Abstract
Background:
Current guidelines suggest utilizing continuous glucose monitoring (CGM) to improve hemoglobin A1c (HbA1c) in patients with diabetes. Financial cost remains a barrier to implementation. Medicare coverage criteria include all patients with diabetes treated with at least one injection of insulin per day, while Washington Medicaid is more restrictive. There remains a paucity of literature examining effectiveness of CGMs on clinical outcomes among patients with type 2 diabetes with lower incomes.
Methods:
This is a single-center, retrospective, observational study including adults with type 2 diabetes receiving institutional financial assistance for CGMs. A cohort with no CGM use is included for comparison. The primary outcome is change in HbA1c approximately three months after CGM implementation from baseline. Secondary outcomes include mean differences in number of antidiabetic agents and changes in insulin dose prior to and after CGM implementation.
Results:
Among the CGM cohort, most patients were of Hispanic ethnicity (77%) and a majority had no insurance (77%). The average HbA1c prior to CGM implementation was 8.3% and three months post-CGM was 7.7%, with a mean difference of −0.6% (P = .004). There were no statistically significant differences in the average number of antidiabetic agents, total daily dosages of insulin, or mean differences in the number of emergency room visits or hospitalizations prior to and post-implementation of a CGM.
Conclusion:
Overall, there is a statistical and clinical improvement in HbA1c before and after implementation of CGMs in patients with type 2 diabetes who meet Medicaid criteria for CGM coverage receiving financial assistance.
Introduction
According to the Centers for Disease Control and Prevention, an estimated 38.4 million people in the United States had diabetes in 2021 1 with a total cost of diabetes estimated to be around $327 billion in 2017. 2 With the discovery of insulin as a primary hormone in glucose regulation and its eventual mass production, insulin has been a cornerstone of diabetes therapy. 3 The use of insulin therapy provides blood glucose control with beneficial macrovascular and microvascular outcomes in both patients with type 1 and type 2 diabetes mellitus while carrying a risk of hypoglycemia.4-6 Current American Diabetes Association (ADA) guidelines recommend that patients with diabetes, and especially those on insulin, self-monitor blood glucose (SMBG). The ADA guideline suggests that either real-time continuous glucose monitoring (CGM) or intermittently scanned CGM should be offered to patients with diabetes on insulin, 7 with existing literature to support the use of CGM in improving hemoglobin A1c (HbA1c) as a measure of blood glucose control in both patients with type 1 and type 2 diabetes.8-12 Continuous glucose monitoring provides advantages over SMBG that include the reduction of finger pricks for glucose readings, the availability of the immediate glucose level to the user, and the accessibility of glycemic data and trends over time for a more extensive glimpse into glycemic control. 13 However, one of the major barriers to CGM access is the financial cost of the devices.
The prevalence of diabetes is highest among adults in the United States with family income levels that are below the federal poverty level, with about 13.1% having diabetes in 2021. 1 Currently, under Medicare, individuals with diabetes treated with at least one injection of insulin a day or individuals not using insulin but with a history of symptomatic hypoglycemia are eligible for CGM coverage. 14 Under Washington Medicaid, CGM coverage criteria are more restrictive for individuals with type 2 diabetes than the Medicare criteria. With Medicaid, individuals need to be on intensive insulin therapy, defined as at least three injections of insulin per day, and testing SMBG at least four times daily or experiencing severe hypoglycemia defined as blood glucose less than 50 mg/dL while adherent to intensive insulin therapy and SMBG testing. 15 Real-world evidence suggests CGMs provide improved clinical outcomes in patients with type 1 and type 2 diabetes including those with public insurance.16,17 Indeed, one study of pediatric patients with type 1 diabetes and Medicaid demonstrated lower A1c and increased percent time in range with the use of CGMs. 18 However, there remains a paucity of literature examining the effectiveness of CGMs among adult patients with type 2 diabetes with lower incomes that would meet eligibility criteria for Medicaid.
Washington state requires all hospitals to provide financial assistance to individuals and families that meet certain income requirements and do not have other means of medical coverage. Harborview Medical Center, a large county hospital in Seattle, Washington, has provided CGM coverage through their financial assistance program since September 2022. To qualify for the institutional financial assistance program, patients with type 2 diabetes must meet certain income requirements, with financial assistance offered up to 400% of the federal poverty level. In addition, patients need to be on at least three injections of insulin per day and utilize SMBG four times daily, consistent with Washington state Medicaid CGM coverage criteria. This study will aim to look at an underserved, uninsured population receiving financial assistance who meet criteria for CGM and evaluate the use of CGMs on glycemic control efficacy and adherence outcomes. Implications of this study may provide further evidence of CGM benefits in individuals with lower incomes and the need to expand access to CGM through financial assistance programs and reform current Washington Medicaid coverage criteria.
Methods
This was a single-center, retrospective observational study conducted at an urban and tertiary academic medical center. All patients (aged ≥ 18 years) with type 2 diabetes mellitus prescribed at least three injections of insulin per day and receiving institutional financial assistance for the Freestyle Libre 2 CGM between September 2022 and September 2023 were included in the CGM cohort. The comparator group consisted of patients from the same clinic with type 2 diabetes mellitus prescribed insulin receiving institutional financial assistance without any CGM use in the same time period.
Data were collected from electronic health records. Date of CGM implementation was patient-specific and based on dispense date at the institution’s outpatient pharmacy. Outcomes data closest to the implementation date were used as baseline and compared with data at months 3 and 6 after initiation of CGM use. For the comparator arm, the first date within the study timeframe with a collected HbA1c served as the baseline, and outcomes data were compared at month 3 (primary outcome only) and month 6 from baseline.
The primary end point was the change in HbA1c three months after CGM implementation (post-CGM) from baseline HbA1c. Secondary end points included mean difference in number of antidiabetic agents, mean change in total daily insulin dosage, mean difference in number of emergency department (ED) visits and hospitalizations related to diabetes mellitus, and mean difference of blood glucose readings within target based on SMBG reports pre-CGM and CGM reports post-CGM.
Descriptive statistics were used to summarize baseline characteristics. Statistical analyses were performed within the cohort using the Wilcoxon signed-rank test for continuous paired data. Formal statistical analyses were not performed between the two cohorts.
This study was approved by the academic health center institutional review board.
Results
A total of 22 patients were included in the CGM cohort with a median age of 58.5 years and 59% female. Thirty patients were included in the comparator group with a median age of 55.5 years and 47% female. The CGM population was predominantly white (77%) with most patients being of Hispanic ethnicity (77%). Among this population, 77% of patients had no insurance. The comparator group was also predominantly white (63%) with most patients being of Hispanic ethnicity (77%), and 83% of all patients had no insurance. All patients included had type 2 diabetes mellitus and the median time since their diagnosis of diabetes mellitus was 17 years (CGM cohort) and 13 years (comparator group). A majority of the CGM patients had hypertension (73%) and dyslipidemia (50%) as comorbidities, while less common comorbidities included chronic kidney disease (27%), heart failure (18%), and non-alcoholic steatohepatitis (18%). Similarly, the most common comorbidities were hypertension (53%) and dyslipidemia (37%) among the comparator group (Table 1).
Patient Characteristics.
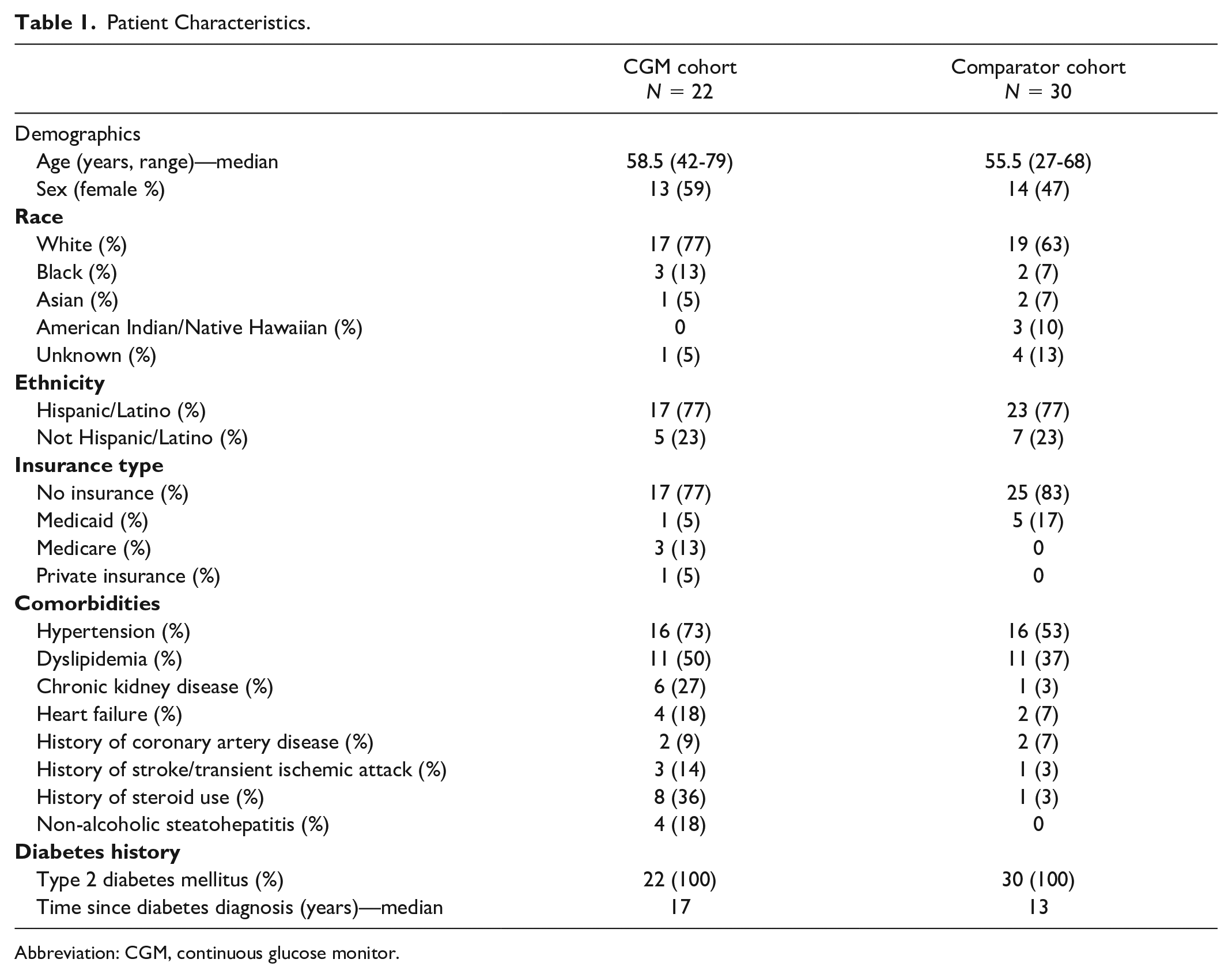
Abbreviation: CGM, continuous glucose monitor.
The average HbA1c prior to CGM implementation was 8.3%, and the average HbA1c both three and six months post-CGM implementation were 7.7%. There was a statistical difference in the primary outcome of change in HbA1c three months post-CGM implementation from baseline HbA1c (mean difference of −0.6%, P = .004) (Table 2). For the comparator group, average HbA1c at baseline was 8.8%, with the average HbA1c being 7.9% and 8%, three and six months from baseline, respectively. There was a statistical difference in the change in HbA1c three months from baseline (mean difference of −0.9%, P = .005) in the comparator group (Table 2).
Primary Outcome.

Abbreviations: CGM, continuous glucose monitor; HbA1c, hemoglobin A1c.
For secondary outcomes, there was no statistically significant difference in the average number of antidiabetic agents patients were on three months (2.45 agents, P = .484) or six months (2.5 agents, P = .374) post-CGM compared with pre-CGM (2.36 agents). Among the comparator cohort, there was also no statistically significant difference in the average number of antidiabetic agents patients were on six months from baseline (2.4 agents, P = .3978) compared with baseline (2.57 agents). All patients in the CGM cohort were on insulin, with the next most prescribed agent being metformin followed by a glucagon-like peptide-1 receptor agonist (GLP-1 RA) (Table 3). A majority of patients in the comparison group were on insulin at baseline (83%), with the next most prescribed agent being metformin followed by a sodium-glucose cotransporter-2 (SGLT-2) inhibitor and GLP-1 RA (Table 3). In terms of the average total daily dose of insulin, there was no statistically significant difference three months post-CGM (77.3 units, P = .219) or six months post-CGM (74.8 units, P = .575) compared with pre-CGM (74.6 units). Over half the patients were on insulin glargine for basal insulin and most patients were on either insulin lispro or insulin aspart for bolus insulin (Table 4). Among the comparison group, there was a statistical difference in the average total daily dose of insulin six months from baseline (mean difference of −11.4 units, P = .0069). The majority of patients (67%) were on insulin glargine for basal insulin, and a majority (77%) were not on any short-acting insulin (Table 4). There was no statistically significant difference in the total number of ED visits and hospitalizations related to diabetes within the health system at three months (0 visits, P = .346) or six months (one visit, P = .773) post-CGM compared with pre-CGM (two visits).
Concomitant Antidiabetic Agents Over Time.
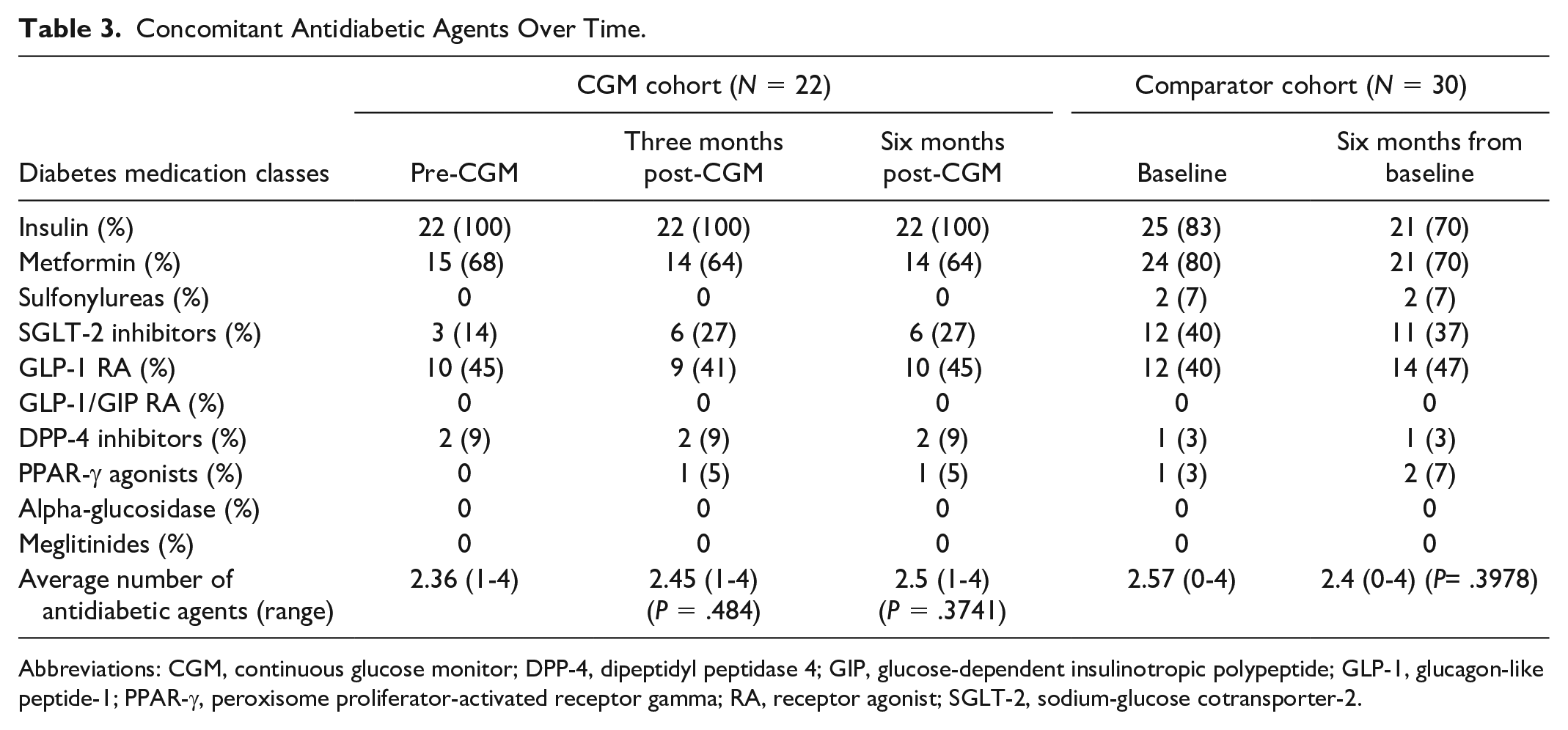
Abbreviations: CGM, continuous glucose monitor; DPP-4, dipeptidyl peptidase 4; GIP, glucose-dependent insulinotropic polypeptide; GLP-1, glucagon-like peptide-1; PPAR-γ, peroxisome proliferator-activated receptor gamma; RA, receptor agonist; SGLT-2, sodium-glucose cotransporter-2.
Insulin Usage Over Time. a
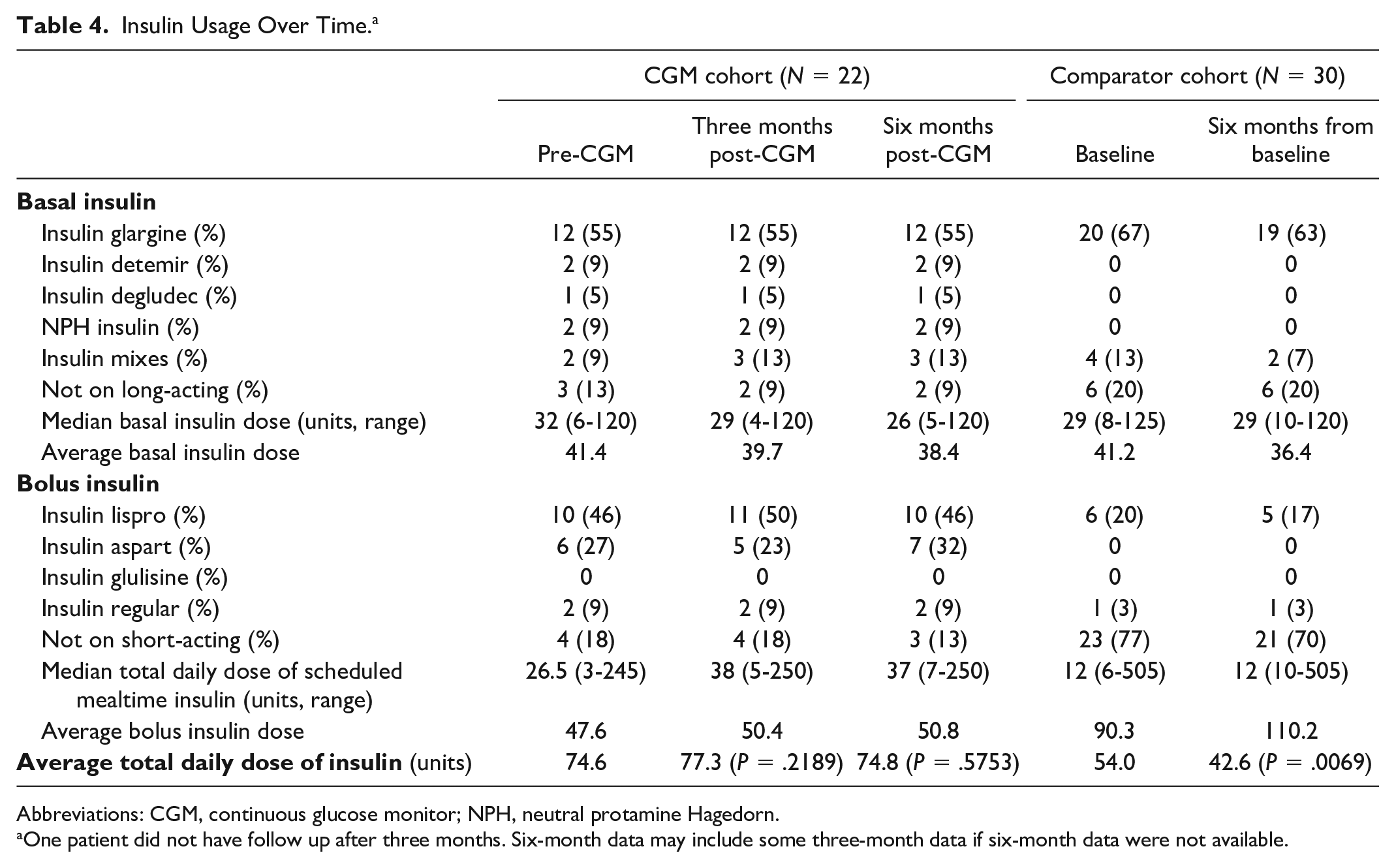
Abbreviations: CGM, continuous glucose monitor; NPH, neutral protamine Hagedorn.
One patient did not have follow up after three months. Six-month data may include some three-month data if six-month data were not available.
Of 22 patients, 15 patients had an SMBG report prior to CGM implementation and 20 patients had three- and six-month post-CGM reports for analysis. Self-monitor blood glucose readings within, above, and below target were compared with CGM time in range, above, and below target, respectively. There appears to be a statistically significant difference in the average blood glucose readings above and within target when comparing three or six months post-CGM to pre-CGM. More readings were within target after the implementation of a CGM at 48.9% (P = .017) at three months and 50.2% (P = .002) at six months compared with 18.9% pre-CGM. Fewer readings were above target after implementation of a CGM at 50.3% (P = .011) at three months and 49.4% (P = .002) at six months compared with 80.1% pre-CGM. Readings that were below target were not statistically different at three or six months after CGM implementation (Table 5).
Average Blood Glucose Readings Over Time. a
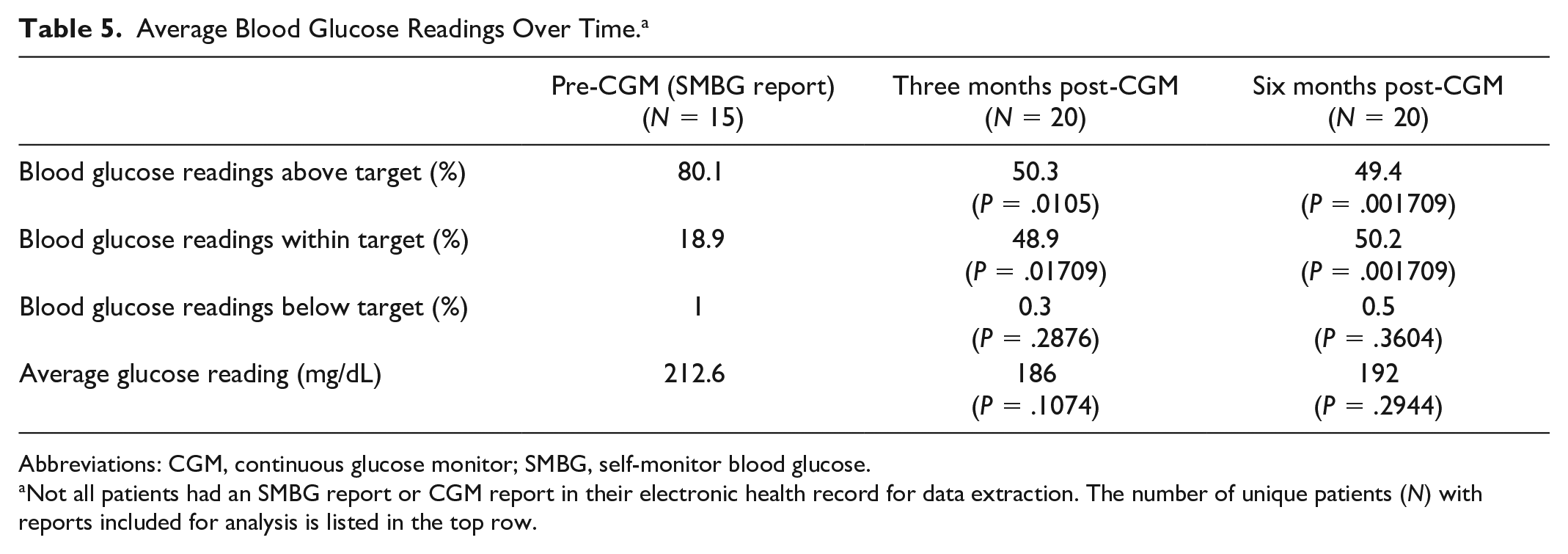
Abbreviations: CGM, continuous glucose monitor; SMBG, self-monitor blood glucose.
Not all patients had an SMBG report or CGM report in their electronic health record for data extraction. The number of unique patients (N) with reports included for analysis is listed in the top row.
Discussion
To the best of our knowledge, this is the first study evaluating the effectiveness of CGMs among patients with type 2 diabetes with lower incomes receiving institutional financial assistance for CGM coverage. Among a population predominantly with no insurance, there was a statistically significant difference in the change in HbA1c approximately three months after CGM implementation from baseline HbA1c. The mean difference in HbA1c of −0.6% after implementation of CGM is clinically relevant as previous trials have demonstrated reductions in mean HbA1c by 0.4% to 0.6%.8,19,20
There also appears to be a statistically significant decrease in the average blood glucose readings above target and increase of values within target at three and six months post-CGM compared with pre-CGM implementation. Numerically, fewer readings were above target, and more readings were within target after CGM implementation. While there were very few blood glucose readings below target pre-CGM, reflecting minimal time in a hypoglycemic state, these readings remained low three and six months after CGM implementation. From a safety perspective, the minimal time spent with hypoglycemia highlights an advantage of CGMs to bring awareness to patients as to when and how often the patients may be hypoglycemic relative to their insulin therapy.
Simultaneously, there were no statistically significant differences in potential confounders that may also influence HbA1c, such as the average number of antidiabetic agents or the average total daily dose of insulin with the study population. Despite no substantial differences in the average number of antidiabetic agents prescribed, there was a 13% increase in the use of SGLT-2 inhibitors at three months post-CGM compared with pre-CGM, which could contribute to the substantial decrease in HbA1c seen in the CGM cohort.
Overall, these results may reflect greater patient agency over their own diabetes management. Having access to real-time glucose levels and trends allows patients to track the effects of their lifestyle, such as types of foods or activity levels, on their blood glucose. Consequently, this knowledge may help patients become more aware and engaged in their own care.9,21 While this study is not a randomized controlled trial and we cannot solely attribute the reduction in HbA1c to the implementation of the CGM, the lack of demonstrated effect of confounding factors that could influence glycemic control provides additional support for the impact of CGM implementation on HbA1c.
A group comprising patients from the same clinic in the same timeframe who were on institutional financial assistance and not on CGMs were identified to serve as a comparator cohort. The mean difference in HbA1c of −0.9% three months from baseline HbA1c was also statistically significant despite no CGM use. Unlike the CGM cohort, there was a statistical difference in one of the confounders. The average total daily dose of insulin substantially decreased at six months along with the percentage of patients on insulin and metformin. Simultaneously, there were more patients on GLP-1 RAs six months from baseline. The decrease in insulin use along with an increase in GLP-1 RA usage may explain the substantial reduction in HbA1c seen among this cohort, as GLP-1 RAs have known efficacy in reducing HbA1c by 1% to 2%, 22 correlating well with the mean difference of −0.9% three months from baseline. Furthermore, there may be other factors contributing to the decrease in HbA1c without the use of a CGM. This comparator cohort appears to be slightly younger with less comorbidities and time since diabetes diagnosis, which may reflect a slightly healthier population and one that has a lower degree of insulin resistance than with the CGM cohort.
Limitations of this study include a small sample size conducted in a retrospective manner that limits definitive conclusions regarding the sole impact of CGM implementation as an intervention on HbA1c. In addition, only single-center records were reviewed, restricting the scope of safety data collected, namely, ED visits and hospitalizations that may be underrepresented should patients have had a diabetes-related ED visit or hospitalization outside the health system. Due to the limitations of retrospective chart review, other confounders that can have effect on HbA1c, such as lifestyle changes, were not adequately captured and examined in this study.
Strengths of this study include being the first study evaluating the effectiveness of CGMs among patients with type 2 diabetes with lower incomes receiving institutional financial assistance for CGM coverage. This study also provides further pragmatic evidence of the impact of CGMs in historically underrepresented racial and ethnic groups. Indeed, most patients qualifying for the financial assistance program were of Hispanic ethnicity or black race.
Future studies could consider a comparison group via propensity-score matching to compare HbA1c and other diabetes outcomes among patients with lower incomes with CGM implementation to those without CGM implementation. Despite the health-system financing these CGMs at 100%, there may potentially be overall cost avoidance with better glycemic control.23,24 Subsequently, additional studies should also attempt to assess the cost-benefit of an institution covering the CGMs at 100%.
In conclusion, implementation of CGMs in patients with type 2 diabetes mellitus on lower incomes receiving institutional financial assistance has a significant benefit to lower HbA1c, without changes in diabetes medications, and without apparent increase in hypoglycemia. In addition, the implementation of a CGM appears to have more glucose readings within range and fewer above range than pre-CGM implementation. There remain significant disparities in the initiation and use of CGMs, with lower use in historically underrepresented groups even after adjusting for socioeconomic status, education level, insurance, and health literacy. 25 Cost remains a significant barrier to the equitable use of CGMs. Harborview Medical Center’s financial assistance program covering 100% of the CGM cost aims to reduce one barrier for patients to promote health equity, with demonstrated improvement in glycemic control.
Footnotes
Abbreviations
ADA, American Diabetes Association; BMI, body mass index; CGM, continuous glucose monitor; DPP-4, dipeptidyl peptidase 4; ED, emergency department; eGFR, estimated glomerular filtration rate; GIP, glucose-dependent insulinotropic polypeptide; GLP-1, glucagon-like peptide-1; HbA1c, hemoglobin A1c; MD, medical doctor; NPH, neutral protamine Hagedorn; PharmD, pharmacist; PPAR-γ, peroxisome proliferator-activated receptor gamma; RA, receptor agonist; RN, registered nurse; SGLT-2, sodium-glucose cotransporter-2; SMBG, self-monitor blood glucose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tracy S. Tylee is a consultant for Abbvie and Monod Bio. All other authors have no financial or personal disclosures.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
