Abstract

Type 1 diabetes mellitus (T1D) patients admitted to the non–critical care setting (NCC) are at increased risk to develop hypoglycemia and may benefit from inpatient continuous glucose monitoring (CGM). The 2011 Endocrine Society CGM guidelines recommend against inpatient CGM due to insufficient evidence for accuracy and safety. 1 A 2016 expert consensus opinion acknowledges that CGM in the NCC could detect hypoglycemia earlier and allow for more aggressive glycemic control. 2
There are only 4 documented T1D cases where inpatient CGM was initiated in the NCC. A 2009 case report presented a T1D patient found comatose who had been hypoglycemic for the preceding 4.5 hours on blinded CGM. 3 Three T1D and 23 type 2 diabetes mellitus patients were included in an observational study that noted increased detection of nocturnal hypoglycemia with CGM, albeit blinded during admission. 4
We started nonblinded CGM in a T1D patient during admission to a NCC and prevented recurrent inpatient hypoglycemia.
Case
A 33-year-old T1D male was admitted to the hospital with new-onset seizures. He was diagnosed with T1D at age 13, complicated by gastroparesis and end-stage renal disease requiring dialysis.
On admission, in the setting of altered mental status, swallowing dysfunction, and hypoglycemia unawareness, his OmniPod insulin pump was disconnected and continuous tube feeding was initiated. He was initially given glargine insulin 8 units twice daily, regular insulin 3 units every 6 hours, and concurrent correction regular insulin. On hospital day 4, tube feeding was discontinued in the setting of emesis, leading to labile glycemic control. On hospital day 5, he experienced profuse diaphoresis associated with a point-of-care glucose value of 26 mg/dL. On hospital days 6 and 7, he repeatedly experienced point-of-care glucose values less than 55 mg/dL.
CGM was subsequently initiated to prevent severe hypoglycemia. We obtained informed consent from family prior to using the DexCom G4 CGM device, as the patient had persistent altered mental status. We instructed family and nurses on how to use and calibrate the CGM device. The hypoglycemia threshold alarm was set to 80 mg/dL. The hyperglycemia threshold alarm was set to 250 mg/dl. The CGM results were not blinded during admission, which factored into our treatment recommendations. During CGM use, no acetaminophen was administered, nor was MRI or CT scan imaging obtained, which would be contraindicated with DexCom CGM use. 5 The CGM device did not experience technical difficulty.
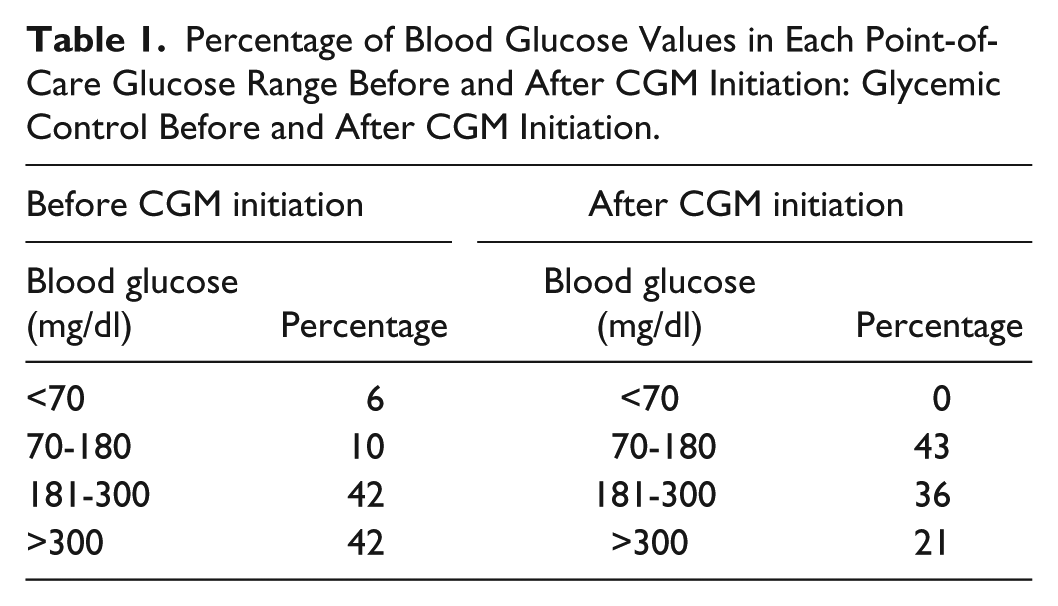
Before CGM initiation, 52 point-of-care glucose values were measured, with a mean value of 278 mg/dL, and 6% of glucose values below 70 mg/dL. After CGM initiation, 1,071 interstitial glucose levels were measured, with a mean value of 222 mg/dL, and no point-of-care glucose values below 70 mg/dL (Table 1). The CGM hypoglycemia alarm was triggered once for an interstitial glucose value of 72 mg/dL, corresponding to a point-of-care glucose value of 80 mg/dL, not warranting glucose administration. CGM was used for 4 days until discharge home.
Percentage of Blood Glucose Values in Each Point-of-Care Glucose Range Before and After CGM Initiation: Glycemic Control Before and After CGM Initiation.
Conclusion
Our case demonstrates that inpatient CGM in the NCC could prevent severe T1D hypoglycemic episodes, limiting morbidity. Unlike prior cases, we used nonblinded CGM.
Footnotes
Abbreviations
CGM, continuous glucose monitoring; NCC, non–critical care setting; T1D, type 1 diabetes mellitus.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
