Abstract
Background:
We aimed to identify the normal range of glucose rates of change (RoC) observed in health and assess whether existing metrics of temporal glycemic variability (GV-timing), such as mean absolute glucose change (MAG) and continuous overlapping net glycemic action (CONGA), are predictive of abnormally rapid RoC in type 1 diabetes (T1D).
Methods:
We identified the normal range of RoC over one-hour intervals from continuous glucose monitoring (CGM) data of healthy individuals. Rapidly rising glucose was defined as RoC values above percentiles 99% (level 1, L1) or 99.9% (level 2, L2), and rapidly falling glucose as below 1% (L1) or 0.1% (L2). The percentage of time these thresholds are exceeded in a given individual is referred to as time in fluctuation (TIF). In a separate CGM dataset of 736 T1D individuals, we calculated TIF-L1 and TIF-L2, and compared them against corresponding values of MAG and CONGA.
Results:
The extremum percentiles of RoC observed in health are 0.1%: −80 mg/dL/h, 1%: −50 mg/dL, 99%: +56 mg/dL/h, and 99.9%: +89 mg/dL/h. The T1D individuals spend significantly more TIF at rates exceeding these thresholds (TIF-L1: median, 16.7% [interquartile range, 12.7-21.5], TIF-L2: 5.0% [3.1-7.8]) than healthy individuals (TIF-L1: 1.4% [0.6-2.8], TIF-L2: 0.0% [0.0-0.2]). Both MAG and CONGA are highly correlated with TIF-L1 and TIF-L2 (r > .95 in each pairwise comparison).
Conclusions:
Individuals with T1D spend significant time with glucose RoC exceeding those observed in health. Existing GV-timing metrics are strongly correlated with time with abnormal RoC. Incorporation of a GV-timing metric in clinical practice is recommended.
Keywords
Introduction
Glycemic variability (GV) is recognized as a potential causative factor of vascular complications in diabetes, either per se or via hypoglycemia as an intermediate step. 1 Several reviews and perspectives are available on GV, focusing on its measurement,2-7 pathophysiological mechanisms, 8 and association with vascular complications.1,9-11 The GV can manifest on short (minutes to days) and long (months) timescales. This article is focused on short-term GV, which we refer to simply as GV.
GV is characterized by 2 components: magnitude and timing.2,12 The GV-magnitude relates to the extent of glycemic deviations from a mean or baseline value. The GV-timing relates to the frequency of glucose fluctuations and the rate of change (RoC) of glucose during these fluctuations. Both magnitude and timing are recognized as clinically significant; 2 however, the only GV metrics currently used in clinical practice (as determined by international consensus)13,14 are measures of magnitude: the glucose standard deviation (SD) and the percentage coefficient of variation (CV) (Supplementary Table 1). As DeVries 15 highlighted over a decade ago, both of these metrics are independent of the frequency of fluctuations. This is illustrated in Figure 1a, which shows two hypothetical glucose profiles, P1 and P2, whereby mean glucose and SD are identical, but P2 exhibits a higher frequency of fluctuations.
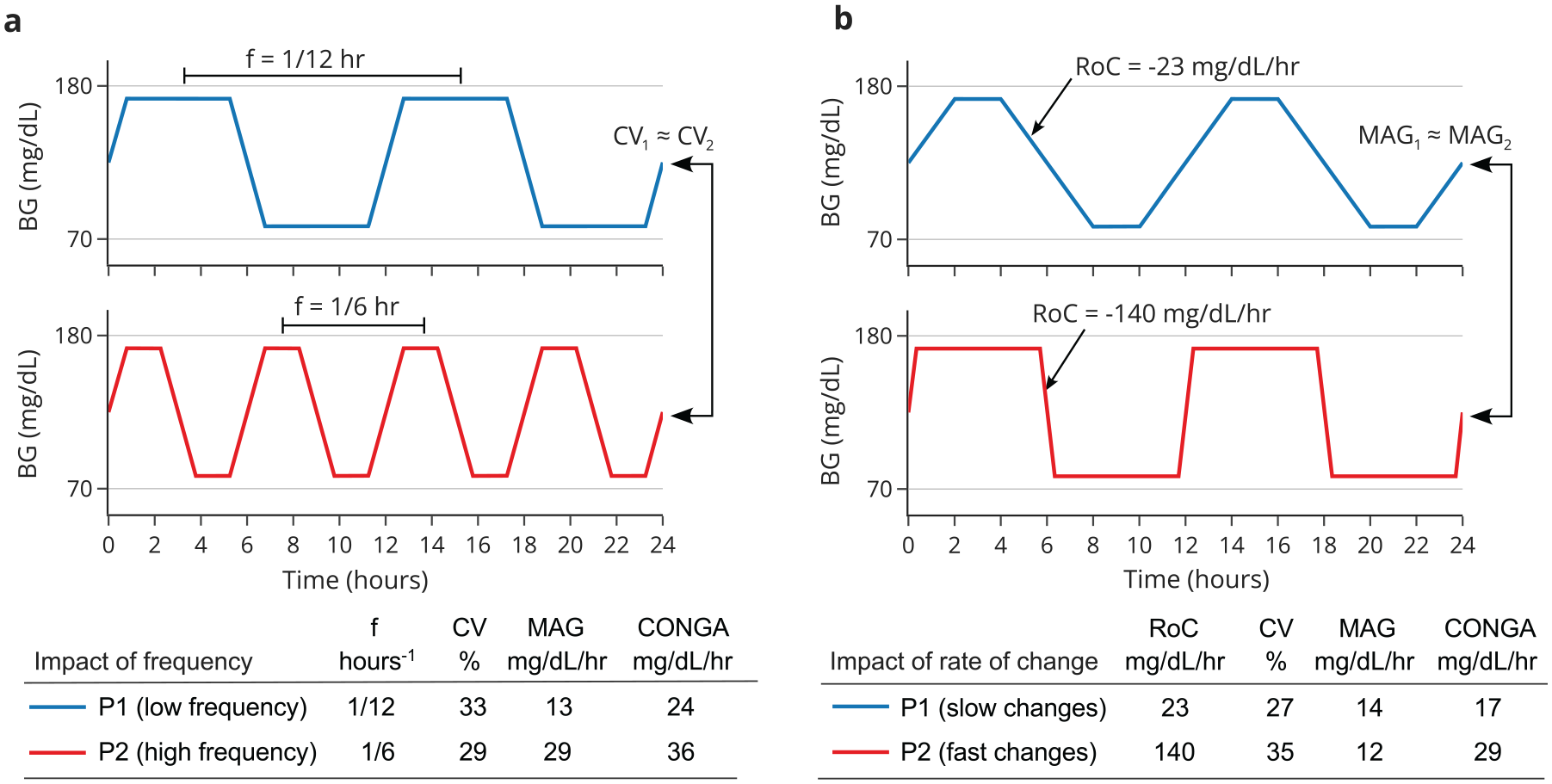
Impact of (a) frequency of fluctuations and (b) rate of change (RoC) on glycemic variability metrics (CV, MAG, CONGA). (a) Patient P2 exhibits more frequent fluctuations than P1, and thus greater variability. This is reflected by increased MAG and CONGA (measures of timing) but not by CV (a measure of magnitude). (b) Patient 2 (P2) exhibits faster rates of change in glucose than P1 (but with the same frequency and magnitude). This is not reflected by MAG, which is approximately equal in each case, since MAG exclusively measures the mean RoC across the entire period (ie, including both the periods of transition and the in-between periods of stability). It is partly reflected by CONGA, which has some sensitivity to outliers in RoC; however, the sensitivity is modest: a 500% increase in RoC results in only 60% increase in CONGA.
Several GV-timing metrics have been proposed in the literature.16-22 The most established of these are mean absolute glucose change (MAG) 23 and continuous overlapping net glycemic action (CONGA) 24 (Supplementary Table 1). The MAG is defined as the average absolute change of glucose over one-hour intervals. The CONGA is defined as the SD of the blood glucose changes over n-hour intervals, such that CONGA-1, CONGA-2, and CONGA-4 are the SDs of glucose differences measured 1, 2, and 4 hours apart, respectively. In this article, we will only consider one-hour intervals, and so CONGA-1 is referred to simply as CONGA.
As measures of GV-timing, both MAG and CONGA are sensitive to the frequency of glucose fluctuations; this is shown in the aforementioned Figure 1a. However, as both metrics primarily reflect the average RoC, they are theoretically not necessarily sensitive to infrequent, rapid glucose changes. This is illustrated in Figure 1b, which shows that, for a given frequency of fluctuations, MAG is in fact entirely insensitive to RoC, and CONGA is only mildly sensitive to RoC. This is because the fast changes during the periods of transition in profile P2 are offset by the stable periods in between, resulting in a mean RoC that is identical to that of profile P1. Put another way, MAG in fact measures the glucose throughput, and so whether transitions between glucose levels happen relatively slowly (P1) or quickly (P2), the throughput is unchanged. The CONGA, on the contrary, does exhibit some sensitivity to RoC, due to the nonlinear (quadratic) term in its mathematical formula (Supplementary Table 1 and Supplementary Figure 1). However, this sensitivity is modest; for example, in Figure 1b, a 500% increase in RoC only results in a 60% increase in CONGA. This example is contrived for illustrative purposes; however, such dynamics could arise in practice. For example, profile P2 in Figure 1b could arise if a patient consumes fast-acting carbohydrates or misses an insulin dose (resulting in a fast glucose rise) and later applies a large correction dose (resulting in a fast glucose fall). This highlights a potential limitation of existing GV-timing metrics: despite the general consensus that slow steady changes in glucose are preferable to rapid changes, existing GV-timing metrics do not necessarily penalize a profile like P2 (fast changes) more than a profile like P1 (slow changes). Thus, an objective of the present study is to establish whether existing GV-timing metrics are in fact correlated with rapid RoC values in real-world data of T1D individuals, despite this not being guaranteed theoretically.
It is generally believed that fluctuations within the normal range of glucose levels (ie, between 70 mg/dL [3.9 mmol/L] and 140 mg/dL [7.8 mmol/L] or even 180 mg/dL [10.0 mmol/L]) are clinically benign. This is based on the observation that healthy individuals exhibit fluctuations in that range. However, a relatively unexplored question is whether fluctuations in diabetes occur at rates exceeding those observed in healthy individuals. Studies have shown that metrics including MAG and CONGA are higher in diabetes than in health, 24 with little or no overlap between the lowest percentile of individuals with diabetes and the highest percentile of nondiabetic individuals. However, those findings do not necessarily prove that individuals with diabetes reach higher RoC values, only that the average RoC is higher. Last, a recent study proposed using the percentage of time in which glucose changes over 15-minute intervals exceed a certain threshold as a metric of temporal GV; however, that study did not quantify the equivalent percentiles of 15-minute changes observed in nondiabetic individuals, and so the physiological significance of the chosen threshold is unclear. Thus, another objective of the present study is to quantify and compare the distributions of RoC in T1D and nondiabetic subjects.
In summary, the objectives of this article are twofold: first, to compare the distributions of RoC values, and not just the mean RoC, between the T1D and nondiabetic populations; second, to determine whether existing GV-timing metrics reflect abnormally high RoC in T1D. We achieve this by defining the time in fluctuation (TIF) as the percentage of time spent at RoC exceeding thresholds routinely observed in health. We then compare the TIF values between the T1D and nondiabetic subjects, and use regression analyses to evaluate correlations between TIF and GV-timing metrics, MAG and CONGA.
Methods
Datasets
Two open-source CGM datasets were retrospectively analyzed in this study: a nondiabetic dataset 25 and a T1D dataset, T1DiabetesGranada. 26 Descriptive statistics of each dataset are shown in Table 1. The nondiabetic dataset consists of 153 individuals aged 7 to 80 years, with between three and ten days of data per person. Data were measured with Dexcom G6 sensors. The original remit of this dataset was to benchmark normal glycemia to support future studies. The T1D dataset consists of 736 individuals aged 12 to 81 years, with up to four years of CGM data per patient. Data were measured with Freestyle Libre 1/2 sensors. Further details of these datasets can be found in their associated publications.25,26 These sensors each have different measurement intervals and data persistence intervals: Libre 1 measures every five minutes and persists every 15 minutes, Dexcom G6 measures and persists every five minutes, and Libre 2 measures every minute and persists every five minutes along with any minute that the device is scanned. To ensure consistency in the current analysis, both datasets were resampled to five-minute granularity. Missing data for contiguous periods of up to 15 minutes were imputed via linear interpolation.
Descriptive Statistics of Patient Characteristics and Glycemic Metrics.
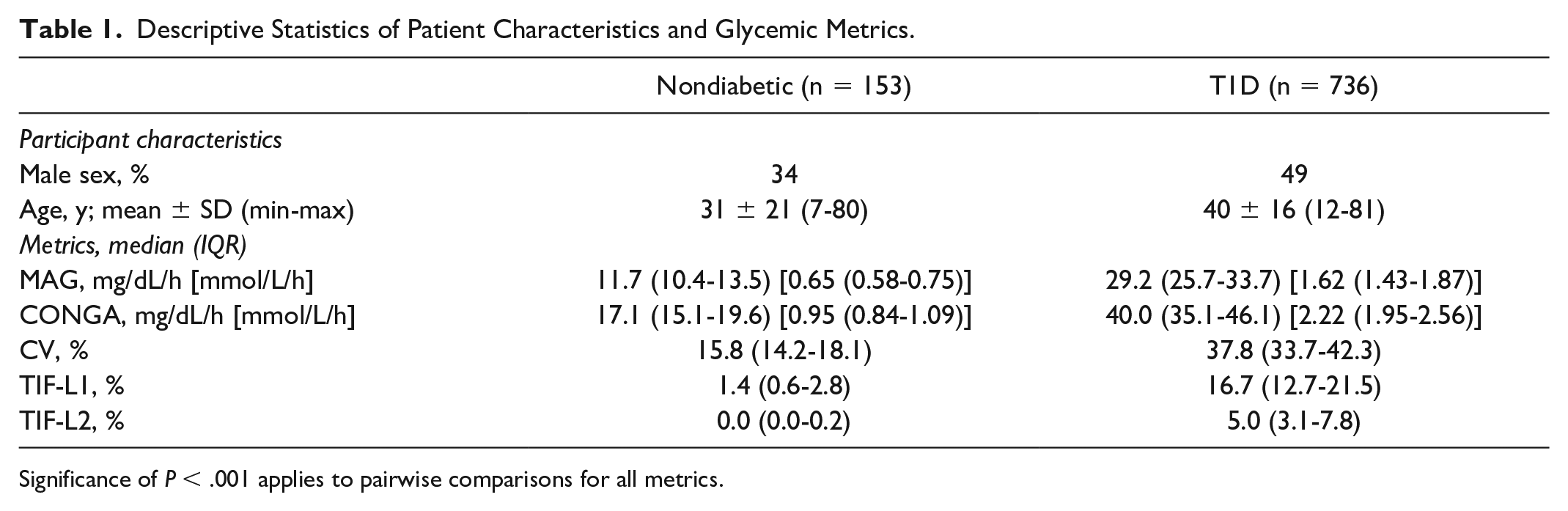
Significance of P < .001 applies to pairwise comparisons for all metrics.
Identification of Normal Range of RoC
A normal range of RoC was defined based on percentiles of the nondiabetic CGM data. The RoC was calculated as the change in blood glucose over hourly intervals,
Calculation of Metrics
Glycemic metrics MAG and CONGA for each individual in the T1D dataset were calculated based on their published formulae (Supplementary Table 1). The TIF for each individual in either dataset was defined as the percentage of time RoC values were below the RoC values corresponding to 0.1% (L2) and 1.0% (L1), or above the RoC values corresponding to 99.0% (L1) or 99.9% (L2) of the nondiabetic dataset.
Statistical Analyses
Descriptive statistics were calculated for the patient characteristics and glycemic metrics in each dataset. The datasets were compared using independent sample t-tests. Univariate linear regression analyses were performed to examine the associations of MAG, CONGA, and CV with TIF-L1 and TIF-L2. The associated Pearson correlation coefficient and 95% confidence intervals were also calculated. For comparison, the same analyses were performed on the relationship between mean glucose and TAR-L1 and TAR-L2.
Analysis Software
The Python programming language was used for all stages of the analysis, including data processing and calculation of glycemic metrics (Pandas), 27 and statistical analysis (SciPy). 28
Results
Normal Range of RoC
The extremum percentiles of RoC observed in health are 0.1%: −80 mg/dL/h (−4.4 mmol/L/h), 1%: −50 mg/dL/h (−2.8 mmol/L/h), 99%: +56 mg/dL/h (+3.1 mmol/L/h), and 99.9%: +89 mg/dL/h (+4.9 mmol/L/h). These RoC values are used to define the thresholds for abnormal rates of change, namely L1: <−50 or >55 mg/dL/h (<−2.8 or >3.1 mmol/L/h) and L2: <−80 or >89 mg/dL/h (<−4.4 or >4.9 mmol/L/h). The percentiles of RoC in each dataset are shown in Table 2, and histograms of the full distributions are shown in Figure 2. Note that only the nondiabetic dataset (Figure 2a) was used to define the thresholds of abnormal RoC, whereas both Figure 2a (nondiabetic dataset) and Figure 2b (T1D dataset) show those RoC thresholds as vertical lines. Notably, significantly more than 1% and 0.1% of the T1D data falls in the abnormal ranges of L1 and L2, respectively.
Percentiles of One-Hour Rates of Change in the Nondiabetic and T1D Groups.

Extremum percentiles in the nondiabetic group which define the thresholds for normal RoC (L1 and L2) are shown in bold.
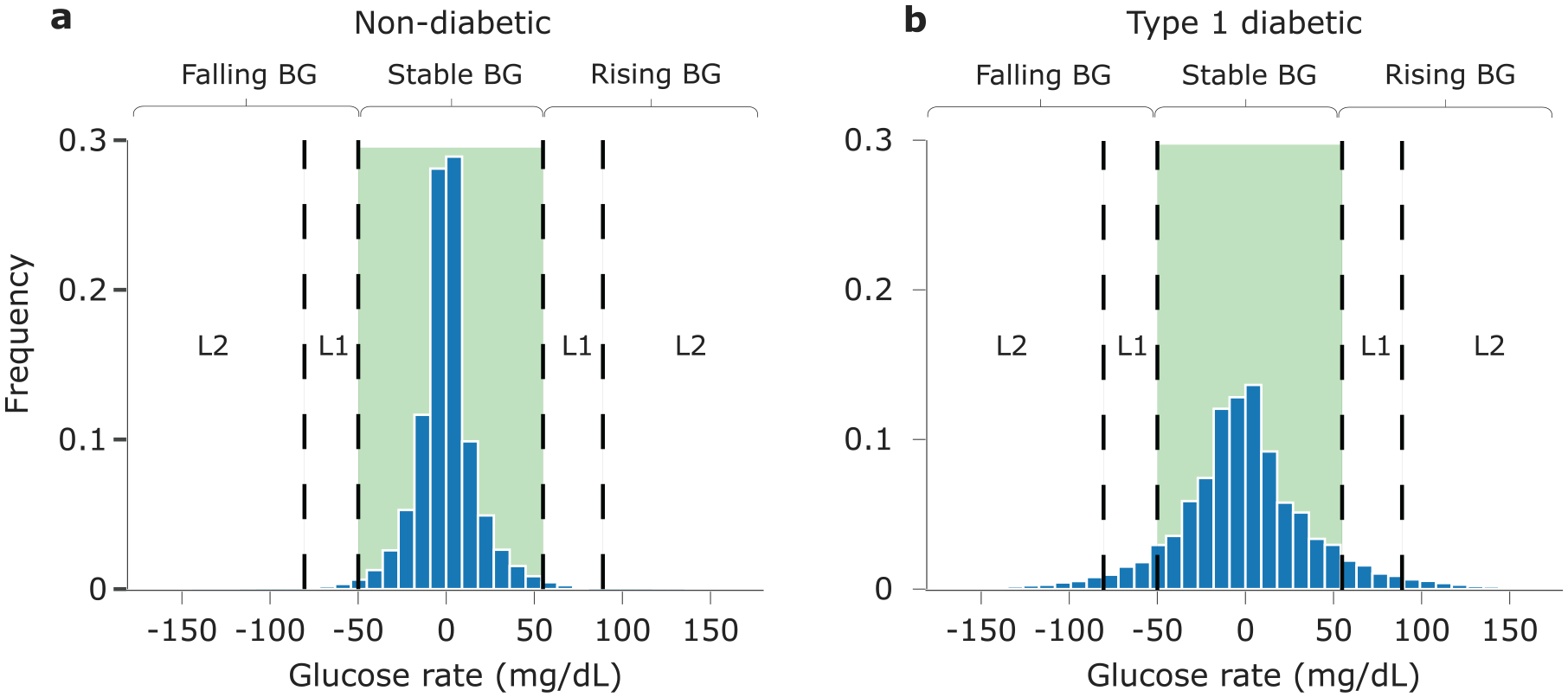
Histograms of one-hour rates of change in healthy (a) and T1D (b) populations. a, Healthy population. The vertical dashed lines indicate RoC values at percentiles 0.1%, 1.0%, 99.0%, 99.9% in this dataset, which define the boundaries for levels of abnormal fluctuation, L1 and L2. b, T1D population. The L1 and L2 thresholds derived from the data in (a) are shown mapped onto the T1D patient dataset (b).
Descriptive Statistics
Descriptive statistics of patient characteristics and GV metrics are shown in Table 1. The T1D group had significantly higher values of all GV metrics than the nondiabetic group. In the T1D group, the median TIF-L1 (16.7%) was more than ten times higher than that of the nondiabetic group (1.4%). The median TIF-L2 in the nondiabetic group was 0.0%, indicating that most healthy individuals never exceed the L2 thresholds. In contrast, in the T1D group, the median TIF-L2 was 5.0%, and the interquartile range was 3.1% to 7.8%, indicating that most T1D individuals spend a significant percentage of time fluctuating at rates almost never observed in health.
Correlation Between GV Metrics and TIF
Figure 3 shows the associations between GV metrics and TIF. A tabular summary of these associations is also shown in Supplementary Table 2. The MAG is very highly correlated with TIF-L1 (r = .99) and TIF-L2 (r = .96). The MAG and CONGA are very highly correlated with each other (r = .99), and thus CONGA exhibits similar correlations as MAG with TIF-L1 (r = .99) and TIF-L2 (r = .98). By comparison, CV exhibits significantly lower correlation with TIF-L1 (r = .58) and TIF-L2 (r = .57). For context, Supplementary Table 2 also shows the correlation between mean glucose level and TAR-L1 (r = .96) or TAR-L2 L1 (r = .95). These correlations are similarly strong to those between MAG or CONGA with TIF-L1 or TIF-L2.
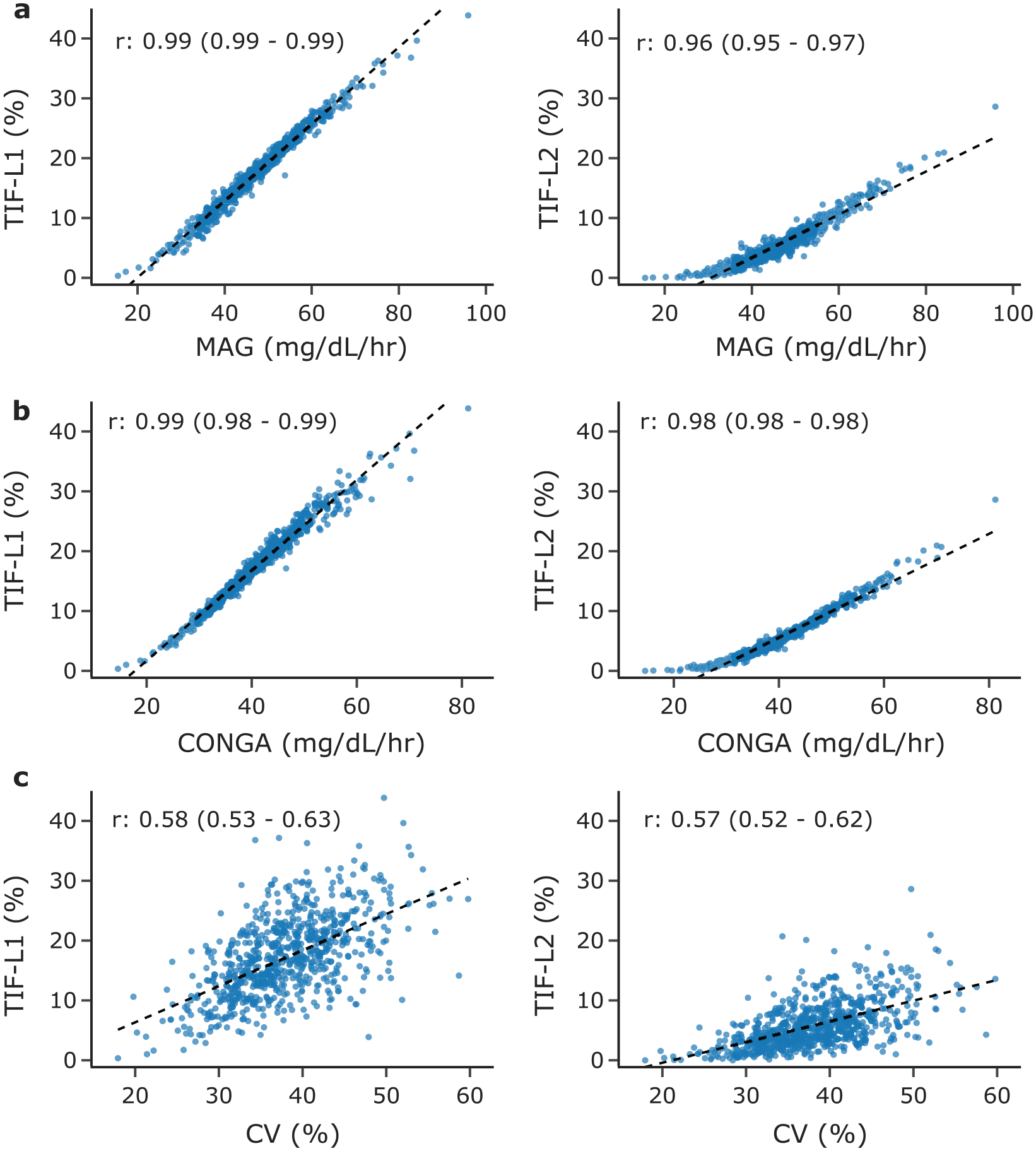
Scatterplots of TIF against metrics of GV. MAG (a) and CONGA (b) are measures of GV-timing. CV (c) is a measure of GV magnitude. The dashed black lines indicate the line of best fit; the blue dots indicate individual datapoints.
Discussion
There are two main findings from this study. First, the results indicate that T1D subjects have significantly higher TIF-L1 and TIF-L2 than healthy individuals. This finding was expected, but had not previously been quantified. Second, despite not being guaranteed theoretically, MAG and CONGA are very highly correlated with TIF-L1 and TIF-L2. We can therefore rely on these GV-timing metrics as indicators of abnormally fast RoC in T1D.
It is worth highlighting the analogy between TIF, a measure of out-of-range glucose RoC, and time below range (TBR) or time above range (TAR), which are established measures of out-of-range glucose levels. Table 3 shows a summary of analogous relationships between metrics of glucose levels and metrics of glucose RoC. It is clinically understood that the risk associated with blood glucose levels increases nonlinearly with the degree of deviation from the normal range, 7 and so TBR and TAR are recognized as more significant than mean glucose per se. Analogously, we postulate that mean RoC is not clinically significant per se; rather, time spent at high rates of change, those rarely or never observed in health, might carry significance. In other words, a glucose profile with phases of stability punctuated by phases of abnormally rapid fluctuation might be harmful, whereas a glucose profile with constant gradual fluctuations within the range of RoC observed in health is less likely to be so.
Analogous Relationships Between Metrics of Glucose Levels and Metrics of Glucose Rates of Change.
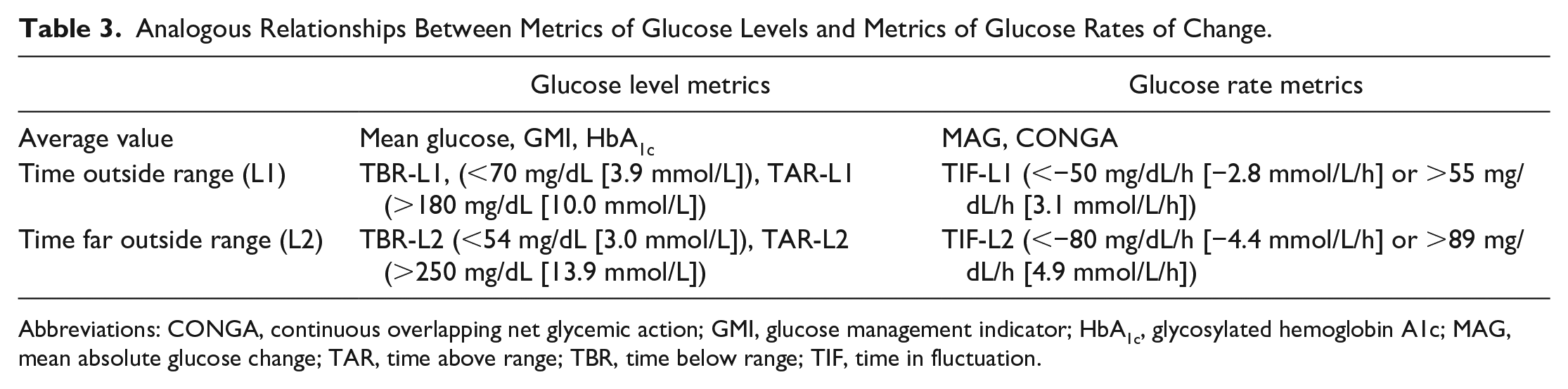
Abbreviations: CONGA, continuous overlapping net glycemic action; GMI, glucose management indicator; HbA1c, glycosylated hemoglobin A1c; MAG, mean absolute glucose change; TAR, time above range; TBR, time below range; TIF, time in fluctuation.
A key advantage of metrics that discretize data into bands (such as TBR, TAR, and TIF) is that they are intuitive to understand and interpret. This is a potential reason to consider a metric like TIF as an alternative to MAG or CONGA as a candidate GV-timing metric for clinical practice. However, a disadvantage of such “time in range” metrics is that they discard much of the information contained in the data; for example, in the case of TIF, any fluctuations that are even marginally below the defined threshold are discarded. Whether these advantages outweigh those of metrics such as MAG or CONGA is a matter of debate. However, the observation in this study that all three metrics are very highly correlated with each other (r = 0.95-0.99 in all their pairwise comparisons), and by extension with abnormal glucose RoC, suggests that any one of them would suffice as a candidate GV-timing metric. This is analogous to the observation made in previous studies29-31 that mean glucose level is highly correlated with TAR, and thus knowledge of mean glucose alone may be sufficient to indicate the degree of hyperglycemia, or vice versa.
Interestingly, CONGA exhibits very slightly higher correlation than MAG with TIF-L2. In other words, CONGA is more sensitive than MAG to the most extreme glucose rates of change. This is expected theoretically given their mathematical formulae (Supplementary Table 1): the SD equation of CONGA weights outlier values nonlinearly, whereas the linear equation defined by MAG is insensitive to outliers. Supplemental Figure 1 illustrates this further, by showing that MAG and CONGA can be interpretated as continuous penalties applied to glucose derivatives, analogous to the established low and high blood glucose risk index for glucose levels. 2 This behavior is also exemplified by the example given in the introduction (Figure 1b). However, the relatively subtle difference in correlations observed in the current dataset suggests this difference is not sufficient to strongly support one metric over the other in practice.
The observation that CV is not highly correlated with TIF (or, by extension, MAG or CONGA) is an additional reason to consider inclusion of a GV-timing metric, such as MAG or CONGA, in clinical practice, since it implies that additional information would be added by including such a metric. Previous studies have noted that the existing core metrics can be simplified into two groups, each exhibiting high within-group correlation: metrics correlated with hyperglycemia (such as mean glucose and TAR) and metrics correlated with hypoglycemia (such as TBR and CV).29-31 The fact that neither of these groups are very highly correlated with MAG or CONGA indicates that GV-timing would constitute a third such group. This was retrospectively investigated by a principal component analysis (PCA) applied to the T1D group, implemented using the scikit-learn PCA function in Python. 32 This showed that two components were sufficient to explain 96.0% of the variance for the core international consensus metrics, whereas three principal components were required to achieve a similar level of explained variance (96.4%) when TIF-L1 and TIF-L2 were included in the set of metrics.
There are a number of reasons a GV-timing metric has yet to be included in the consensual core glycemic metrics. First, it is clear that some practical issues still need to be addressed, including determining the most pertinent timescale(s) over which to measure glucose differences and establishing a clear threshold value to define good versus poor control. Second, there remains a lack of definitive evidence from randomized controlled trials for a causal relationship between short-term GV and adverse clinical outcomes. However, as noted by Monnier et al, 1 an absence of definitive evidence does not imply evidence of absence, and indeed several studies do support the hypothesis of such a relationship.33-36 The current study, while not providing any direct evidence for a relationship between GV and diabetes complications, does reveal that glucose RoC in diabetes routinely exceeds values observed in healthy subjects. This observation should provide additional motivation to address the remaining barriers to using GV-timing metrics in clinical practice.
The current study has some limitations. First, we chose to focus exclusively on T1D, but similar analyses could be applied to T2D. Second, although this study introduces a potential new metric, there remain limitations of CGM metrics more generally, including imperfect correlation between 14-day values and long-term control, 37 and the need to consider nuances of hypoglycemia not easily captured by metrics. 38 Third, we used separate CGM datasets, which were collected using different sensors for nondiabetic individuals (Dexcom G6) and T1D individuals (Freestyle Libre 1 or 2). Although both sensors are considered very accurate, studies have shown that CGM metrics can differ between sensors in individuals wearing these sensors at the same time, and even in the same sensor using different versions of CGM software for postprocessing. 39 It is possible, therefore, that the glucose RoC values used in this study would differ for this reason. However, the effect of RoC differences is most significant on timescales close to the measurement sample rate (5-15 minutes) and so the choice of one-hour time steps in the present study should mitigate this. One-hour timesteps are also the size most widely used by existing GV-timing metrics, including MAG and CONGA. Last, and related to the above, it is possible that RoC values measured across shorter timescales, such as 5 to 15 minutes, are of equal or greater clinical significance than one-hour timescales, since even consistent smaller magnitude fluctuations may be less preferable than a stable higher glucose concentration. Therefore, a comparison of RoC values between healthy and T1D groups on such timescales would be insightful. The same sensor type would need to be used in each group in that case. Alternatively, it would be desirable if CGM manufacturers in future enabled access to the unfiltered/unsmoothed data to improve comparisons between devices more generally.
Conclusions
The normal range of blood glucose (70-180 mg/dL [3.9-7.8 mmol/L]) is familiar to anyone involved in diabetes research or clinical care, but this study highlights that there are normal ranges of other aspects of glycemia that have not been quantified. Specifically, normal ranges of blood glucose RoC were identified at two levels (L1: −50 to +55 mg/dL/h [−2.8 to +3.1 mmol/L/h], L2: −80 to +89 mg/dL/h [−4.4 to +4.9 mmol/L/h]). The percentages of time spent outside these ranges were defined as TIF (TIF-L1 and TIF-L2), in analogy to TAR. The T1D individuals were found to have significantly higher TIF than healthy individuals. This finding was expected, but had not previously been quantified. Despite not being guaranteed theoretically, MAG and CONGA were found to be very highly correlated with TIF, and so we can rely on these GV-timing metrics as indicators of abnormally fast RoC in T1D. If we accept the premise that deviations from patterns observed in health carry a greater possibility of harm, then these findings should motivate the eventual inclusion of a GV-timing metric in clinical practice.
Supplemental Material
sj-docx-1-dst-10.1177_19322968241298248 – Supplemental material for Do Metrics of Temporal Glycemic Variability Reveal Abnormal Glucose Rates of Change in Type 1 Diabetes?
Supplemental material, sj-docx-1-dst-10.1177_19322968241298248 for Do Metrics of Temporal Glycemic Variability Reveal Abnormal Glucose Rates of Change in Type 1 Diabetes? by Robert Richardson in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
CONGA, continuous overlapping net glycemic action; CV, coefficient of variation; GMI, glucose management indicator; HbA1c, glycosylated hemoglobin A1c; MAG, mean absolute glucose change; GV, glycemic variability; TAR-L1, time above range (>180 mg/dL); TAR-L2, time above range (>250 mg/dL); TBR-L1, time below range (<70 mg/dL); TBR-L2, time below range (<54 mg/dL); TIF-L1, time in fluctuation (<−50 mg/dL/h or >56 mg/dL/h) ]); TIF-L2, time in fluctuation (<−80 mg/dL/h or >89 mg/dL/h).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
