Abstract
Smart pen technology has evolved over the past decade with new features such as Bluetooth connectivity, bolus dose calculators, and integration with mobile apps and continuous glucose monitors. While similar in appearance to a traditional insulin pen, smart pens have the ability to record and store data of insulin injections. These devices have the potential to transform diabetes management for clinicians, and patients with type 1 and type 2 diabetes on insulin therapy by improving adherence, glycemic control, and addressing barriers to diabetes management. Smart pens can also highlight the relationship between insulin, food, and physical activity, and provide insight into optimizing insulin regimens. Education of clinicians and patients, and more clinical studies showing the benefits of smart pens and cost-effectiveness, are needed.
In 2019, it is estimated that 463 million adults, aged 20-79, had diabetes worldwide, and by 2045, this number is anticipated to rise to 700 million. 1 Approximately 10% of all adults with diabetes have type 1 diabetes (T1D) and 7.5% have type 2 diabetes (T2D) requiring insulin therapy.1,2 Insulin pens are the most common insulin administration method used worldwide. 3
The first insulin pen, NovoPen by Novo Nordisk (Bagsværd, Denmark), was launched in 1985 and with advances in insulin pen technology, most insulin currently available on the market offers the option for insulin pens. 4 Clinicians and patients tend to prefer insulin pens over vials and syringes since they have many advantages.3,4 These include improvement in adherence, convenience, dosing accuracy, and social acceptability.3-5
Although insulin pens have been shown to be beneficial, patients with T1D or T2D on insulin therapy may continue to struggle with diabetes management and achievement of glycemic goals. 6 Insulin therapy is challenging due to its narrow therapeutic range and risk of hypoglycemia. Patients may be on complex multiple daily injection (MDI) insulin regimens and have to decide every day the doses of insulin based on amount of carbohydrates, food choices, and physical activity. These challenges are reflected by the small percentage of patients reaching target A1c <7.0%. 7 Furthermore, patients may not be adherent to insulin therapy for reasons such as forgetting to administer insulin, injecting the incorrect type of insulin, fear of hypoglycemia, and/or administering insulin more than once.
Thus far, there has not been a way to objectively evaluate dosing practices in patients with diabetes who self-inject insulin using syringes or pens. Unreliable glucose logbooks, multiple glucometers, patients not performing self-monitoring of blood glucose, and/or having difficulties with carbohydrate counting can make assessment of insulin therapy adherence challenging and prevent patients from achieving optimal glycemic control.8-11
Clinicians have to make decisions on a patient’s insulin regimen based on the presumption that the patient is following the insulin prescription. This presumption may lead to under or over treatment, particularly if nonadherence is frequent. In a recent study using Bluetooth-enabled insulin pen cap technology, nonadherence to insulin in patients with T1D and T2D was recorded for 24% of bolus insulin administration and 36% of basal insulin administration. 12 Therefore, for patients with diabetes on insulin, missing an insulin dose is not uncommon and can negatively impact glycemic control.13-15
To address the challenges of insulin pens, “smart pens” were developed and introduced in 2007 with the ability to store data such as the date, time, and amount of previous insulin doses. 16 Smart pens are similar in appearance to insulin pens and require the patient to prime the needle, set the insulin dose, and use the depressing device for insulin delivery. Over the past decade, smart pens have evolved and recently have added connectivity features such as Bluetooth or near-field connectivity (NFC) technology to display data on applications and to download records. The InPen (Companion Medical; San Diego, CA) is the first and only FDA-approved Bluetooth-enabled wireless smart pen available in the United States, and recently, has been acquired by Medtronic (Dublin, Ireland). 11 In Europe, ESYSTA BT Pen (Emperra; Potsdam, Germany) and Pendiq 2.0 (Pendiq; Moers, Germany) are available and CE-marked.17,18 NovoPen 6 (Novo Nordisk) is not yet available, but is anticipated to launch soon in Europe. 16
All of the available connected smart pens have the ability to record and store data of the amount and timing of insulin injections, which can be reviewed by the patient in a timely manner, and provide downloadable reports to the patient and healthcare provider. 19 The InPen has additional features such as bolus dose calculator, detection of prime dose versus actual dose, reminder alerts, active insulin on board (IOB) monitoring, and integration with continuous glucose monitors. 11 A comparison of the devices is shown in Table 1.
Comparison of Approved Connected Smart Pens.
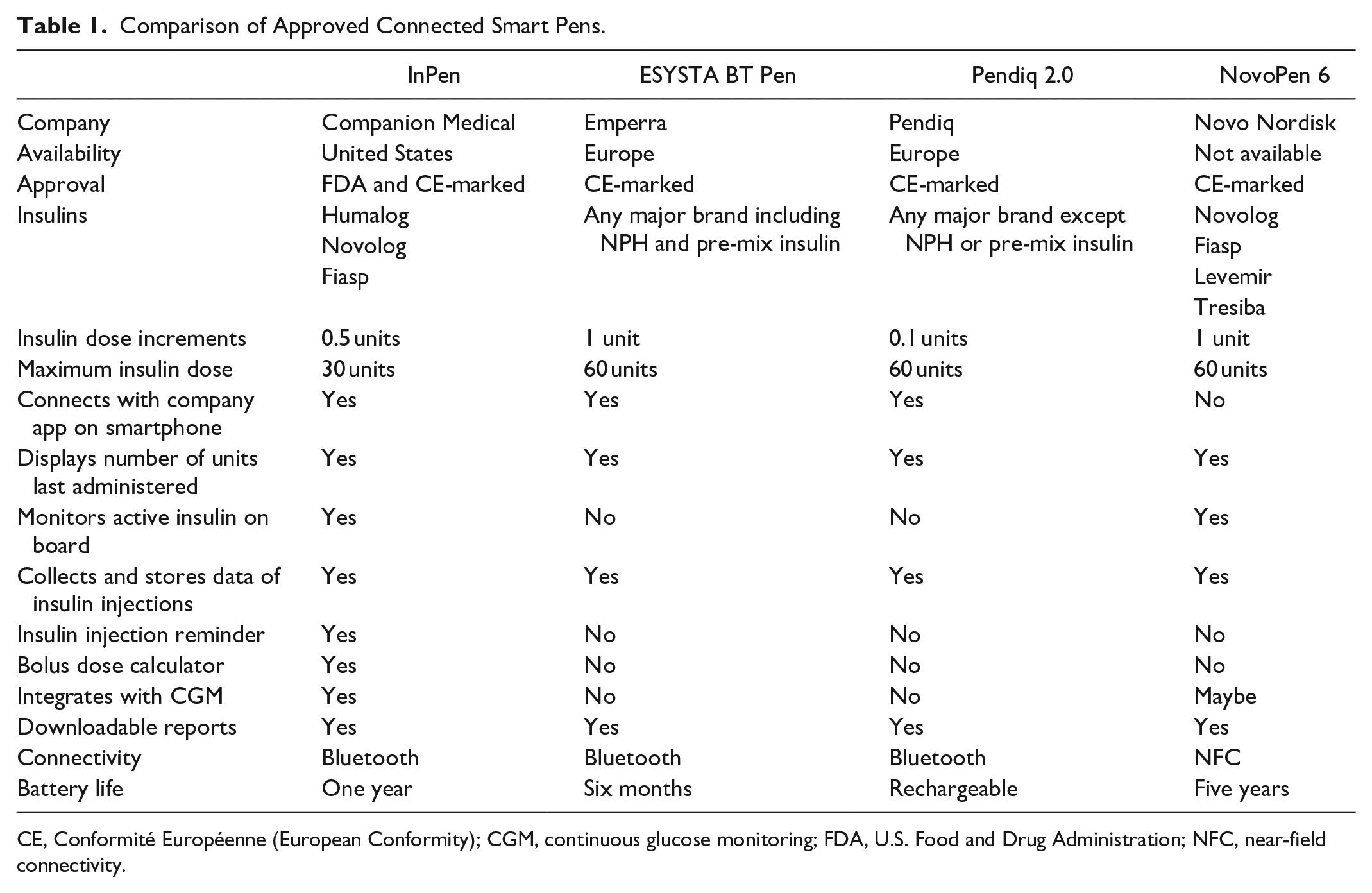
CE, Conformité Européenne (European Conformity); CGM, continuous glucose monitoring; FDA, U.S. Food and Drug Administration; NFC, near-field connectivity.
The Benefits of Using Connected Smart Pens
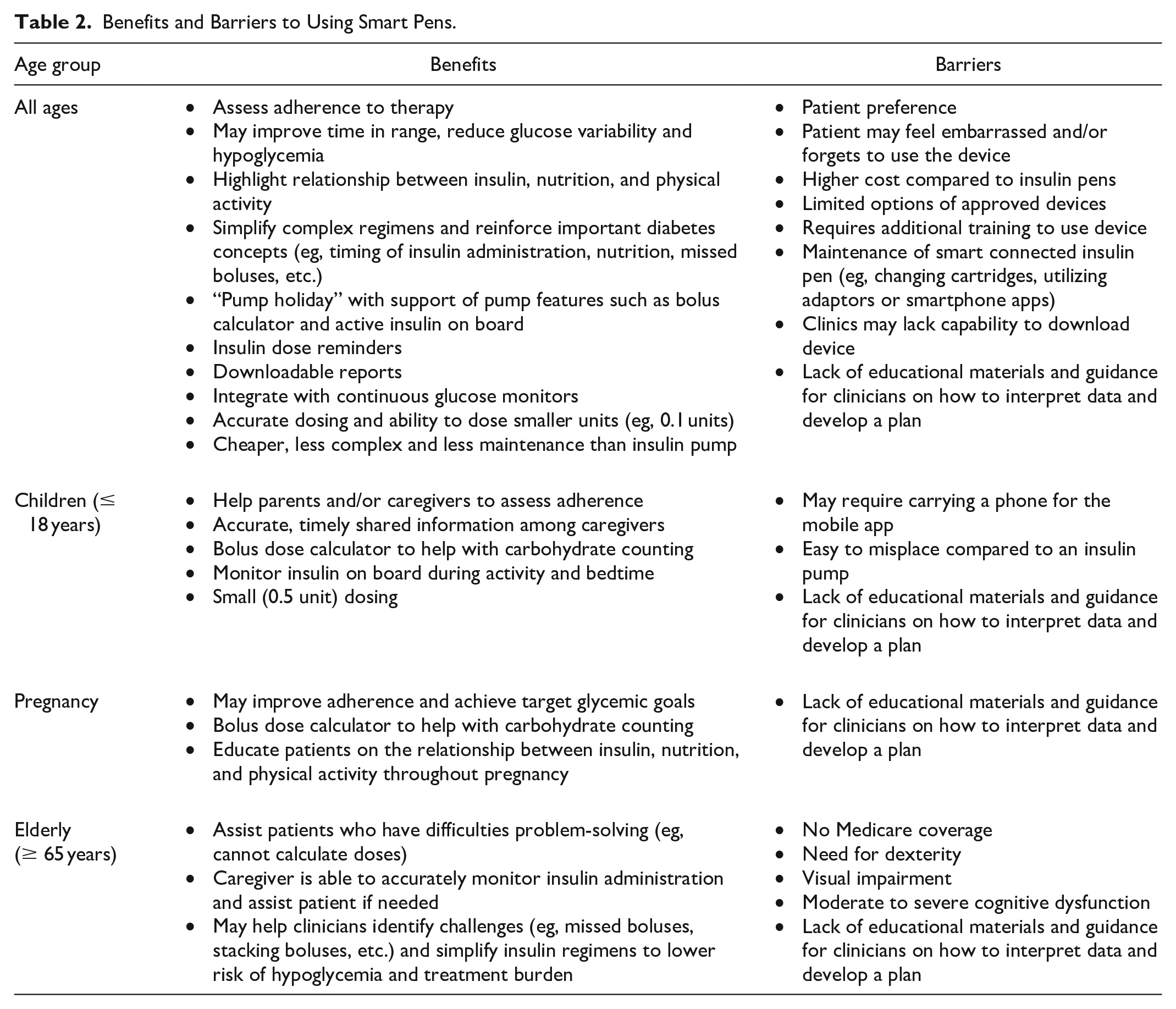
Connected smart pens have many benefits and may be useful for patients of all ages with T1D or T2D on insulin therapy (Table 2).
Benefits and Barriers to Using Smart Pens.
Improve Adherence and Glycemic Control
With the ability to record insulin injections, smart pens may help patients monitor their adherence to insulin therapy and assist clinicians in clinical decision making. Smart pens when combined with continuous glucose monitoring (CGM) can show patients, in real-time, the consequences of behaviors such as missing insulin doses or insulin stacking, and this provides an opportunity for targeted diabetes education (Figure 1). A recent proof-of-concept study using NovoPen6 in patients with T1D on MDI with CGM showed that smart pens can help improve adherence to insulin therapy. 20 Participants had a 43% reduction in missed bolus doses at follow-up (≥180 days) compared to baseline using the smart pen. This study also found a significant increase of 1.9 hours/day for time in range (TIR) with a reduction in time spent in hyperglycemia (>180 mg/dL) and hypoglycemia (<54 mg/dL). Improving adherence to insulin therapy not only has a positive effect on glycemic control but may also reduce healthcare costs and utilization.21,22
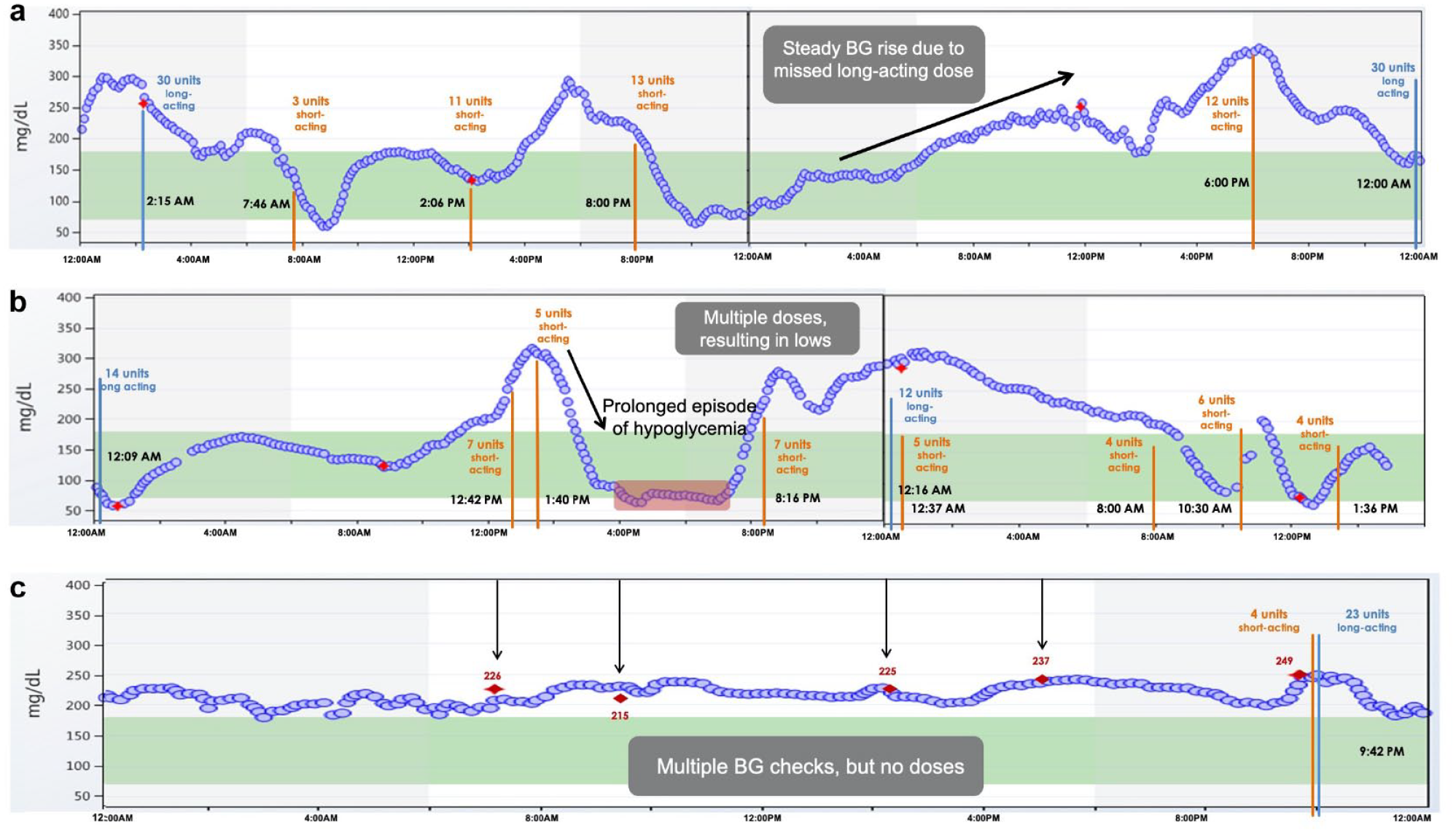
Smart pens plus CGM can reveal nonadherence to insulin regimens and may help healthcare providers create individualized treatment plans and provide targeted education.
Highlight Challenges Between Insulin and Food
The timing of mealtime insulin boluses is key to achieving optimal post-prandial glycemic control. Clinical studies have shown that injecting rapid-acting insulin 15-20 minutes pre-meal reduced hyperglycemia by 30% and hypoglycemia compared to injection immediately before the meal.23,24 With smart pen features such as dose reminder alerts and the ability to view active IOB, patients may be more successful in timing the dose of insulin boluses in relation to meals and avoid insulin stacking.
Meal composition (fat, protein, and carbohydrates) can also significantly affect post-prandial glucose. Previous studies have shown that dietary fat can lead to sustained post-prandial hyperglycemia up to five hours. 25 Using Bluetooth-enabled insulin pen cap technology and CGM in patients with T1D on MDI, 37% of mealtime boluses resulted in elevated three-hour post-prandial glucose levels >180 mg/dL despite insulin being injected at the appropriate time. 26 This study also showed that late or missed mealtime boluses occurred frequently and resulted in post-prandial glucose excursions. Therefore, smart pens combined with CGM may be used as an education tool to teach patients and give immediate feedback on dosing and timing of insulin and meal choices (Figure 2). It can also inform healthcare providers whether insulin to carbohydrate ratios (ICR) and insulin sensitivity factors (ISF) are appropriate.
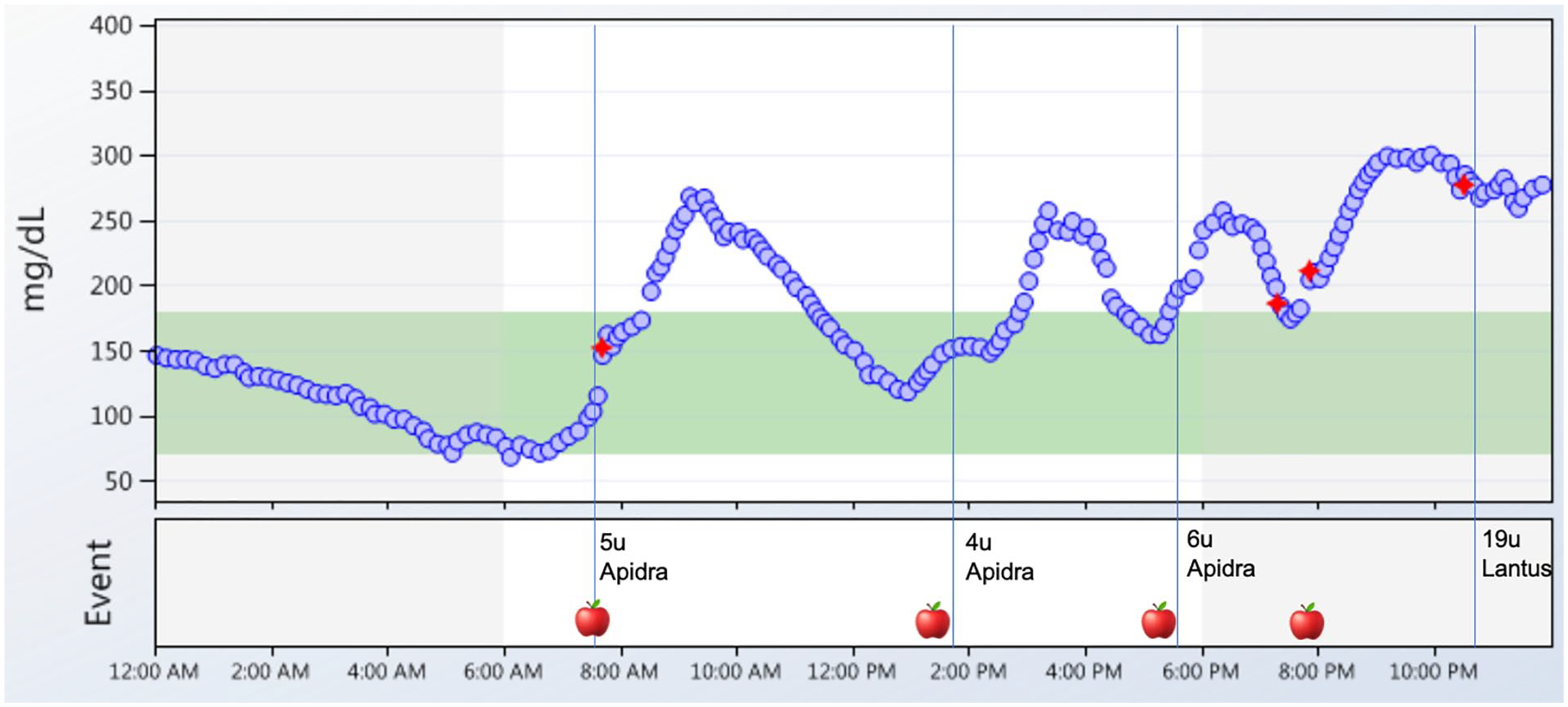
Smart pens plus CGM can highlight the relationship between insulin and food. A daily glucose report shows that a patient consumed four meals per day (apple symbol) and injected rapid acting insulin (Apidra) to cover meals and long-acting insulin at bedtime (Lantus). Data from the pen revealed that the patient was injecting rapid-acting insulin immediately before or shortly after eating, and consequently the patient experienced post-prandial hyperglycemia. There was one episode of a missed mealtime insulin dose at 8:00 PM (presence of apple symbol without Apidra administration). The patient had a consistent pattern of post-breakfast hyperglycemia and a review of dietary intake uncovered that the patient was eating a donut regularly at breakfast time. The red diamond symbol indicates fingerstick calibrations for the CGM and blue vertical lines indicate insulin administration.
Address Barriers to Diabetes Management in Different Diabetes Populations
For patients with T1D, smart pens offer similar features as insulin pumps such as bolus calculator and IOB but are less expensive, complex, and are easier to maintain. Smart pens can be helpful for children and their parents, and young adults to assist with carbohydrate counting, teach the impact of exercise on glucose control, and educate them on important diabetes concepts (eg, timing of insulin, relationship between insulin and food, etc.). Moreover, in children with more than one caregiver involved, smart pens offer the ability to share data on insulin injections. In addition, in young adults with T1D on MDI who are at increased risk of diabetic ketoacidosis and severe hypoglycemia due to insulin omission or over stacking, smart pens can assist with bolus reminders and active IOB. The downloadable data can also identify these challenges and help clinicians and patients devise a personalized plan to mitigate hypoglycemia and hyperglycemia (Figure 1). 27
For those patients who want to transition from MDI to an insulin pump, smart pens may help to optimize insulin doses, ICR, and ISF. Conversely, the InPen can be considered for patients who are looking for a “pump holiday” but want to maintain the features of a pump such as IOB, and bolus and correction dose calculator. In addition, smart pens when combined with CGM may help patients adjust insulin when physically active as shown in a recent study of professional cyclists with T1D. 28 Smart pen and CGM data were used during a five-day race to adjust bolus and basal doses for physical activity and macronutrient intake.
Smart pens may also be advantageous in older adults with T1D or T2D on MDI to assess adherence.12,29 Older adults with diabetes are at increased risk of developing cognitive dysfunction and this can interfere with their ability to follow an insulin plan. 30 Smart pens can help provide reminders for insulin administration and assist patients who have difficulties with problem-solving (eg, cannot calculate insulin doses). Moreover, using smart pens with CGM may help clinicians simplify insulin regimens to reduce the risk of hypoglycemia, lower the treatment burden, and ensure that the patient and/or caregiver can follow the insulin plan consistently.
Patients with diabetes who have challenges with numeracy tend to have worse glycemic control. 31 Smart pens could help address this barrier with the use of a bolus dose calculator. The InPen syncs with a built-in dose calculator on the mobile app that can advise how much rapid-acting insulin and/or correction dose to administer based on either number of carbohydrates, meal size estimation (small, medium, or large), or fixed dosing. 32 Another advantage of smart pens is that insulin can be accurately delivered in small doses such as 0.1-0.5 unit increments.
In addition, smart pens may play a role in diabetes management of pregnant patients with T1D, T2D, or gestational diabetes. A multicenter randomized trial demonstrated that in pregnant patients with T1D, MDI plus CGM users compared to pump plus CGM users were more likely to achieve target A1c and have increase TIR by 24 weeks gestation.33,34 Therefore, smart pens may have the potential to improve adherence and achieve glycemic goals, which are important for maternal and fetal outcomes. Clinicians can utilize the smart pen downloadable reports to educate patients on the relationships between insulin, nutrition, and physical activity throughout their pregnancy.
Furthermore, smart pens may be beneficial for patients with T1D or T2D who are hospitalized or admitted to nursing homes to provide accurate dosing and reduce the risk of errors. Insulin administration errors are common in hospitals and nursing homes, and can lead to serious consequences.35-37 Smart pens could help nursing staff record insulin delivery and administer the appropriate insulin at the correct dose and time. If used along with CGM, clinicians may also be able to better address glycemic excursions that occur with frequent changes in health status.
Challenges of Adopting Connected Smart Pens Into Clinical Practice
While smart pens have been available for the past decade, patients and the healthcare community have been slow to adopt and embrace this new technology. Currently, there are few clinical studies with published data showing benefits in adherence, improvement in A1c, and TIR. 20 Other barriers to using smart pens include limited availability of the devices worldwide, higher cost compared to traditional insulin pens, additional training costs, and technology inertia. 3 In addition, clinicians need to be able to download the smart pen data, have the necessary skills to interpret the data, and formulate an appropriate insulin plan. Furthermore, clinicians and patients need to take into account the user experience of smart pens and their respective apps. There are also patient-specific barriers that can restrict the use of smart pens (Table 2).
However, the use of smart pens is gaining recognition. The American Diabetes Association recently published guidelines recommending the use of smart pens in diabetes management. 38 In order to disseminate the use of smart pens, there need to be education and guidelines for patients and healthcare providers on how to use the technology, interpret the data, and implement appropriate management plans as well as the benefits of smart pens. Healthcare providers also need to recognize the advantage of combining smart pens with CGM for real-time data to create individualized treatment plans for their patients. In addition, smart pens would be more readily adopted if costs were substantially reduced or subsidized and covered by Medicare. Finally, more clinical studies are needed to investigate the benefits of smart pens and their impact on diabetes outcomes, cost-effectiveness, and quality of life.
Conclusion
Connected smart pens have the potential to help patients with T1D or T2D on insulin therapy improve adherence, glycemic control, and address challenges related to diabetes management. Further education of healthcare providers and patients, cost reduction, integration with CGM, and large clinical studies are needed to advance this technology for widespread use. The technology of smart pens is still in the early development phase with untapped potential that can enhance diabetes care and change the way how MDI therapy is used in the future.
Footnotes
Abbreviations
CE, Conformité Européenne; CGM, continuous glucose monitoring; FDA, U.S. Food and Drug Administration; ICR, Insulin to carbohydrate ratio; IOB, Insulin on board; ISF, Insulin sensitivity factor; MDI, multiple daily injections; NFC, Near-field communication; TIR, Time in range; T1D, Type 1 Diabetes; T2D, Type 2 Diabetes.
Author(s) contributions
All authors accept responsibility for all aspects of the work.
Conception of the work: SS and ET
Drafting and revising: SS, MM and ET
Final approval of the version: SS, MM and ET
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ET is a consultant for Medtronic. MM is a consultant for Sanofi and Lilly. SS declares that there are no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: ET and MM’s time is covered by NIH DP3 Grant (1DP3DK112214-01). SS does not receive financial support for research, authorship, and/or publication of this article.
