Abstract

Keywords
Incretin-based therapies such as dipeptidyl peptidase-4 inhibitors (DPP-4is: incretin enhancers) and glucagon-like peptide-1 receptor agonists (GLP-1-RAs; incretin mimetics) have been increasingly playing an important role in the therapy of diabetes mellitus and obesity. Since their respective first agents, sitagliptin and exenatide, respectively, were approved by the regulatory agencies, many drug candidates are on the market, and new ones are still in the pipeline. Among people with diabetes mellitus—including those treated with incretin-based drugs—self-monitoring of blood glucose is still a fundamental technology for diabetes self-management.1,2 Global guidelines for interference testing are established, and the blood glucose meter (BGM) systems have to be continuously checked for potential interferences.3 -5 This is even more important as some antidiabetic drugs are known to interact with BGM systems, so that diabetes management could be affected.3 -6
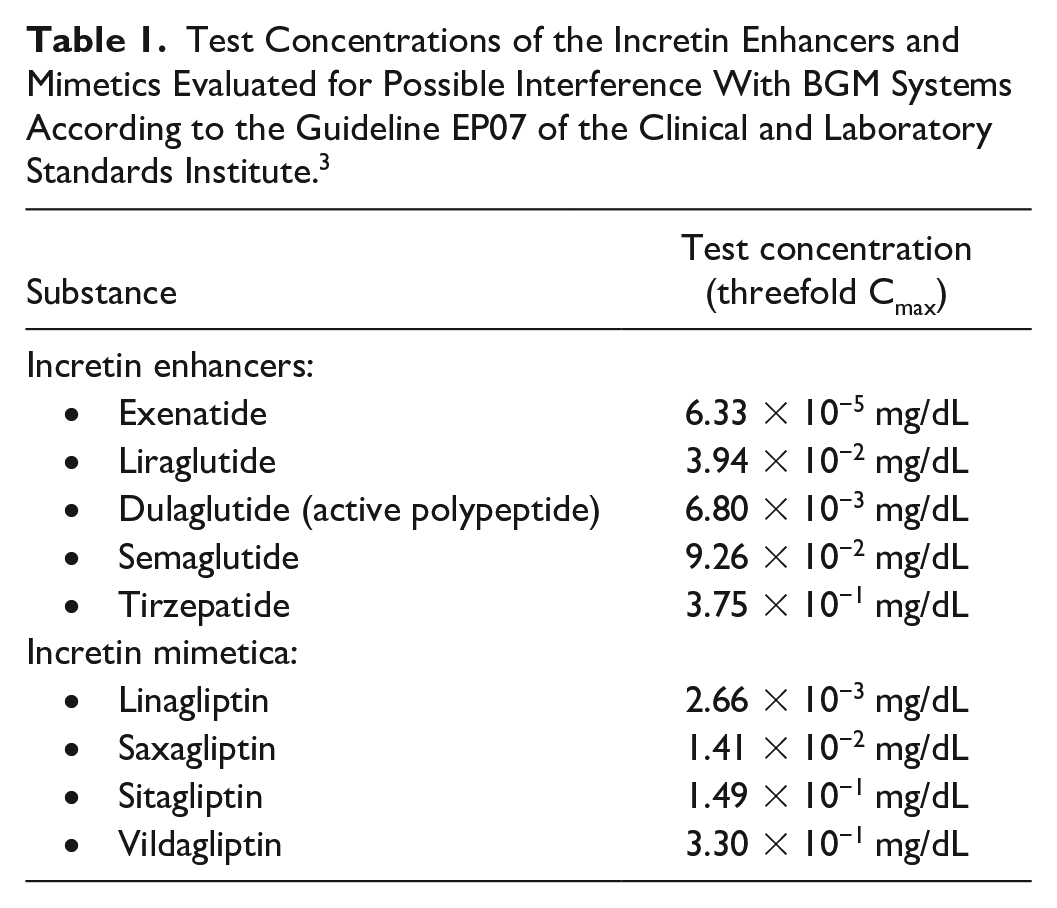
With this in mind, five GLP-1-RAs and four DPP-4is (Table 1) were evaluated regarding their performance using three BGM systems: Accu-Chek Active, Accu-Chek Guide, and Accu-Chek Instant (Roche Diabetes Care, Indianapolis, Indiana). In order to simulate high plasma concentrations, threefold the clinically relevant plasma concentrations (Cmax) with the highest possible daily dose of the substances (according to the literature available from the author) were used for interference testing (Table 1).
Test Concentrations of the Incretin Enhancers and Mimetics Evaluated for Possible Interference With BGM Systems According to the Guideline EP07 of the Clinical and Laboratory Standards Institute. 3
Three different test strip lots were tested each for the BGM systems. All substances were evaluated in a whole blood matrix at three target glucose concentrations (60, 120, and 250 mg/dL) using the paired difference test.4,5 For each drug at each glucose level, the bias between the test and control sample (with or without addition of one of the investigated incretin-based drugs) was calculated, and these results were evaluated against the acceptance criteria defined in the standard 15197:2013 of the International Organization for Standardization (ISO). 4 For all three BGM systems, a substance is considered as “non-interfering” if the mean bias between the test and control results for each drug at each glucose level is within ±10 mg/dL for glucose levels <100 mg/dL or ±10% for glucose levels ≥100 mg/dL. 4
The three evaluated BGM systems show excellent performance for the nine incretin enhancers and incretin mimetics tested, meeting the specified acceptance criteria: a mean bias to control values within ±10 mg/dL for glucose levels below 100 mg/dL and within ±10% for glucose levels equal to or greater than 100 mg/dL. All systems comply with ISO 15197:2013 standards and the Food and Drug Administration requirements for testing relative (mg/dL) bias, percent bias, and lower and upper limits within the 95% confidence intervals for each concentration of potential interferent.4,5
Overall, these results show that none of the tested incretin-based therapies do interfere with the tested BGM systems. Thus, we can conclude that these meters can be safely and reliably used by people treated with incretin-based therapies.
Footnotes
Acknowledgements
The authors are grateful Dr Katharina Scheibner for the intellectual contribution of the conception of the study and the review of the manuscript. The authors thank Susanne Moser for her support in writing the manuscript.
Abbreviations
BG, blood glucose; BGM, blood glucose meter; Cmax, maximal plasma concentration; DPP-4i, dipeptidyl peptidase-4 inhibitors; GLP-1, glucagon-like peptide-1; GLP-1-RA, glucagon-like peptide-1 receptor agonist; ISO, International Organization for Standardization.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JKM is a member in the advisory boards of Abbott Diabetes Care, Becton-Dickinson/Embecta, Biomea, Eli Lilly, Medtronic, Novo Nordisk, Pharmasens, Roche Diabetes Care, Sanofi, and Viatris; received speaker honoraria from Abbott Diabetes Care, A. Menarini Diagnostics, Becton-Dickinson/Embecta, Dexcom, Eli Lilly, MedTrust, Novo Nordisk, Roche Diabetes Care, Sanofi, and Ypsomed; and is a shareholder of decide Clinical Software GmbH and elyte Diagnostics. BH, RS, GB, and JR are the employees of Roche Diabetes Care GmbH and Roche Diabetes Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication has been sponsored by Roche Diabetes Care GmbH.
