Abstract
Background:
Type 2 diabetes (T2D) is a phenotypically heterogeneous disease. The use of insulin is required in a significant portion of people with T2D, despite recent developments in antidiabetic medications. This study analyzes glycemic outcomes in automated insulin delivery (AID) users with T2D with different insulin requirements.
Methods:
This is a retrospective, real-world analysis including MiniMed 780G (MM780G) data uploaded to CareLink Personal (January 2020 to April 2024). Four cohorts were identified based on phenotypes of T2D: (A) users with total daily dose of insulin (TDD) ≥ 100 IU, (B) users with self-reported T2D, (C) users with self-reported T2D and TDD ≥ 100 IU, and (D) users with self-reported T2D and TDD <100 IU. Glycemic outcomes and insulin use were assessed post-AID, pre-AID versus post-AID, and six-month longitudinal post-AID.
Results:
A total of 26 427 users were included in this study, of which 18 466 in cohort A, 10 795 in cohort B, 2 834 in cohort C, and 7 961 in cohort D. Mean time in range (TIR) was 71.1% ± 12.2 for cohort A, 75.1% ±14.1 for cohort B, 72.2% ± 15.0 for cohort C, and 76.1% ± 13.6 for cohort D. Mean time below range (TBR) <70 mg/dL was ≤1% in all cohorts. The users in cohort C using the recommended optimal settings (glucose target [GT] of 100 mg/dL and active insulin time [AIT] of two hours) had a greater TIR with 78.7% ± 10.8. All cohorts increased ≥10% post-AID compared with pre-AID.
Conclusions:
The use of this AID is associated with effective therapy outcomes, as indicated by over 70% TIR, and appears to be safe, as demonstrated by a low TBR in a large cohort of real-life users with self-reported T2D and high or low TDD.
Introduction
Type 2 diabetes mellitus (T2D) is a phenotypically heterogeneous chronic disease. 1 One way to characterize T2D is according to the varying therapeutic pathways. These pathways are currently determined by clinical features such as degree of hyperglycemia, body weight, and the presence of cardiorenal complications, including chronic kidney disease, vascular atherosclerotic disease, and congestive heart failure. Although there is an increase of utilization of newer classes of medications, mainly glucagon-like peptide-1 receptor agonist (GLP-1 RA) and sodium-glucose cotransporter 2 (SGLT2) inhibitors, it is associated with delayed initiation of insulin therapy, 2 . Eventually, insulin is still required in 18.9% of those with T2D in Europe (ie, ~6.6 million), 3 of these 30% (ie, ~2.3 million) are on multiple daily insulin injections. 4 It is also apparent that most of those using insulin do not reach the current international consensus treatment goal of HbA1c <7%.5,6
Automated insulin delivery (AID) systems have demonstrated significant success in achieving good glycemic outcomes in individuals with type 1 diabetes (T1D). Studies on the MiniMed 780G (MM780G) system have shown consistency in results among patients of various ages7,8 and geographies7,9-11 and adaptability to diverse clinical circumstances such as lifestyle changes 11 and pregnancy.12,13 People with T2D requiring insulin therapy present heterogeneous pathophysiological phenotypes spanning from beta-cell failure driving insulin deficiency to insulin resistance with consequently hyperinsulinemia.1,14,15 Therefore, it is essential to examine the glycemic outcomes observed in different AID users with T2D requiring different doses of daily insulin in a real-world setting.
Methods
This is a retrospective, global, real-world analysis including MM780G data uploaded to CareLink Personal from January 2020 to April 2024 by individuals who provided consent and resided in countries where local data privacy regulation permitted the use of anonymized data in aggregated format. Data were extracted according to a validated methodology described by Van den Heuvel et al, which has been utilized in other CareLink Personal real-world analyses on the performance and safety of MiniMed systems. 16
The cohorts were defined by self-reported T2D and by total daily dose of insulin (TDD): (A) users with a TDD of ≥100 IU, (B) users who self-reported to have T2D regardless of TDD, (C) users who self-reported T2D and have a TDD of ≥100 IU, and (D) users who self-reported T2D and have a TDD of <100 IU. Outcomes were assessed in three analyses, in line with previous publications. 7 In analysis 1, outcomes were assessed after AID was initiated for the first time (post-AID analysis). In analysis 2, the outcomes were compared before and after initiation of AID (pre- vs post-AID analysis). In analysis 3, the sustainability of glycemic outcomes was evaluated for each month over a six-month period (longitudinal analysis). Cohort C was selected for this analysis as the users in this cohort are regarded as those most likely living with T2D and insulin resistance.
To be included in the analyses, users needed to have a minimum of 10 days of sensor glucose (SG) data in each period evaluated; for analysis 3, this referred to each month following AID initiation. This requirement (ie, ≥10 days of SG data) was based on a previous publication validating a similar duration of continuous glucose monitoring (CGM) metrics to determine long-term glycemic control and has been used consistently in real-world analyses from CareLink Personal. 6 To avoid selection bias, all data available post-AID initiation was included, regardless of whether the system was in AID or in open loop (eg, following an AID exit triggered by either the system or the user).
Apart from the three main analyses, a prespecified subanalysis was performed based on the use of system settings. The percentage of time that a glucose target (GT) of 100 mg/dL (5.5 mmol/L), 110 mg/dL (6.1 mmol/L), or 120 mg/dL (6.7 mmol/L) was used as well as the percentage of time an active insulin time (AIT) of two, two to three, three to four, or four or more hours was set, was calculated per user. Glycemic outcomes were then analyzed for those users using the recommended optimal settings (ROS; that is, both the GT = 100 mg/dL and AIT of two hours for at least 95% of the time). Of note, the latter analysis was performed for cohort C only in accordance with the rationale previously outlined.
Endpoints
Demographic data, including age and gender, are reported. Glycemic outcomes, including the mean percentage of time in range 70 to 180 mg/dL (3.9-10.0 mmol/L) (TIR), time in tight range 70 to 140 mg/dL (3.9-7.0 mmol/L) (TITR), hypoglycemia level 1: time below 70 mg/dL (<3.9 mmol/L) (TB54), hypoglycemia level 2: time below 54 mg/dL (<3.0 mmol/L) (TB70), hyperglycemia level 1: time above 180 mg/dL (>10.0 mmol/L) (TA180), and hyperglycemia level 2: time above 250 mg/dL (>13.9 mmol/L) (TA250), were calculated. The mean SG, standard deviations hereof (SD), and glucose management indicator (GMI) were assessed, as well as the time the system was used, the percentage of time spent in AID, and the percentage of time using specific system settings, as described above. Furthermore, the TDD, percentage of insulin delivered automatically by the system (auto basal and autocorrection bolus), and user-initiated manual bolus were assessed, as well as number of manual boluses per day, mean insulin-to-carbohydrate ratio (ICR), and amount of carbohydrates entered per day. Details on steps from raw data to endpoints, and the logics used, have been described elsewhere. 16
Statistics
Descriptive analysis was conducted using the mean and SD for continuous variables, or the median and interquartile range (IQR; 25th-75th percentiles). For categorical variables, number of cases and proportions (%) were used.
Results
Analysis 1: Post-AID Initiation
Population
A total of 26 427 MM780G users from 73 countries were included in this analysis. Figure 1 provides a detailed description of user identification in four cohorts: In total, 18 466 users had a TDD of ≥100 IU (A) and 10 795 users self-reported to have T2D in CareLink Personal (B). Among cohort B, 2834 users were identified to have a TDD of ≥100 IU (C) and 7961 had a TDD of <100 IU (D).
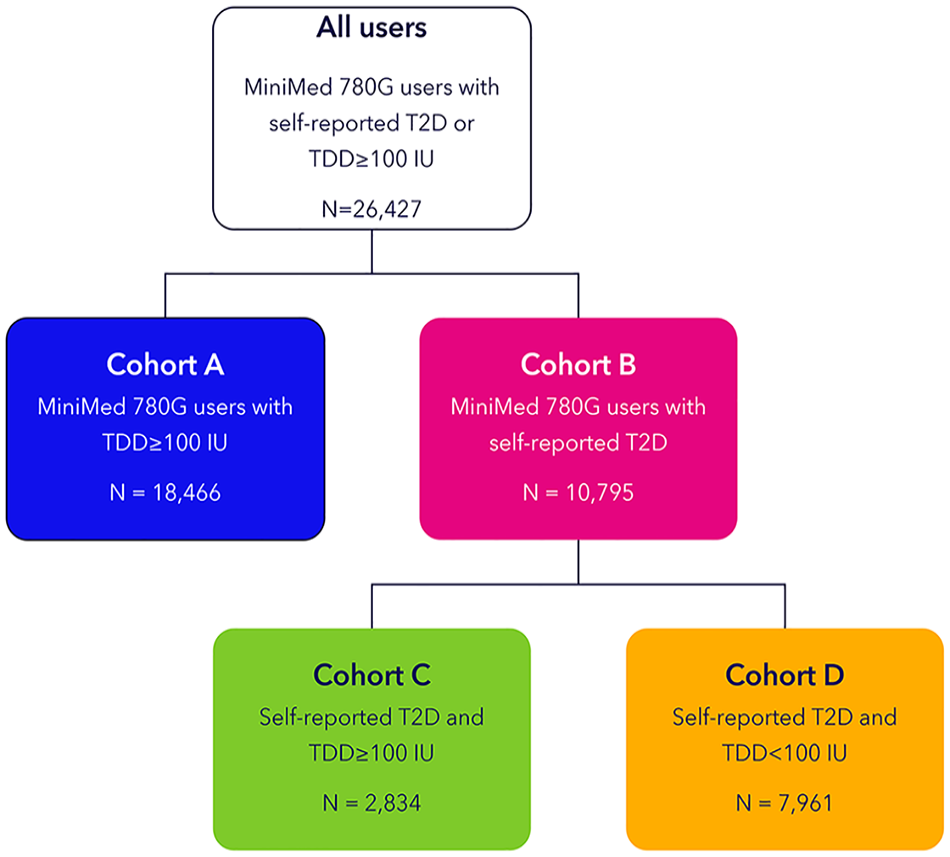
Flowchart of AID user identification in four selected cohorts.
Supplemental Table S1 provides user characteristics in terms of age groups and gender. In cohort A (TDD ≥ 100 IU), 44.3% reported to be ≥43 years (21.8% aged 43-55 years; 22.5% aged ≥56 years). About 62.8% of users reported to be male. In the cohort of self-reported T2D users (cohort B), 73.1% reported to be ≥43 years (22.2% aged 43-55 years; 50.9% aged ≥56 years). About 55.1% reported to be male. Similar age and gender distributions were seen in those with self-reported T2D and a TDD ≥ 100 IU (C) or a TDD <100 IU (D).
Glycemic outcomes
Figure 2 shows the glycemic outcomes after initiation of this AID for all four cohorts of users. In cohort A, the mean observation period was 213.1 (±203.4) (median: 158.0; IQR: 72.3-269.7) days. The mean TIR was 71.1% (±12.2), mean TITR was 44.5% (±11.8), and GMI was 7.1% (±0.4). About 57.4% of users achieved the international consensus target of TIR >70%, and 96.4% of users achieved the target of TB70 <4%. About 43.9% achieved the composite treatment target of TIR >70%, TB70 <4%, and GMI <7% (Figure 3).
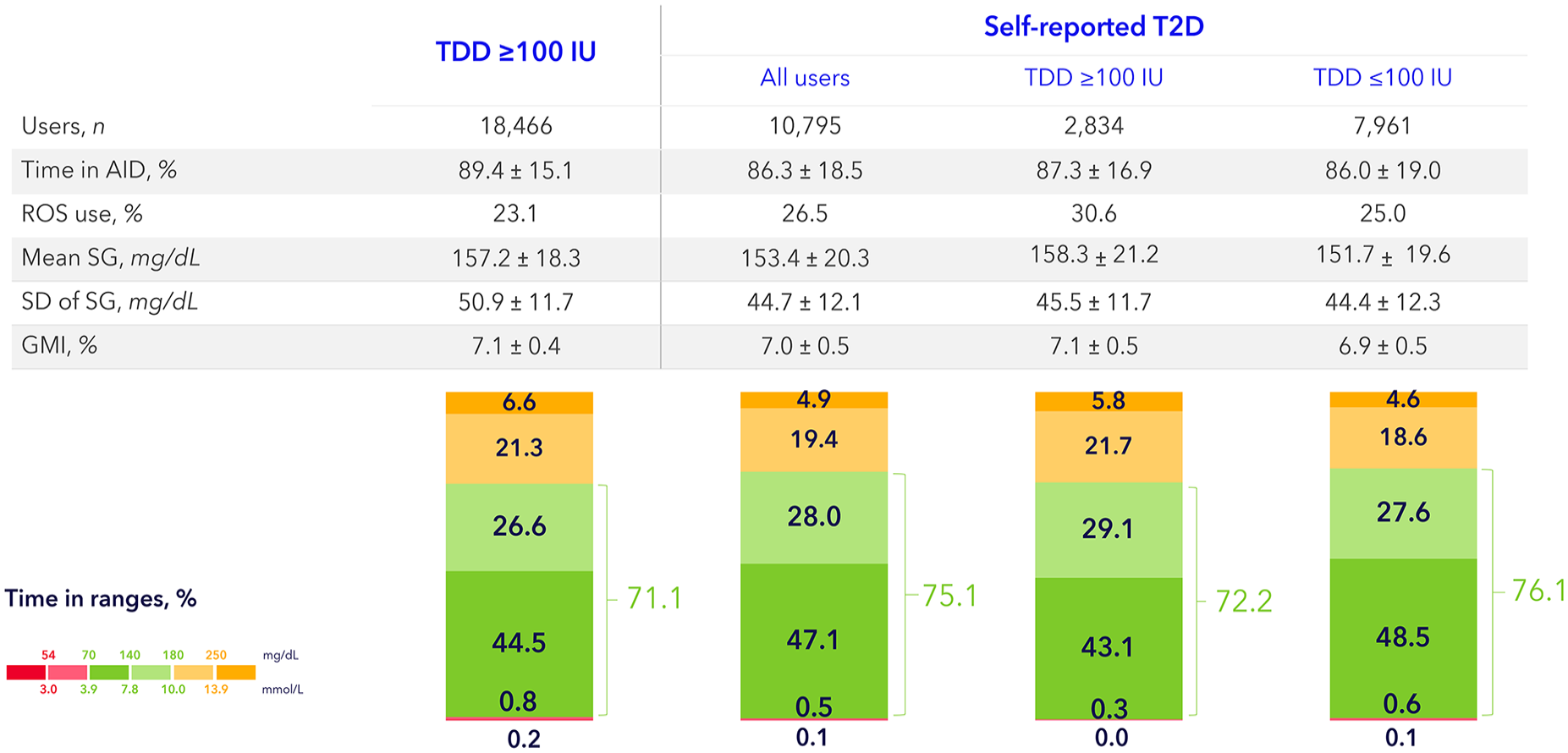
Glycemic outcomes of real-world AID users with self-reported T2D with or without a TDD of >100 IU.
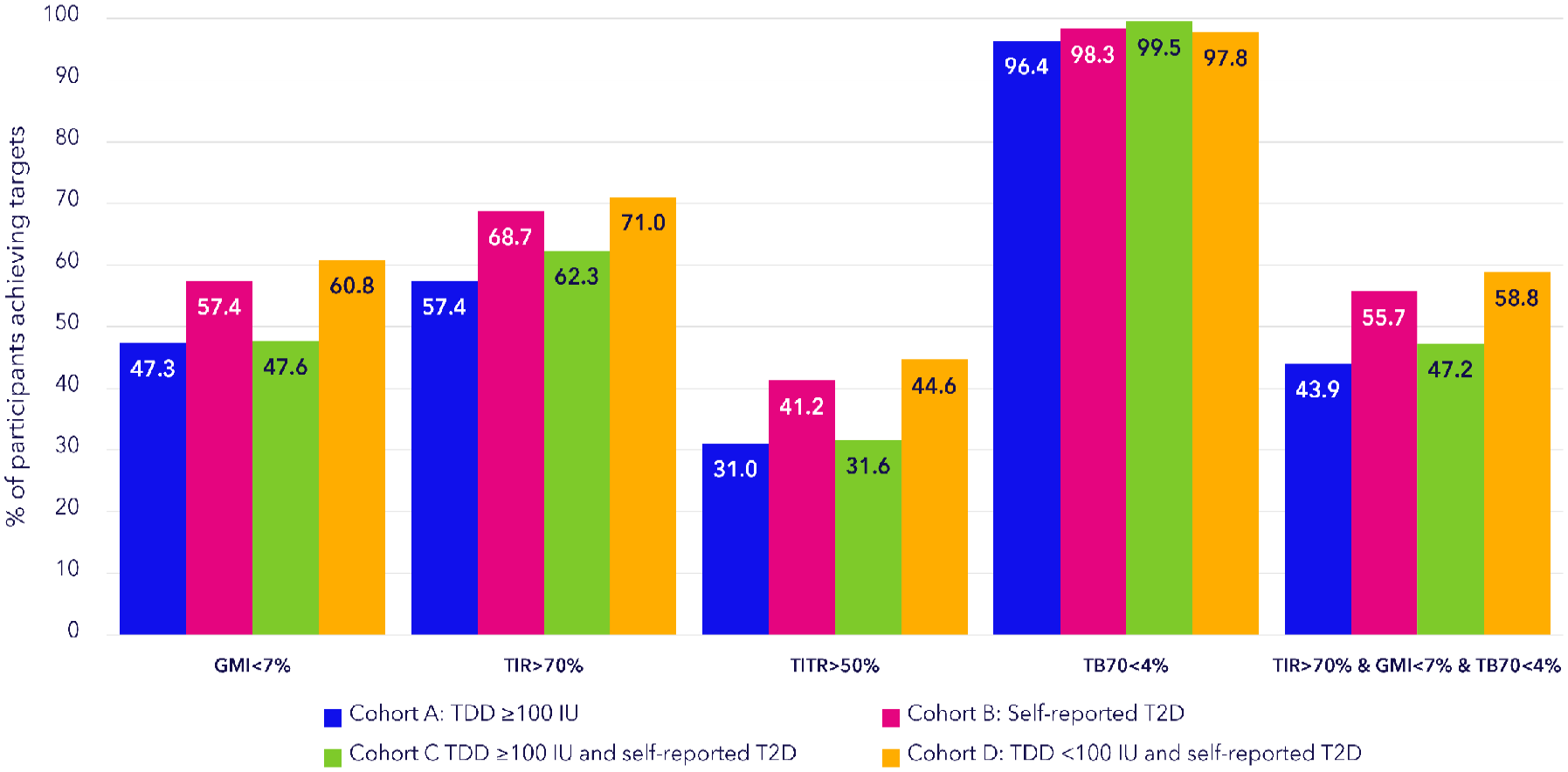
Percentage of users achieving international consensus treatment targets by real-world AID users with self-reported T2D with or without a TDD of ≥100 IU.
In cohort B, those users who self-reported to have T2D, the mean observation period was 148.0 (±144.0) (median: 111.4; IQR: 55.0-196.0) days. The mean TIR for this cohort was 75.1% (±14.1), mean TITR was 47.1% (±15.0), and GMI was 7.0% (±0.5) (Figure 2). About 68.7% of users achieved the international consensus target of TIR >70%, and 98.3% of users achieved the target of TB70 <4% (Figure 3). In cohort C and D, mean TIR was 72.2% (±15.0) and 76.1% (±13.6), respectively. The mean TITR was 43.1% (±14.5) and 48.5% (±15.0), respectively (Figure 2).
System use and Recommended Optimal Settings (ROS)
Cohort A spent on average 89.4% (±15.1) of time in AID. The recommended optimal GT of 100 mg/dL was used on average 61.8% (±42.9) of time, and the AIT of two hours on average 58.9% (±46.1) of time (Figure 4). About 23.1% of users used the combination of ROS for ≥95% of the time.
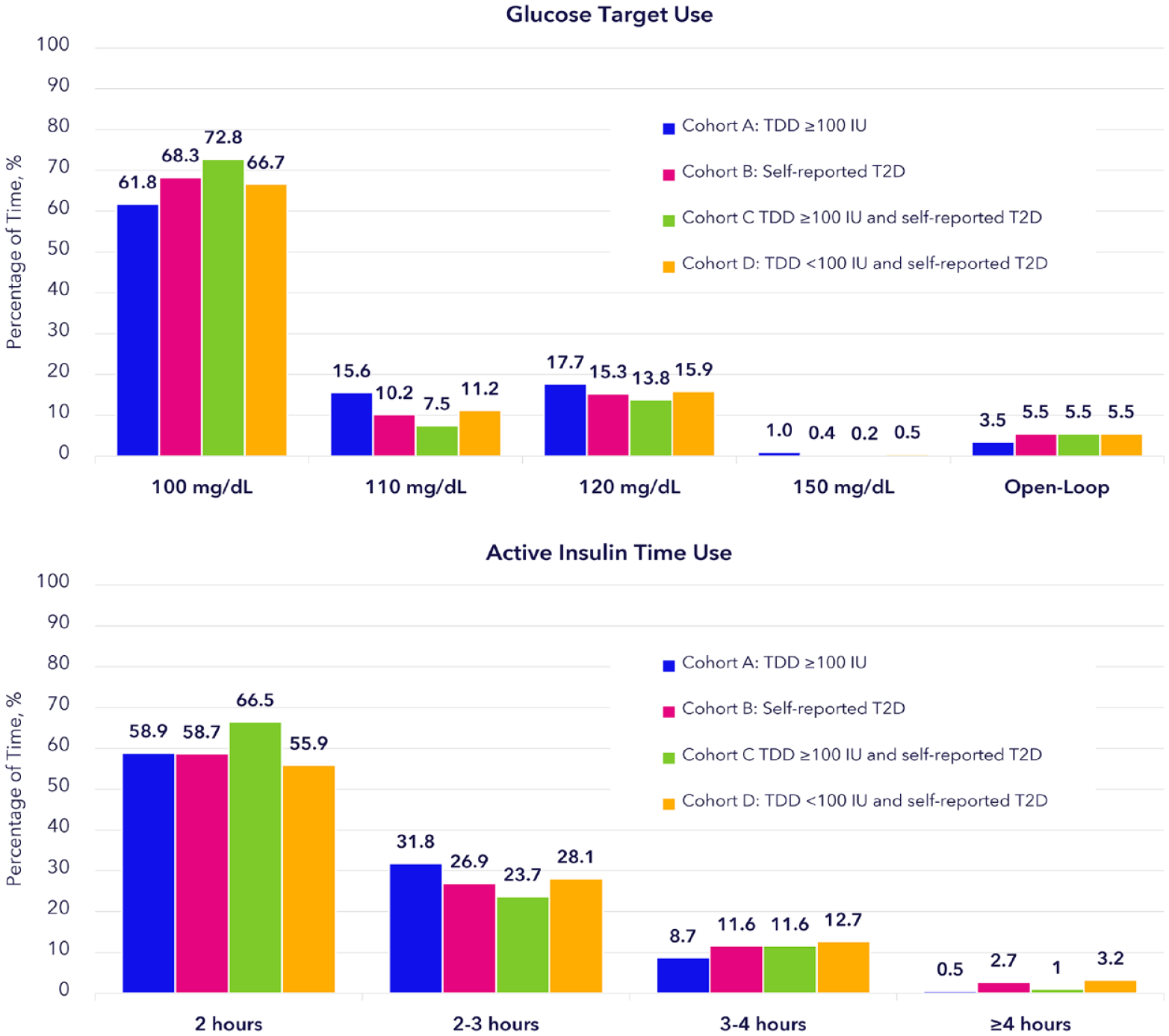
Percentage of time system settings of GT and AIT are used by real-world AID users with self-reported T2D with or without a TDD of ≥100 IU.
Cohort B spent on average 86.3% (±18.5) of time in AID. The use of ROS was similar to cohort A with GT of 100 mg/dL used on average 68.3% (±41.3) of time, and the AIT of two hours on average 58.7% (±46.4) of time (Figure 4). About 26.5% of users used the combination of ROS for ≥95% of the time. Similar system use and use of ROS was seen in cohort C and D, with average time in AID 87.3% (±16.9) and 86.0% (±19.0) of time; GT of 100 mg/dL used 72.8% (±38.9) and 66.7% (±42.0) of time, respectively; and AIT of two hours used 66.5% (±43.8) and 55.9% (±47.0) of time, respectively.
Glycemic outcomes for those who used the ROS are provided in Supplemental Figure S1 for cohort C only. Of 2834 users in cohort C, 868 used the ROS ≥95% of the time. Compared with the cohort not restricted to ROS users, the TIR was greater with 78.7% (±10.8) versus 72.2% (±15.0) as well as the TITR with 49.8% (±12.3) versus 43.1% (±14.5). The GMI was lower with 6.9% (±0.3) versus 7.1% (±0.5). The percentage of users achieving the international consensus target of TIR >70% was greater with 80.8% versus 62.3% (Supplemental Figure S2).
Total daily dose, insulin delivery, and bolusing behavior
Figure 5 shows the distribution of insulin delivery and TDD. In cohort A, the mean TDD was 133.9 IU (±34.4), of which 65.4 (±13.3) was delivered automatically by the system, that is, 45.7% (±9.1) by auto basal and 19.7% (±8.3) by auto correction bolus. About 34.6% (±13.3) of TDD was delivered as a user-initiated bolus. On average, this cohort entered a mean carbohydrate amount of 200.7 g/day (±116.6).
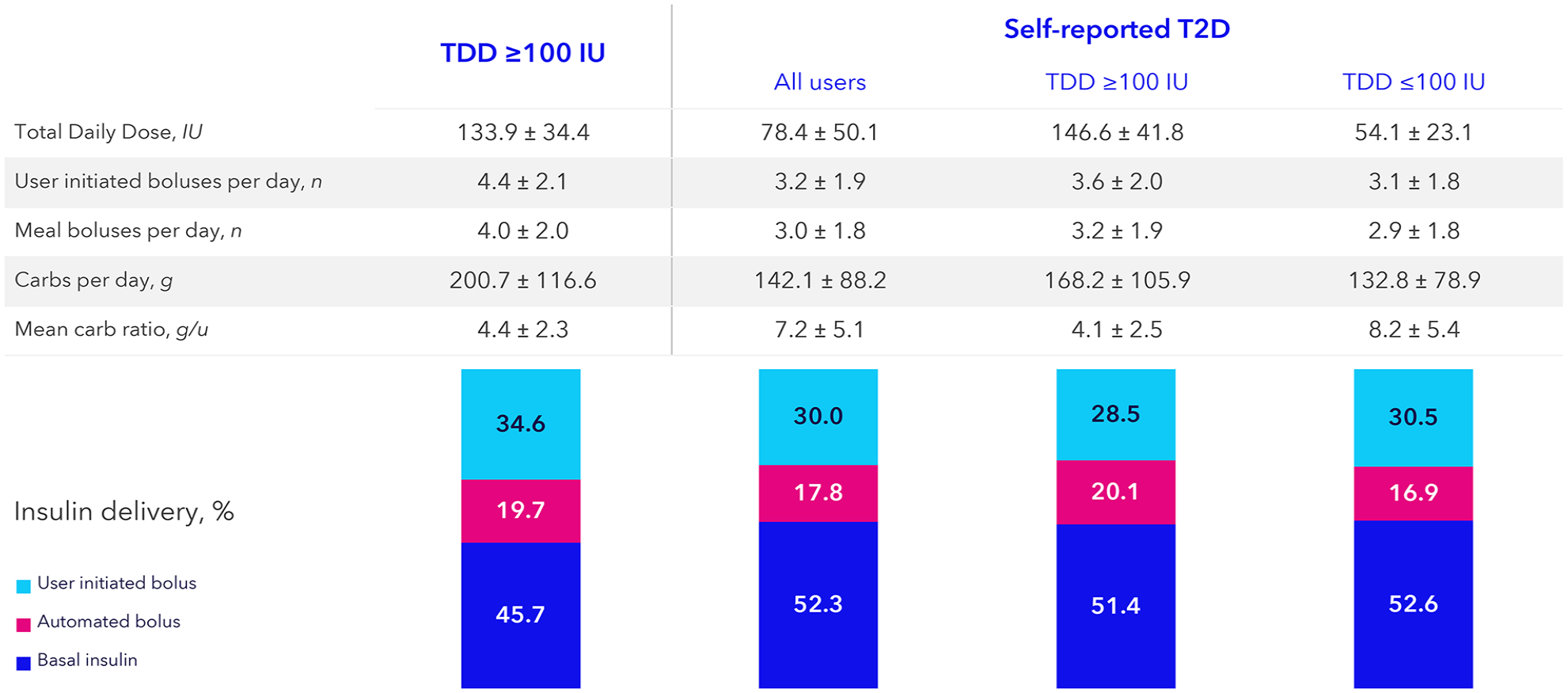
Total daily dose, insulin delivery mode, and bolus behavior in real-world AID users with self-reported T2D with or without a TDD of ≥100 IU.
In cohort B, the mean TDD was 78.4 IU (±50.1), of which 70.0 (±15.1) of insulin was delivered automatically by the system, that is, 52.3% (±12.3) by auto basal and 17.8% (±8.8) by auto bolus. Cohorts C and D had a mean TDD of 146.6 IU (±41.8) and 54.1 IU (±23.1), respectively. The insulin delivery of cohorts C and D was similar, with 71.5 (±13.9) and 69.5 (±15.4), respectively, delivered automatically by the system. The carbohydrate amount entered per day for cohorts B to D ranged from 142.1 (±88.2), 168.2 (±105.9), and 132.8 (±78.9) g/day, respectively.
Analysis 2: Pre-AID Versus Post-AID
In total, 896 users in cohort A, 810 users in cohort B, 190 users in cohort C and 620 users in cohort D, respectively, were included in this analysis (Figure 6). In cohort A, the mean TIR increased with 15.9%-points (±14.1) from 54.1% (±18.8) pre-AID to 70.0% (±13.0) after AID initiation. The GMI decreased from 7.6% (±0.7) to 7.1% (±0.5). In cohort B, the mean TIR increased from 60.0% (±21.2) to 72.1% (±14.7) (+12.1%-point), and GMI decreased from 7.4% (±0.8) to 7.1% (±0.5). Similar differences were shown in cohorts C and D where TIR increased from 54.0 (±23.4) to 69.7 (±16.0) (+15.7%-points) and from 61.8 (±20.2) to 72.9 (±14.3) (+11.1%-points), respectively. The GMI decreased from 7.7% (±0.9) to 7.2% (±0.5) and from 7.4% (±0.8) to 7.0% (±0.5), respectively.
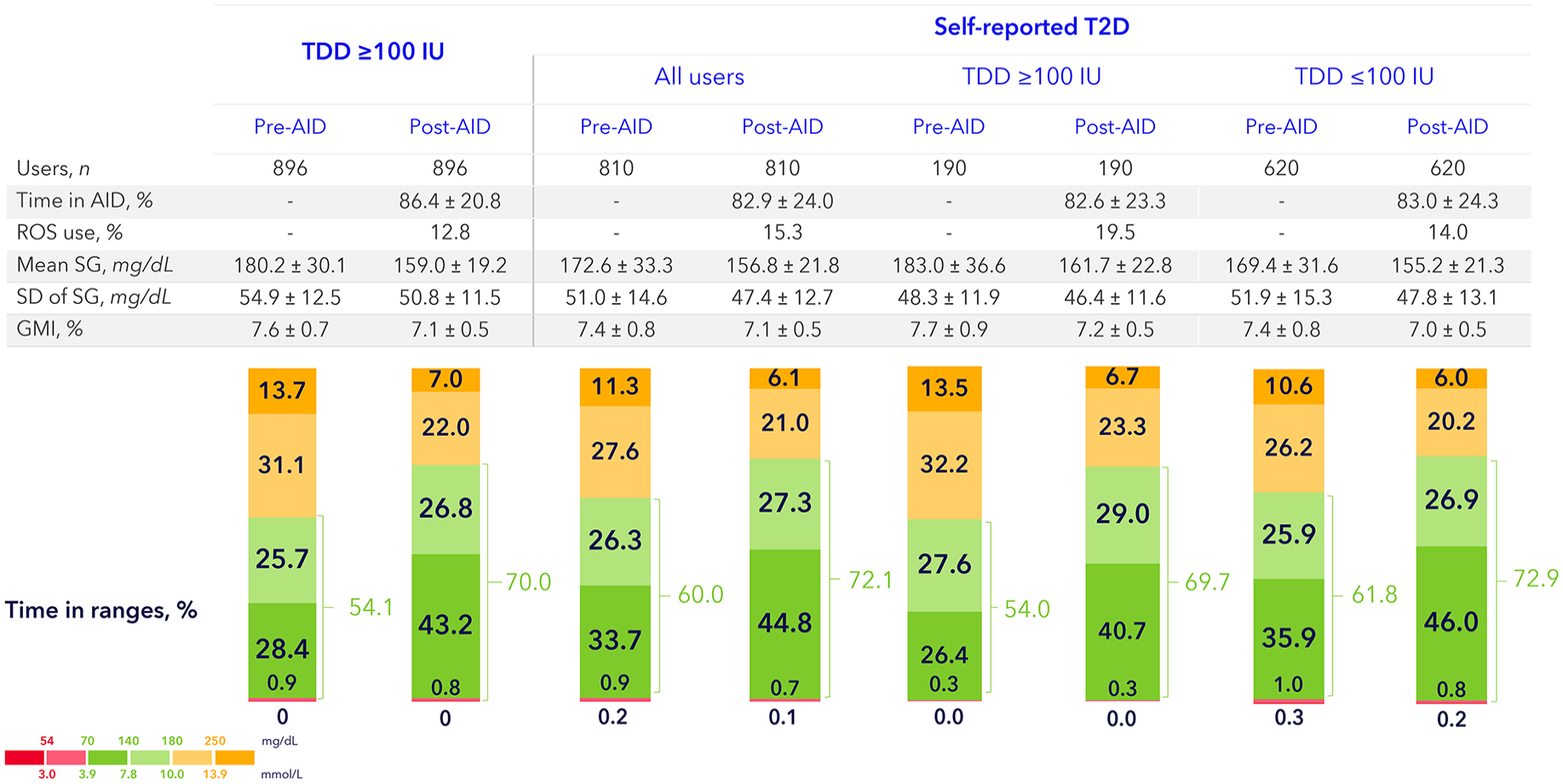
Glycemic outcomes preinitiation and postinitiation of AID in real-world AID users with self-reported T2D with or without a TDD ≥100 IU.
Analysis 3: Longitudinal
Longitudinal glycemic outcomes over six months after initiation of AID are shown in Supplemental Figure S3. In total, 862 users in cohort C were included in this analysis. Consistent results throughout six months were found, with the mean TIR ranging from 74.7% to 75.5%. TB70 consistently reaches 0.4%. The use of the ROS increased from 25.2% in month 1 to 41.2% from month 2 and onward.
Discussion
This is the first real-world evidence describing the performance of MM780G in people with self-reported T2D and different insulin requirements (as determined by TDD).
To characterize different phenotypes of T2D in this real-world data cohort, the population was grouped by different insulin requirements defined by a TDD >100 IU (cohort A), or self-reported T2D (cohort B). The group of users who self-reported to have T2D was further divided into those users having a TDD >100 IU (cohort C) likely representing a phenotype with greater insulin resistance, and those with a TDD <100 IU (cohort D) representing a phenotype with lower insulin requirements. All selected user cohorts on average achieved the international consensus target for the treatment of T2D in terms of TIR. 6 Furthermore, all cohorts improved more than 10%-points in TIR after AID initiation and stayed well below the target of <4% of TB70, while TA180 was reduced by 10% to 15% after initiation of AID in all cohorts. Longitudinal data for those with self-reported T2D and TDD >100 IU demonstrated that glycemic outcomes were consistent over at least six months.
These results are in line with previous large cohort real-world analyses in MM780G users with T1D. 7 Choudhary et al, for example, reported outcomes in >100 000 users with T1D with an average TIR of 72.3% and average TB70 of 2.0%. 8 In addition, the current results are also in line with the results of a single-arm prospective study comparing baseline SAP or HCL therapy versus AID in T2D, where AID users achieved on average 79.8% TIR at the end of the study. 17
While comparing clinical trials conducted is difficult because of selection biases as well as different devices, investigators, and subject populations, doing so provides a high-level view of how systems compare. Davis et al reported results of a clinical study where OmniPod 5 system reached a mean TIR of 58.6%. 18 Daly et al reported outcomes of a randomized controlled trial with the CamAPS HX algorithm, a full closed-loop system without meal announcement, in which people with T2D achieved a mean TIR of 66.3%. 19 Studies evaluating the outcomes in PWD using the Control-IQ system showed a mean TIR of 71% in a single-arm, prospective study 20 and a mean TIR of 72% in real-world data. 21 A small pilot study (n = 17) evaluating the DBLG1 system (per protocol) reported a mean TIR 76%. 22 Although results from our study show better achievements in glycemic control in T2D and high insulin requirements, no firm conclusions can be made as the algorithms are different, as is study population, study design, baseline control, and the type of sensor. Interestingly, all systems demonstrated a significant and clinically relevant improvement in glycemic metrics, and only a low percentage of TB70. This shows that AID systems provide for improved and safe glycemic control in people with T2D requiring intensive insulin therapy.
The MM780G was designed for use with the ROS, being a GT of 100 mg/dL and AIT of two hours. 23 Data in T1D demonstrated that the best glycemic control were reached using ROS, with a mean TIR up to 80%, and 92.7% reaching the treatment goal of TIR >70%. 7 The present study (cohort C) provides evidence that also in T2D greater glycemic control is reached when ROS are employed, with a TIR of 78.7%, and 80.8% of users reaching the treatment goal of TIR>70%.
Users with a TDD of more than 100 IU, regardless of whether they self-reported to have T2D or not, exhibited a higher carbohydrate intake per day and greater number of meal boluses per day than the other cohorts. Nonetheless, the proportion of insulin delivered automatically by the system remained consistent across all cohorts, averaging around 65% to 70% of TDD. Compared with insulin delivery data with this AID system in T1D, the proportion of insulin delivered automatically by the system is slightly higher in this cohort. In T1D, on average 55% of TDD is delivered automatically by the system, and 45% delivered by user-initiated boluses. 7 This suggests that the accuracy of mealtime announcements and carbohydrate assessment (that determines the percentage of user-initiated bolus) might play a smaller role and autocorrections can compensate inaccuracies for glycemic management of people with T2D and high insulin requirements versus people with T1D.24-28
The data presented suggest that this AID system can provide additional benefits to those with T2D; however, the data cannot provide information on the concomitant use of other therapies. Recent data from a single-arm study suggest, however, that the use of this AID system is rather additive to the use of GLP-1 RAs and SGLT2 inhibitors. 29
Limitations
Limitations of this study pertain to the data constraints of CareLink Personal. Apart from being restricted to those users who provided consent, sociodemographic variables, such as gender, age, and diabetes type, are self-reported due to privacy regulations, with age information grouped. In addition, the nature of the data does not allow for the collection of diabetes duration, duration of insulin pump use, HbA1c and weight, kidney function, nor the use of concomitant glucose-lowering medications. Further correlations of these outcomes should be assessed in repositories evaluating the long-term effects of automation. Despite this, demographic descriptions of the selected cohorts provide some confidence that the cohorts represent distinct phenotypes, that is, the cohort with T2D and >100 IU are predominantly older males, while those with >100 IU but without self-reported T2D are younger. The mean TDD in the group with self-reported T2D significantly differs from previous real-world data of users with type 1 diabetes, that is, on average 45 IU in Europe, the Middle East, and Africa, and 58 IU in the United States.7,10 As weight data are unavailable to calculate TDD in U/kg, the criterion of TDD >100 IU was selected to represent users with high insulin requirements. We cannot exclude that a significant percentage of patients present with T1D with a high daily carbohydrate intake, explaining a TDD of >100 IU.
Second, despite the strict adherence to the principles of real-world evidence, the potential impact of missing data on outcomes remains uncertain. Third, safety data are restricted to CGM readings, with no information on adverse events like severe hypoglycemia and diabetic ketoacidosis.
Strengths
The use of unbiased user uploads, coupled with a robust methodology for analyzing real-world data that have been validated and widely described, establishes confidence that the large cohort of 26 427 users provides credible information.
Conclusion
In conclusion, the use of this AID system is associated with effective therapy outcomes, indicated by over 70% TIR, and appears to be safe, as demonstrated by a low time below range (TBR) in a large cohort of real-life users with self-reported T2D with or without high daily insulin requirements.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251318373 – Supplemental material for Performance of an Automated Insulin Delivery System in People Living With Type 2 Diabetes and Insulin Resistance: First Real-World Evidence in 26 427 Users
Supplemental material, sj-docx-1-dst-10.1177_19322968251318373 for Performance of an Automated Insulin Delivery System in People Living With Type 2 Diabetes and Insulin Resistance: First Real-World Evidence in 26 427 Users by Isabeau Thijs, Arcelia Arrieta, Javier Castañeda, Michael Joubert, Francesco Giorgino, Benedikt Voelker, Tim Van den Heuvel, Jeremy Basset-Sagarminaga, Goran Petrovski, John Shin, Robert Vigersky and Ohad Cohen in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
AID, automated insulin delivery; AIT, active insulin time; CGM, continuous glucose monitoring; GLP-1 RA, glucagon-like peptide-1 receptor agonist; GT, glucose target; IQR, interquartile range; IR, insulin resistance; IU, units of insulin; MM780G, MiniMed 780G system; ROS, recommended optimal settings; SD, standard deviation; SG, sensor glucose; SGLT2, sodium-glucose cotransporter 2; T1D, type 1 diabetes; T2D, type 2 diabetes; TAR, time above range; TA180, time above 180 mg/dL; TA250, time above 250 mg/dL; TBR, time below range; TB54, time below 54 mg/dL; TB70, time below 70 mg/dL; TIR, time in range; TITR, time in tight range; TDD, total daily dose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: I.T., J.C., J.B.-S., A.A., B.V., T.v.d.H., G.P., J.S., R.V., and O.C. are full-time employees of Medtronic. M.J. declares consultant and/or speaker fees and/or research support from Abbott, Air Liquide Santé International, Amgen, Asdia, AstraZeneca, Bayer, BMS, Boehringer-Ingelheim, Dexcom, Dinno Santé, Glooko, Insulet, Lifescan, Lilly, LVL médical, Medtronic, MSD, Nestle HomeCare, Novo Nordisk, Organon, Orkyn, Roche Diabetes, Sanofi, Tandem, Vitalaire, Voluntis, and Ypsomed. F.G. has received grants from Eli Lilly, Roche Diabetes Care; consulting fees from; Eli Lilly, Novo Nordisk; support for attending meetings from AstraZeneca, Boehringer-Ingelheim, Eli Lilly, Lifescan, Merck Sharp & Dohme, Medtronic, Novo Nordisk, Roche Diabetes Care, Sanofi Aventis; Eli Lilly, Sanofi Aventis; serves or has served on the advisory panel for AstraZeneca, Boehringer-Ingelheim, Eli Lilly, Lifescan, Merck Sharp & Dohme, MedImmune, Medtronic, Novo Nordisk, Roche Diabetes Care, Sanofi Aventis; EASD/EFSD, Società Italiana di Endocrinologia (SIE), Fo.Ri.SIE (unpaid leadership); AstraZeneca, Eli Lilly, Novo Nordisk, Sanofi Aventis (support for medical writing and statistical analysis).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
