Abstract
Background:
Lipohypertrophy is a common skin complication in individuals with insulin-treated diabetes, but this condition in those using insulin pumps (continuous subcutaneous insulin infusion, CSII) remains poorly understood. This study aimed to identify and summarize scientific evidence regarding the risk factors and clinical consequences of lipohypertrophy in people using CSII.
Methods:
Medical databases (MEDLINE/PubMed, Embase, CENTRAL) were searched to identify relevant studies published in English from 1990 to March 19, 2024. If possible, extracted data were cumulated in meta-analyses. This systematic review was registered on PROSPERO (CRD42024554127).
Results:
Nine studies reporting risk factors for lipohypertrophy or its consequences in people treated with CSII were identified. In the included studies, only individual risk factors were reported, which in most cases prevented the conduct of a meta-analysis. Meta-analyses could be performed for two factors, that is, improper cannula site rotation and male sex. The odds of developing lipohypertrophy were higher in individuals incorrectly rotating (prevalence odds ratio, pOR = 2.59 [95% Confidence interval, CI: 1.39-4.83]), whereas gender had no impact on the prevalence of lipohypertrophy (pOR = 1.13 [95% CI: 0.62-2.06]). Due to the limitations of the available data, it was not possible to draw conclusions about the clinical consequences of lipohypertrophy in people on CSII.
Conclusions:
This systematic review demonstrated that studies on the risk factors and clinical consequences of lipohypertrophy in CSII users are limited and that the currently published data are insufficient to draw definitive conclusions. There is a need for more comprehensive and well-designed clinical studies to better understand this issue in CSII users.
Introduction
Lipohypertrophy is a common skin complication in people with insulin-treated diabetes, 1 characterized by the abnormal accumulation of subcutaneous fat tissue at skin sites at which insulin is administered. 2 This condition not only affects the aesthetic appearance of the skin but also has significant implications for diabetes management, as it can lead to erratic insulin absorption and unpredictable glycemic control resulting in an elevated risk of both hyperglycemia and hypoglycemia.3,4 The high prevalence of lipohypertrophy in people with insulin-treated diabetes (41.8% [95% confidence interval, CI: 35.9-47.6]) 5 underscores its clinical importance, necessitating a thorough understanding of its risk factors and consequences.
Among people with diabetes who are using syringes or pens for insulin administration, clinical data on the risk factors for lipohypertrophy and its potential impact on glycemic control are relatively well studied. For instance, clinical studies have identified several risk factors, such as improper rotation of injection sites,6 -9 reuse of needles,10 -13 and long duration of insulin therapy. 14 Recent meta-analyses showed that lack of injection site rotation (prevalence odds ratio (pOR) = 8.85 [95% CI: 5.10-15.33]) and repeated use of insulin needles (3.20 [1.99-5.13]) are the strongest risk factors for lipohypertrophy. 15 Furthermore, those with lipohypertrophy are more frequently overweight or obese compared to those without this condition.10,15 The potential clinical consequences of lipohypertrophy in this group include a seven times higher odds of unexplained hypoglycemia (pOR = 6.98 [95% CI: 3.30-14.77]), higher Hemoglobin A1c (glycated hemoglobin) levels (mean difference (MD) = 0.55% [95% CI: 0.23-0.87]), and increased insulin requirements (total daily dose: 7.68 IU [5.31-10.06] and = 0.06 IU/kg bodyweight [0.01-0.12]). 16 It is not well understood whether similar risk factors and clinical consequences exist in those people with diabetes using insulin pumps for continuous subcutaneous insulin infusion (CSII) therapy. Given the different methods of insulin delivery, there may be distinct risk factors and clinical consequences associated with lipohypertrophy in those administering insulin by injections versus those using insulin pumps. Specifically, CSII infuses insulin continuously at the very same skin site over several days, which might affect the reaction of the subcutaneous adipose tissue differently compared to insulin injections. Additionally, there may also be differences between “classical” tubed insulin pumps and the patch pumps. Such insulin pumps are also used in systems for automated insulin delivery (AID). 17 Identifying such differences in risk factors is important for optimizing personalized insulin therapy management and might be of help to improve clinical outcomes. Infusion of insulin in skin areas with lipohypertrophy might hamper the success of this treatment option. 18 This knowledge gap is particularly concerning given that recent meta-analyses have indicated a high prevalence of lipohypertrophy in people on CSII, with an estimated frequency of 36% [95% CI: 19.3-52.6%]. 5 Despite the high prevalence, the identification of specific risk factors and the impact of lipohypertrophy on treatment outcomes in this group are underexplored.
Therefore, our study aimed to identify and summarize the published data in people using CSII to determine, by conducting a systematic review and meta-analysis, the risk factors and clinical consequences of lipohypertrophy in this user group. This research aims to identify current gaps in knowledge and support the development of better clinical practices to manage and prevent lipohypertrophy in the growing population of people living with diabetes who use CSII.
Methods
Selection Criteria and Search Strategy
This systematic review followed the PRISMA (Preferred Reporting Items for Systematic Review and Meta-Analyses) 2020 guidelines. 19 The protocol of the systematic review was registered in the PROSPERO (The International Prospective Register of Systematic Reviews) database (CRD42024554127). The systematic literature search was conducted on March 19, 2024 in MEDLINE (via PubMed), Embase, and CENTRAL (via The Cochrane Library) databases using keywords and queries related to diabetes and lipohypertrophy as shown in the Supplemental Appendix (Supplemental Tables 1-3). A hand-search of the references of the included studies was also performed to retrieve any other relevant publications not indexed in searched medical databases.
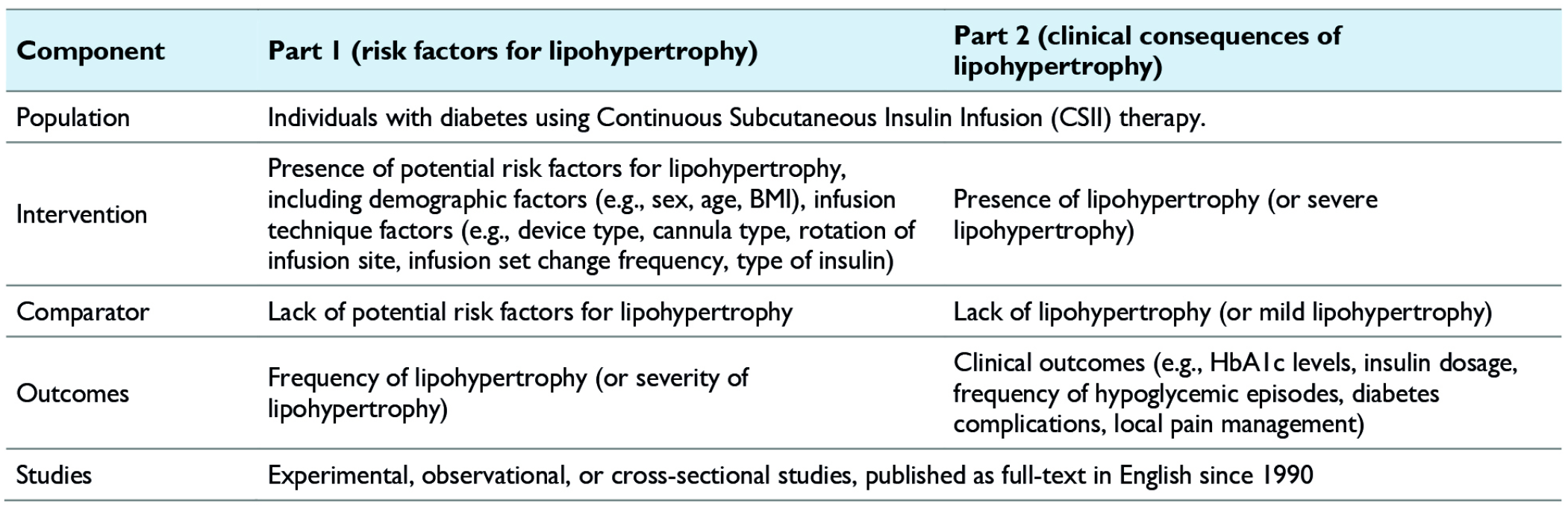
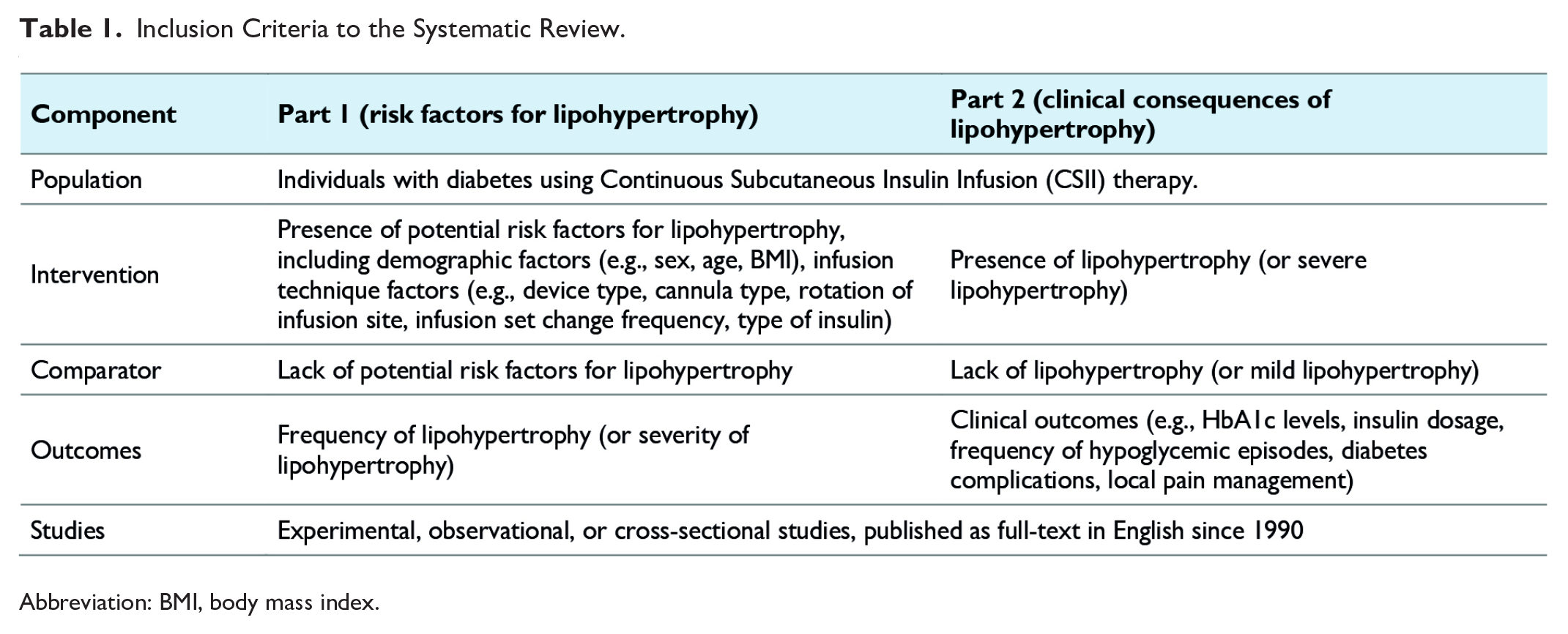
In the first part of the systematic review, we searched for studies reporting any potential risk factors for lipohypertrophy in people with diabetes treated with CSII (Table 1). Studies of primary interest compared the frequency of lipohypertrophy in groups with and without potential risk factors related to individual characteristics (eg, sex, age, body mass index (BMI), duration of CSII and diabetes) and infusion technique (eg, device type, cannula type, rotation of infusion sites, infusion set changes). Studies reporting risk factors as continuous data in groups of patients with and without lipohypertrophy were also considered. In the second part of the systematic review, we looked for studies that describe potential clinical consequences of lipohypertrophy (eg, its impact on glycemic control (eg, HbA1c levels), continuous glucose monitoring (CGM) data, insulin dosage, and frequency of hypoglycemic episodes). Due to the anticipated limited number of available studies, we allowed the inclusion of studies that evaluated the degree of lipohypertrophy severity based on the presence of potential risk factors, as well as possible consequences of lipohypertrophy in groups of people with diabetes with varying levels of its severity.
Inclusion Criteria to the Systematic Review.
Abbreviation: BMI, body mass index.
No restrictions on study design were made, allowing for the inclusion of both experimental, observational, and cross-sectional studies. Exclusion criteria included studies published in languages other than English or only as conference proceedings. Additionally, only research conducted and published after 1990 was included to ensure relevance to current clinical practices, as earlier studies predominantly involved animal-derived insulin and types of insulin pumps/insulin infusion sets, which are no longer the standard in diabetes management.
Data Analysis
The review process was carried out by two independent analysts (AST, MM). Any disagreements during the full-text selection stage were resolved by consensus. The same two reviewers independently extracted data for meta-analysis. Any discrepancies between reviewers were discussed and resolved by consensus. The extracted items included the methodology of the studies, patients’ characteristics with an emphasis on details of CSII performance, risk factors for lipohypertrophy, and their clinical consequences. When available, we also extracted the results of comparisons and P-values reported by the study authors. The quality of studies was assessed using Joanna Briggs Institute (JBI) tools for cross-sectional studies 20 and case series. 21
Calculated results were presented either as pOR for the percentage of patients with lipohypertrophy among risk factor groups or as MD for variables expressed by means and standard deviations. All results were given with 95% CI. Meta-analyses, if conducted, used a fixed-effects model (inverse variance) as between-study heterogeneity for all available comparisons was non-significant (P-value for Cochrane Q test >0.10 and I2 < 50%). The assessment of publication bias, subgroup analyses, and sensitivity analyses were not conducted due to the insufficient number of studies included in the meta-analyses. For all statistical analyses, Sophie Version 1.5.0 software was used (validated with STATA Version 10.0).
Results
Search Results
Of the 4295 records identified during databases and references search, 272 full-text articles were selected for further analysis. Among these, nine studies (in alphabetical order: Barlas et al, 22 Berg et al, 23 Binder et al, 24 Deeb et al, 25 Pickup et al, 26 Rabbone et al, 27 Schober and Rami, 28 Ucieklak et al, 29 van Munster et al 30 ) met the inclusion criteria for this systematic review, and 263 papers were excluded. Detailed reasons for exclusion and the PRISMA flow diagram describing the selection process are given in the Supplemental Appendix (Supplemental Table 4, Supplemental Figure 1).
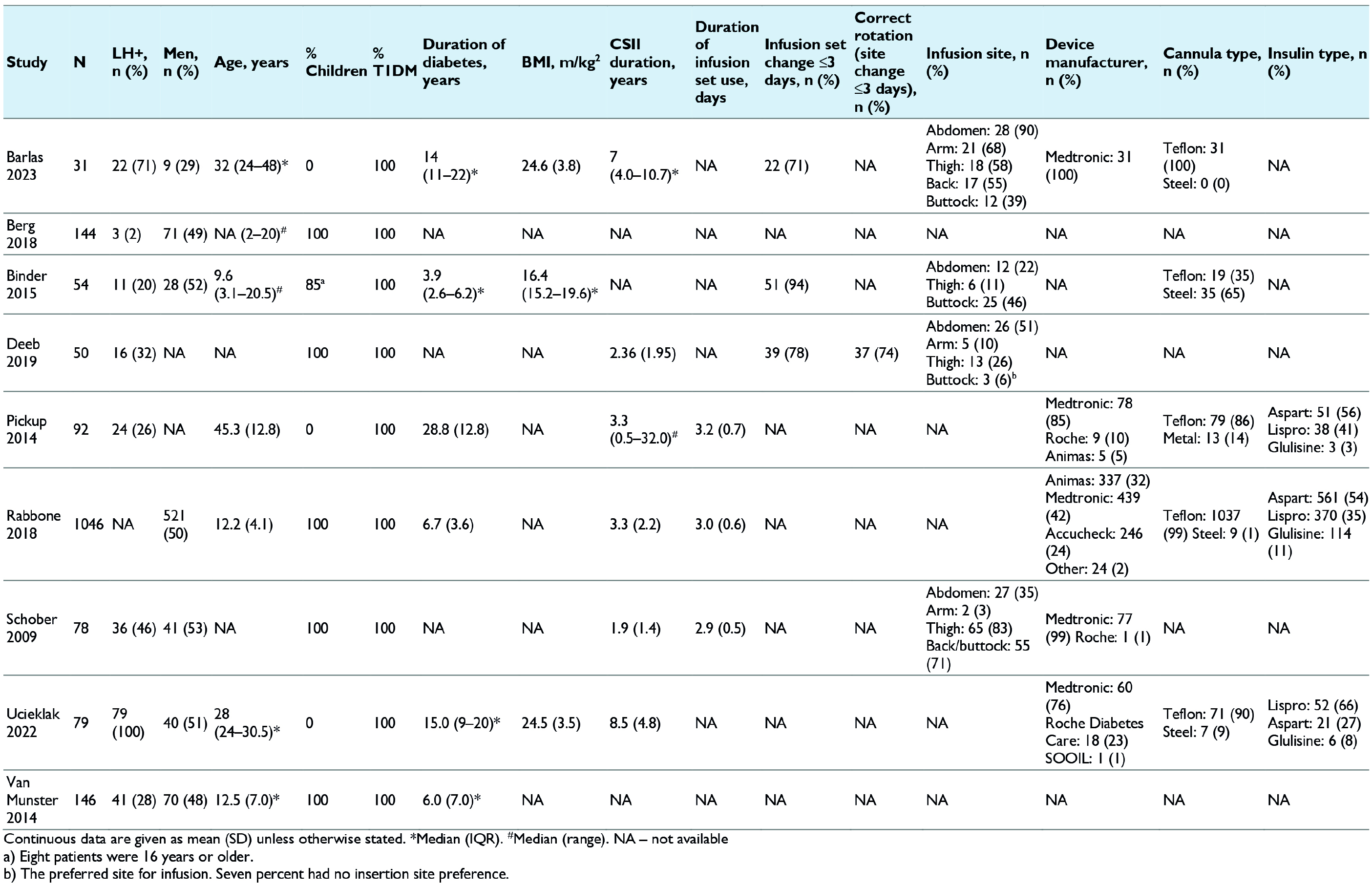
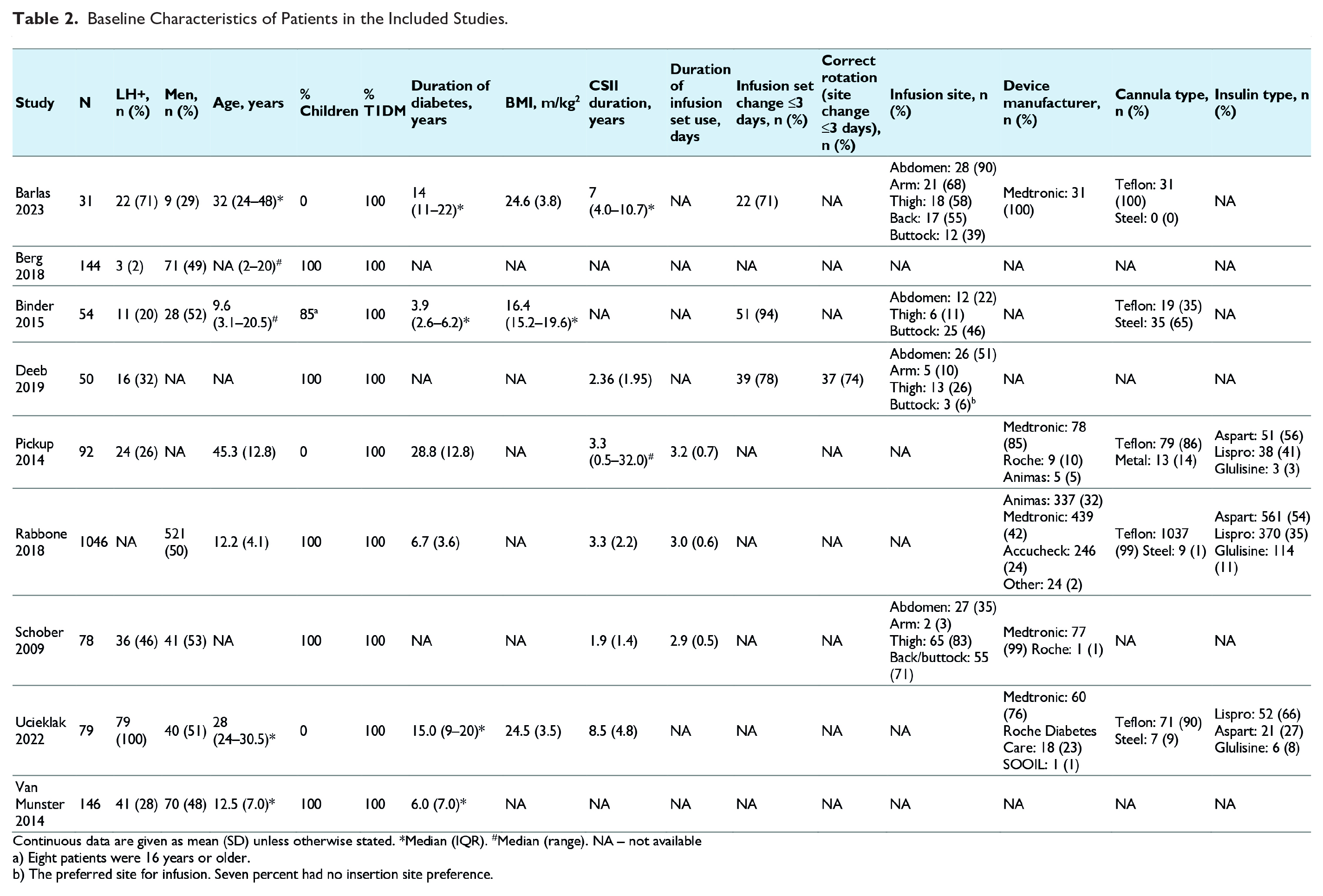
Most of the nine included studies were cross-sectional and single-center. The only prospective, multi-center, observational study was Rabbone et al’s study. 27 Seven studies were conducted in Europe (Denmark, England, Italy, Austria, Poland, and the Netherlands). Sample sizes varied from 31 to 1046 participants. Except for the largest study (Rabbone et al), 27 all others recruited fewer than 150 patients. Six studies included pediatric and/or adolescent populations while the remaining three focused on adults with type 1 diabetes. Detailed patient characteristics were infrequently reported (Table 2).
Baseline Characteristics of Patients in the Included Studies.
The quality of the included studies varied, with scores ranging from 3/8 to 8/8 on the JBI scale for cross-sectional studies. However, in most of the studies, the assessment of potential risk factors for lipohypertrophy and its consequences was not the primary focus of the research. Nonetheless, no study was excluded from this review due to methodological issues. Additional information on the methodology of studies and their quality assessment is available in the Supplemental Appendix (Supplemental Tables 5-8).
Risk Factors for Lipohypertrophy
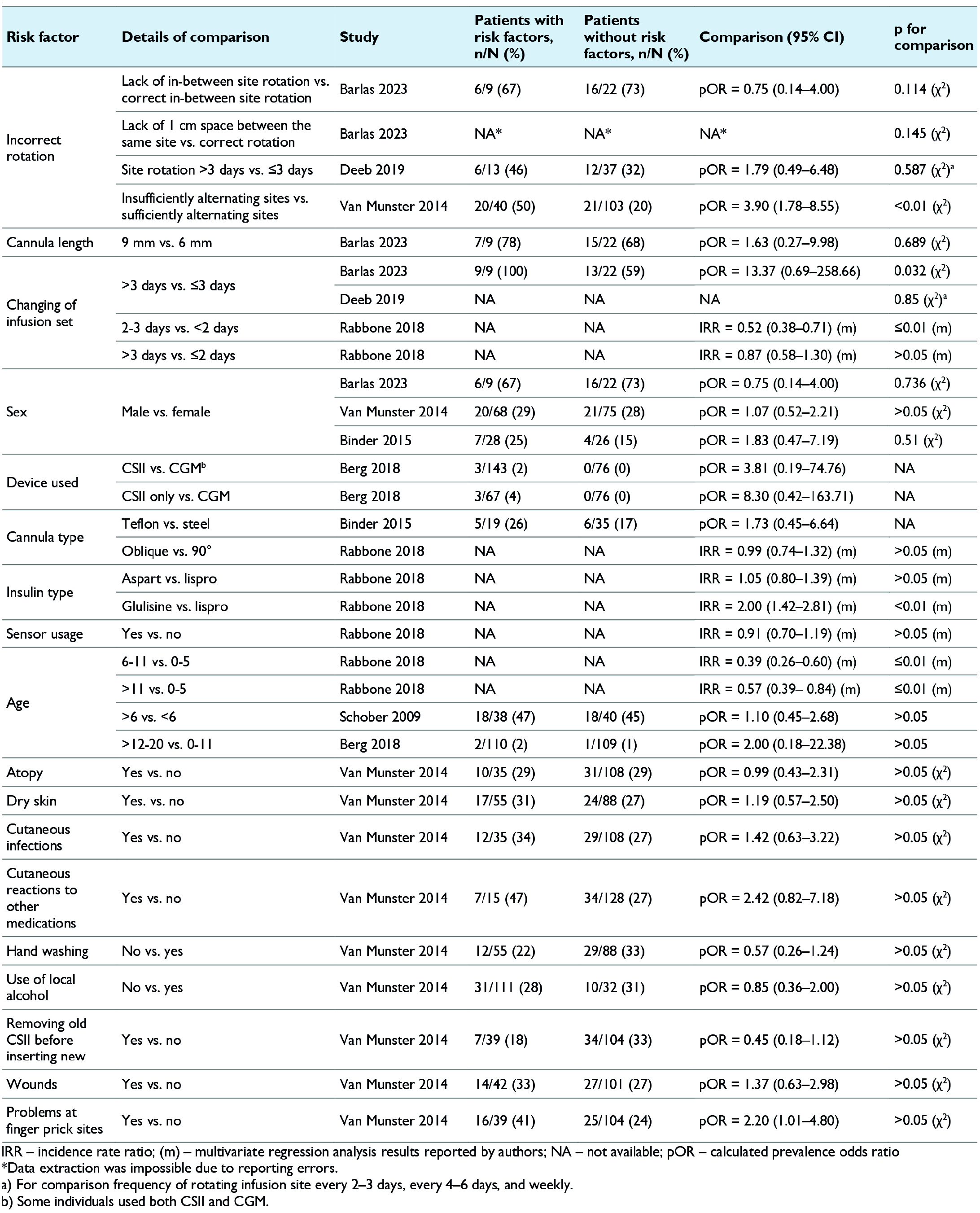
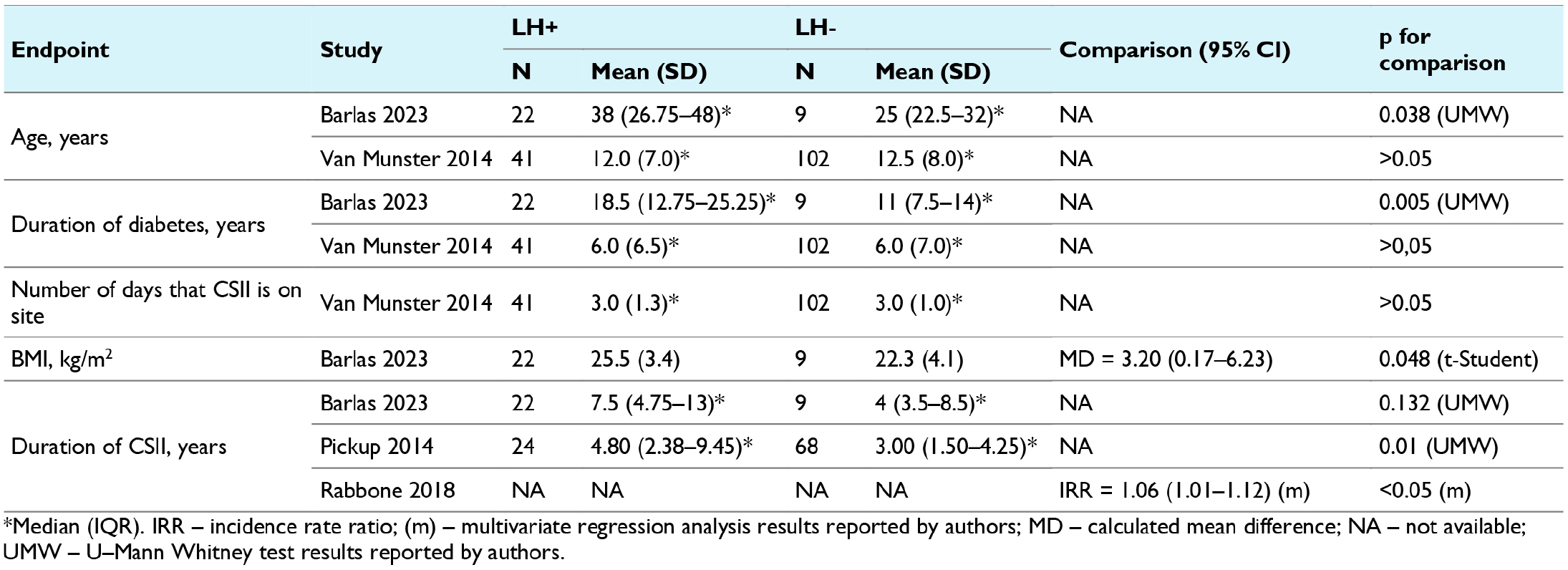
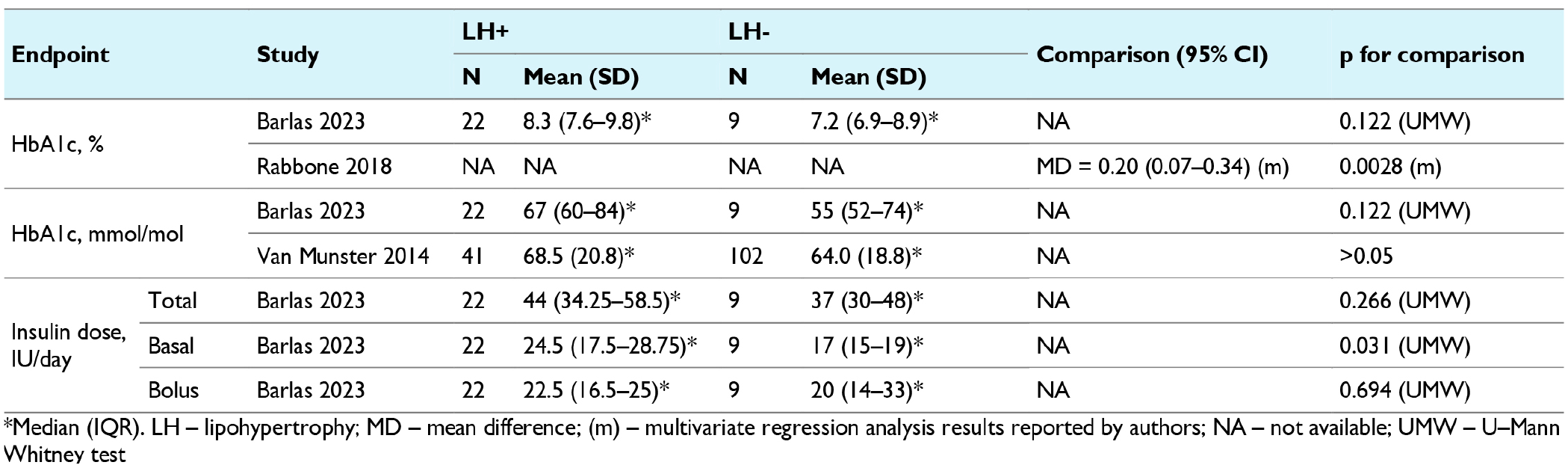
Regarding demographic risk factors, three studies included (Barlas et al, 22 Binder et al, 24 van Munster et al 30 ) found no significant differences in the frequency of lipohypertrophy between genders (Table 3) with a meta-analysis result showing pOR = 1.13 [95% CI: 0.62-2.06] for male and female comparison (Figure 1). In the Schober and Rami 28 and Berg et al’s 23 studies, lipohypertrophy occurred at similar rates across the analyzed age groups. However, Rabbone et al 27 reported a significantly lower incidence rate ratio (IRR) for lipohypertrophy in older age groups compared to younger ones (6-11 years and >11 vs 0-5 years). The median age of people with lipohypertrophy was however higher than those without this condition (38 vs 25 years, P = .038) in the van Munster et al’s 30 study. Barlas et al 22 noted a higher BMI in individuals with lipohypertrophy (MD = 3.20 kg/m2 [95% CI: 0.17-6.23]; Table 4). The median duration of diabetes therapy in people with lipohypertrophy was significantly longer compared to individuals without lipohypertrophy in the Barlas 2023 study (18.5 vs 11.0 years, P = .005), while van Munster et al 30 noted comparable medians in both analyzed groups (Table 4).
Risk Factors for Lipohypertrophy—Dichotomous Data.
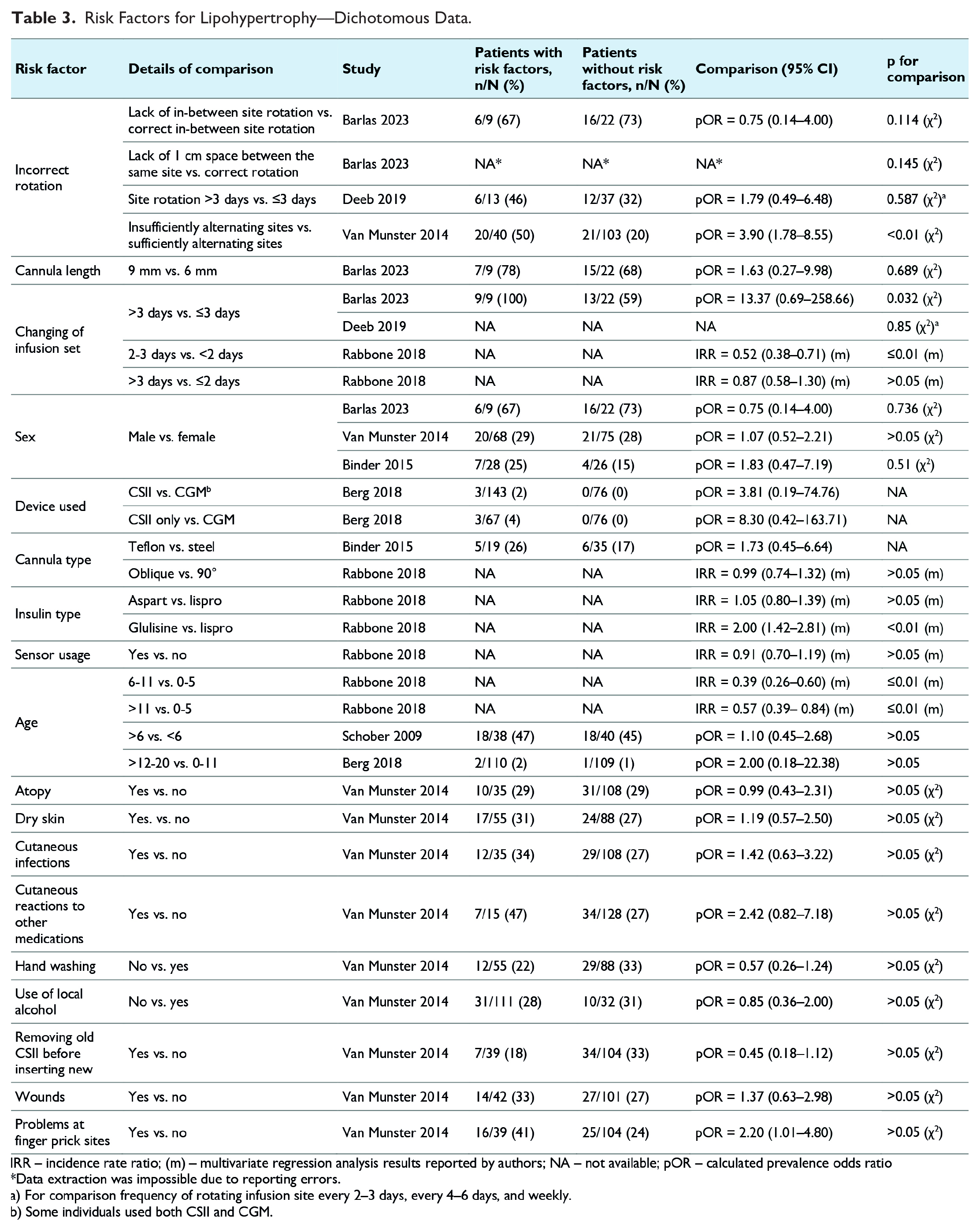
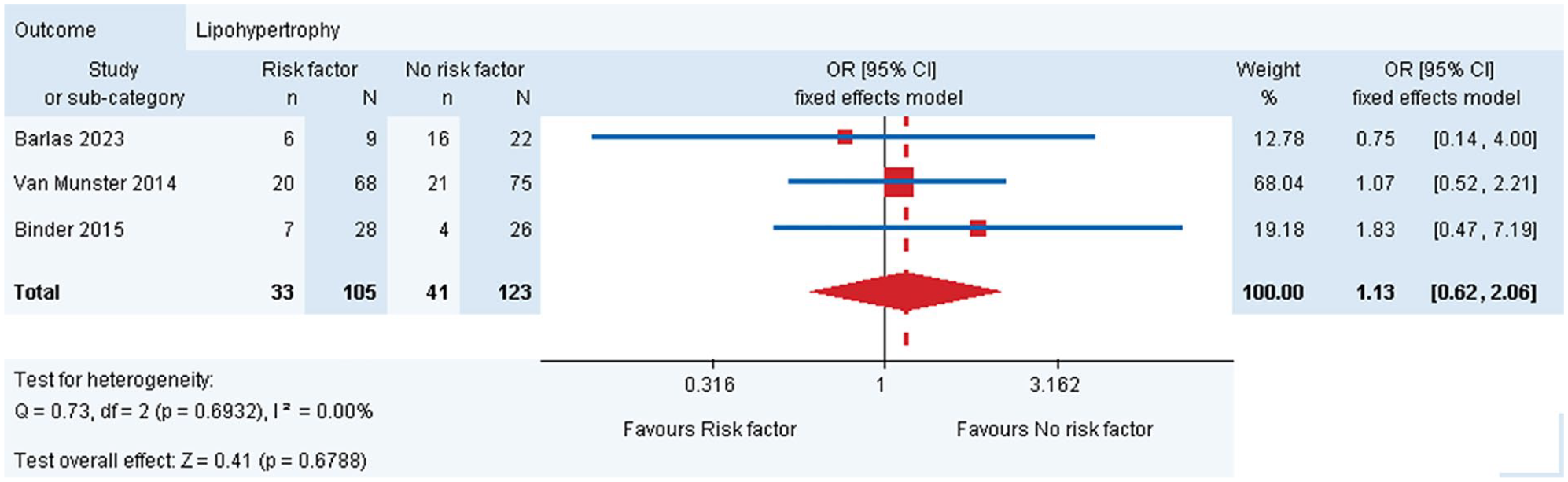
Risk factors of lipohypertrophy—male sex.
Risk Factors for Lipohypertrophy—Continuous Data.
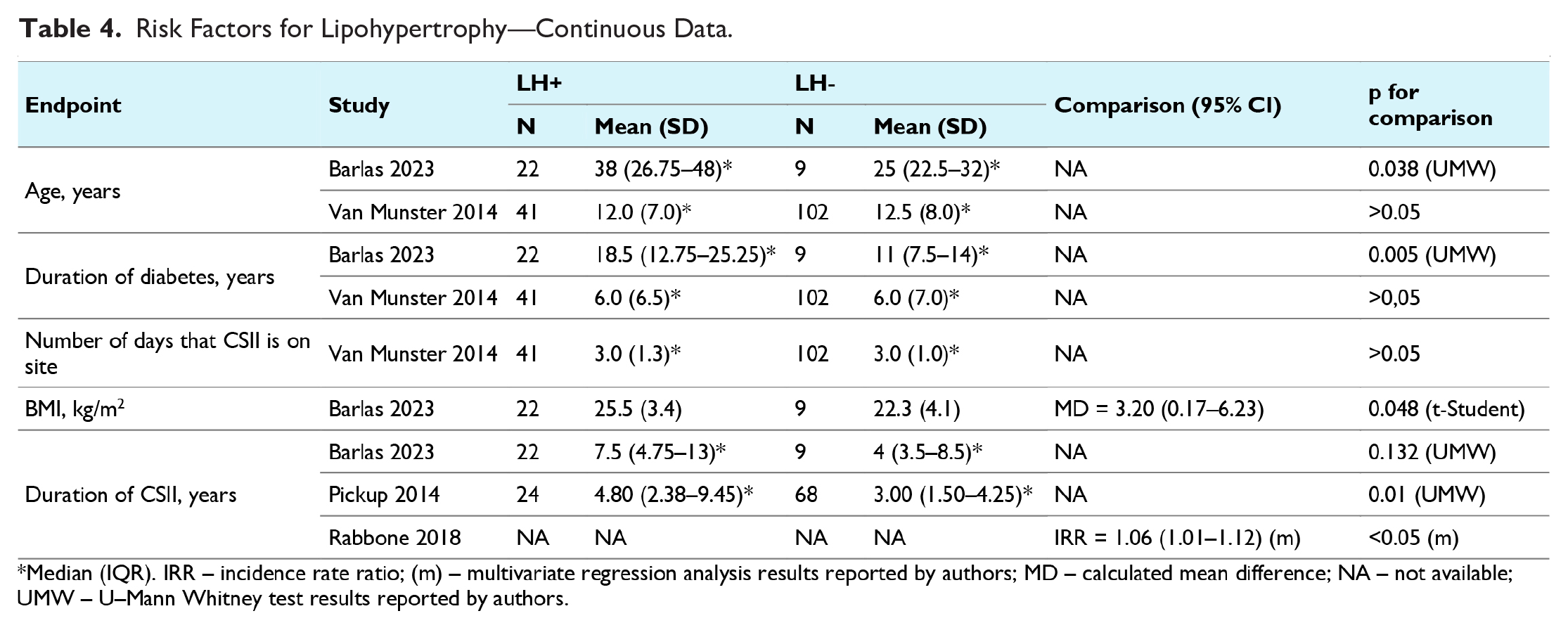
Three studies (Barlas et al, 22 Deeb et al, 25 van Munster et al 30 ) reported results on the impact of infusion site rotation on the frequency of lipohypertrophy. A meta-analysis (Figure 2) revealed that lipohypertrophy occurs significantly more often in those people with diabetes who do not rotate their infusion sites properly compared to those who do (pOR =2.59 [95% CI: 1.39-4.83]). In the study conducted by Rabbone et al, 27 it was also noted that the IRR was significantly lower in CSII users who changed their infusion set daily compared to those who changed it every two to three days ([95% CI: 0.38-0.71], P < .01; Table 3). Interestingly, comparisons allowing the use of infusion sets for up to three days versus more than three days did not show a significant impact on the prevalence of lipohypertrophy, or the results were on the borderline of statistical significance (Table 3). A higher IRR for lipohypertrophy was also observed by Rabbone et al 27 with the use of glulisine insulin compared to lispro insulin (IRR = 2.00 [95% CI: 1.42; 2.81]). The length and type of the infusion set cannula, concurrent use of CGM systems, hand washing, use of alcohol for disinfection at the injection site, and removal of the old infusion set before inserting a new one did not significantly impact the frequency of lipohypertrophy in the studies reviewed. The presence of other skin conditions (eg, atopy, dry skin, infections, wounds) also did not increase the frequency of lipohypertrophy. However, those people with diabetes with lipohypertrophy had a longer duration of CSII usage compared to those without this condition (Table 4).
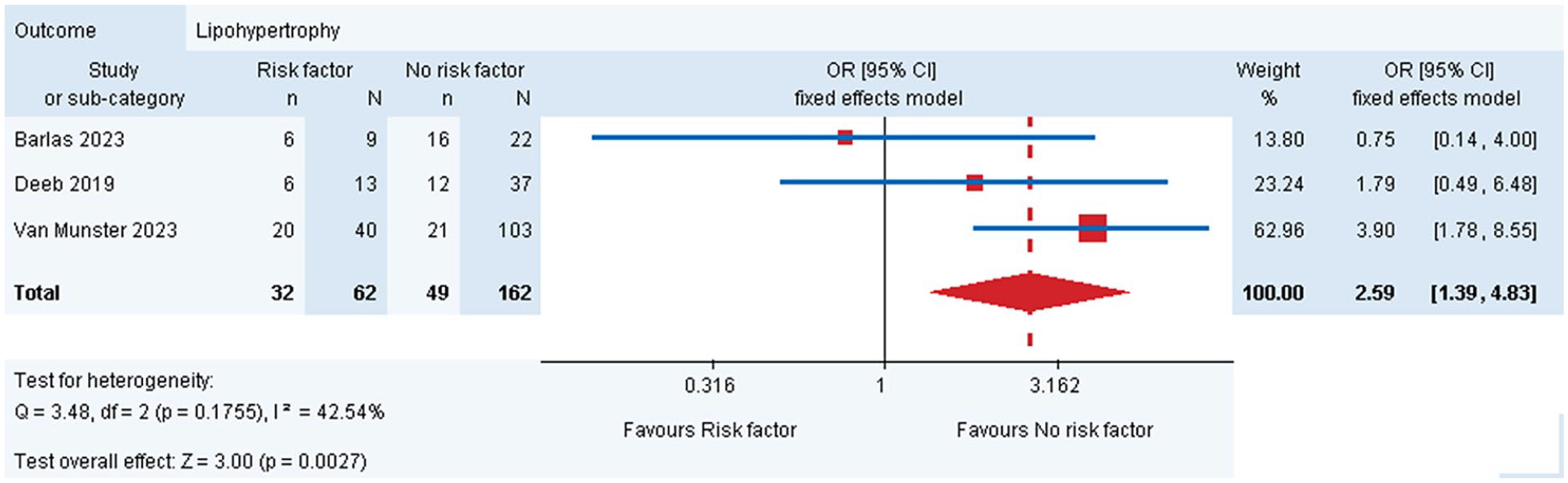
Risk factors of lipohypertrophy—incorrect infusion site rotation.
In the study that assessed the impact of risk factors on the severity of lipohypertrophy in people with diabetes (Ucieklak et al 29 ), it was observed that those who use an infusion set with a cannula length ≥9 mm had more than three times higher odds of experiencing advanced lipohypertrophy compared to those using shorter cannulas. Regarding other risk factors (eg, gender, hypothyroidism, changing of infusion sets), no significant impact on the severity of lipohypertrophy was noted (results presented in the Supplemental Appendix; Supplemental Tables 9 and 10).
Clinical Consequences of Lipohypertrophy
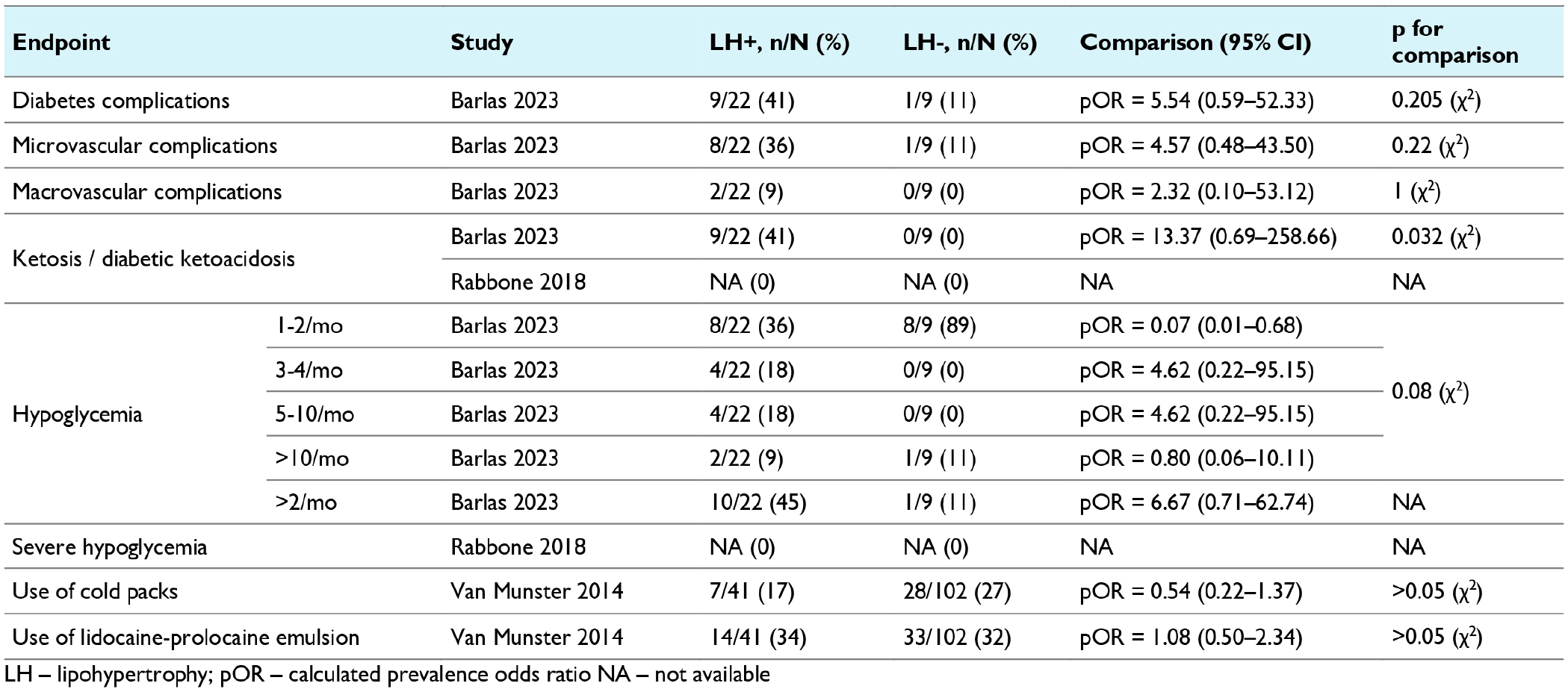
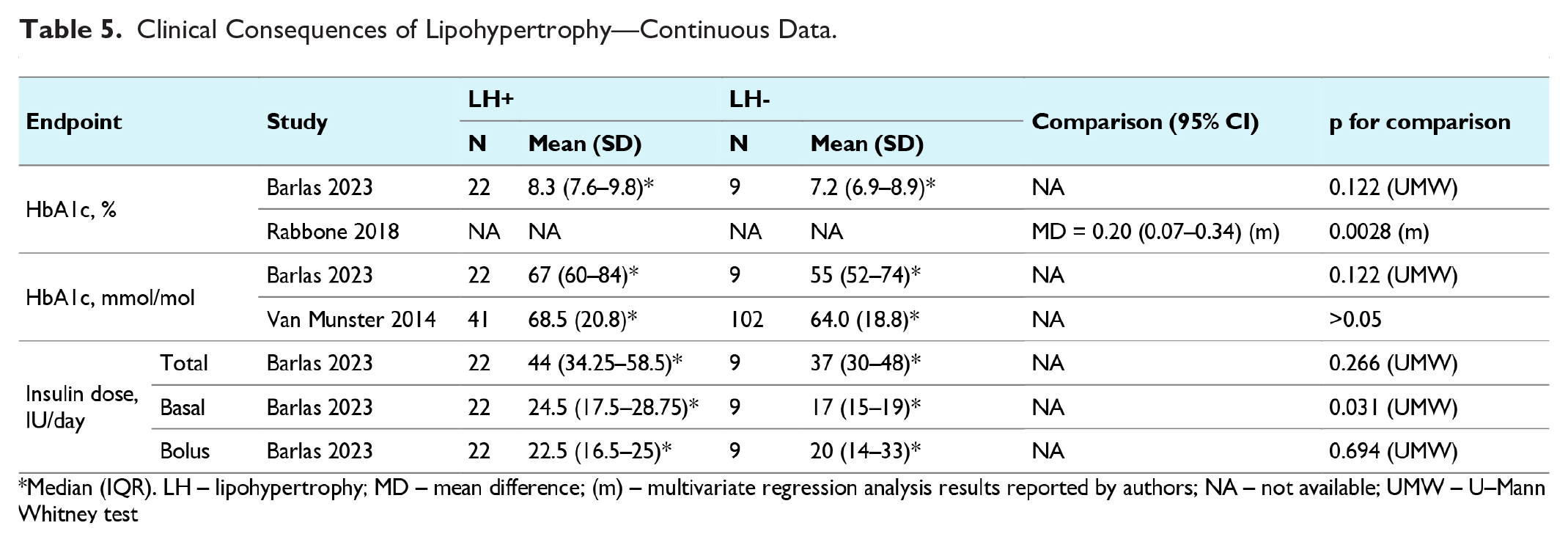
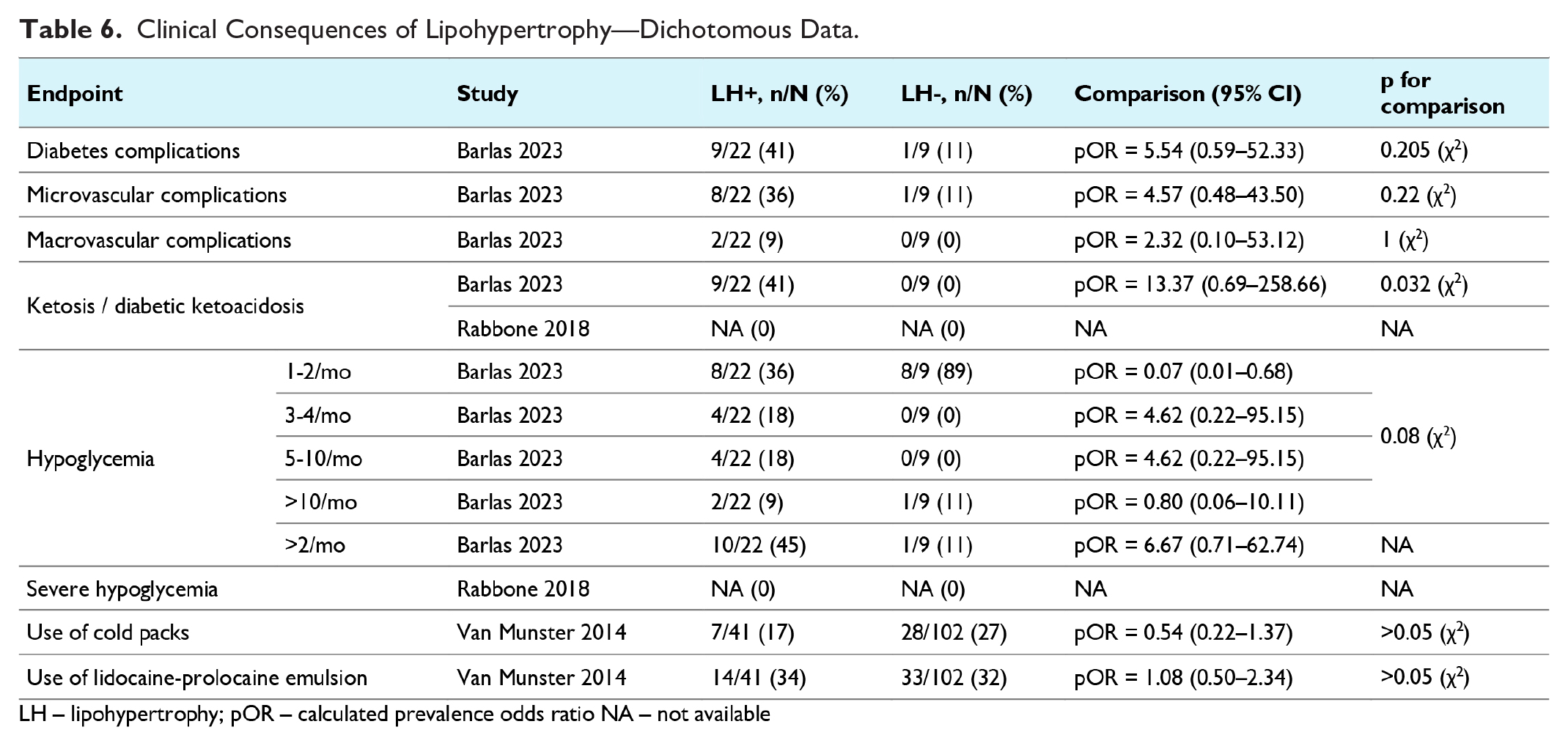
Only three studies reported data on the potential clinical consequences of lipohypertrophy in CSII users (Barlas et al, 22 Rabbone et al, 27 van Munster et al 30 ). In one study (Rabbone et al 27 ), it was noted that the HbA1c levels were higher in people with lipohypertrophy compared to those without (MD = 0.20% [95%: 0.07-0.34]). However, in the other studies,22,30 this difference was not statistically significant (Table 5). No significant differences were also found regarding the frequency of diabetes complications, hypoglycemic episodes, or the need for local pain management such as cold packs or lidocaine-prilocaine emulsion (Table 6). However, the median basal insulin dose was higher in those with lipohypertrophy in comparison to those without this skin condition in the Rabbone et al’s 27 study (24.5 vs 17 IU, P = .031; Table 5).
Clinical Consequences of Lipohypertrophy—Continuous Data.
Clinical Consequences of Lipohypertrophy—Dichotomous Data.
In the study where data on the potential consequences of lipohypertrophy were available based on its severity (Ucieklak et al 29 ), no differences were found in HbA1c levels between those with more severe versus less severe lipohypertrophy. However, it was noted that high doses of insulin, that is, >0.7 IU/kg, were more frequently used in those with more severe lipohypertrophy (data available in the Supplemental Appendix; Supplemental Tables 11 and 12).
Discussion
To our knowledge, this systematic review is the first that aimed at exploring and elucidating the risk factors and clinical consequences of lipohypertrophy in patients using CSII. Previous research, for example, conducted by Wang et al 5 has only estimated the prevalence of lipohypertrophy among insulin pump users without examining its risk factors and outcomes in this group of patients.
Our systematic review identified nine studies that described risk factors and/or potential clinical consequences of lipohypertrophy in CSII users. In contrast, more than 50 studies15,16 have been conducted on people with diabetes using syringes or pens for insulin administration, with new publications emerging annually. Given that a significant proportion of the people with type 1 diabetes uses insulin pumps (also as a component of AID systems), and the increasing number of people with type 2 diabetes using CSII, 31 the scarcity of high-quality data for insulin infusion provides the impetus to conduct additional studies. It is estimated that about 40% to 80% of children and adolescents with type 1 diabetes are using insulin pumps, and their popularity among people with insulin-treated diabetes has been increasing over time.31,32
In other words, the major outcome of our literature search and meta-analysis is the limited number of identified studies, which highlights a significant gap in the existing literature.
The studies included in this systematic review were not primarily designed to assess the risk factors and consequences of lipohypertrophy in this user population. Most studies involved relatively small sample sizes from single centers (up to 150 participants) and varied in research quality. Population characteristics were often insufficiently detailed in the publications to perform a thorough assessment of homogeneity between studies, and the risk factors and clinical outcomes were usually limited to single variables, which were inconsistent across studies. As a result, available data did not allow conduction of comprehensive quantitative synthesis in most cases. Another issue noted was the method of assessing skin changes in the included studies. In two studies (Rabbone et al, 27 Schober and Rami 28 ), the assessment method was not specified, while in three others (Berg et al, 23 Binder et al, 24 Pickup et al 26 ), lipohypertrophy was self-reported by patients or caregivers based on photographs, without additional ultrasonographic or palpation examination. This raises doubts about the accuracy of lipohypertrophy diagnoses in the included studies.
Regarding the results of our meta-analysis, most analyzed risk factors and clinical consequences did not show a statistically significant association with lipohypertrophy, possibly due to the small sample sizes of the studies included in this systematic review. Continuous subcutaneous insulin infusion research often involved fewer than 150 individuals, with meta-analyses including a maximum of just over 200 patients. In contrast, a recent meta-analyses for people with diabetes using insulin injections involved over 14,000 individuals for the impact of needle reuse and 11,000 individuals for the impact of lack of rotation on lipohypertrophy occurrence. 15 Despite the small sample sizes, our meta-analysis suggests that lack of infusion site rotation is a significant factor in the development of lipohypertrophy not only in insulin-injecting people, but also in those using CSII (pOR = 2.59 [95% CI: 1.39-4.83]. This result can be attributed to the prolonged exposure of subcutaneous tissue to insulin at a single site, which promotes localized tissue changes and hypertrophy. 2 Additionally, repeated mechanical trauma from inserting the infusion set in the same location may exacerbate these effects, creating an environment conducive to the development of lipohypertrophy. 2 These findings highlight the importance of regular rotation of infusion sites in preventing this complication.
Other risk factors related to infusion technique were generally not significantly different in the identified studies, but the low numerical values suggest the need for further research into factors such as the size and type of the infusion set cannula used, frequency of infusion set changes, type of insulin used, and duration of insulin therapy. Regarding the potential consequences of lipohypertrophy for insulin pump usage, further studies are particularly needed to conclude its impact on insulin levels, hypoglycemic and hyperglycemic episodes, and insulin requirements.
The main “limitation” of our systematic review is the small number and variable quality of the identified clinical studies as well as small sample sizes, which prevent drawing definitive conclusions about the risk factors and consequences of lipohypertrophy in people with diabetes on CSII. Nevertheless, from the perspectives of researchers, diabetologists, and practicing physicians, this review is relevant as it identifies current knowledge gaps in the care of people with diabetes who are using this treatment option. By highlighting this gap, this review underscores the need for more robust and well-designed studies to better understand the prevalence, risk factors, and clinical consequences of lipohypertrophy in people with diabetes who are using insulin pumps or AID systems.
To address these gaps effectively, an international collaboration among diabetologists and/or insulin pump manufacturers could be initiated to gather data through a multi-center, multinational patient registry for people with diabetes using insulin pumps or AID systems, similar to the World Injection Technique Study.33,34 The research would collect detailed and consistent data across various user populations, allowing for clearer conclusions and the development of new, evidence-based guidelines to optimize insulin pump management. A key priority for this collaboration would be to establish a standardized, universally accepted definition of lipohypertrophy, as well as consistent protocols for its measurement and reporting. The lack of such a definition has led to variability in studies, making it challenging to compare results or draw robust conclusions. A defined framework would ensure uniformity in future research, enabling clinicians and researchers to address this issue more effectively. Further research on lipohypertrophy could also incorporate the use of optical coherence tomography to enhance our understanding of the condition and its clinical implications in the context of using pumps for insulin therapy. 35
By addressing these foundational gaps, an international consortium would create a robust infrastructure for investigating lipohypertrophy, facilitating high-quality, comparable data collection. Only through this systematic approach will clinicians and researchers be able to fully understand the condition, optimize insulin therapy, and improve outcomes for patients worldwide. This collaborative effort would mark a significant step forward in managing lipohypertrophy and enhancing the care of people with diabetes.
Conclusions
In conclusion, while this systematic literature review highlights the limited and variable quality of existing research on lipohypertrophy in people with diabetes using insulin pumps or AID systems, it also underscores the importance of this issue. Further research is needed to better understand the risk factors and clinical consequences of lipohypertrophy in people with diabetes who are using insulin pumps, which could lead to improved management and outcomes. The findings from this review should serve as a call to action for researchers and clinicians to conduct more comprehensive and high-quality studies on this topic.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251319806 – Supplemental material for Existing Knowledge Gaps in Risk Factors and Consequences of Lipohypertrophy in People Using Continuous Subcutaneous Insulin Infusion Therapy: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-1-dst-10.1177_19322968251319806 for Existing Knowledge Gaps in Risk Factors and Consequences of Lipohypertrophy in People Using Continuous Subcutaneous Insulin Infusion Therapy: A Systematic Review and Meta-analysis by Julia K. Mader, Riccardo Fornengo, Ahmed Hassoun, Lutz Heinemann, Bernhard Kulzer, Magdalena Monica, Trung Nguyen, Jochen Sieber, Eric Renard, Yves Reznik, Przemysław Ryś, Anita Stożek-Tutro and Emma G. Wilmot in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-2-dst-10.1177_19322968251319806 – Supplemental material for Existing Knowledge Gaps in Risk Factors and Consequences of Lipohypertrophy in People Using Continuous Subcutaneous Insulin Infusion Therapy: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-2-dst-10.1177_19322968251319806 for Existing Knowledge Gaps in Risk Factors and Consequences of Lipohypertrophy in People Using Continuous Subcutaneous Insulin Infusion Therapy: A Systematic Review and Meta-analysis by Julia K. Mader, Riccardo Fornengo, Ahmed Hassoun, Lutz Heinemann, Bernhard Kulzer, Magdalena Monica, Trung Nguyen, Jochen Sieber, Eric Renard, Yves Reznik, Przemysław Ryś, Anita Stożek-Tutro and Emma G. Wilmot in Journal of Diabetes Science and Technology
Footnotes
Correction (April 2025):
This article has been updated to include the confidence interval (CI) information in both the Results section of the main text and the abstract.
Abbreviations
CGM, continuous glucose monitoring; HbA1c, glycated hemoglobin; n/N - n/N – the number of patients with the event (n) / of the total number of patients analyzed (N). AID, automated insulin delivery; CI, confidence interval; CSII, continuous subcutaneous insulin infusion; IQR, interquartile range; JBI, Joanna Briggs Institute; LH+, patients with lipohypertrophy; LH−, patients without lipohypertrophy; MD, mean difference; T1DM, type 1 diabetes mellitus; pOR, prevalence odds ratio; SD, standard deviation.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JKM is a member of the Advisory Board of Abbott Diabetes Care, Becton, Dickinson and Company (BD), Boehringer Ingelheim, Eli Lilly, embecta, Medtronic, Novo Nordisk A/S, Pharmasens, Roche Diabetes Care, Sanofi-Aventis, Viatris and received speaker honoraria from A. Menarini Diagnostics, Abbott Diabetes Care, BD, embecta, Boehringer Ingelheim, Dexcom, Eli Lilly, Medtrust, MSD, Novo Nordisk A/S, Roche Diabetes Care, Sanofi, Servier, and Ypsomed. BK is a member of the advisory board of embecta. ER reports personal fees as a consultant/advisor and/or speaker honoraria for Abbott, AstraZeneca, Boehringer Ingelheim, Dexcom, Eli Lilly, Embecta, Insulet, Lifescan, Novo Nordisk, Roche Diagnostics, Sanofi-Aventis, and Tandem, and research grant/material support from Dexcom, Insulet, and Tandem. JS was an employee of embecta as this research was conducted. He is a stockholder of embecta and BD. TN is an employee of embecta. LH is a consultant for several companies developing novel diagnostic and therapeutic options for diabetes treatment. He is a shareholder of the Profil Institut für Stoffwechselforschung GmbH, Neuss, Germany. YR declares consultant/speaker fees from Medtronic, Insulet, embecta, Abbott, Novo Nordisk, Eli Lilly, Sanofi, and Air Liquide Santé International. AH is a member of the advisory board of embecta. AST, MM, PR, and RF declare no conflict of interest. EGW has been a member of the advisory board of Abbott Diabetes Care, Eli Lilly, embecta, Insulet, Medtronic, Novo Nordisk, Roche Diabetes Care, Sanofi-Aventis, and Sinocare; has received research support from Abbott Diabetes Care, embecta, Insulet, Novo Nordisk, and Sanofi-Aventis; and has received speaker honoraria from Abbott Diabetes Care, AstraZeneca, Dexcom, Eli Lilly, embecta, Glooko, Insulet, Medtronic, Novo Nordisk, Sanofi, and Ypsomed.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was sponsored by embecta. The sponsor had a role in the study design, interpreting data, writing the report, and in the decision to submit the paper for publication.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.