Abstract

Continuous glucose monitoring (CGM) has become the standard for glucose monitoring in people with type 1 diabetes or type 2 diabetes with intensified insulin therapy. 1 The commercially available CGM systems differ in the nominal wear times of these sensors (7-14 days). To our knowledge, no data have been published yet about the actual wear time of the glucose sensors in the daily life of people with diabetes.
We asked adult people with type 1 or type 2 diabetes who are members of an online panel (www.dialink-diabetes.de) to take part in an online survey. To assess wear time, participants were asked to enter the number of days that they usually wear their glucose sensors. They were also asked for reasons why their glucose sensor usually has a shorter wear time.
Data from 342 participants were analyzed of which 288 (85.2%) had type 1 diabetes. The mean age was 57.6 (SD 13.4) years, 47.1% were women, and all of them were using insulin. Most participants used FreeStyle Libre 3 (44.4%), 26.9% used Dexcom G6, 10.8% used Dexcom G7, 9.9% used the Medtronic Guardian 4, and 5.6% used the Medtronic Guardian 3 (2.3% used another system).
The wear times of the different glucose sensors are displayed in Figure 1a. Most people (87.5%) using Libre 3 reported an average wear time of 14 days, with 11.8% reporting a shorter wear time, with 1.3% reporting only an average wear time of 7 days. Around 83.7% of G6 users reported an average wear time of 10 days and 81.1% of G7 users reported an average wear time of 10 to 11 days. Only a small fraction of G6 (11.9%) and G7 (16.2%) users reported a wear time <10 days. Most Medtronic users (Guardian 3: 84.2%; Guardian 4:85.3%) reported a wear time of 7 days, with 10.5% (Guardian 3) and 11.8% (Guardian 4) reporting a shorter wear time.
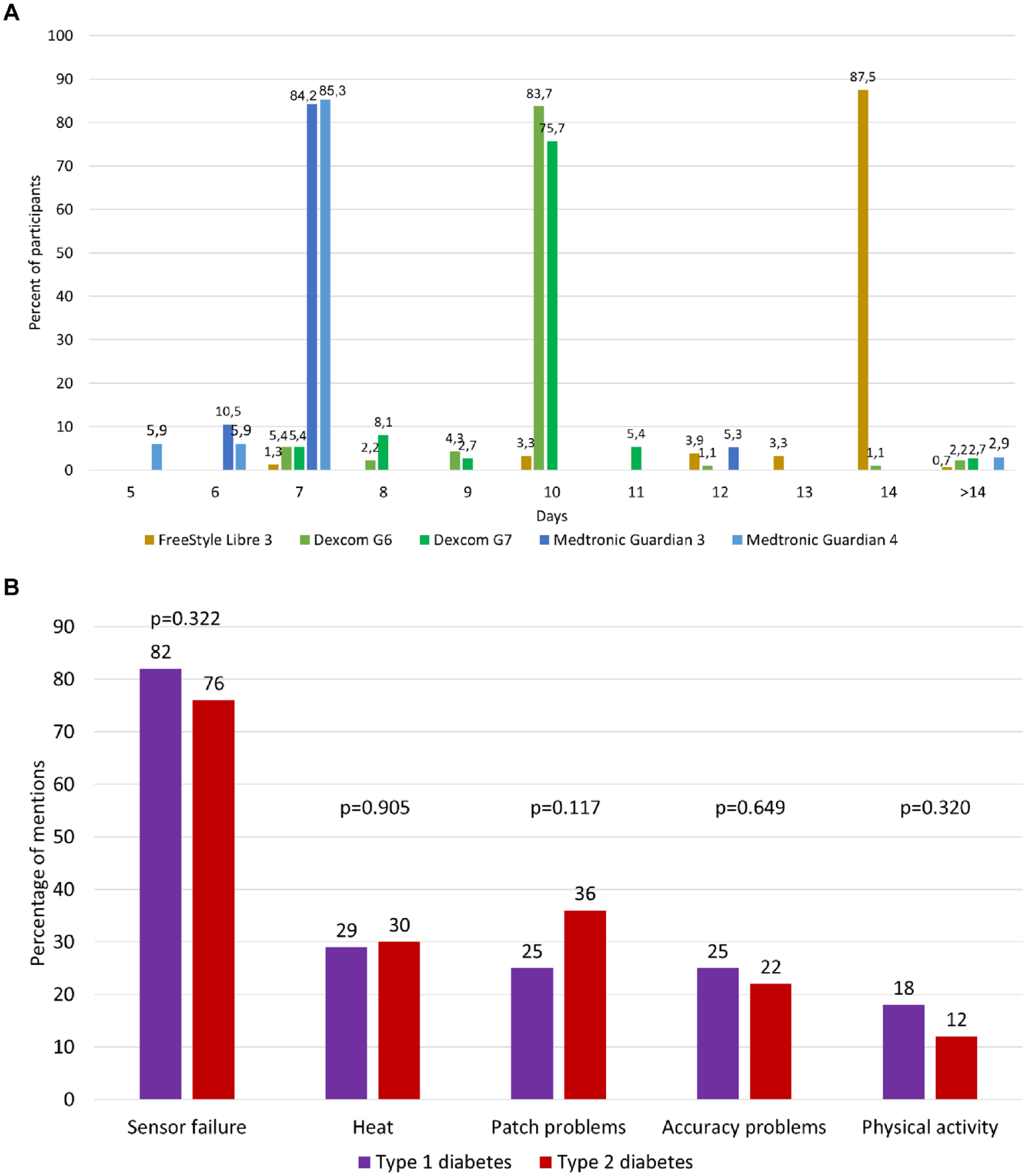
Wear time of different glucose sensors (a) and reasons for a shorter wear time (b).
The most reported reason for a shorter wear time was sensor failure (81%), followed by heat (30%), patch problems (27%), accuracy issues (25%), and physical activity (17%). There were no significant differences between people with type 1 and type 2 diabetes (Figure 1b; independent sample t-tests). Sensor failure was reported more often by men compared to women (86% vs. 76%; P = 0.022).
The results showed that across different CGM systems, approx. 10% to 15% of glucose sensors did not reach the intended wear time. This result matches with the findings from Marsters et al, who report premature sensor loss in 18% of cases in their randomized control trial. 2 In our study, glucose sensor failures and heat were the most common reasons for a shorter wear time. Although the wear time of CGM systems is reported in clinical trials (but not specifically for glucose sensors), this reflects the adherence to the study protocol and not real-world conditions. 3 The data from this survey reflects the daily-life-reported experiences with glucose sensors.
Footnotes
Acknowledgements
The authors thank the dia·link members who took part in this study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DE reports Advisory Board member fees from Dexcom Germany and Roche Diabetes Care as well as honoraria for lectures from Berlin Chemie AG, Sanofi-Aventis, Dexcom Germany, Boehringer Ingelheim, Eli Lilly, and Roche Diabetes Care. BO reports Advisory Board member fees from Abbott Diabetes Care as well as honoraria for lectures from Astra Zeneca, Eli Lilly and Evivamed. TR reports honoraria for lectures from Berlin Chemie AG. NH reports Advisory Board member fees from Abbott Diabetes Care and Insulet as well as honoraria for lectures from Berlin Chemie AG, Becton Dickenson, Sanofi Germany, Roche Diabetes Care, and Dexcom Germany. BK reports Advisory Board member fees from Abbott Diabetes Care, Embecta, Roche Diabetes Care, Novo Nordisk, Berlin Chemie AG, and Dexcom Germany as well as honoraria for lectures from Sanofi Germany, Novo Nordisk, Abbott Diabetes Care, Roche Diabetes Care, Berlin Chemie AG, Embecta, Dexcom, and Feen. In addition, he reports support for travel and fees for scientific meetings from Sanofi, Roche Diabetes Care and Berlin Chemie AG as well as unpaid obligations as workshop leader and member of working groups of the German Diabetes Association. LH is a consultant for Abbott, Lifecare (also a member of the Board of Directors), Medtronic EU Advisory Board, Dexcom Germany, Roche Diagnostics, Liom, and Perfood. He is part owner of the Profil Institut für Stoffwechselforschung in Neuss, Germany, Science Consulting in Diabetes GmbH, Düsseldorf, Germany, and diateam GmbH, Bad Mergentheim, Germany.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
