Abstract
Background:
Type 2 diabetes mellitus (T2DM) and dementia are two of the leading chronic diseases in aging and are known to influence each other’s disease progression. There is well-established evidence that T2DM increases the risk for cognitive decline and dementia. At the same time, people with cognitive changes or dementia can find it difficult to manage their diabetes, resulting in hyper- or hypoglycemic events which can exacerbate the dementia disease progression further. Monitoring of glucose variability is, therefore, of critical importance during aging and when people with T2DM develop dementia. The advent of continuous glucose monitoring (CGM) has allowed the monitoring of glucose variability in T2DM more closely. The CGM seems to be highly feasible and acceptable to use in older people with T2DM and has been shown to significantly reduce their hypoglycemic events, often resulting in falls. Less is known as to whether CGM can have a similar beneficial effect on people with T2DM who have cognitive impairment or dementia in community or hospital settings.
Aims:
The current perspective will explore how CGM has made an impact on T2DM management in older people and those with comorbid cognitive impairment or dementia. We will further explore opportunities and challenges of using CGM in comorbid T2DM and dementia in community and hospital settings.
Introduction
Diabetes—Background, Pathophysiology, Dementia Linkage
Type 2 diabetes mellitus (T2DM) and dementia are aging-associated chronic and progressive conditions.1,2 The T2DM is a complex metabolic disorder affecting 483 million adults worldwide 3 with high mortality and morbidity rates. 4 The pathophysiology of T2DM is attributed to insulin resistance as a subset of the metabolic syndrome, 5 including obesity and dyslipidaemia, 6 due to an imbalance between energy intake and expenditure to affect the metabolism of nutrient-storage. Not surprisingly, lifestyle factors (eg, obesity, low physical activity, unhealthy diet) are a major contributor to the risk of insulin resistance and T2DM. 7 Furthermore, T2DM has significant short-term and long-term health effects. 8
Key short-term effects of T2DM in aging are hypoglycemia and hyperglycemia. Hypoglycemia, defined as plasma glucose levels below 70 mg/dL or 3.9 mmol/L, is highly prevalent in aging. In the United Kingdom, the prevalence of both mild, moderate, and severe hypoglycemia is 20%. 9 Hypoglycemia often results in changes to cognition, confusion, falls, fractures,10,11 and anxiety, and in severe cases delirium, comas, cardiovascular complications, and even death. 12 Similarly, hyperglycemia can also result in life-threatening acute consequences such as diabetic ketoacidosis (DKA), hyperglycaemic hyperosmolar syndrome (HHS), coma, and death. 13 Long-term T2DM effects are related to microvascular and macrovascular conditions, 8 such as myocardial infarction, stroke, retinopathy, nephropathy, and neuropathy.14,15 Of particular interest in terms of long-term risk of T2DM, dementia has emerged as a key factor in recent years. It is now well established that T2DM can increase the risk of cognitive impairment and dementia significantly in the long-term.16,17
Dementia—Background, Pathophysiology, and Type 2 Diabetes Mellitus Linkage
Dementia is a clinical syndrome associated with changes to the cognition, including memory, which affects everyday activities,18,19 with Alzheimer’s disease (AD) the most common form. An estimated 55 million people worldwide have dementia, which is predicted to increase to ~130 million by 2050. 20 Dementia is thought to be caused by the accumulation of amyloid-β proteins and phosphorylated tau, which can be exacerbated by insulin, cardiovascular, and inflammatory risk factors. 21 The largest risk factors for dementia are age, high blood pressure, diabetes, cerebrovascular disease, and obesity. 1 Lifestyle factors such as nutrition and physical activity status are now regarded as some of the biggest risk factors for future AD, with interventions targeted toward reducing cardiovascular and metabolic risk factors. Of particular interest in this regard is that people with T2DM receiving healthy lifestyle advice including a balanced diet and physically active lifestyle show a 30% lower risk for cognitive decline, 22 even just by adhering for six months to a multimodal lifestyle intervention that targeted improved dietary habits and regular physical activity behaviors. 23
It is well established that T2DM increased the risk for dementia, even though the exact pathophysiology links are still being established. Still, T2DM is associated with 1.5- to 2.5-fold higher risk of dementia.24,25 Similarly, higher HbA1c levels are attributed with higher risk of dementia incidence among patients with T2DM. 26 It is estimated that 13% to 20% of people with dementia have comorbid diabetes, 27 although an exact prevalence has not been established, due to many people with dementia having undiagnosed T2DM. At the same time, people with cognitive impairment or dementia are at higher risk for hypoglycemia, 28 since it is often not clear whether cognitive or behavior changes are due to the dementia or hypoglycemia 11 with older people with T2DM most vulnerable for severe hypoglycemia. 24 Furthermore, hypoglycemia increases the risk of falls and fractures in older people with T2DM, 29 which can further exacerbate cognitive decline due to potential delirium.10,11 Finally, people with both T2DM and dementia experienced more hypoglycemic events with coma and used insulin more often than those without dementia. 30 Similarly, people who attended hospital by ambulance due to severe hypoglycemia were significantly older adults. 31 From the above, it emerges that diabetes management is critical not only for T2DM and its complications but also that people with cognitive impairment or dementia who are particularly vulnerable to significant changes in glucose variability (GV) resulting in hyperglycemic or hypoglycemic complications.32-34 Although annual neurocognitive tests such as Montreal Cognitive Assessment (MoCA) is suggested to older people having type 2 diabetes, 35 Mini Mental State Examination (MMSE), Abbreviate Mental Test (AMT), Addenbrookes Cognitive Examination (ACE) could be also recommended to the same population who are at relatively higher risk for dementia.
Glucose Variability
Glucose variability refers to fluctuations in plasma glucose levels throughout the day.36,37 Although HbA1c has been widely accepted as a gold standard biochemical parameter for glycemic control, it is now accepted that it mostly reflects average glycemia over the last three months.38,39 Based on this, HbA1c is now more commonly regarded as indicative of long-term dysglycemia, whereas GV is indicative of more short-term fluctuations in blood glucose.37,40 The GV has, therefore, has emerged as a key factor in managing short-term dysglycemia, and its measurement has become increasingly crucial. The current gold standard is still to measure blood glucose via finger pricks. However, finger pricks are only conducted a few times a day which might miss GV and potential adverse events in between those measurements. Instead, continuous glucose measurements became increasingly popular to monitor GV (Figure 1).36,41
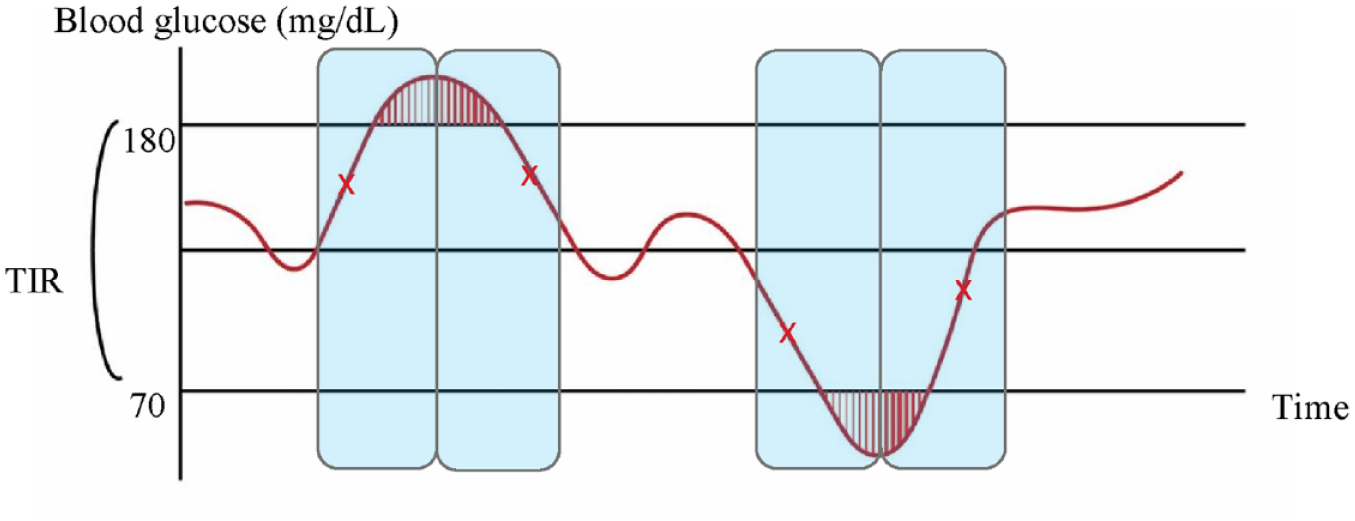
The blood glucose fluctuations with the time both captured by finger pricks and for CGM alarms/alerts. Diagram illustrating differences in glucose fluctuations via finger prick measurements or CGM. The red shaded area shows the dysglycemia. The red “x” refers the finger prick measurements., TIR = time in range, time spent in targeted blood glucose (70-180 mg/dL). The blue-shaded area shows the time that CGM receiver or smartphone sends alarms/alerts when it detects the interstitial glucose is upgoing/descending trend in a short time. The CGM can send notifications when the interstitial glucose of the user is increasing or decreasing rapidly (it also provides the speed with two modes) even if they are in euglycemia.
Continuous Glucose Monitoring
Continuous Glucose Monitoring (CGM) devices allow real-time monitoring of interstitial glucose. Even though CGM sensors are now widely used by people with type 1 diabetes, they are only more recently being adopted for T2DM, with 70% of people with type 1 diabetes using them in the United States, whereas only 13% of people with T2DM used CGM.42,43 Relatively higher costs including initial set up and specially maintenance costs without financial support from government emerge as one of the main obstacles of increased frequency of using CGM sensors. 44 According to a recent research, older adults with type 1 diabetes using CGM were more likely to report impaired awareness of hypoglycemia and less likely to have cognitive impairment who benefit from diabetes technologies including insulin pumps, 45 and reduced nocturnal hypoglycemia may improve the cognition in adults with type 1 diabetes. 46
Despite the low uptake in T2DM, it is now well established that CGM is highly feasible and acceptable to people diagnosed with T2DM over 65 years, while simultaneously improving glycemic control and the detection of hypoglycemic episodes.27,47,48 There are now numerous studies that have recommended the use of CGM sensors in the management of T2DM,37,49 in particular as they are more effective than finger prick results in capturing asymptomatic hypoglycemia events, 50 and can support reducing HbA1c in the long-term. 4 For instance, people with T2DM using CGM for 16 weeks showed lower HbA1c levels compared to those using only finger pricks. 51 Furthermore, the use of CGM sensor reduced overall hospitalization, including due to DKA, diabetes-related coma.52-54 Finally, since older age is one of the highest risk factors for dementia, it has been suggested to use CGM for T2DM management in older people might not only improve glucose control but also future dementia risk. 55 Unfortunately, due to the paucity of CGM data in older people with T2DM, it is yet to be established whether CGM might reduce future dementia risk, compared to existing finger prick glucose measurement methods.
Continuous Glucose Monitoring in Comorbid Type 2 Diabetes Mellitus and Dementia
Monitoring glycemic levels in people with diabetes and cognitive impairment/dementia is becoming increasingly important. Specifically, conducting finger prick testing and remembering to conduct those tests while also remembering whether medication has been administered, clearly poses a challenge to people with dementia, since their memory is commonly affected from the early stage of the diseases. In addition, both people with dementia and their carers might not recognize or mistake dysglycemia symptoms with changes to their cognition. The CGM emerges, therefore, as an ideal method to monitor and mitigate GV changes in people with T2DM and dementia. 47 There is also now evidence that using CGM is feasible and acceptable for people with comorbid diabetes-dementia, as well as their carers. 27
Carers are playing a significant role in the management of diabetes among people having cognitive impairment as well as providing an environment to interact and understand the cognitive needs of people with dementia. Hence, cognitive support by carers becomes crucial. 56 In the research including person-centered interventions on persons with dementia, educational support to carers and internal care team is found key in enhancing and maintaining quality of life. 57
Most of the existing CGM studies in this aspect have been conducted in the prodromal forms of dementia (eg, Mild Cognitive Impairment) or early stage dementia, at which stage people might still be able to monitor and respond to hyperglycemic and hypoglycemic level CGM alerts. Less clear is whether people with moderate or severe cognitive dysfunction or dementia might still benefit from CGM. This is of particular interest, as people in the moderate or severe stages of dementia might benefit most from using CGM which can then inform their carer on whether glycemic levels need to be optimized. 58 In the interest of safety, Diabetes UK recommends that glucose reading less than 4 mmol/L should be treated in the frail population. 59 In addition to this, the suggested HbA1c target for people with comorbidities, cognitive impairment, and who are living in a care home is 59 to 69 mmol/mol, whereas glycemic target is addressed 4.9 to 7.2 mmol/L (90-130 mg/dL).60,61
In this regard, there is, to our knowledge, also no evidence yet whether carers of people with comorbid T2DM-dementia might play a role in monitoring CGM alerts—similar to parents for children with type 1 diabetes. In the following section, we highlight briefly, existing studies in patients comorbid T2DM-dementia in community and in-hospital settings.
Community-Based Continuous Glucose Monitoring in Comorbid Type 2 Diabetes Mellitus Dementia
In the community, CGM has shown that large glycemic fluctuations just after a 72-hours’ period can contribute to poorer cognitive outcomes. 62 Fasting glucose levels, postprandial peak glucose, and 24-hour area under the glucose curve obtained from CGM sensors have also been found to relate to cognitive function. 63 Importantly, glucose metrics as measured via CGM sensors were more closely associated with cognitive impairment than HbA1c or other GV factors. 64 Moreover, GV and glycemic excursions were strongly linked with cognitive impairment. 65
Time in range (TIR; accepted glycemic target: 70-180 mg/dL or 3.9-10 mmol/l) and time above range (TAR; time spent in hyperglycemia) obtained from CGM sensors are associated with cognitive performance; especially in executive function and working memory tests. 66 Higher time below range (TBR; time spent in hypoglycemia) and TAR (>13.9 mmol/L) were related to worse cognition in terms of memory, visuospatial ability, and executive functioning. 67 Hypoglycemia captured by CGM was very common in older people, even though the clinical relevance of such low glucose readings remains still unclear. 68 Nearly half of the hypoglycemic episodes in older adults are asymptomatic and emerging during the night, between dinner and breakfast.69,70 Similarly, community-based studies in older people, some diagnosed with dementia, using CGM sensors showed that most hypoglycemic episodes were missed during the night via finger pricks. Recently provided smart insulin pen, as another diabetes technology product, also supports CGM with improving diabetes management and providing less risk for hypoglycemia via its memory recording missed or extra doses, in particular people with cognitive impairment. 71
The CGM allows to capture those episodes and potentially mitigate adverse outcomes at night, such as falls.27,50,72 Furthermore, the failure to capture night-time hypoglycemia might be particularly exacerbated in older people who experience delirium, which is common in dementia, and in turn can lead to significant progression of the dementia or even death.73,74
Although there was no direct causality between TIR and cognitive impairment, there is evidence that well-managed TIR can improve cognition and other chronic complications via frequent glucose monitoring using CGM sensors. 75 A community-based CGM feasibility study involving older adults with cognitive impairment and diabetes, resulted in the high acceptability and feasibility of sensors perceived by the study population and carers according to their experiences. Despite the short study duration of two weeks, the study highlights the potential benefits of CGM usage for comorbid diabetes-dementia in the community. 27 Less clear is whether such benefits can also be seen in hospital settings, where a high percentage of in-patients are diagnosed with either diabetes, dementia, or both.
In-Patient Continuous Glucose Monitoring in Comorbid Type 2 Diabetes Mellitus-Dementia
Studies to date using CGM in comorbid T2DM-dementia have been exclusively conducted in the community, with hospital-based studies virtually non-existent. Current hospital guidelines on T2DM management are all based on finger prick GV monitoring, whereas CGM usage has been little explored. This is not trivial, as dysglycemia in this population might be even more difficult to detect and treat within hospitals.47,76 Not surprisingly, in-patient hypoglycemia has been associated with increased adverse in-patient outcomes, 77 such as prolonged length of stay, mortality, and readmission.
A recent study, using CGM in a hospital setting, indicated those with frailty managed with insulin or sulfonylurea showed the highest risk of delirium (4.8%), hypoglycemia (3.5%), cardiovascular disease (CVD) (20.2%), and hospitalization (49%). 78 The use of CGM might allow the detection of potential dysglycemic events in comorbid T2DM-dementia patients much earlier and alert the patient or health care professionals of such events in case of hypoglycemia particularly during the night. Despite the advantages, there are also challenges in using CGM within hospital settings, which we outline in Table 1.
Benefits and Challenges of Using Continuous Glucose Monitoring Sensors Among Individuals With Cognitive Impairment in Hospital Setting and After Hospitalization.

This table defines some benefits, opportunities, and challenges of CGM use in individuals with cognitive impairment in hospital setting.44,58,78,79
Abbreviation: CGM, continuous glucose monitoring.
The CGM sensors as a part of diabetes management have key benefits for people with cognitive dysfunction in hospital settings.80,81 In particular, it may not only improve cognitive outcomes in hospitalized people with cognitive impairment 81 but also hospital outcomes in terms of length of stay, readmissions, or even mortality. Therefore, using CGM should be considered as a part of usual care in order to improve functional and mental abilities and as a support system for each patient. 82 Future research in this direction is, therefore, urgently needed to establish the evidence base for CGM in hospital settings.
Conclusion
In summary, management of T2DM in older adults with cognitive impairment is critical for providing better clinical outcomes including less diabetes-related complications and improved cognitive function. Such management requires close, real-time monitoring of GV, for which CGM is the ideal candidate. This is particularly pertinent for people with dementia who have more moderate or severe cognitive impairment, which makes it more challenging to identify symptoms of hypoglycemia. Existing community-based studies in T2DM-dementia using CGM show promising potential for better management of T2DM in those people. By contrast, there are virtually no studies on CGM in hospitalized people diagnosed with comorbid T2DM-dementia despite its potential benefits.
The CGM has a significant potential impact to reduce cognitive impairment and dementia risk via improving diabetes management and increasing glycemic awareness. It also has key opportunities to improve glucose management for health care professionals and carers during hospitalization and after discharge. Importantly, CGM sensors have been found feasible, useful, and significantly effective in capturing hypoglycemic events, allowing health professionals to manage the T2DM more closely and mitigate any adverse health or hospital outcomes. However, if usage of CGM in hospitals would be adopted, it would require a significant education program for health care professionals, which currently does not exist.
For the future, there is an urgent need to establish additional evidence for the benefits and challenges of CGM usage in the community and in-hospital settings for comorbid T2DM-dementia. Specifically, CGM usage in those populations and their long-term feasibility are needed and should be complemented by randomized controlled trials in this area. The potential benefits of using CGM in comorbid T2DM-dementia are, therefore, highly promising but underexplored.
Footnotes
Abbreviations
AD, Alzheimer’s disease; ADA, American Diabetes Association; BG, blood glucose; CGM, continuous glucose monitoring; DKA, diabetic ketoacidosis; GV, glucose variability; HbA1c, hemoglobin A1c; HHS, hyperglycemic hyperosmolar syndrome; SMBG, self-monitoring of blood glucose; T2DM, type 2 diabetes; TAR, time above range; TBR, time below range; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
