Abstract
Background:
Diabetes technologies are valuable tools to reduce burden and enhance glycemic control, especially during adolescence. The current study sought to understand the factors associated with parent and adolescent diabetes device satisfaction.
Methods:
This study used cross-sectional data from 175 adolescents living with type 1 diabetes and 176 parents. Adolescent ages ranged from 12 to 19 (Mage=14.7, SD=1.89) and were balanced by gender (48% male). Kendall’s W examined concordance between parent and adolescent satisfaction and bivariate correlations and paired
Results:
There was low concordance (Kendall’s W = 0.13) between parent and adolescent device satisfaction. Automated insulin delivery (AID) use (vs non-use) was related to higher satisfaction for adolescents (4.52 [0.71] vs 4.20 [0.87],
Conclusions:
Adolescents and their parents have discrepant levels of satisfaction with devices. Although both adolescent and parent satisfaction are linked to use of automated technology and better psychosocial functioning, only parent satisfaction is associated with glycemia. This pattern suggests adolescents and parents hold varying priorities when it comes to device use. Acknowledging and addressing these differences may enhance the uptake and continued use of devices.
Introduction
Adolescence is a period of marked deterioration in glycemic control1-3 with less than one in four adolescents living with type 1 diabetes meeting the American Diabetes Association (ADA) guidelines for target hemoglobin A1c (HbA1c).4,5 Diabetes technologies can improve glycemic and psychosocial outcomes6-8 that are at risk during this developmental stage. Although adolescents with type 1 diabetes have a relatively high utilization rate of insulin pumps, 3 they are more likely than other age group to discontinue device use. 9 Measures of device satisfaction have been developed for insulin delivery 10 and glucose monitoring 11 among adults, showing that higher satisfaction is related to better glycemic control, self-management, and insulin pump use. Such measures have not yet been developed/adapted for device use among adolescents with type 1 diabetes or their parents. Understanding how satisfied adolescents and parents are with current devices and the factors related to satisfaction levels has important implications for diabetes technology uptake and ongoing use during this critical developmental stage.
Research on adolescent and parent perspectives on diabetes technologies has identified several important factors that can influence uptake and continued use. The most commonly endorsed barriers to device use among adolescents are cost, wear-related issues (eg, skin irritation), and the hassle of wearing devices. 12 The most commonly endorsed reasons for discontinued device use are related to device visibility and adhesion problems, 13 which may be particularly pertinent during adolescence when worries about self-image and fear of unwanted attention are prominent. Most parents of youth living with type 1 diabetes hold highly positive attitudes toward technology and have minimal barriers. 14 However, little research has compared adolescent and parent perspectives within the parent-adolescent dyad or explored the clinical, psychosocial, and contextual factors that may be linked to device satisfaction. Such factors may be avenues to optimize device use benefits and satisfaction to offset barriers and reasons to discontinue use.
The current study explored adolescent and parent diabetes device satisfaction including (1) concordance in satisfaction rates within parent-adolescent dyads, (2) differences in satisfaction based on current device use, and (3) glycemic, psychosocial, and demographic correlates of satisfaction. We hypothesized moderate agreement in satisfaction ratings between adolescents and parents within dyads and that device (insulin pump, automated insulin delivery [AID] system) use would be associated with higher satisfaction. We also hypothesized that both parent and adolescent satisfaction would be related to glycemic control, whereas only adolescent satisfaction would be closely linked to better psychosocial functioning. The results from the study can inform efforts to promote device uptake and ongoing use among adolescents living with type 1 diabetes.
Methods
This study drew cross-sectional data collected at the baseline of a randomized controlled trial that evaluated the “Insul-In This Together” program, a family-based intervention for adolescents with type 1 diabetes and their parents. Potential participants were recruited through the Stanford Pediatric Diabetes Clinics, nationwide through diabetes organizations, online forums, and social media posts. A total of 606 families completed online screenings, of which 383 (64%) met inclusion criteria of: adolescent ages 12 to 19 years with type 1 diabetes diagnosis for at least 6 months, participating parent currently living with adolescent, both English proficient, access to smartphone and/or Wi-Fi via computer, no developmental, cognitive, or psychiatric limitations that would compromise participation in survey completion and/or intervention session, and current US resident. Of the eligible participants, 48% provided informed parental consent and adolescent consent (for ages 18+ years) or assent (for ages under 18 years).
Once enrolled in the study, participants provided several types of data: online surveys, continuous glucose monitor (CGM) data, and HbA1c results. Online surveys were collected through the HIPAA-compliant and secured Stanford REDCap platform (http://redcap.stanford.edu). These platform services at Stanford are subsidized by (1) the Stanford School of Medicine Research Office and (2) the National Center for Research Resources and the National Center for Advancing Translational Sciences, National Institutes of Health, through grant UL1 KL2 TR001085. Longitudinal data were collected but the current study used only baseline data collected before randomization. The study protocol conforms to recognized standards and was approved by the Stanford University Institutional Review Board (#52851 and #58372).
Statistical Analysis
Kendall’s W was used to test concordance between adolescent and parent device satisfaction with parent-adolescent dyads. Kendall’s W was selected as it measures the agreement among raters (rank correlation). A combination of bivariate correlations and independent
Measures
Diabetes device satisfaction
A single item was used to measure both parents’ and adolescents’ satisfaction with devices: “How satisfied are you with your [teen’s] current diabetes devices?” The response scale ranged from 1 (unsatisfied) to 5 (very satisfied).
Diabetes device use
Current diabetes device use was measured by parent reports of whether their adolescent was currently using a CGM, insulin pump, and AID.
Glycemic measures
Hemoglobin A1c
When available, participants would provide a copy of an HbA1c result from a recent diabetes clinic visit (from within a month of survey data collection). If a clinic test was not conducted in the necessary time frame, then participants were sent an at-home HbA1c test kit to collect a sample and send it to the Advanced Research and Diagnostics Laboratory at the University of Minnesota, which provided the result.
Percent time-in-range, time in hypoglycemia, and time in hyperglycemia
The CGM data were collected through online accounts. All participants, except one, were already using CGM at baseline (99.4%). The participant who was not already using CGM was provided a FreeStyle Libre 14 day device to use by the study team for data collection purposes. Most of the data collected (96.2%) were from Dexcom glucose monitoring devices, whereas 2% were from Libre devices and 2% from Medtronic devices. Fourteen days of data were collected, when available, with no less than 10 days of data within a 14-day period. Data were analyzed for percent TIR (70-180 mg/dL), time in hypoglycemia (mg/dL <70), and time in hyperglycemia (mg/dL >250).
Psychosocial factors
Diabetes-specific health-related quality of life
Adolescent and parent HRQOL were measured via the T1DAL survey.15,16 The adolescent version (ages 12-17 years) 15 includes 23 items. and the late adolescence/young adult version (ages 18-25 years) 16 includes 27 items. Both surveys have questions focused on diabetes-related psychosocial functioning, support, and challenges across life domains (sample item: “I can do activities I like for fun even with diabetes”) The parent version 17 consists of 30 items (sample item: “Our family manages my teen’s diabetes as a team”). Response options range from 1 (no, not at all true) to 5 (yes, very true). Item scores are converted (1 = 0, 2 = 25, 3 = 50, 4 = 75, 5 = 100) and then averaged to create a total score with a possible range of 0 to 100. In the current sample, Cronbach’s alpha was 0.90 for ages 12 to 17 years, 0.74 for ages 18+ years, and 0.87 for parents. The adolescents and 18+ groups were collapsed in current analyses, per the measure design.
Diabetes distress
Adolescent diabetes distress was measured with the Problem Areas in Diabetes—teen version.18,19 This survey includes 14 items that describe various possible problem areas with diabetes and asked how much each bothered the participant within the past month (sample item: “Feeling overwhelmed with my diabetes regimen”). The response scale ranges from 1 (not at all) to 6 (serious problem). All item scores are summed with a possible range of 14 to 84 with a higher total score reflecting greater diabetes distress. Cronbach’s alpha in the current sample was 0.94.
Parent diabetes distress was measured with the Problem Areas in Diabetes—parents of teens version.18,20 This measure includes 15 items that describe possible problem areas that parents of youth with diabetes may have and ask how much each item bothered the participant during the past month (sample item: “Feeling overwhelmed with my teen’s diabetes regimen”). The response scale ranges from 1 (not a problem) to 6 (serious problem). The item scores were summed into a final score with a possible range of 15-90 with higher values reflecting higher parental diabetes distress. Cronbach’s alpha in the current sample was 0.96.
Diabetes-related parental warmth
The Diabetes Family Behavior Scale (DFBS)21,22 Warmth-Caring Subscale (teen and parent versions) asked the respondent to report on the parent/child who was participating in the study with them. Each scale had 15 items about the frequency of certain dynamics occurring within the family (sample item: “My parent understands how I feel about having diabetes” [reverse scored]). Response options range from 1 (all the time) to 5 (never). Item scores are summed with a possible range from 15 to 75 with higher scores indicating greater diabetes warmth/caring. In the current study, Cronbach’s alpha for the adolescent version was 0.71 and 0.62 for the parent version.
Diabetes-related family conflict
Family conflict related to diabetes, specifically, was measured using the Diabetes Family Conflict Scale (DFCS)23,24 teen and parent versions. Both versions asked participants to report on how much they argued or hassled with their parent/teen (who was participating in the study with them) over the past month about specific aspects of diabetes management presented in each of the 19 items (sample item: “Remembering to give shots or to bolus”). Response options include 1 (never argue), 2 (sometimes argue), and 3 (always argue). Item scores are summed into a total score with a possible range from 19 to 57 with higher scores indicating greater conflict. Cronbach’s alpha was 0.91 for the adolescent report and 0.85 for the parent report.
Demographic characteristics
Adolescent age was calculated based on adolescent self-report of date of birth and the baseline survey completion date. Adolescent race/ethnicity was based on adolescent self-report. For analytic purposes, the various racial/ethnic categories were collapsed to create a binary variable representing non-Hispanic white vs all other racial/ethnic groups (reported in Table 1). Parent report of annual household income was used to create a binary variable of annual household income of $100,000 or higher vs under $100,000. Finally, parents reported on insurance type (private vs public/unspecified).
Baseline Characteristics of Study Sample.
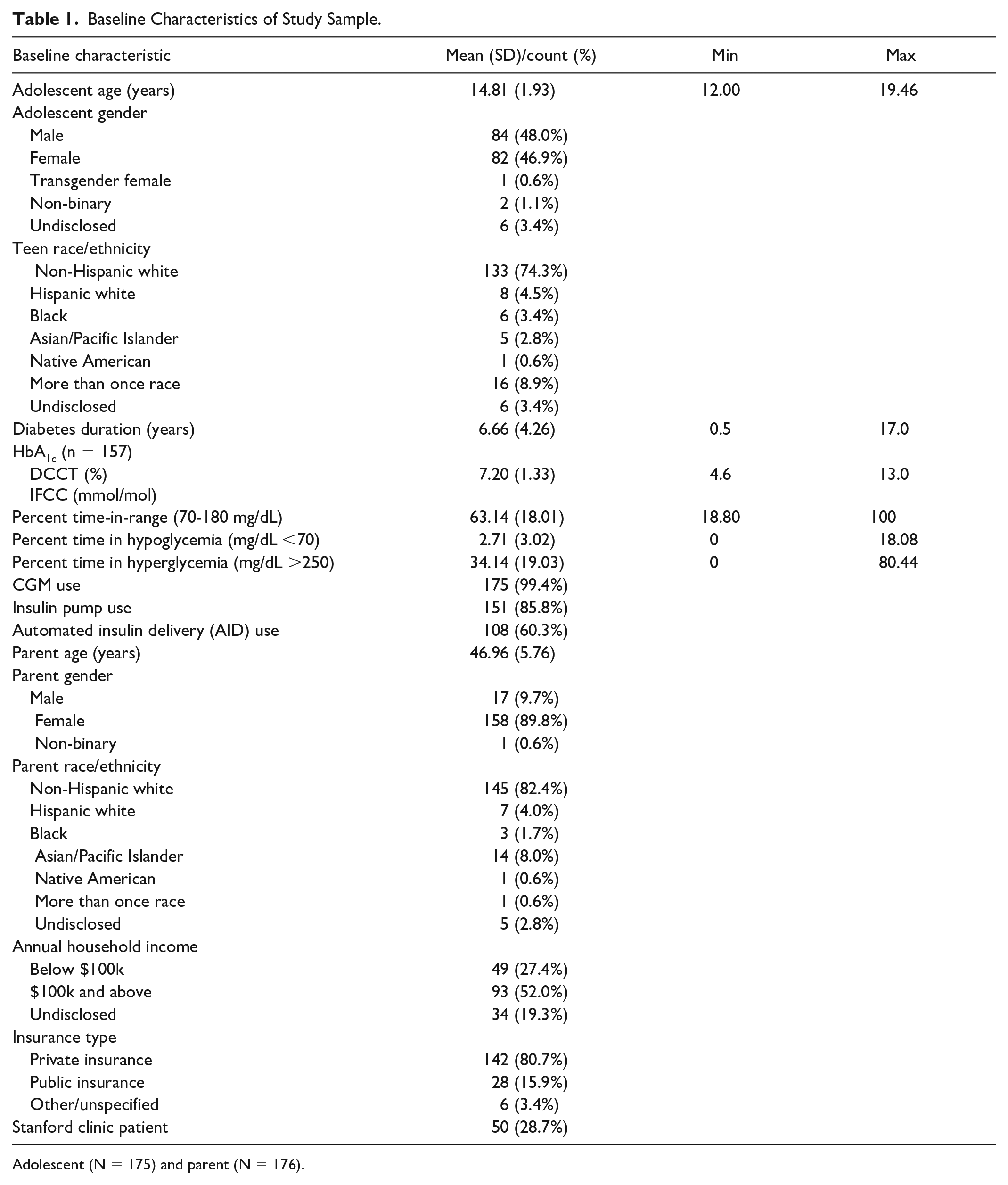
Adolescent (N = 175) and parent (N = 176).
Results
Our sample included 175 adolescents living with type 1 diabetes and 176 parents of adolescents drawn from the baseline data of a randomized controlled trial (Table 1). The sample included 172 parent-adolescent dyads as well as 3 adolescents and 4 parents who were the only ones within their dyad to complete baseline data collection. The adolescent sample was balanced by gender (48.0% male), whereas most parents identified as female (89.8%). The sample was predominantly non-Hispanic white (74.3% for adolescents, 82.4% for parents) with over half (52.0%) having an annual household income at or above $100,000. Most (80.7%) were privately insured and nearly a third (29.7%) received diabetes care at the Stanford Pediatric Diabetes Clinic.
Average diabetes device satisfaction was lower and had more variability among parents (M = 4.04, SD = 0.89) compared to adolescents (M = 4.39, SD = 0.79) (histograms in Figure 1.)
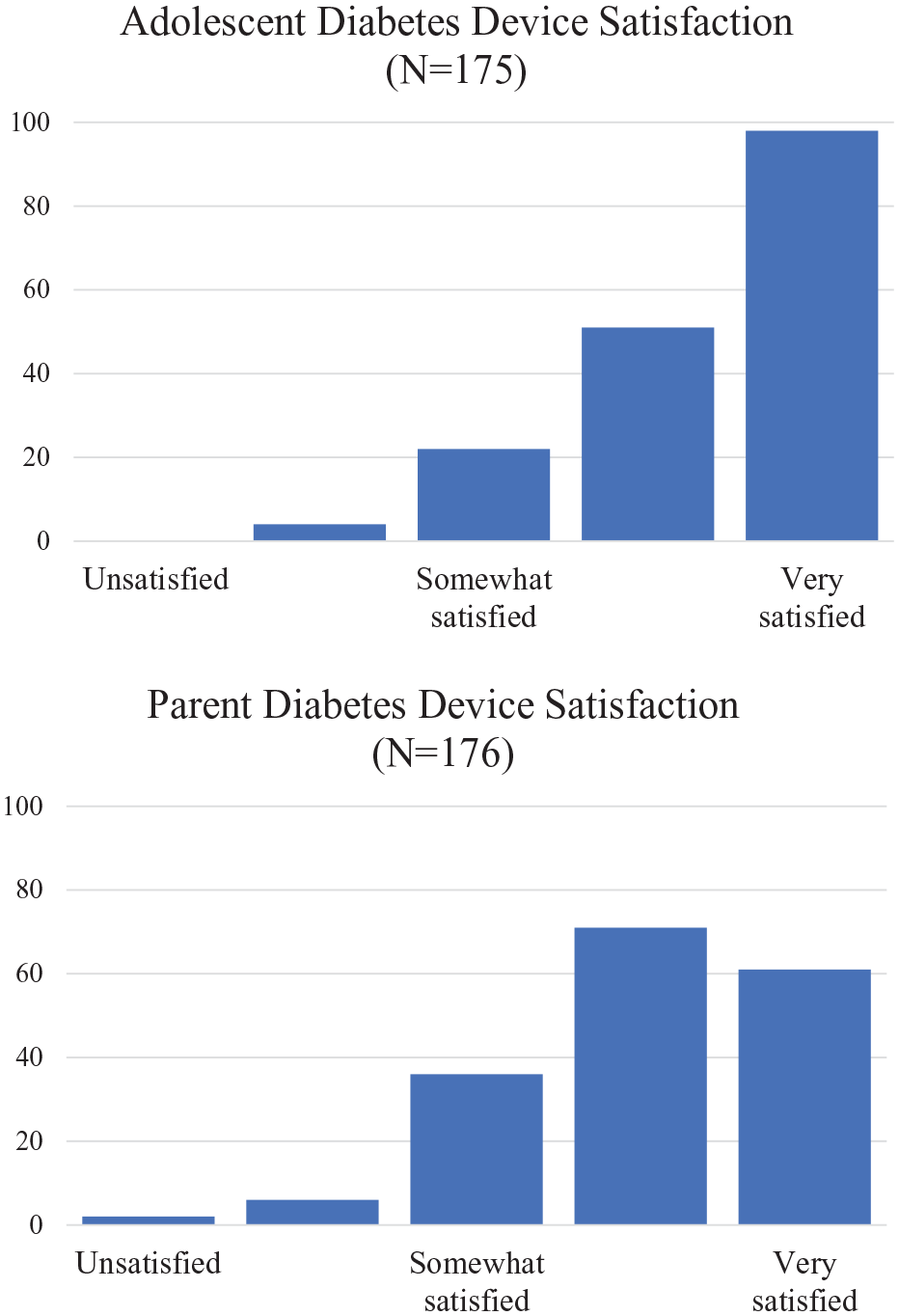
Histograms of diabetes device satisfaction variables (5 = very satisfied, 3 = somewhat satisfied, 1 = unsatisfied).
Satisfaction Concordance
Concordance between adolescent and parent diabetes device satisfaction was low with Kendall’s W = 0.132 (0 = no agreement, 1 = complete agreement).
Correlates of Device Satisfaction
Device use
Given that only one participant was not using CGM, this could not be tested as a correlate. Independent
Glycemic correlates
All correlations between device satisfaction and continuous variables are reported in Table 2. Several glycemic variables were found to significantly correlate with parent device satisfaction, including HbA1c (R = −0.301,
Correlations Between Continuous Variables and Device Satisfaction.
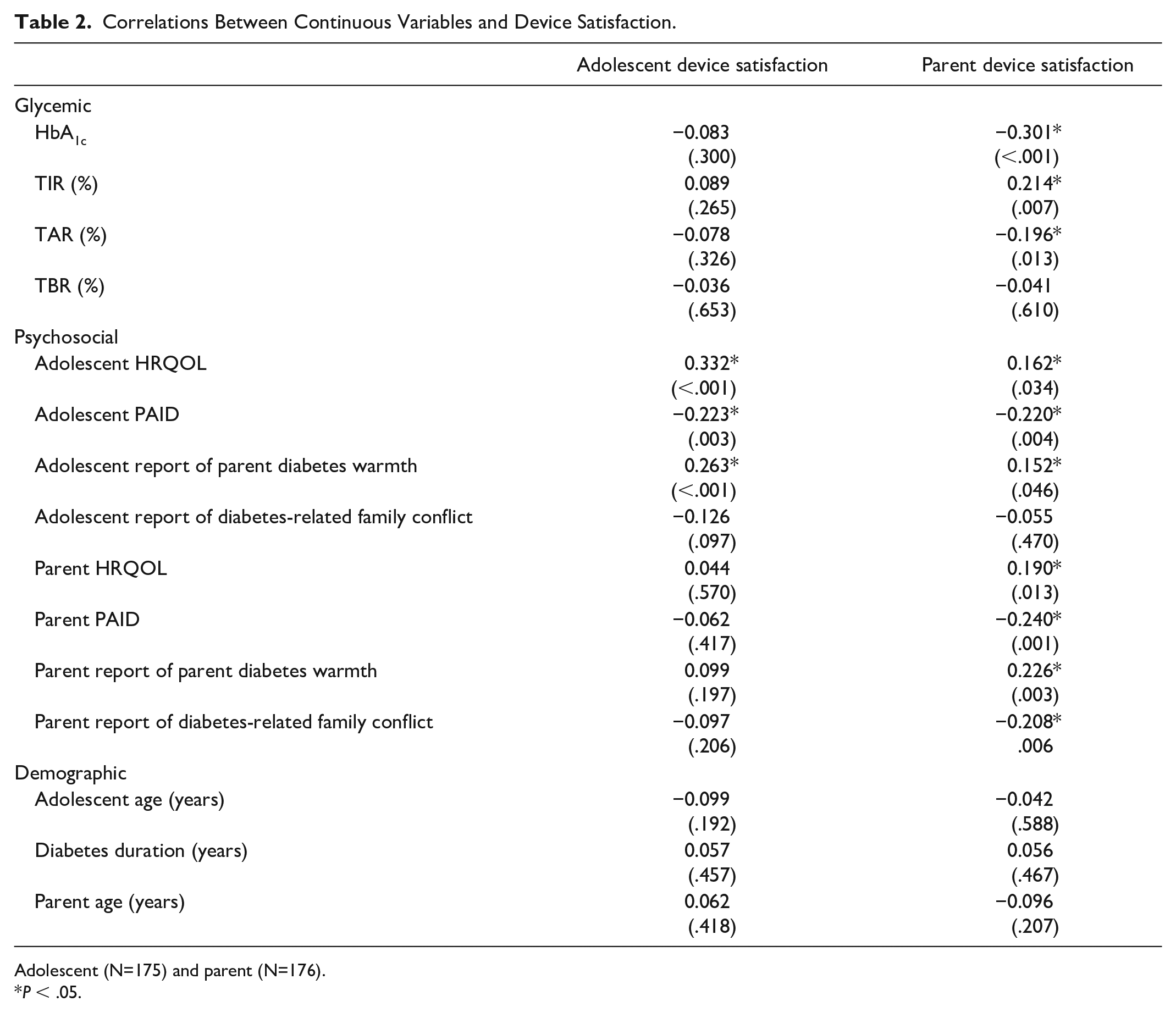
Adolescent (N=175) and parent (N=176).
Psychosocial correlates
A distinct pattern emerged among psychosocial correlates of adolescent and parent satisfaction. Specifically, adolescent and parent device satisfaction were both positively correlated with adolescent HRQOL (R = 0.332,
Demographic correlates
No demographic characteristics were found to significantly correlate with either adolescent or parent device satisfaction (
Regression Analysis
Adolescent device satisfaction was significantly and uniquely predicted by AID use (β = 0.16,
Discussion
Diabetes technologies provide a variety of glycemic and psychosocial benefits.6-8 Such benefits can be particularly impactful during adolescence, a period of marked deterioration in self-management and glycemic control.1-3 The current study explored adolescent and parent device satisfaction and their correlates to expand our empirical understanding of priorities and experiences. Findings suggest that more automated technologies are closely tied to adolescent and parent device satisfaction and that parents prioritize glycemic outcomes in their evaluation of devices, whereas youth do not. Acknowledging these differences can enhance device education and communication as well as breed opportunities to amplify adolescents’ perspectives and agency in the device decision-making process.
The low concordance in device satisfaction between adolescents and their parents may be expected for several reasons. First, the adolescents in this study are the ones living with diabetes and are likely to experience and evaluate technologies differently from their parents. Prior research on open-source AID has found that adults living with diabetes experience a broad range of psychosocial benefits from this technology, whereas parents of children living with diabetes only experience benefits related to sleep quality and fear of hypoglycemia. 25 Another contributing factor could be discrepancies in priorities between these groups. Research has shown that adolescents are primarily concerned with short-term consequences, whereas parents prioritize both short- and long-term consequences of diabetes management. 26 As such, parents’ satisfaction may be more closely tied to glycemic measures, whereas youth might be more influenced by psychosocial functioning that they experience in real-time. Finally, dyads may be different in attributional style. Adolescents may focus more on negative and internal factors, such as their management missteps, impacting their diabetes self-management compared to parents. 27 For this reason, parents may be more likely to attribute glycemic outcomes to current diabetes technology, whereas adolescents may hold themselves solely accountable for their glycemia and minimize or dismiss the role of their current technology use.
In addition to the discrepancy within dyads, it is important to acknowledge that the largest correlation observed for parent satisfaction was with HbA1c whereas the largest correlation observed for adolescent satisfaction was with adolescent HRQOL. For these reasons, emphasizing both glycemic and psychosocial benefits in diabetes technology education and acknowledging these as areas of concern among parents and adolescents, respectively, could increase interest while validating that parent and adolescent priorities might vary.
The observed correlations between device satisfaction and diabetes distress may bring a new perspective to previously mixed findings related to technology use and diabetes distress.28-32 Understanding device satisfaction may be critical to ensure that people are using the devices that are the best match for them before expecting a strong impact on diabetes distress. Resources such as DiabetesWise.org can provide unbiased and unbranded user-focused education on diabetes technologies to promote device uptake based on personal preferences and priorities.33,34 The current findings reinforce the use of such person-oriented approaches as well as openly acknowledging differences in priorities, such as those observed between adolescents and their parents.
Prior findings have indicated that automatized technologies offer the potential for greater independence in adolescents and reduce monitoring needs and management burdens for parents.35,36 The current findings indicate that both adolescents and parents are significantly more satisfied with their devices when using automated technologies (such as AID) than not, with no difference in satisfaction with devices among those using vs not using insulin pumps.
This study included several methodological strengths, including a large sample size, psychometrically validated measures, and a combination of self-report and biomedical measures. Study limitations include the use of a single item to measure device satisfaction. The use of a psychometrically sound survey that has been validated among adolescents with type 1 diabetes and parents would enhance the nuance and robustness of the current findings. Another limitation is the lack of representativeness in the study sample. Only one participant was not using CGM at the time of enrollment, limiting our understanding of device satisfaction among adolescents not using CGM and their parents. In addition, most of the sample was non-Hispanic white and of generally high socioeconomic status, which limits generalizability to the broader population of adolescents with type 1 diabetes and their parents. This area of research would benefit from future studies that focus on adolescents from marginalized backgrounds, lower socioeconomic status, and limited diabetes technology exposure.
Diabetes technology continues to advance at a rapid rate creating increased opportunities to improve glycemic control and ease psychosocial burdens among people living with type 1 diabetes and their family members. Understanding adolescents’ and parents’ perspectives and priorities is necessary to take advantage of these opportunities, maximize the potential benefits, and reduce device attrition.
Footnotes
Abbreviations
HbA1c, Hemoglobin A1c; AID, automated insulin delivery; HRQOL, health-related quality of life; CGM, continuous glucose monitoring; TIR, time-in-range; TAR, time-above range; TBR, time-below-range.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by NIH grant K23-DK121771 and Stanford University’s Maternal and Child Health Research Institute award to JJW.
