas et al 42 ) to 13 289 (Frid et al 45 ) patients; however, the majority of them included less than 500 participants (38 studies). Most of the included studies were conducted on a mixed population in terms of age and type of diabetes. Only 13 studies exclusively targeted people with type 1 diabetes, and 14 studies concentrated on those with type 2 diabetes. Notably, seven studies included a pediatric type 1 diabetes population. All studies included participants on insulin therapy, only two studies featured a small subset of patients on GLP-1 analogs (Frid et al, 45 Kiraç et al 18 ). As a result of the systematic search, no studies allowed assessment of the risk factors in those on GLP-1 agonists therapy.
Regarding the risk of bias assessment, most included studies scored at least six points on the JBI scale, indicating good methodological quality. Only individual studies (De Coninck et al, 43 Gentile et al, 25 Kakourou et al, 47 Omar et al, 52 and van Munster et al 59 ) presented average quality with four to five points on JBI scales. Nonetheless, no study was excluded from the systematic review due to methodological issues. Detailed methodology, patient characteristics, and JBI scores are shown in the supplementary appendix (Supplementary Tables 5-9).
Performed meta-analyses showed that incorrect rotation of injection sites was the strongest factor associated with an increased prevalence of lipohypertrophy (pOR = 8.85 [95% CI: 5.10-15.33]) in people with diabetes (Figure 1). The second most significant contributor to lipohypertrophy presence was needle reuse (3.20 [95% CI: 1.99-5.13]). Higher frequency of lipohypertrophy was also observed among those who used longer needles, that is, ≥8 mm (2.00 [95% CI: 1.35-2.97]) or ≥6 mm (1.52 [1.20-1.92]) and who injected insulin more than two times per day (2.27 [1.58-3.25]). The mean difference in daily injection between patients with lipohypertrophy and those without this complication was 0.40 [95% CI: 0.24-0.56], indicating a higher number of daily injections in the lipohypertrophy group (Figure 2). The type of injection device (pen, syringe) had no impact on lipohypertrophy prevalence (0.75 [95% CI: 0.49-1.14]). Interestingly, individuals treated with insulin analogs had lower odds of having lipohypertrophy than those treated with human insulins (0.67 [95% CI: 0.48-0.94]). The mean daily insulin dose was about 7.86 [95% CI: 5.57-10.15] units higher in patients with lipohypertrophy than in those without this condition.
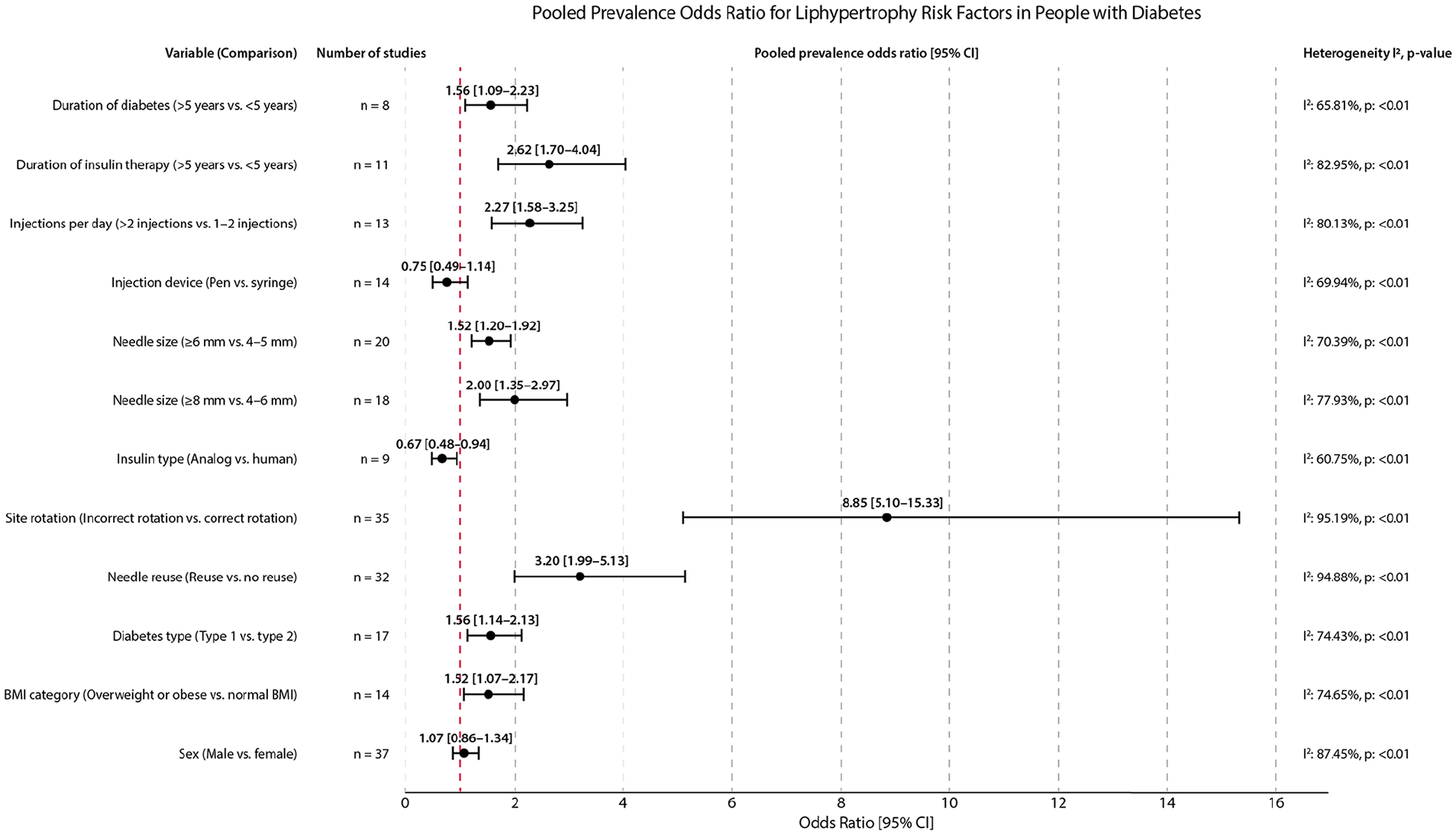
The pooled prevalence odds ratio for lipohypertrophy risk factors in people with diabetes.
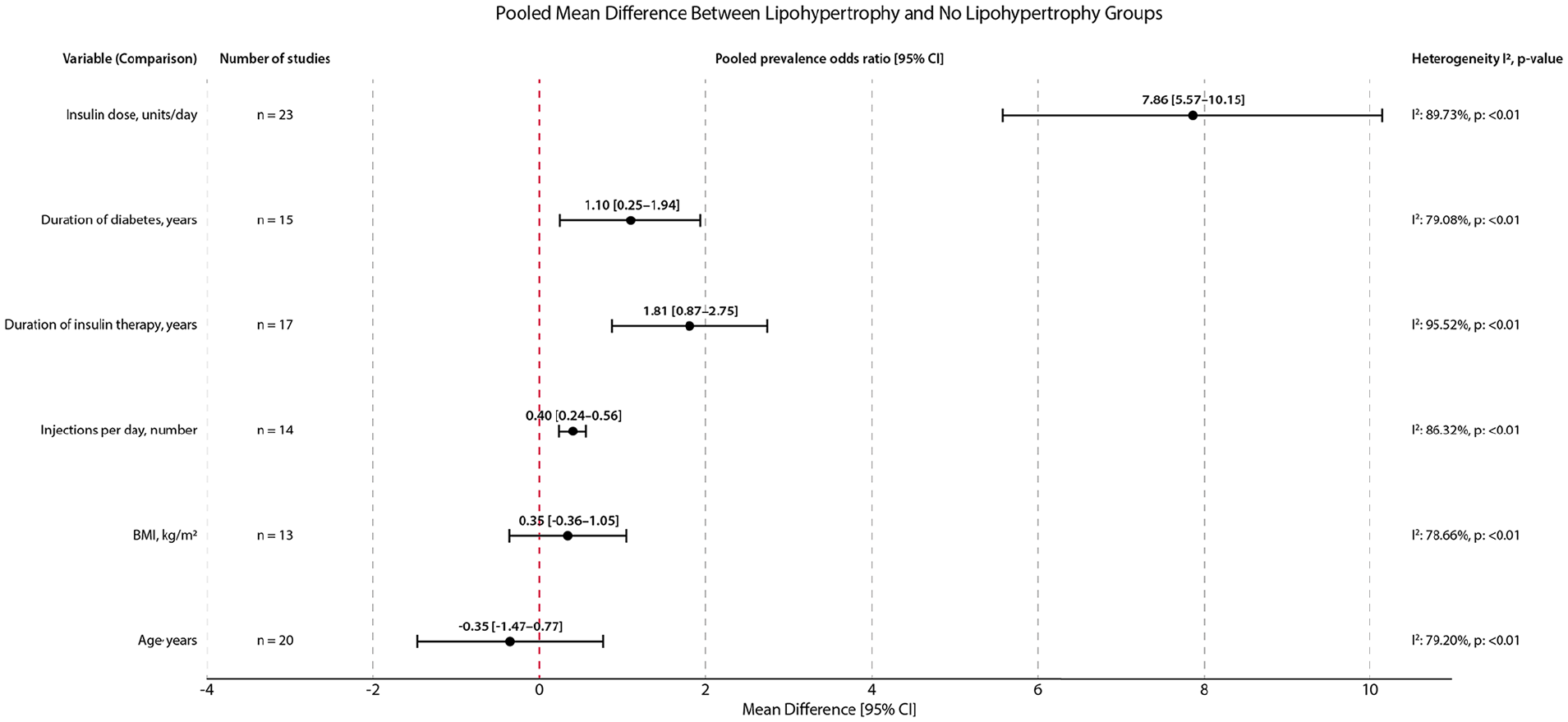
The pooled mean difference between lipohypertrophy and no lipohypertrophy groups.
Among variables related to patients’ characteristics, both longer (>5 years) duration of diabetes and insulin therapy were associated with increased odds of lipohypertrophy (1.56 [95% CI: 1.09-2.23] and 2.62 [1.70-4.04]). Mean differences in duration of diabetes and insulin therapy were higher among patients with lipohypertrophy, that is, 1.10 [95% CI: 0.25-1.94] years and 1.81 [0.87-2.75] years, respectively. Lipohypertrophy was slightly more prevalent in patients with type 1 diabetes compared with those with type 2 diabetes (1.56 [95% CI: 1.14-2.13]) and in overweight/obese individuals compared with those with normal BMI (1.52 kg/m2 [1.07-2.17]). Nonetheless, patients with and without lipohypertrophy did not differ in mean BMI (MD = 0.35 kg/m2 [95% CI: -0.36 to 1.05]). Sex and age were insignificant risk factors for lipohypertrophy development (Figures 1 and 2).
In the subgroup analyses, significant interactions for lipohypertrophy odds ratios were observed for specific comparisons depending on factors such as geographical region, the proportion of patients with type 1 diabetes, publication date, type of lipohypertrophy diagnosis (clinical, ultrasonographical), age of the population, and the number of patients in the studies. In most cases, interactions were noted for comparisons involving subgroups of a single study, suggesting that the observed discrepancies are more likely to be incidental rather than indicative of clear trends. Results of subgroup analyses were shown in the supplementary appendix (Supplementary Tables: 12, 15, 19, 23, 27, 31, 35, 38, 41, 44, 48, 52).
In many cases, meta-regression was conducted on fewer studies than the original comparisons, rendering these findings exploratory (Supplementary Tables: 13, 16, 20, 25, 28, 32, 36, 39, 42, 45, 49, 53). Nonetheless, the conducted meta-regression, in most cases, did not reveal any significant associations between the analyzed variables and the results of comparisons for specific risk factors regarding the frequency of lipohypertrophy occurrence. This suggests an independent nature of analyzed risk factors from other variables. However, the effect size of risk factors such as needle reuse and incorrect site rotation could be influenced by study size. In addition, as the odds ratio for incorrect rotation increased, the odds for reuse also rose, suggesting a co-occurrence of these injection technique-related issues among patients (Supplementary Table 13).
No publication bias was observed in funnel plots (Supplementary Figures: 3, 23, 38, 40, 52, 54, 68, 82, 84, 97, 99, 112, 125, 136, 149, 151, 166, 168).
Discussion
Lipohypertrophy is a recognized skin complication of subcutaneous insulin therapy, and understanding the risk factors is crucial for optimizing diabetes care. This meta-analysis, including 51 studies, is the first to systematically examine a wide range of potential risk factors associated with lipohypertrophy.
Our results indicate that incorrect rotation of injection sites and needle reuse are the two strongest contributors to lipohypertrophy, as evidenced by high odds ratios, emphasizing a robust and non-random association. These modifiable risk factors underscore the importance of patient education and health care provider guidance in promoting proper injection techniques and minimizing the risk of lipohypertrophy. Other factors with statistical significance for increased lipohypertrophy odds included type 1 diabetes, being overweight or obese, longer needle size (≥6 mm, ≥8 mm), a higher number of daily injections, and longer duration of insulin or diabetes. In contrast, using insulin analogs seemed to have a protective impact on lipohypertrophy development. However, it is crucial to note that most of these associations, while statistically significant, exhibit relatively low odds ratios (<2). Considering also the moderate quality of the studies, caution is warranted in interpreting these results. Sex, age, and insulin device (pens, syringe) were not associated with increased odds of lipohypertrophy in this meta-analysis.
While extended exposure to insulin therapy in terms of longer insulin therapy duration and frequent injections may seem intuitive as contributors to lipohypertrophy, explanations for other risk factors are not as straightforward. For instance, the relationship between type 1 diabetes and slightly increased lipohypertrophy prevalence raises questions about whether these patients are more susceptible to developing lipohypertrophy due to different pathogenesis or because of generally longer duration of insulin therapy or higher use of multiple daily injection schemes than patients with type 2 diabetes. Alternatively, it is also possible that lipohypertrophy is more easily identifiable in individuals with type 1 diabetes due to higher awareness of potential complications and better diabetes management education stemming from an early diagnosis in childhood, as suggested by our own clinical experience. Explaining the causes of these differences, however, requires further research to be thoroughly investigated and validated.
Similarly, higher occurrence of lipohypertrophy in individuals with increased BMI may result from excess sensitivity of adipose tissue itself or the fact that first signs of subcutaneous nodules in leaner patients may be visually detectable at an early stage, enabling taking preventive actions much earlier. Interestingly, this meta-analysis also showed that using shorter needles and insulin analogs is associated with a reduced prevalence of lipohypertrophy. However, it remains unclear whether these decreased odds are attributable to specific pharmacological properties of insulin analogs and mechanical advantages of shorter needles or if patients with access to modern interventions also receive better diabetes education with lipohypertrophy prevention. Further research, especially well-designed prospective studies, is needed to provide a more comprehensive understanding of these risk factors.
Previous meta-analyses15,61 focused on the pooled prevalence of lipohypertrophy among people with diabetes. Only Wang et al 15 attempted to identify risk factors for lipohypertrophy using meta-regression to assess factors such as sex, age, diabetes type, duration of insulin therapy, needle reuse, needle size, site rotation, HbA1c levels, insulin dose, and mean number of daily injections on pooled lipohypertrophy prevalence. However, their approach did not consider variations in the prevalence of these factors between patients with and without lipohypertrophy. Instead, they used a cumulative score for the entire study population as a confounding variable. As a result, they did not identify any significant influence of analyzed factors on the development of lipohypertrophy. Only the duration of insulin therapy was found to have a notable impact on its prevalence (β coefficient = 0.124 [0.026-0.221], P = 0.013). This contradicted the findings from several individual studies, which consistently indicated the presence of other risk factors, such as incorrect site rotation and needle reuse. Our systematic review examined the characteristics of patients with and without lipohypertrophy separately, which allowed the calculation of the odds ratio for each considered factor. Furthermore, our review encompassed a greater number of studies compared with Wang et al, enhancing the comprehensiveness of the systematic review.
Despite providing a comprehensive literature review and analysis of potential risks, this meta-analysis has certain limitation. The primary limitation lies in the composition of the included studies, which predominantly consisted of cross-sectional studies. Although these studies provide valuable insights into the potential risk factors, they cannot establish causality. This concern is particularly relevant when examining the associations between insulin dose and lipohypertrophy. It remains challenging to determine whether increased insulin requirements result from impaired insulin absorption from lipohypertrophic lesions or whether higher insulin doses accelerate the development of lipohypertrophy. Most likely, the relationship between insulin dose and lipohypertrophy is bidirectional, as evidence from prospective studies suggests that educational intervention aimed at improving injection technique can reduce both lipohypertrophy and insulin dose requirements. Grassi et al 62 reported that correct injection techniques consisting of single-use of 4-mm needles and avoiding injection into lipohypertrophy decreased insulin dose by two units in the three months. In a randomized clinical trial conducted by the AMD-OSDI Study Group, 63 educational intervention significantly reduced insulin demand in patients with diabetes by 20.7%. These findings support the results of our meta-analysis.
Another significant challenge is the substantial heterogeneity observed across various analyses. To elucidate the potential sources of the heterogeneity, a series of subgroup meta-analyses and meta-regression were conducted considering confounding variables such as diabetes type, patient age, lipohypertrophy measurement, and other related to patient characteristics and injection techniques. Unfortunately, due to limited studies and data availability, only one confounding variable could be assessed at the time, often using fewer studies than initially included in the primary meta-analysis. Despite considerable efforts, most of the univariable analyses did not entirely explain the observed variability of results. Nonetheless, the subgroup and meta-regression analyses suggested that several factors may contribute to the observed effect size, including sample size. While ancillary analyses provide valuable hypotheses for further investigation, they should be interpreted cautiously. Specifically, generalizing results for subgroups consisting of one study could be inappropriate, as the publication of new studies in the future may dramatically change the effect size.
Furthermore, only research conducted and published after 1990 was included to ensure relevance to current clinical practice, given the shift from animal-derived insulin to modern insulin formulations. This approach was necessary to reflect contemporary treatment standards but inherently limits comparisons with older data. Further uncertainties in the results related to reporting errors were found in some publications included in the systematic review. For example, some data from Strollo et al 33 study were omitted in extraction due to incorrect calculations of percentages of patients with lipohypertrophy in the specific risk groups. In addition, variations in definitions of lipohypertrophy, methods of assessment, and different clinical practices across diabetes centers, including qualifications and technical skills of individuals assessing lipohypertrophy, may have influenced the meta-analysis results. Despite a comprehensive literature review, no evidence could be found describing risk factors for lipohypertrophy in patients treated with GLP-1 analogs. Future research should focus on this patient group. Moreover, it is important to emphasize that the results of this meta-analysis should not be directly extrapolated to patients using insulin pumps. This caution is due to the distinct nature of insulin delivery and the potential unique risk factors associated with continuous subcutaneous insulin infusion systems, which were not specifically addressed in our study. Further research is needed to understand lipohypertrophy in the context of insulin pump therapy and its potential differences from those receiving insulin via injections.
Conclusions
In conclusion, this study has identified several modifiable risk factors associated with lipohypertrophy in insulin-injecting patients. Incorrect rotation of injection sites, needle reuse, needle size, and insulin type are targets for educational interventions. Widespread education on safe and correct injection practices is a clear option to reduce the prevalence of lipohypertrophy and improve the overall quality of diabetes care.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251325569 – Supplemental material for Risk factors for Lipohypertrophy in People With Insulin-Treated Diabetes: A Systematic Meta-Analysis
Supplemental material, sj-docx-1-dst-10.1177_19322968251325569 for Risk factors for Lipohypertrophy in People With Insulin-Treated Diabetes: A Systematic Meta-Analysis by Julia K. Mader, Riccardo Fornengo, Ahmed Hassoun, Lutz Heinemann, Bernhard Kulzer, Magdalena Monica, Trung Nguyen, Jochen Sieber, Eric Renard, Yves Reznik, Przemysław Ryś, Anita Stożek-Tutro and Emma G Wilmot in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-2-dst-10.1177_19322968251325569 – Supplemental material for Risk factors for Lipohypertrophy in People With Insulin-Treated Diabetes: A Systematic Meta-Analysis
Supplemental material, sj-docx-2-dst-10.1177_19322968251325569 for Risk factors for Lipohypertrophy in People With Insulin-Treated Diabetes: A Systematic Meta-Analysis by Julia K. Mader, Riccardo Fornengo, Ahmed Hassoun, Lutz Heinemann, Bernhard Kulzer, Magdalena Monica, Trung Nguyen, Jochen Sieber, Eric Renard, Yves Reznik, Przemysław Ryś, Anita Stożek-Tutro and Emma G Wilmot in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
CI, confidence interval; GLP-1, glucagon-like peptide-1; HbA1c, glycated hemoglobin; JBI, Joanna Briggs Institute; LH+, patients with lipohypertrophy; LH-, patients without lipohypertrophy; MD, mean difference; pOR, prevalence odds ratio; SD, standard deviation, T1DM, type 1 diabetes; T2DM, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JKM is a member of the Advisory Board of Abbott Diabetes Care, Becton-Dickinson/embecta, Boehringer Ingelheim, Eli Lilly, embecta, Medtronic, Novo Nordisk A/S, Roche Diabetes Care, Sanofi-Aventis, Viatris; and received speaker honoraria from A. Menarini Diagnostics, Abbott Diabetes Care, AstraZeneca, Becton-Dickinson/embecta, Boehringer Ingelheim, Dexcom, Eli Lilly, Medtrust, MSD, Novo Nordisk A/S, Roche Diabetes Care, Sanofi, Servier, and Ypsomed. BK is a member of the advisory board of embecta. ER is a member of the Advisory Board of Eli Lilly, Novo Nordisk, and received speaker honoraria from embecta. JS was an employee of embecta as this research was conducted. He is stockholder of embecta and Becton&Dickinson. TN is an employee of embecta. LH is a consultant for several companies developing novel diagnostic and therapeutic options for diabetes treatment. He is a shareholder of the Profil Institut für Stoffwechselforschung GmbH, Neuss, Germany. YR declares consultant/speaker fees from Medtronic, Insulet, embecta, Abbott, Novo Nordisk, Eli-Lilly, Sanofi, and Air Liquide Santé International. AH is a member of the advisory board of embecta. AST, MM, PR, and RF declare no conflict of interest. EGW is a member of the advisory board of Abbott Diabetes Care, Eli Lilly, embecta, Insulet, Medtronic, Novo Nordisk, Roche Diabetes Care, and Sanofi-Aventis. Research support from Abbott Diabetes Care, embecta, Insulet, Novo Nordisk, and Sanofi-Aventis, and has received speaker honoraria from Abbott Diabetes Care, AstraZeneca, Dexcom, Eli Lilly, embecta, Glooko, Insulet, Medtronic, Novo Nordisk, Sanofi, and Ypsomed.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was sponsored by embecta. The sponsor had a role in the study design, interpreting data, writing the report, and in the decision to submit the paper for publication.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
