Abstract

Self-monitoring of blood glucose (SMBG) is still the most widely used method for glucose monitoring, especially in patients with type 2 diabetes mellitus (DM). 1 Nowadays many of such patients use sodium-glucose transport protein 2 (SGLT2) inhibitors for their diabetes therapy. Since Dapagliflozin was approved by the European Medical Agency (EMA) in 2012 as the first drug in the new class of oral drugs, a number of SLGT2 inhibitors have come to the market. For those patients who perform SMBG, it is important to know that these drugs have no impact on the glucose measurement per se, ie, that no interferences hamper a reliable and accurate measurement result. Different systems for SMBG are constantly evaluated for interactions with potentially interfering substances.2,3 Such clinically relevant interferences show up with a number of widely used drugs, to a degree that has implications for diabetes management of patients with DM.2,4-6 To ensure safety and efficacy of SMBG, interferences should be tested especially for newer classes of clinically used drugs patients with DM might be exposed to.
Therefore, four different SGLT2 inhibitors were evaluated with respect to their impact on measurement results obtained with different SMBG systems based on potentiometric and colorimetric technology (different Accu-Chek® systems [see below], Roche Diabetes Care, Mannheim, Germany). Clinically relevant plasma concentrations corresponding to the highest daily drug dose were depicted from the literature (literature with the authors). To mimic high drug exposure, fivefold maximal reported plasma concentrations (Cmax) were used during the evaluations:
Canagliflozin (Invokana®) 1.7 mg/dL,
Dapagliflozin (Farxiga®) 0.1 mg/dL,
Empagliflozin (Jardiance®) 0.3 mg/dL, and
Ertugliflozin (Steglatro®) 0.2 mg/dL.
Three different test strip lots were tested each for the Accu-Chek® Aviva, Accu-Chek® Guide, Accu-Chek® Instant and Accu-Chek® Performa blood glucose monitoring systems. In addition, two strip lots were tested for the Accu-Chek® Active system in two different testing modes. The samples used for the evaluation were prepared in whole blood at three target glucose concentrations using the paired difference.4,5
The bias between the test and control sample (with or without addition of a given SGLT2 inhibitor) was calculated for each drug at each glucose level. The bias results were then evaluated against the acceptance criteria given in International Organization for Standardization (ISO) 15197:2013.
A substance is defined as “interfering” if the mean bias exceeds 10 mg/dL for glucose concentrations <100 mg/dL or if it exceeds 10% for glucose concentrations ≥100 mg/dL. 5
For each tested SMBG system, which is based either on amperometric or on potentiometric technology, the bias was <10 mg/dL or 10% at the blood glucose concentrations tested (Figure 1).
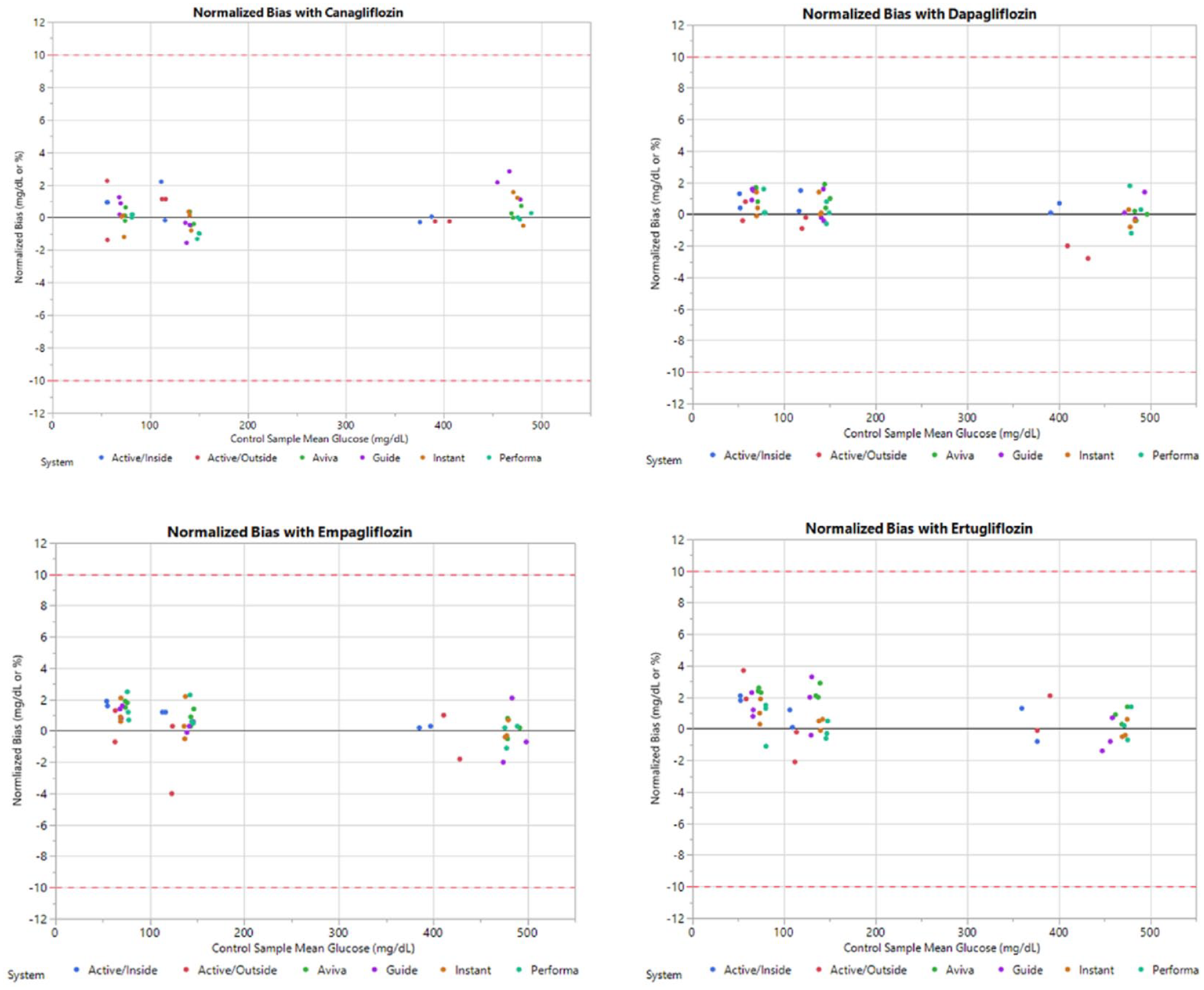
Comparison of the mean bias from each SMBG system evaluated for the different SGLT2 inhibitors tested. The mean bias at each glucose level with each strip lot (or strip lot and testing mode for the Accu-Chek Active) is plotted versus the mean glucose results of the control sample. Abbreviations: SGLT2, sodium-glucose transport protein 2; SMBG, self-monitoring of blood glucose.
All systems met ISO 15197:2013 criteria and FDA requirements for testing of relative (mg/dL) bias, percent bias, lower and upper limits within the 95% confidence intervals for each concentration of potential interferent. 5
In summary, patients with DM using the widely used SGLT2 inhibitors studied (canagliflozin, dapagliflozin, empagliflozin, and ertugliflozin) can safely and accurately measure their BG using up-to-date SMBG systems without any concern of interference.
Footnotes
Abbreviations
ADA, American Diabetes Association; BGM, blood glucose meter; Cmax, maximal plasma concentration; DM, diabetes mellitus; EMA, European Medical Agency; HCP, health care professional; ISO, International Organization for Standardization; SGLT2, sodium-glucose transport protein 2; SMBG, self-monitoring of blood glucose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Both authors are full-time employees of Roche Diabetes Care GmbH.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Roche Diabetes Care GmbH.
