Abstract

Keywords
Continuous glucose monitoring (CGM) devices are increasingly being used in clinical studies to evaluate the effectiveness of therapeutic interventions for type 1 diabetes. 1 For these studies, CGM data are downloaded from the commercial platforms for analysis; however, the time interval between glucose readings varies between devices, e.g., 5 minutes for Dexcom G6 and 15 minutes for Freestyle Libre. 2 Despite this recognized variability, no prior research has evaluated the effect of these differences on key glycemic metrics, as defined by the American Diabetes Association’s (ADA) International Consensus on the Use of Continuous Glucose Monitoring. 3 Understanding these effects is crucial to ensure the reliability and comparability of CGM data across studies.
We analyzed data from 496 adults in the T1DEXI study, who wore the Dexcom G6 for 28 days. 4 To simulate 15-minute recording intervals, we modified the data set by removing two-thirds of the 5-minute data points. Metrics from the ADA’s International Consensus were calculated for both data sets using Diametrics software. 5 After excluding participants with <70% data availability, 477 participants were included. Paired t-tests compared metrics between 5-minute and simulated 15-minute intervals. Code is openly available at https://github.com/cafoala/recording-interval-cgm.
The cohort was predominantly White European (91%) and female (73%), with a median age of 33 years, body mass index (BMI) of 24.5 kg/m2, diabetes duration of 16 years, and hemoglobin A1c (HbA1c) of 6.6%. Most participants used insulin pumps (38%) or closed-loop systems (45%). Glycemic control was generally tight, with a mean time in range (TIR, 70-180 mg/dL) of 74.4% and a mean glucose of 146.1 mg/dL. Participants experienced a mean of 22.9 hypoglycemic episodes and 52.2 hyperglycemic episodes over the study, based on the 5-minute data.
Most glycemic metrics were consistent between 5-minute and 15-minute recording intervals (Figure 1). No significant differences were observed in mean glucose, coefficient of variation (CV), or TIR. However, significant reductions were noted in the detection of hypoglycemic and hyperglycemic episodes with 15-minute intervals:
Hypoglycemic episodes decreased by 4.4 episodes per person (95% CI = 4.1-4.7; P < .001), a 19.2% reduction.
Severe hypoglycemia (level 2) episodes decreased by 1.2 episodes (95% CI = 1.1-1.4; P < .001), a 27.9% reduction.
Hyperglycemic episodes decreased by 3.9 episodes per person (95% CI = 3.6-4.1; P < .001), a 7.5% reduction.
Severe hyperglycemia (level 2) episodes decreased by 0.9 episodes (95% CI = 0.8-1.0; P < .001), a 6.7% reduction.
This significant underreporting of glycemic episodes has important implications. In research, such underreporting may undermine conclusions regarding the efficacy of interventions, complicate comparisons across studies using different CGM devices, and challenge the synthesis of findings in systematic reviews or meta-analyses. Clinically, reduced detection of glycemic episodes could delay recognition of necessary treatment adjustments.
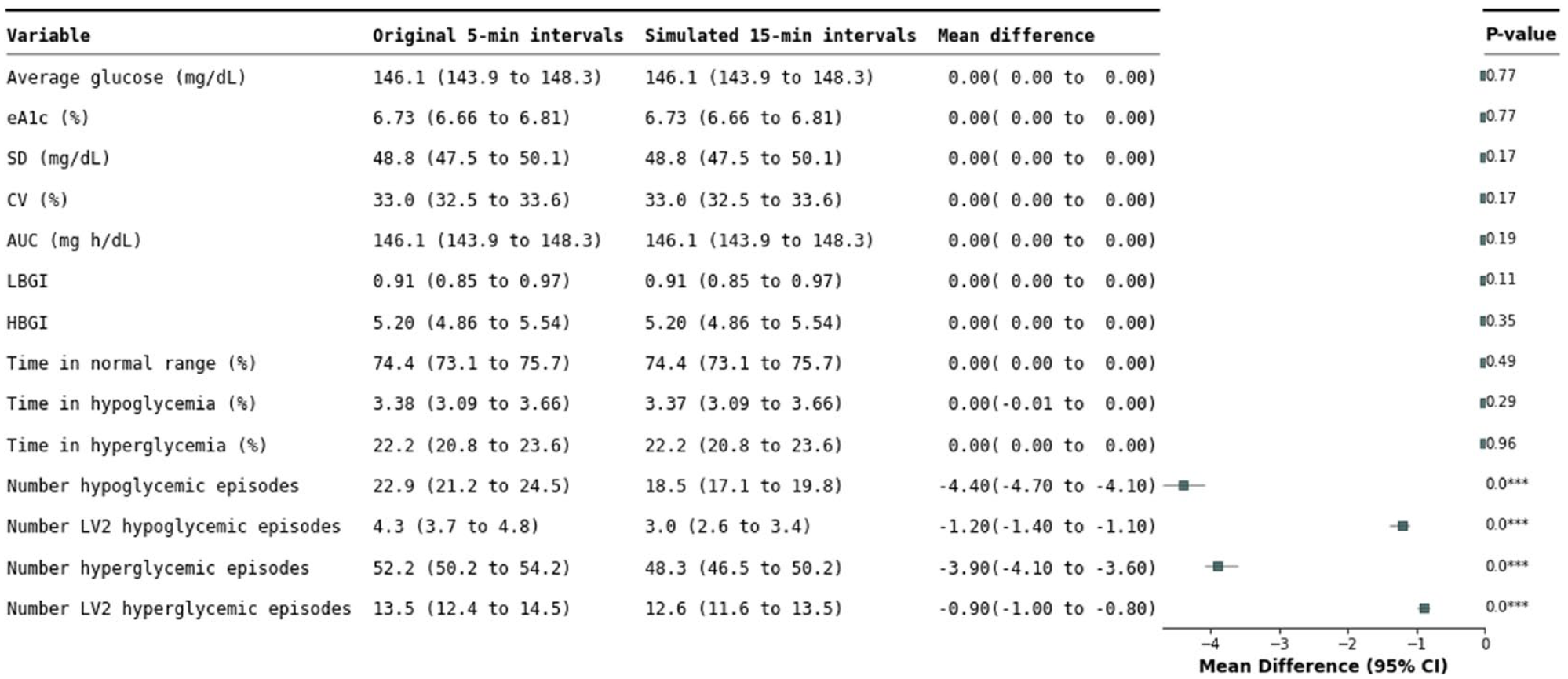
Comparative analysis of glycemic control metrics using 5-minute and 15-minute interval CGM data. This table displays the mean values of various glycemic control metrics calculated from the CGM data collected at 5-minute intervals and from the same data set modified to reflect 15-minute intervals by removing two-thirds of the data points. The analysis includes 28 days of data from 477 participants from the T1DEXI study, providing a direct comparison of the effect of recording interval frequency on key glycemic indicators. Time in normal range: 70-180 mg/dL (3.9-10 mmol/L), time in hypoglycemia: <70 mg/dL (<3.9 mmol/L), and time in hyperglycemia: >180 mg/dL (>10 mmol/L). Units for the glycemic episodes are per person over the 28 days. Mean differences with 95% confidence intervals are provided, alongside P-values to determine statistical significance.
These results highlight the need to standardize CGM data reporting and analysis. Future research should refine methodologies to address recording interval variations, ensuring reliable and comparable findings to support robust conclusions and optimize CGM technology’s clinical utility.
Footnotes
Acknowledgements
The authors thank the participants of the Type 1 Diabetes EXercise Initiative (T1DEXI) study. Without their invaluable contribution, this research would not have been possible. This publication is based on research using data from the T1DEXI study that has been made available through Vivli, Inc. Vivli has not contributed to or approved, and is not in any way responsible for, the contents of this publication. All authors were involved in the conception, design, and conduct of the study, as well as the analysis and interpretation of the results. C.L.R. wrote the code and the first draft of the manuscript. M.J.A. served as the technical advisor for code and data analysis. R.C.A. acted as the clinical advisor, and R.M.P. provided expertise as the public health advisor. All authors edited, reviewed, and approved the final version of the manuscript. C.L.R. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. The authors have no conflicts of interest. During the course of preparing this work, the authors used ChatGPT, Perplexity, ChatPDF, Scholarcy, and Copilot for the purpose of summarizing research papers, text editing, and providing code snippets. Following the use of these tools, the authors formally reviewed the content for its accuracy and edited it as necessary. The authors take full responsibility for all the content of this publication.
Abbreviations
ADA, American Diabetes Association; AUC, area under the curve; BMI, body mass index; CGM, continuous glucose monitoring; CV, coefficient of variation; eA1c, estimated HbA1c; HbA1c, hemoglobin A1c; HBGI, high blood glucose index; LBGI, low blood glucose index; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.L.R.’s PhD was supported by the Expanding Excellence in England (E3) fund. M.J.A. receives research funding from the National Institute for Health Research (NIHR) Applied Research Collaboration, South West Peninsula. E.C. receives funding from the NIHR School for Primary Care Research. R.C.A. has received remuneration from Novo Nordisk, AstraZeneca, and Eli Lilly for conducting educational talks on diet and exercise for health care professionals.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
