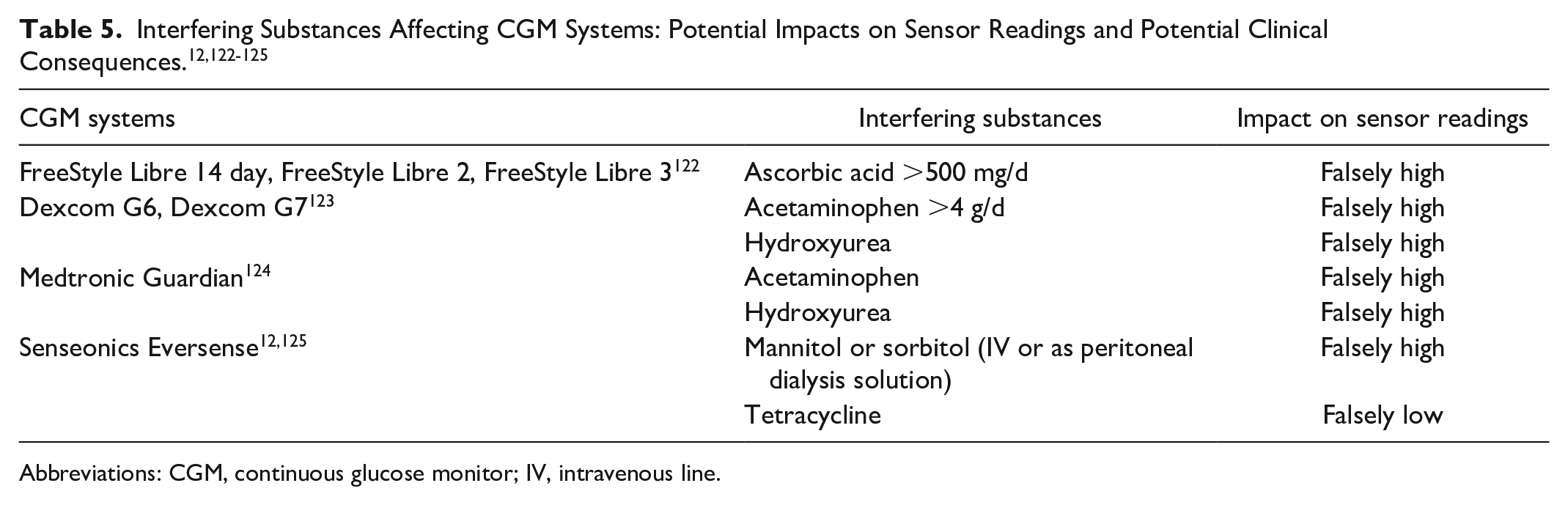Abstract
This report represents the conclusions of 15 experts in nephrology and endocrinology, based on their knowledge of key studies and evidence in the field, on the role of continuous glucose monitors (CGMs) in patients with diabetes and chronic kidney disease (CKD), including those receiving dialysis. The experts discussed issues related to CGM accuracy, indications, education, clinical outcomes, quality of life, research gaps, and barriers to dissemination. Three main goals of management for patients with CKD and diabetes were identified: (1) greater use of CGMs for better glycemic monitoring and management, (2) further research evaluating the accuracy, feasibility, outcomes, and potential value of CGMs in patients with end-stage kidney disease (ESKD) on hemodialysis, and (3) equitable access to CGM technology for patients with CKD. The experts also developed 15 conclusions regarding the use of CGMs in this population related to CGMs’ unique delivery of both real-time information that can guide monitoring and management of glycemia and continuous and predictive data in this population, which is at higher risk for hypoglycemia and hyperglycemia. The group noted three major clinical gaps: (1) CGMs are not routinely prescribed for patients with diabetes and CKD; (2) CGMs are not approved by the United States Food and Drug Administration (FDA) for patients with diabetes who are on dialysis; and (3) CGMs are not routinely available to all of those who need them because of structural barriers in the health care system. These gaps can be improved with greater stakeholder collaboration, education, and awareness brought to the use of CGM technology in CKD.
Keywords
Introduction
Chronic kidney disease (CKD) is defined as decreased kidney function with an estimated glomerular filtration rate (eGFR) of <60 mL/min/1.73 m2 and/or the presence of kidney damage (ie, pathologic abnormalities ascertained by biopsy or imaging, urinary sediment abnormalities, or increased albuminuria) for ≥3 months.1,2 Regarding the natural history of CKD, the stages of this disease are presented in Table 1. Advanced CKD requiring dialysis or kidney transplantation is referred to as end-stage kidney disease (ESKD), although more recently the term kidney failure (defined as an eGFR <15 mL/min/1.73 m2 or treatment with dialysis) has been recommended as a more patient-centered description. 3 The various manifestations of kidney diseases include acute kidney injury (AKI), which is defined as (1) an abrupt decline in kidney function manifested by an increase in serum creatinine, urea, and other nitrogenous waste products; (2) dysregulation of extracellular volume and electrolytes; and (3) development of oliguria. Patients with underlying CKD are at heightened risk of developing AKI4,5 and vice-versa. 6
Stages of Chronic Kidney Disease (CKD).
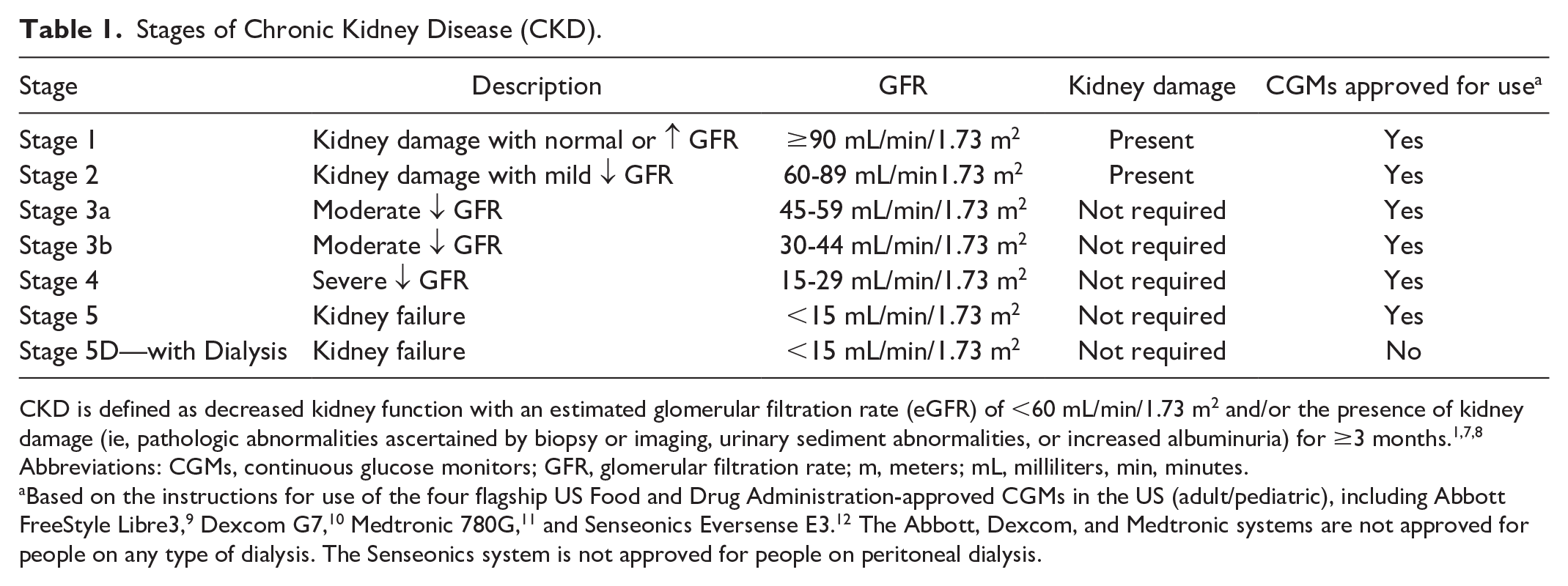
CKD is defined as decreased kidney function with an estimated glomerular filtration rate (eGFR) of <60 mL/min/1.73 m2 and/or the presence of kidney damage (ie, pathologic abnormalities ascertained by biopsy or imaging, urinary sediment abnormalities, or increased albuminuria) for ≥3 months.1,7,8
Abbreviations: CGMs, continuous glucose monitors; GFR, glomerular filtration rate; m, meters; mL, milliliters, min, minutes.
Based on the instructions for use of the four flagship US Food and Drug Administration-approved CGMs in the US (adult/pediatric), including Abbott FreeStyle Libre3, 9 Dexcom G7, 10 Medtronic 780G, 11 and Senseonics Eversense E3. 12 The Abbott, Dexcom, and Medtronic systems are not approved for people on any type of dialysis. The Senseonics system is not approved for people on peritoneal dialysis.
Diabetes mellitus (DM) is the leading cause of CKD worldwide, 13 and it dominantly contributes to the high morbidity and mortality (ie, third fastest-growing cause of death globally) of kidney diseases. The term diabetic kidney disease (DKD) is used to describe a clinical diagnosis that is based on presence of albuminuria and/or impaired kidney function in patients with diabetes, but does not per se indicate the specific underlying pathologic phenotype of kidney damage ensuing from diabetes.14-16 In contrast, the term diabetic nephropathy has traditionally been used to describe the presence of albuminuria in a setting of retinopathy (typically in patients with type 1 diabetes [T1D]), as a sign of diabetic glomerulopathy characterized by glomerular basement membrane thickening, endothelial damage, mesangial expansion and nodules, and loss of podocytes. Given that various forms of CKD occur because of diabetes (ie, tubulointerstitial disease, nonclassical glomerular lesions, etc), the term DKD aids in clarifying that the underlying pathologic phenotype is often unknown.
Compared to those with other causes of CKD, patients with DKD have a more rapid decline in kidney function and substantially higher death risk. 17 Hence, addressing the high morbidity and mortality of DKD requires a multifaceted and multidisciplinary approach, as shown in Figure 1, in which adequate glycemic monitoring and control are key cornerstones of management. 18 It is well known that traditional glycemic metrics have limitations in patients affected by advanced CKD with respect to accuracy, convenience, and provision of a complete representation of glycemic status.19,20 Hence, there has been growing recognition of the critical role of continuous glucose monitors (CGMs) in addressing clinical and research gaps, although the widespread use of this tool remains nascent in the DKD population. 21
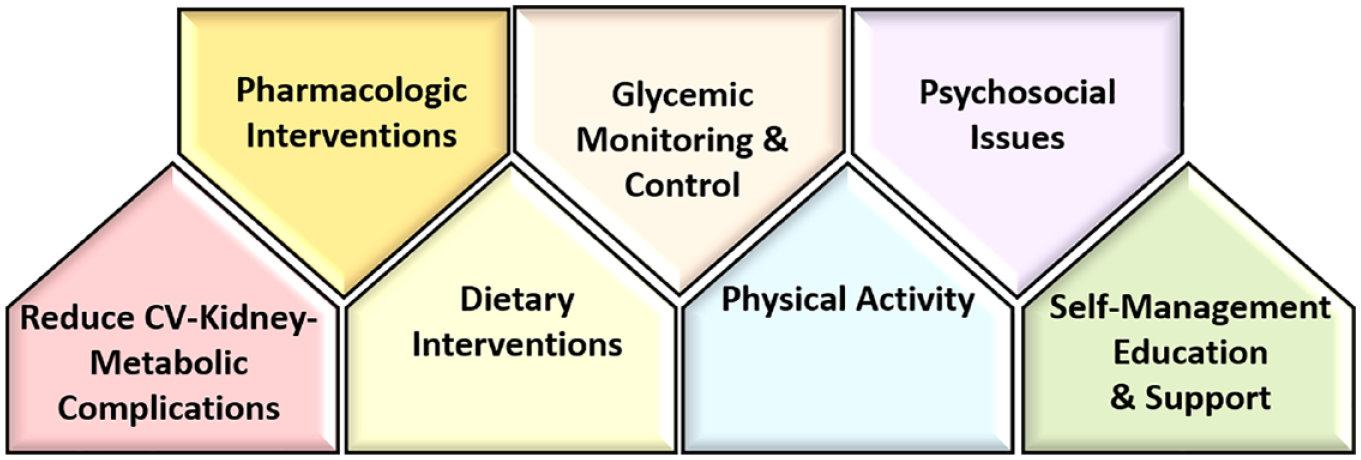
A multifaceted and multidisciplinary approach to DKD management that includes adequate glycemic control and monitoring, which are a key cornerstone.
To further inform the use of CGMs in patients with diabetes and CKD, including those receiving dialysis, Diabetes Technology Society (DTS) convened a group of experts in nephrology and endocrinology which met on January 30, 2024, May 31, 2024, and August 21, 2024 to discuss existing data, based on their knowledge of key studies and evidence in the field, about using CGM in diabetes patients with CKD with respect to accuracy, indications, clinical endpoints, patient-reported outcomes, barriers to widespread use, current knowledge gaps, and future research directions. The group formulated 15 key conclusions on the use of CGMs in patients with diabetes and CKD, and also highlighted several major and high-priority clinical gaps necessitating further collaboration among key stakeholders in order to advance the clinical implementation and further research of CGM technology toward the goal of improved outcomes in the DKD population. These gaps and conclusions are described in this report.
Epidemiology of Chronic Kidney Disease and End-Stage Kidney Disease and Diabetes in the United States and Globally
The number of patients affected by DKD has been rising worldwide, in parallel with the increase in the number of patients with DM.
In different countries, and in populations with different socioeconomic characteristics, the epidemiology of DKD has been affected by some trends that are increasing and other trends that are decreasing the incidence and prevalence of DKD.
The overall incidence of ESKD attributable to DM increased almost three-fold between 2000 and 2015.
Regarding the epidemiology of CKD approximately 37 million adults in the United States (14% of the US adult population) have CKD, among whom 90% are unaware of their condition. 22 In Europe, approximately 10% of the population has CKD. 23 As many as 850 million people worldwide are estimated to have kidney disease, including non-dialysis dependent CKD, ESKD, and AKI. 24
Moreover, type 2 diabetes mellitus (T2D) has developed into a pandemic, currently affecting 537 million adults worldwide, with numbers projected to rise to 643 million by 2030 and to 783 million by 2045. 25 T2D has spread to all parts of the world, with (1) 75% of affected adults being residents of low-to-middle income countries, (2) approximately one trillion US dollars being spent for annual worldwide health care expenditures, and (3) 6.7 million deaths worldwide being attributed to this disease in 2021. 25
One of the most common complications of T2D is CKD, and T2D is the single most important risk factor for CKD. 26 In parallel with the increasing numbers of individuals with T2D, the total number of those developing CKD attributable to DM has also been increasing worldwide: new CKD cases have shown a 74% rise, increasing from 1.4 million in 1990 to 2.4 million in 2017. 27 Although it is clear that the rising number of people with diabetes and the increase in the world’s population have fueled an increase in the overall number of patients with DKD, the population dynamics of DKD are also affected by other phenomena, which often exert competing effects on the incidence and prevalence of DKD. For example, changes in diagnostic practices (e.g., increasing efforts to detect albuminuria) could lead to more people being diagnosed with DKD and hence increase its overall incidence, whereas changes in preventative efforts (e.g., better glucose and blood pressure control in patients with diabetes without CKD) could lead to fewer people being diagnosed with DKD. Changes in treatment practices (e.g., the introduction of new medications) could lead to better clinical outcomes in patients with DKD, resulting in a higher overall disease prevalence. To further complicate matters, these various changes may occur at different times and to varying extents across different countries and may even differ within the same country for people with heterogeneous characteristics (e.g., unfavorably affecting those with lower socioeconomic status), which explains why the temporal and geographic population dynamics of DKD have shown significant heterogeneity.
The reported prevalence of CKD among patients with T2D varies from 24% in Denmark 28 to 28% in Spain 29 and the Netherlands, 30 38% in the United States, 31 42% in the United Kingdom, 32 and 46% in Japan. 33 Notwithstanding such geographic heterogeneity, the proportion of individuals with DM affected by DKD 31 and the country-specific prevalence of DKD appears to have been stable over time, but important nuances color the picture. A study examining temporal changes in DKD in the US National Health and Nutrition Examination Survey (NHANES) reported a complex picture, with an overall flat prevalence over time, but with discrepant temporal trends for the prevalences of albuminuria (declining over time in younger non-Hispanic White adults) and decreased eGFR (increasing over time in all studied groups). 34 The declining trend over time in albuminuria in select subgroups may have been a result of enhanced use of medications known to reduce albuminuria (e.g., renin-angiotensin-aldosterone system inhibitors [RAASi]) or improved control of hyperglycemia. 34 Recent evidence has demonstrated that other drug classes can decrease albuminuria in CKD, including sodium–glucose cotransporter-2 inhibitors, glucagon-like peptide 1 receptor agonists, mineralocorticoid receptor antagonists, endothelin receptor antagonists, and Janus kinase inhibitors. 35
The worldwide age-standardized incidence rate (ASIR) of DKD was 29.15 per 100,000 in 2017, 27 but higher incidence rates were seen in countries with lower socio-demographic indices. There has been an overall decrease of −0.40%/year in the ASIR of DKD worldwide from 1990 to 2017, 27 but important regional differences have been present, as shown in Figure 2. The largest decrease in the ASIR for DKD was observed in China, whereas the largest increase was in the United States. 27
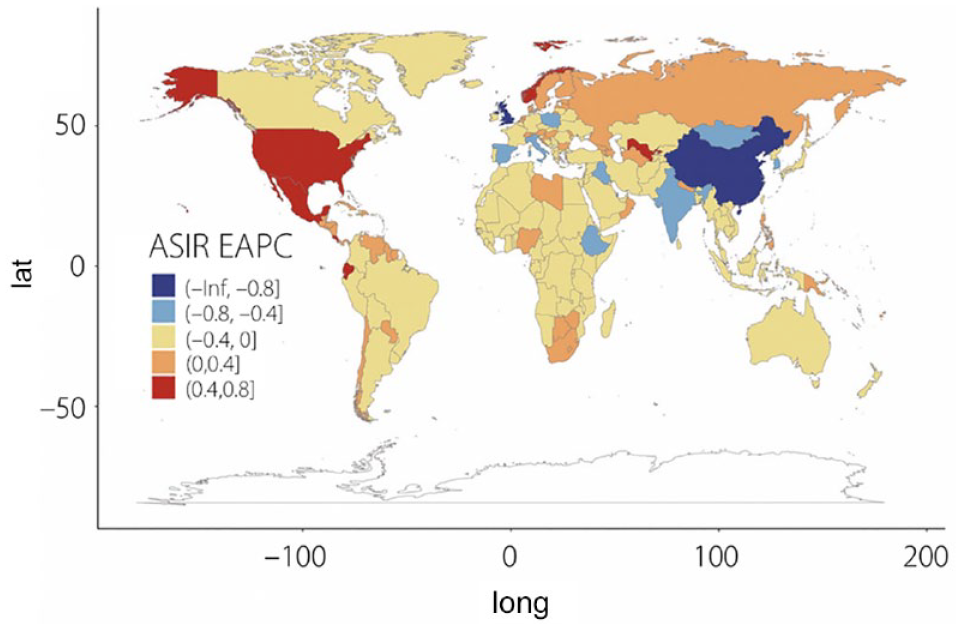
Estimated annual percentage change in age-standardized incidence rates of diabetic kidney disease between 1990 and 2017. Figure reproduced from Li et al 27 under the CC BY-NC license (http://creativecommons.org/licenses/by-nc/4.0/License).
Once kidney disease develops, patients with DKD, compared to patients with CKD from other etiologies, display more rapid loss of kidney function, 36 which may explain the overall higher prevalence of T2D observed in patients with more advanced stages of CKD. 17 Data from NHANES examining temporal trends in the prevalence of T2D among patients with different stages of CKD shows a complex picture, with a decline over time in the prevalence of DM in patients with stages 4 to 5 CKD along with a concomitant increase in the prevalence of DM in patients with earlier stages of CKD. 17 It is possible that these complex trends may have been the results of secular changes in diagnostic and therapeutic practices resulting in earlier disease detection and slower disease progression.
T2D is also the most important cause of ESKD. Between 2000 and 2015, the proportion of ESKD patients with diabetes has increased among all patients with ESKD worldwide from 19 to 30%. 37 During the same time span, the worldwide incidence of ESKD attributed to DM increased almost three-fold (from 375.8 to 1016.0/million). 37 Most of this rise was attributable to T2D, with the incidence of ESKD attributable to T1D remaining stable over time. 38 There are important regional differences in the incidence of ESKD attributable to DM, with the highest incidences observed in the Western Pacific, Asia, and the United States. 37
DKD exerts a considerable burden on health care systems in the form of high health care resource utilization and considerable costs. A population-based analysis of US administrative claims data indicated that patients with incident DKD incurred a total cost of $24 029 per person in the first year after DKD identification. Costs were accentuated in patients with more advanced stages of CKD. Patients who have stage 5 CKD, compared to patients with stage 1 CKD, incur a five-fold higher annual cost and a seven-fold higher inpatient hospitalization rate. 39 The costs of care for DKD in other countries are difficult to compare because of differences in health care systems. 23
Pathophysiology for Dysglycemia in Chronic Kidney Disease, End-Stage Kidney Disease, and Acute Kidney Injury
The kidney is a major locus of glucose regulation under normal conditions, serving as a site for glucose reabsorption, glucose utilization, and gluconeogenesis. These functions are impaired in CKD.
The two main reasons for hyperglycemia in patients with AKI, CKD, and ESKD (with and without DM) are insulin resistance and decreased insulin secretion.
Hypoglycemia is a major risk for individuals with AKI, CKD, and ESKD because of diabetes. Risk factors include impaired kidney gluconeogenesis, a decreased response of hormones that normally raise blood glucose levels (primarily catecholamines), reduced food intake, and a prolonged half-life of medications in patients with DM.
Normal Physiology
Glucose is freely filtered by the kidney glomerulus, and about 90% of it is reabsorbed in the proximal convoluted tubule via the sodium-glucose cotransporter-2 (SGLT2). In the presence of normal glucose levels, the remaining 10% is reabsorbed later in the nephron by the sodium-glucose cotransporter-1 (SGLT1). 40 The glucose transporter protein type 2 (GLUT2) is primarily responsible for glucose exit from proximal tubular cells. In addition, significant glucose uptake occurs via facilitative glucose transporters on the basolateral side of the distal tubular cells, which is mediated by the glucose transporter protein type 1 (GLUT1). The kidney is an important site of glucose utilization (ranging from 5% to 15% of total body glucose utilization) 41 because of its high metabolic demands. In addition, the kidneys may account for as much as 40% of total body gluconeogenesis, occurring in the proximal tubular cells. 42
Hypoglycemia Mechanisms
In patients with diabetes, hypoglycemia is defined as a blood glucose <70 mg/dL (3.9 mmol/L). In patients with kidney disease, an interplay of events may lead to hypoglycemia, 43 as shown in Figure 3. These factors include decreased food intake, reduced kidney gluconeogenesis from loss of functioning proximal tubular cells, 42 and a response of hormones, primarily catecholamines, that normally raise blood glucose levels. Inadequate increases in cortisol, growth hormone, and thyroid hormone in response to hypoglycemia may also play a role. Furthermore, in patients with diabetes, higher circulating concentrations of medications that lower blood glucose (eg, insulin, other hypoglycemic agents) may be present with standard doses because of decreased breakdown of these agents by the kidneys. Finally, dialysis predisposes to hypoglycemia because of decreased glucose concentrations in dialysate, intradialytic shifts, increased uptake by erythrocytes, and prolonged half-life of drugs that lower blood glycemia. 44
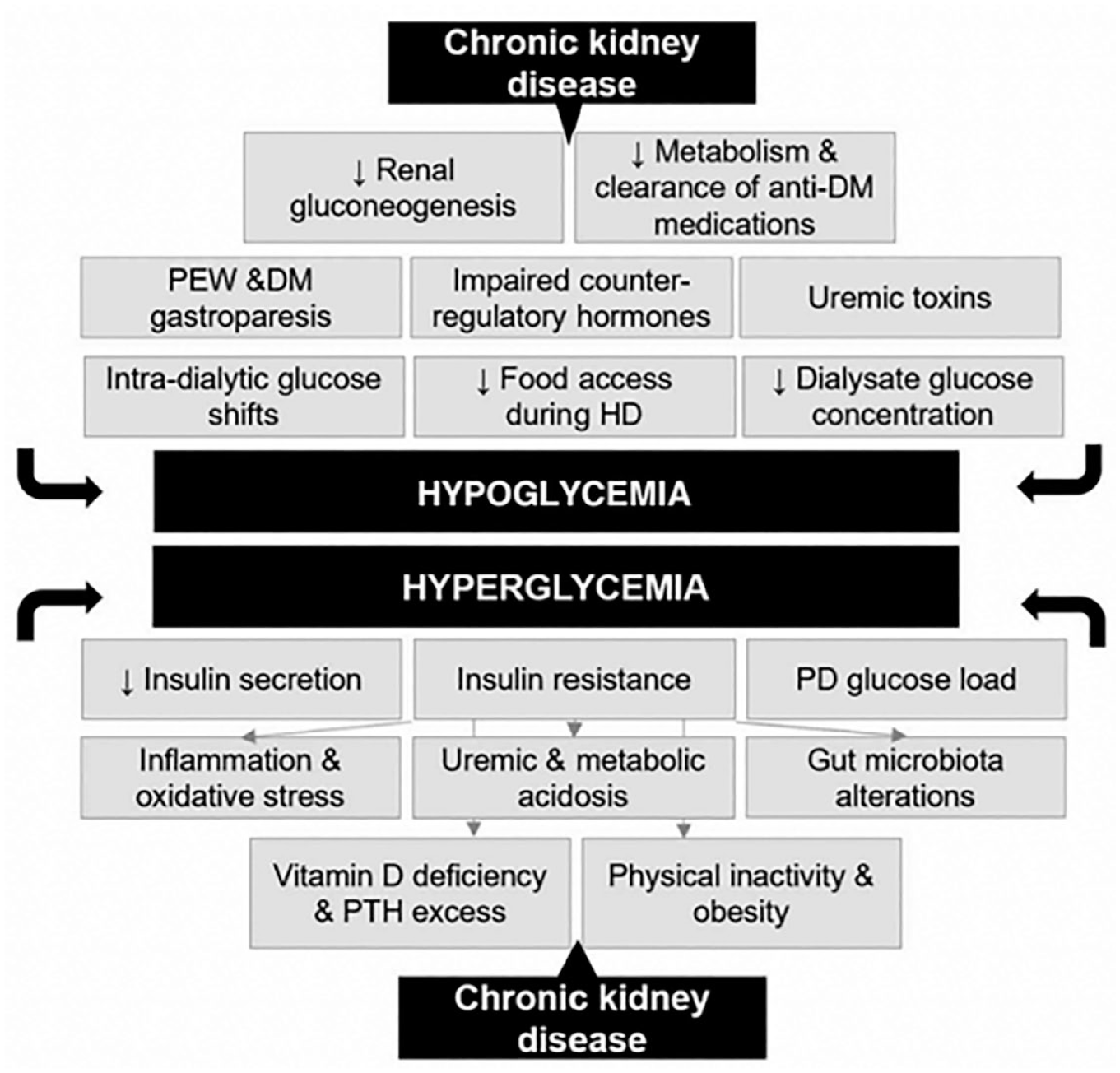
Risk factors for hypoglycemia and hyperglycemia in chronic kidney disease. Figure modified from Rhee et al. 18
Hyperglycemia Mechanisms
Hyperglycemia is defined as blood glucose >125 mg/dL (6.9 mmol/L) if fasting and >180 mg/dL (10 mmol/L) at 2 hours post prandial. 45 Insulin resistance occurs in patients with kidney disease with and without DM. 46 Reasons include increased inflammation, metabolic acidosis, oxidative stress, uremic toxins, physical inactivity, obesity, and other factors which have been shown to lead to impairment or degradation of specific proteins in the insulin signaling pathway (eg, by the ubiquitin proteasome system, which maintains the glomerular filtration barrier). 47 In addition to insulin resistance, decreased insulin secretion has also been observed (which may be mediated at least in part by modest elevations in circulating urea levels). 48
Effects of Chronic Kidney Disease and End-Stage Kidney Disease in Diabetes on Mean Glycemia, Glycated Hemoglobin, Short-Term Mean Glycemic Markers, Glycemic Variability, and Target Continuous Glucose Monitoring Glucose Profiles
Glycated hemoglobin (HbA1c), although considered to be a gold-standard metric of diabetes control in patients with CKD, has major limitations in its accuracy and may underestimate plasma glucose levels in this patient population.
Although glycated albumin (GA) and fructosamine offer particular benefits in assessing blood glucose control in patients with diabetes and advanced CKD and may be superior to HbA1c, neither GA nor fructosamine, are recommended by major guidelines (American Diabetes Association [ADA], European Association for the Study of Diabetes [EASD], American Association of Clinical Endocrinologists [AACE], Kidney Disease Improving Global Outcomes [KDIGO], Kidney Disease Outcomes Quality Initiative [KDOQI]) because of lack of standardization of both assays and lack of robust outcomes data.
Glucose control as assessed by CGM overcomes many disadvantages of traditional metrics; however, data showing correlation between CGM metrics and progression of CKD, morbidity, or mortality are not yet available.
Glycated hemoglobin is the product of a non-enzymatic reaction between glucose and the hemoglobin beta-chain, and in the general population, it is the gold-standard test for assessing long-term glycemic control. The main factors affecting HbA1c values include changes in red blood cell lifespan, glycation rate, and analytical assay interference, all of which are commonly observed in kidney disease. It is noteworthy that even in individuals without CKD essential discordances between HbA1c and mean glucose exist for a subgroup of patients.49,50 These are likely explained by genetic and biologic factors related to glycation rate and glucose transport into erythrocytes, especially in populations where red blood cell lifespan or hemoglobin glycation rates are altered even if analytical accuracy is preserved. Consequently, HbA1c levels may not be accurate in patients with advanced CKD, including those receiving dialysis. HbA1c levels may be falsely low in a setting of anemia,51,52 receipt of blood transfusions that can dilute patient blood with donor blood containing a lower concentration of HbA1c, 53 or any condition that shortens the erythrocyte lifespan, such as hemodialysis.54,55 Eryptosis, or shortened red blood cell lifespan due to suicidal erythrocyte death, causing accelerated erythrocyte loss 56 can occur with receipt of erythropoietin-stimulating agents in patients with CKD. 57
Conversely, HbA1c levels may be falsely elevated in a setting of elevated blood urea nitrogen concentration (ie, exposure to high urea concentrations promotes formation of carbamylated hemoglobin, which cannot be distinguished from HbA1c in certain assays). 58 It should be noted that although metabolic acidosis does not directly affect the rate of hemoglobin glycation, conditions leading to metabolic acidosis, such as CKD and poorly controlled diabetes, can affect glycation rates. 59 On average, HbA1c underestimates plasma glucose levels in the advanced CKD population, when compared with plasma glucose assessed by CGM, GA, or fructosamine. 19 Furthermore, this metric provides no information on glucose variability or hypoglycemia.19,60,61 Yet, the clinical utility of HbA1c is related to its well-known association with diabetes-related complications including mortality, and target HbA1c concentrations are well outlined by national organizations including ADA and KDIGO.
The GA, formed by the non-enzymatic glycation of albumin, reflects the glycemic status within the preceding two to three weeks, given the three-week half-life of albumin. It is not influenced by factors that affect HbA1c through changes in red blood cell lifespan (associated with iron, vitamin B12 and folate deficiency and/or therapy, hemolytic anemia, altered erythropoietin levels), altered hemoglobin glycation rates, or analytical assay interference (present with elevated blood urea nitrogen concentrations). However, conditions of increased protein catabolism such as chronic inflammation, nephrotic range proteinuria, and protein-energy wasting/malnutrition leading to hypoalbuminemia will affect accuracy of GA levels. Additional clinical value of GA is conveyed by the well-documented association of elevated GA levels with progression of microvascular complications and all-cause and cardiovascular mortality for those receiving dialysis. 62 In addition, a meta-analysis of 24 studies with 3928 patients evaluating the correlation between GA or HbA1c against average glucose concentrations reported GA to be superior to HbA1c in assessing average blood glucose concentrations in people with CKD. 63
Fructosamine, a measure of non-enzymatic glycation of all circulating proteins, including albumin, globulins, and lipoproteins, is considered to be an alternative to HbA1c measurement in situations where HbA1c is not reliable. Similarly to GA, fructosamine is influenced by alterations in protein turnover as well as changes in blood concentrations of urea. Neither blood testing of fructosamine nor GA are yet recommended by ADA, EASD, AACE, KDIGO, or KDOQI guidelines for the general population or for CKD patients because of lack of standardization of assays of these substances and robust outcomes.
1,5-anhydroglucitol (1,5-AG) is a metabolically inert polyol that competes with glucose for reabsorption in the kidneys. The 1,5-AG levels in blood change within 24 hours because of glucose’s competitive inhibition of 1,5-AG reabsorption in the kidney tubules and is a marker of short-term glycemic control. When glucose levels rise, urinary loss of 1,5-AG occurs, and circulating levels fall but the converse is not true (ie, when glucose levels fall, circulating levels of 1,5-AG do not rise). The concentration of this substance in blood is not affected by turnover of erythrocytes or proteins. 64
A CGM provides a detailed analysis of patients’ glucose profiles, including metrics on mean sensor glucose, glucose variability, as well as time in range, above range, and below range. It is recognized that a clear link has not yet been established between CGM metrics and progression of CKD, morbidity, or mortality. 21
Specific CGM-determined targets for the amount of time per day spent in various levels of glycemia for patients with CKD, including those receiving dialysis, have not been formulated by ADA, KDIGO, or KDOQI because of a lack of data in the kidney disease population. Target percentages of time can be extrapolated from the 2019 International Consensus on Time in Range Report 65 for high-risk patients (defined as older patients as well as others at high risk of severe hypoglycemia, including those with kidney disease). 65 These targets are presented in Table 2. These goals need to be further evaluated specifically in populations with CKD plus or minus ESKD.
Glycemic Targets for High-Risk Patients Who Are Using Continuous Glucose Monitoring as Recommended in the 2019 International Consensus on Time in Range Report. 65
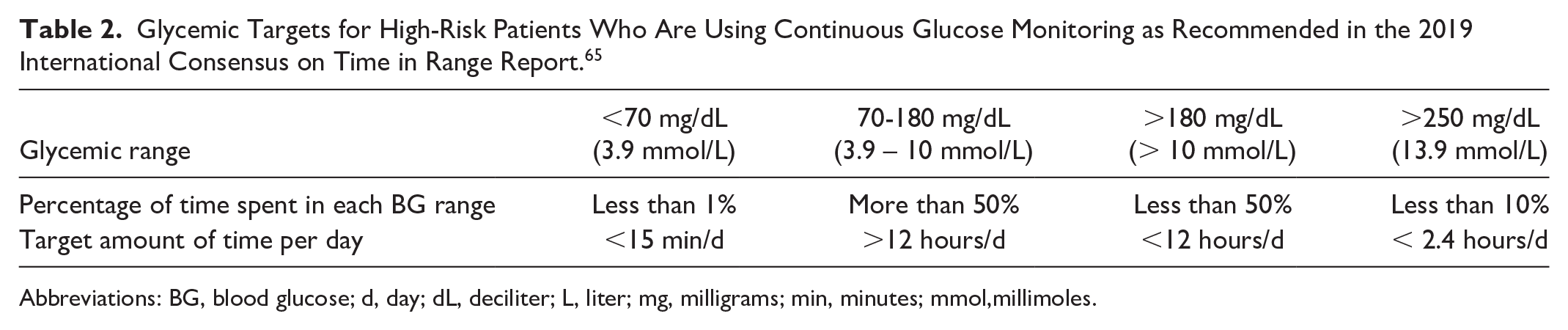
Abbreviations: BG, blood glucose; d, day; dL, deciliter; L, liter; mg, milligrams; min, minutes; mmol,millimoles.
Results From Continuous Glucose Monitor Studies in Chronic Kidney Disease/End-Stage Kidney Disease With Diabetes in Free Living Populations Including Clinical Outcomes and Risk Factors Due to Hypoglycemia and Hyperglycemia
When assessed by CGMs, patients with ESKD are frequently observed to have hyperglycemia, with large excursions occurring most frequently on the day after dialysis.
Hypoglycemic episodes are not the most frequent excursions, however, when they occur, these events are usually severe, prolonged, and associated with high concern/fear by dialysis staff, clinicians, and patients.
When assessed by CGM, patients with ESKD and “burnt-out diabetes,” as defined by previously established T2D and normal HbA1c, are observed to have frequent and prolonged hyperglycemia, with ~4 hours per day of glycemia levels >180 mg/dL (10 mmol/L). This illustrates the limitations of HbA1c to assess glycemic control in patients with ESKD.
As described in the section “Effects of Chronic Kidney Disease and End-Stage Kidney Disease in Diabetes on Mean Glycemia, Glycated Hemoglobin, Short-Term Mean Glycemic Markers, Glycemic Variability, and Target Continuous Glucose Monitoring Glucose Profiles,” there is well-established evidence that HbA1c lacks accuracy in those with ESKD. 66 Newer factory-calibrated CGM devices provide real-time glucose patterns and predictive hypoglycemic glucose alerts. Hence, these sensors have the potential to overcome these limitations.
Most studies of CGMs in the ESKD population have been focused on accuracy, with study durations of less than five days, and most have tested older sensors that are no longer available, as presented in Table 3. Overall, CGM studies in this population have demonstrated a tendency for lower glucose concentrations during hemodialysis, with nadirs toward the end of the dialysis sessions. 67 These studies also lack reports on newer CGM metrics, such as time in different glucose ranges, or glycemic variability metrics. The amount of available published data, however, from newer factory-calibrated sensors is more limited. Overall, studies using factory-calibrated sensors, with seven to 14 days of assessment, and three to five dialysis sessions, compared to studies using older sensors, provide a better assessment of glycemia in ESKD patients. These more recent studies have demonstrated poor glycemic control overall, with time-in-range of 30% to 50% only, and persistently higher time above glucose ranges of >180 (10 mmol/L) and >250 mg/dL (6.9 mmol/L). Surprisingly, time below range in hypoglycemia with glucose levels of <70 mg/dL (3.9 mmol/L) or <54 mg/dL (3 mmol/L) has been less commonly noticed, compared to hyperglycemia.
Results of the Studies Using Continuous Glucose Monitoring in Populations With (a) Stage 5 Chronic Kidney Disease on Dialysis and (b) Stages 3 to 4 Chronic Kidney Disease.
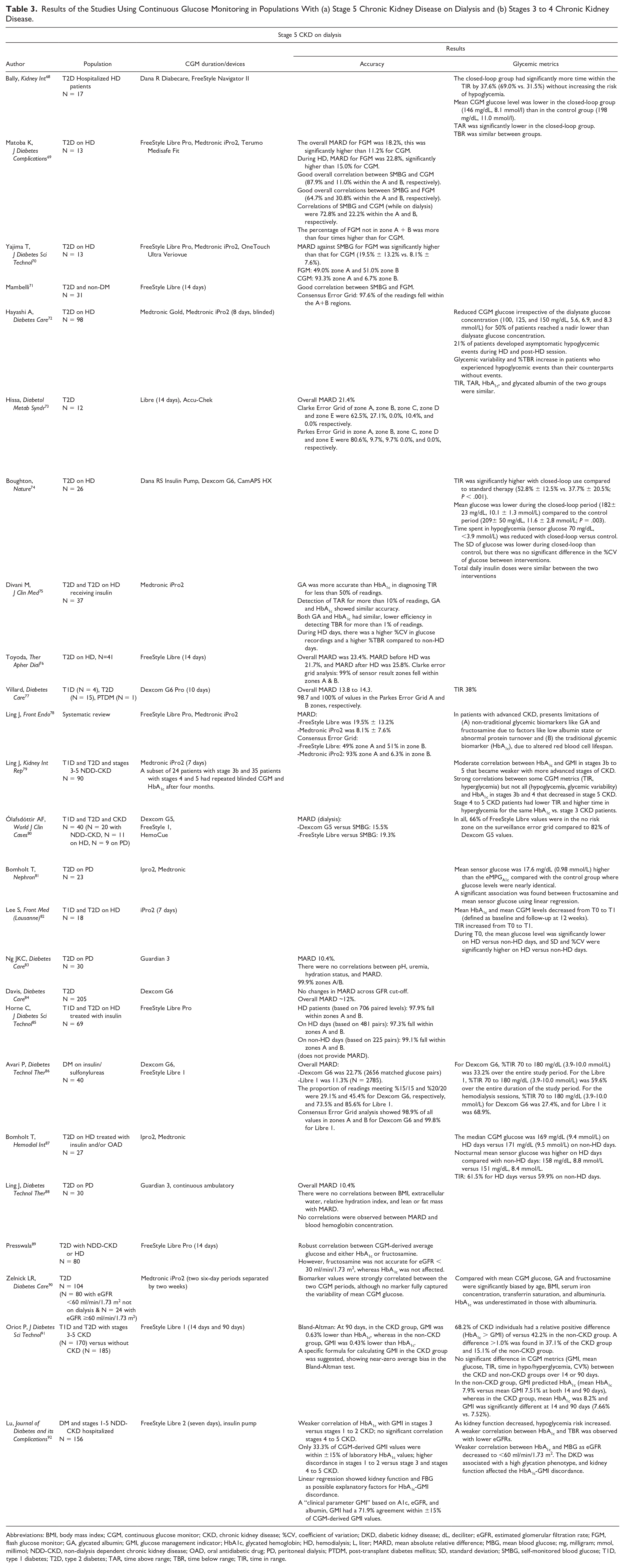
Abbreviations: BMI, body mass index; CGM, continuous glucose monitor; CKD, chronic kidney disease; %CV, coefficient of variation; DKD, diabetic kidney disease; dL, deciliter; eGFR, estimated glomerular filtration rate; FGM, flash glucose monitor; GA, glycated albumin; GMI, glucose management indicator; HbA1c, glycated hemoglobin; HD, hemodialysis; L, liter; MARD, mean absolute relative difference; MBG, mean blood glucose; mg, milligram; mmol, millimol; NDD-CKD, non-dialysis dependent chronic kidney disease; OAD, oral antidiabetic drug; PD, peritoneal dialysis; PTDM, post-transplant diabetes mellitus; SD, standard deviation; SMBG, self-monitored blood glucose; T1D, type 1 diabetes; T2D, type 2 diabetes; TAR, time above range; TBR, time below range; TIR, time in range.
The combination of normoglycemia without antidiabetic treatment in ESKD, as defined by HbA1c levels below 6.5% despite having previously established T2DM, a decline in insulin requirements, and spontaneous hypoglycemia in CKD/ESKD, has sometimes been referred to as “burnt-out diabetes.” The term “burnt-out diabetes” does not correspond to any resolution of underlying pathology and may reflect a decline in kidney insulin clearance, kidney gluconeogenesis, catecholamine release, and food intake due to anorexia. 93 The prognosis is poor when these features of CKD are present. 94 A recent study compared CGM patterns of patients with ESKD on hemodialysis without diabetes versus those with “burnt-out diabetes,” which remains a widely debated term, and patients in both groups had HbA1c concentrations of less than 6.5%. The latter group was observed to have CGM levels time above range of >180 mg/dL (10 mmol/L) for 4.1 hours per day, whereas the former group had time above range levels for 1.1 hours per day. 95 This study demonstrated that the “burnt-out” status is a reflection of the inadequacy of HbA1c in reflecting glycemic status among patients with ESKD on hemodialysis, rather than a true normalization of glycemia.
Moreover, a recent study in patients with T2D demonstrated that CGM metrics, such as mean glucose, glucose management indicator (GMI), or time-in-range 70 to 180 mg/dL (3.9-10 mmol/L), may be considered more appropriate indicators of glycemic control than the traditional HbA1c test in this population. In this study, GMI had a strong correlation with time-in-range (TIR), whereas HbA1c underestimated average glucose, by a mean difference of 0.74%. Furthermore, the authors noticed that 49%, 22%, and 29% of people had a discordance between HbA1c and GMI of >1%, 0.5% to 1%, and <0.5%, respectively. 96
Hypoglycemia is a risk factor for acute cardiac events. This may be of particular importance in patients with ESKD, where many have cardiovascular comorbidities.
In conclusion, emerging data using newer CGM devices has demonstrated that although patients with diabetes or ESKD receiving hemodialysis are at increased risk of severe hypoglycemia events, they are also at heightened risk of hyperglycemia. When assessed by CGMs, large glucose excursions into hyperglycemic ranges are frequently observed, even among those with HbA1c levels in the reference range. Although CGM sensors are an emergent technology in this cohort, there is need for more studies to inform regulatory approval of CGMs in this population.
Role of Continuous Glucose Monitoring in the Nutritional Management of Chronic Kidney Disease/End-Stage Kidney Disease and Diabetes
Dysglycemia is commonly observed in persons with diabetes and advanced CKD or ESKD, in whom alterations in dietary intake may be contributory factors (ie, hypoglycemia and/or hyperglycemia ensuing from appetite changes, protein-energy wasting, malnutrition, dietary restrictions, and/or time-restricted eating patterns related to hemodialysis treatment schedules).
Emerging CGM technologies may be potentially useful tools in informing lifestyle medicine approaches in the treatment of CKD/ESKD patients, including various nutritional management strategies utilized in this population.
Various diets have been proposed and/or studied as tools for CKD management, including the Plant-Dominant Low-Protein Diet (PLADO) and Plant-Focused Nutrition in Patients with Diabetes and CKD (PLAFOND) diets.
Patients with advanced CKD and ESKD including those with diabetes are at-risk for nutrition disorders, including protein-energy wasting, decreased appetite, and anorexia, which may potentially contribute to glycemic derangements in this population. 97 Protein-energy wasting in CKD/ESKD is characterized by excessive losses of protein/energy reserves. These nutritional disorders may be caused by multiple factors including decreased appetite (due to anorexigenic hormones, proinflammatory cytokines, and/or accumulation of uremic toxins), inadequate nutrient intake, catabolic effects of various metabolic abnormalities (eg, inflammation, increased catabolic and decreased anabolic hormones/activity, metabolic acidosis, and/or altered glucose/insulin homeostasis), nutrient losses via dialysis, and oxidant/carbonyl stress and are associated with major morbidity and mortality in this population.97-99 Patients with ESKD treated with dialysis may also be at heightened risk of undernutrition/malnutrition because of various dietary restrictions (ie, restricted dietary potassium, phosphorus, and fluid intake) and time-restricted eating patterns (ie, limited access to food during in-center hemodialysis treatments and/or during transit to and from the dialysis clinics on hemodialysis treatment days).100-102 Hence, CGM technology may be a promising tool in both the glycemic and dietary management of advanced CKD/ESKD patients, given their predisposition to nutrition disorders and a heightened risk of hypoglycemia. Macronutrient intake (including protein, carbohydrates, and fat) dietary composition (including micronutrients, minerals, and vitamins), dietary fiber content, animal versus plant sources of protein, and the balance between processed versus organic foods with respect to cardio-kidney-metabolic health can all affect glycemia.
An ongoing multicenter NIH R01 trial is examining the effects of a PLADO 103 adapted for DKD (NIH “PLAFOND Trial,” 104 in which patients with diabetes and NDD-CKD are being randomized to plant-dominant versus non-plant-dominant low-protein diets to determine the impact on various outcomes including glycemic control assessed by CGMs). 105 PLADO, PLAFOND, and other dietary regimens that have been proposed or tested for CKD management are presented in Table 4. There has also been growing interest in the role of CGM technology in guiding the dietary management of DKD by clinicians (including physicians and dietitians), thus facilitating a more personalized dietary approach consistent with precision medicine and precision nutrition strategies. Further research is needed to determine the role of CGM in informing both research and clinical practice in the nutritional management of DKD patients.
Overview of Diets Prescribed for the Management of Chronic Kidney Disease With or Without Diabetes.

Abbreviations: AKI, acute kidney injury; d, day; DPI, dietary protein intake; ESKD, end stage kidney disease; g, grams; kg, kilograms; mg, milligrams; NDD-CKD, non-dialysis dependent CKD; PKD, polycystic kidney disease; PLADO, Plant-Dominant Low-Protein Diet; PLAFOND, Plant-Focused Low-Protein Nutrition in Diabetes and CKD; RTA IV, renal tubular acidosis type 4.
Use of Continuous Glucose Monitoring in End-Stage Kidney Disease and Diabetes Patients (Hemodialysis and Peritoneal Dialysis, and Kidney Transplantation) to Prevent Hypoglycemia, Hyperglycemia, and Glycemic Variability
The CGM has emerged as a convenient, patient-centered tool for glycemic assessment providing a more comprehensive assessment of glycemic status than is possible with isolated biomarker tests such as HbA1c, fructosamine, GA, and self-monitored blood glucose (SMBG).
Further research is needed to determine the feasibility, efficacy, and safety of CGM in advanced DKD patients, including those receiving dialysis.
Given the high prevalence of hyperglycemia in the early post-transplant period and post-transplant diabetes mellitus among kidney transplant recipients, further research is needed to determine the efficacy and safety of CGM in this population.
A disproportionate burden of diabetes is present among patients with ESKD, in whom dysglycemia is associated with morbidity and mortality risk.19,106,107 Compared to patients without kidney disease, ESKD patients are at heightened risk of hypoglycemia 106 because of impaired kidney gluconeogenesis, decreased metabolism and clearance of insulin and other anti-glycemic medications, co-existing medical comorbidities (eg, diabetic gastroparesis and malnutrition), and accumulation of uremic toxins with glucose-lowering effects.18,19,106,108 Conversely, ESKD patients are also predisposed to hyperglycemia 106 because of increased insulin resistance, impaired insulin secretion, and exposure to high dialysate glucose concentrations in those receiving peritoneal dialysis. Intradialytic and post-dialytic dysglycemia are also frequent and under-recognized. For example, intradialytic hypoglycemia may ensue because of intradialytic glucose shifts into erythrocytes during hemodialysis treatment, secular changes in the use of lower dialysate glucose concentrations over time, and limited access to food during in-center hemodialysis. 44 Subsequently, post-dialysis rebound hyperglycemia may occur because of insulin removal during hemodialysis (vis-à-vis diffusion, convection, or adsorption) and a counter-regulatory hormone response in response to the hypoglycemia during the hemodialysis session. 44
One of the major clinical gaps in the management of patients with ESKD and diabetes is lack of access to a practical and reliable method for frequent glycemic assessment. Strengths and limitations of existing glycemic metrics in advanced CKD and ESKD patients are shown in Figure 4.19,20
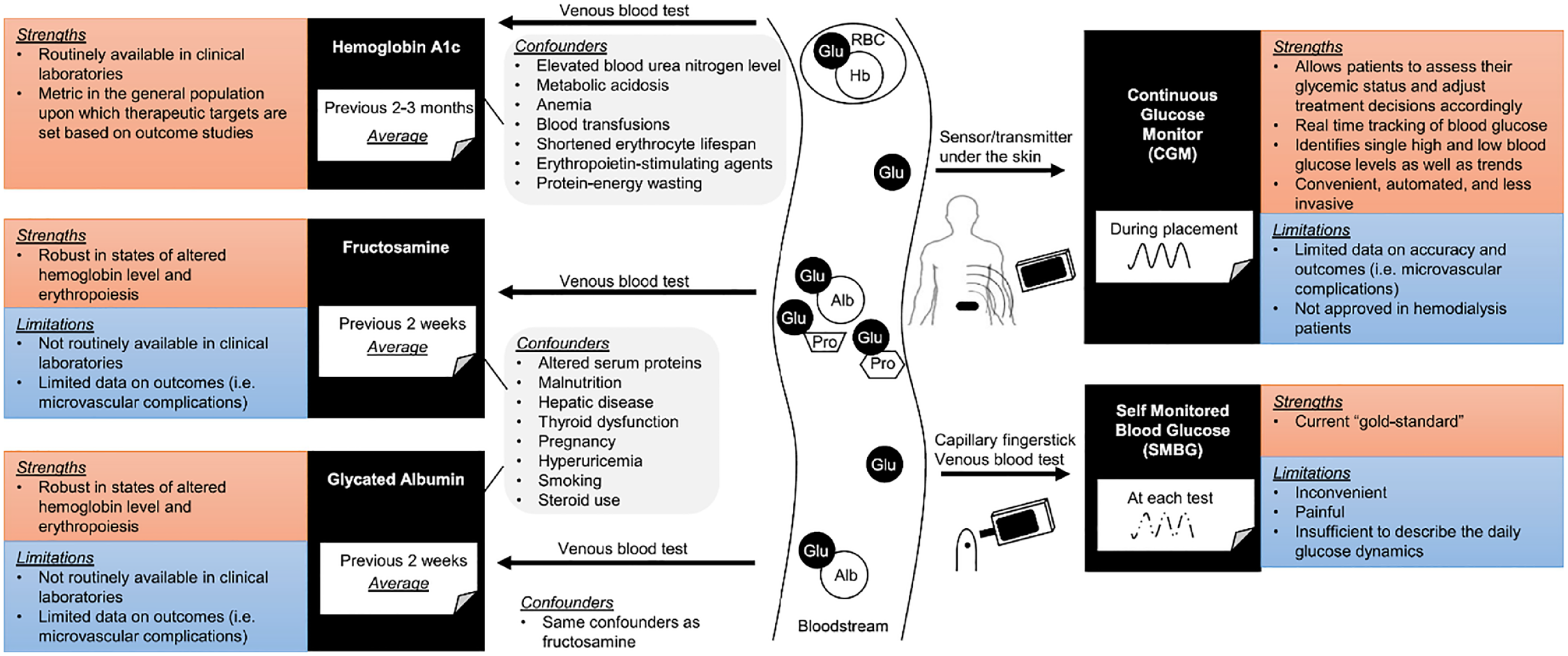
Strengths and limitations of existing glycemic metrics in advanced CKD and ESKD patients. Figure reproduced from Narasaki et al. 20
Although isolated biomarker tests, such as HbA1c, fructosamine, and GA are utilized for assessing long-term and intermediate glycemic status, respectively, as discussed in section “Effects of Chronic Kidney Disease and End-Stage Kidney Disease in Diabetes on Mean Glycemia, Glycated Hemoglobin, Short-Term Mean Glycemic Markers, Glycemic Variability, and Target Continuous Glucose Monitoring Glucose Profiles,” these glycemic metrics are known to have diminished accuracy in ESKD.19,20 Although SMBG or point-of-care (POC) glucose levels are considered to be standard of care for management of most patients with diabetes, these methods may also be affected by sample stability and other factors (ie, anemia, acute illness, medications, etc). However, HbA1c is not considered to be a gold standard in advanced CKD and dialysis patients. This has been concluded in the KDIGO guideline for kidney disease and diabetes, which states that the accuracy and precision of HbA1c measurements decline with advanced CKD (stages 4-5 CKD). This inaccuracy is particularly problematic for patients treated by dialysis, in whom HbA1c measurements have low reliability. Furthermore, frequent capillary fingerstick measurements may be inconvenient and painful for patients and do not provide a comprehensive around-the-clock assessment of glycemic status.
Hence, there is growing interest in the utilization of CGM as a convenient, automated, and comprehensive glycemic assessment method in ESKD patients. Although clinical trials in non-ESKD patients have shown that CGM confers improved glycemic control and clinical outcomes compared with conventional blood glucose monitoring, 109 CGM remains underutilized in ESKD patients in part because of unclear accuracy in this population. 110
There has also been growing interest in evaluating the effects of CGM versus conventional glycemic assessment methods in ESKD patients, particularly in clinical trials. In the “DIALYDIAB Pilot Trial,” which was a cross-over study of SMBG versus CGM conducted in 15 dialysis patients over a 12-week period, the CGM period resulted in more frequent treatment changes and better glycemic control, without an increase in hypoglycemia events. 111 The ongoing multicenter NIH-funded “Continuous Glucose Monitoring in Dialysis Patients to Overcome Dysglycemia Trial (CONDOR trial)” is currently randomizing hemodialysis patients with diabetes to real-time CGM versus usual care to determine the comparative effects of glycemic control, hypoglycemia indices, and patient-reported outcomes.
Although dialysis has been the dominant kidney replacement therapy for ESKD patients, kidney transplantation is considered the “gold-standard” treatment for patients with kidney failure, given its established improvement in quantity and quality of life. However, hyperglycemia in the early post-transplant period (due to surgical stress, infection, and high-dose steroids) and post-transplant DM (diagnosed later in the transplant course when patients have stable kidney function and are on maintenance immunosuppression) is prevalent complications among kidney transplant recipients.
These complications have been associated with worse short-term 112 and long-term 113 outcomes in the transplanted population. Among kidney transplant recipients, there are traditional and transplant-specific risk factors for post-transplant DM, the latter of which include immunosuppression regimens (glucocorticoids, calcineurin inhibitors, and inhibitors of mammalian target of rapamycin), hypomagnesemia, viral infections (cytomegalovirus, hepatitis C), and human leukocyte matching and donor characteristics. These and other risk factors for post-transplant diabetes among kidney transplant patients are shown in Figure 5. Given the high burden and ill effects of post-transplant DM in kidney transplant recipients, there has been increasing interest in the role of CGM in this population. There is limited data on the use of CGM in the post-kidney transplant setting114,115 including 1 study of 61 patients who underwent pancreatic surgery and various types of solid organ transplantation (liver, pancreas, islets of Langerhans, kidney). 116 Further research is needed to determine the accuracy of CGM and implications on clinical outcomes among kidney transplant recipients with or at-risk for post-transplant diabetes.

Risk factors for kidney transplant patients for developing post-transplant diabetes, including risk factors shared with diabetes in non-transplant patients and risk factors specific to kidney transplantation.
Accuracy of Continuous Glucose Monitoring in Populations With Chronic Kidney Disease/End-Stage Kidney Disease and Diabetes and Interfering Substances
Multiple studies have assessed CGM accuracy in patients with advanced CKD, including patients treated with hemodialysis and peritoneal dialysis, and have concluded that CGM is a useful tool for monitoring glucose in this population.
Minimally invasive CGM sensors measure glucose via enzymatic electrochemical reactions that are subject to interference by a variety of substances that can falsely increase or decrease sensor glucose values.
It is important to educate patients and professionals about interfering substances, how CGM readings may be impacted, and the importance of using an alternative glucose monitoring approach when interference is suspected and/or when symptoms do not match CGM glucose values.
Multiple studies have investigated the accuracy of CGM systems in patients with advanced CKD with and without diabetes, including patients undergoing hemodialysis and peritoneal dialysis.70,71,73,76,77,80,83,85,86,88 In a study of 20 US hemodialysis patients who underwent CGM with Dexcom G6-Pro CGM devices versus blood glucose (conducted as POC iSTAT measurements during hemodialysis and SMBG at home) by Villard et al, 77 the mean absolute relative difference (MARD) of CGM versus blood glucose was ~14%, and the majority of CGM values were found to be in zones A/B (zone A defined as no effect on clinical action, and zone B defined as altered clinical action with little or no effect on clinical outcome117) based on consensus grid analysis. 77 In another study by Ng et al83, 30 Hong Kong peritoneal dialysis patients wore a Medtronic Guardian Sensor 3 with Guardian Connect CGM. When CGM readings were compared against venous glucose assessment during an 8-hour in-clinic peritoneal dialysis session, the MARD of the matched CGM-blood glucose pairs was 10%. In another study of 30 US hemodialysis patients who underwent Dexcom G6 versus POC and venous blood glucose testing in the inpatient setting by Rhee et al, 118 the MARD of CGM-blood glucose pairs was ~20%, and consensus error grids showed nearly all CGM values in clinically acceptable zones A and B. In another study evaluating the accuracy of Dexcom G6 and Abbott FreeStyle Libre 1 versus blood glucose in 40 hemodialysis patients in the United Kingdom by Avari et al 86 (“ALPHA study”), the MARDs were ~23% and ~11%, respectively, and consensus error grid analysis showed the majority of CGM values in zones A and B. Examples of the correlations of two CGMs (Dexcom G5 and FreeStyle Libre) with reference data points obtained by a HemoCue capillary reference system are presented on two Bland-Altman scatterplots, where the differences between two measurements are plotted against their averages, in Figure 6, in 40 adults with diabetes and CKD.
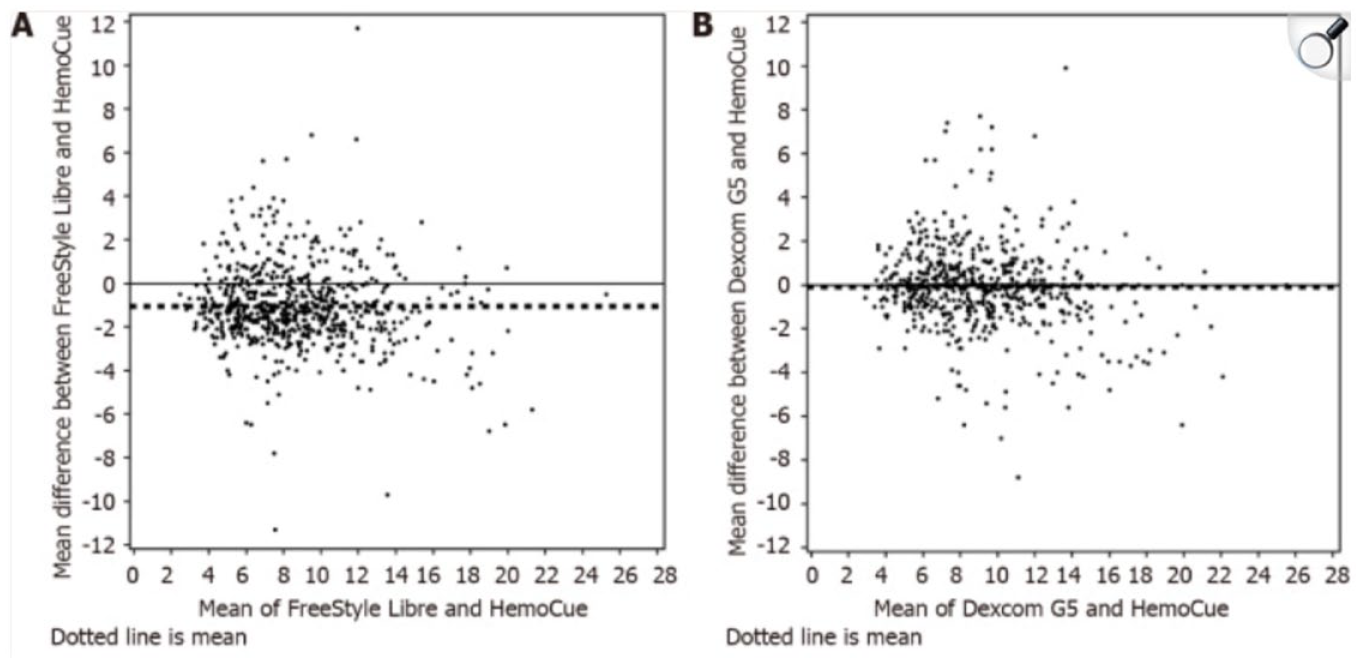
Bland-Altman Plot. A: All individual measurements FreeStyle Libre vs HemoCue; B: All individual measurements Dexcom G5 vs HemoCue. Thick dotted line represents the mean difference. Figure reproduced from Ólafsdóttir et al. 80 under the under the CC BY-NC license (https://creativecommons.org/licenses/by-nc/4.0/)
As presented in Table 3, measures of CGM point and trend accuracy in advanced CKD are generally improved with newer generation CGM systems.70,71,73,76,77,80,83,85,86 Although the reported MARD values in populations with advanced CKD are generally higher than would be expected in a general diabetes population, it is important to consider that CGM accuracy is diminished when there is high glucose variability and at glycemic extremes. Nevertheless, CGM is still a useful tool for monitoring CKD/ESKD patients. Notably, based on current best evidence, the 2022 KDIGO Clinical Practice Guideline for Diabetes Management in CKD concluded that CGM is not known to be biased by CKD or its treatments (eg, dialysis or kidney transplant) and therefore can be considered to (1) inform self-management and treatment decisions when HbA1c is discordant with measured glucose levels or clinical symptoms and (2) help prevent hypoglycemia by predictive alarms, trend arrows, and absolute values, even in the absence of an insulin delivery system. 66
Another consideration that may impact CGM accuracy in a variety of practice settings is interfering substances.119-121 The CGM sensors measure glucose via enzymatic electrochemical reactions that are subject to interference by a variety of substances as presented in Table 5.12,122-125 Depending on the CGM system and interfering substance, sensor readings can be falsely high or low, potentially resulting in missed hypoglycemia or hyperglycemia. 126 Sensor modifications in newer generation CGM systems have helped reduce interference with commonly used substances, such as acetaminophen and ascorbic acid. For example, the addition of a permselective membrane to the Dexcom G6 sensor has reduced acetaminophen interference. 127 Similarly, per Abbott Medical Affairs, FreeStyle Libre 2 Plus sensors have been modified to minimize interference by ascorbic acid. 128 Whereas icodextrin used in peritoneal dialysis 129 can interfere with some enzymes used in blood glucose monitors, 130 this substance does not interfere with the enzymes used in CGM sensors. Nevertheless, it is important that patients are educated about substances that may interfere with readings from their own CGM readings, and they must be guided to use alternative glucose monitoring approaches (e.g., fingerstick glucose monitoring) when interference is suspected and/or when symptoms do not match CGM glucose values.131,132
Impact of Continuous Glucose Monitoring on Patient-Reported Outcomes in Chronic Kidney Disease/End-Stage Kidney Disease and Diabetes
Studies in T1D and T2D populations without CKD have demonstrated short-term benefits of CGM on a number of patient-reported outcome metrics.
CGM may empower patients with diabetes and improve quality of life, with or even without salutary changes in glycemia.
Few studies have evaluated the impact of CGM on patient-reported outcomes in CKD and further research is needed.
Patients with CKD value quality of life, in addition to preservation of kidney function. 133 The CGM, particularly when used in real-time mode, offers patients an opportunity to better understand their glycemic control, such as blood glucose responses to changes in lifestyle and pharmacologic treatments. This knowledge and control may facilitate empowerment, reduce uncertainty and diabetes-related distress, and improve quality of life, with or even without salutary changes in glycemia. Over the long term, improved glycemic control with the use of CGM may also prevent complications, such as retinopathy and neuropathy, that interfere with quality of life. A drawback of CGM for some people who are not tech savvy or who have poor numeracy is that they can become frustrated by data overload. 134
Studies in T1D and T2D populations without CKD have demonstrated short-term benefits of CGM on several patient-reported outcome metrics. For example, in the GOLD trial of patients with T1D using multiple daily insulin injections, significantly higher scores on the Diabetes Treatment Satisfaction Questionnaire, World Health Organization-Five (WHO-5) Well-Being Index, and Hypoglycemic Confidence Questionnaire were achieved with real-time CGM, compared with SMBG by fingerstick. The HbA1c (the primary trial outcome), mean CGM glucose, and glycemic variability were also improved. 135 Observational studies and implementation projects have also demonstrated patient satisfaction with CGM. For example, in the setting of a comprehensive virtual diabetes clinic implemented for patients with T2D that included remote personalized lifestyle coaching and connected blood glucose meters in addition to a real-time CGM (Dexcom G5 or G6) mailed to their home, participants reported high satisfaction with CGM. Over 95% of participants were comfortable with remote insertion and reported improved understanding of eating and diabetes knowledge. The intervention was also associated with decreased diabetes distress and an improvement in HbA1c. 136
Few clinical trials of CGM have been conducted among patients with CKD68,74,111,137to assess patient-reported outcomes in this population. In one trial of 26 outpatient hemodialysis patients with diabetes, there was no difference in diabetes distress and an increased fear of hypoglycemia comparing an automated insulin delivery system to multiple daily insulin injections with blinded CGM, despite improved time in range (primary trial outcome), less time below range, and no difference in severe hypoglycemia. 74 No studies have rigorously evaluated the effect of CGM on symptoms related to diabetes complications in CKD. One cross-sectional study reported that a higher percentage of time in target range was associated with fewer symptoms of neuropathy, which may plausibly be reduced with improved long-term glycemic control, but this study was limited by observational design and cross-sectional analysis. 138
In one study comparing the GMI (which serves as a surrogate for HbA1c in people without CKD) noted that the correlation between GMI and HbA1c decreased with advancing CKD stages and became nonsignificant in stage G5. 78 These correlations by stage are shown in Figure 7.
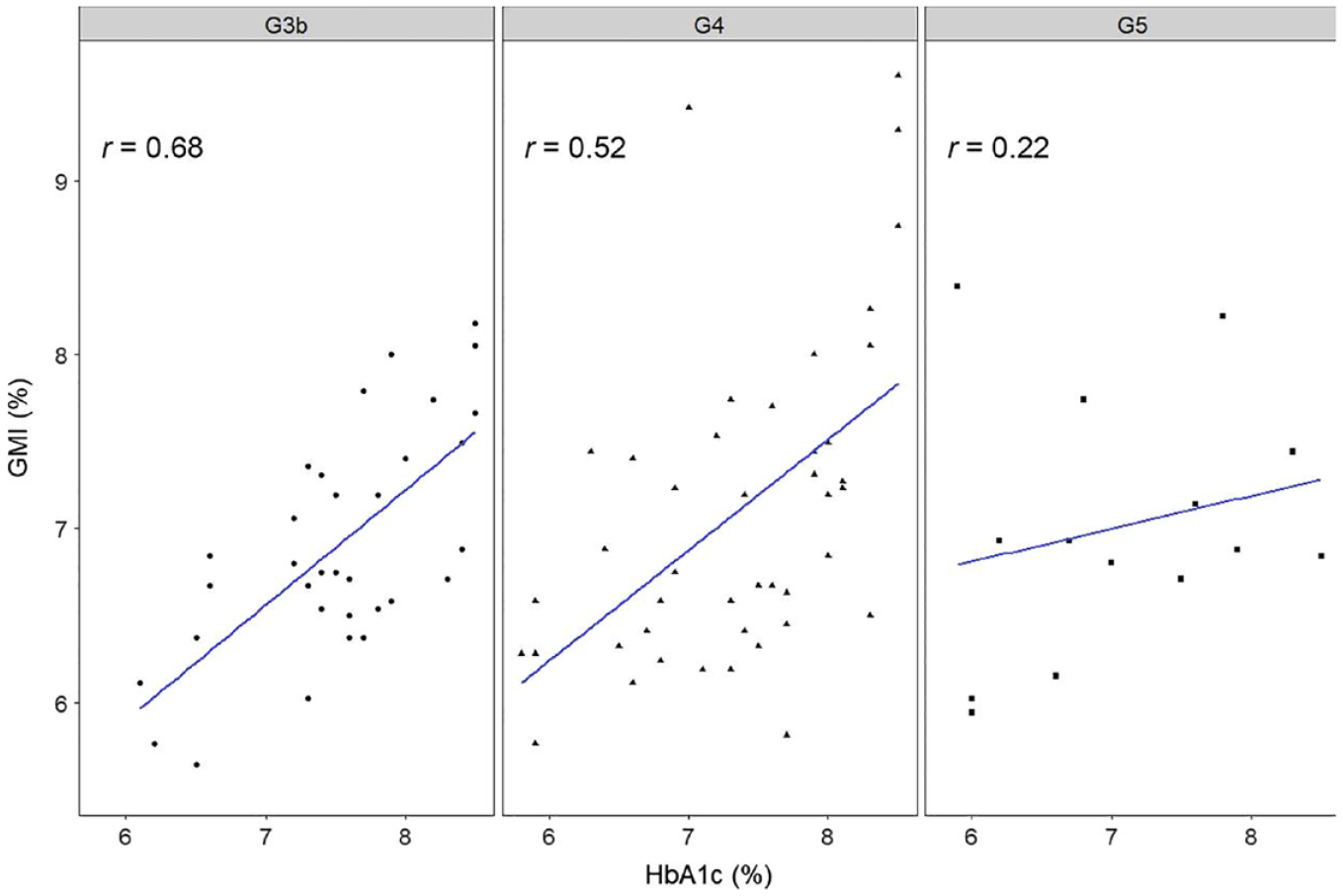
Continuous glucose monitoring metrics in the assessment of glycemia in moderate-to-advanced CKD (in stages 3b, 4, and 5) in diabetes. Figure reproduced from Ling et al 78 under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
Gaps, Controversies, and Recommendations for Research in Chronic Kidney Disease/End-Stage Kidney Disease, Including the Role of Continuous Glucose Monitoring in Managing Chronic Kidney Disease/End-Stage Kidney Disease and Acute Kidney Injury
Glucose control may have less kidney-related preventive effects in advanced CKD, but, on the contrary, these patients are generally at very high risk of other complications. They can benefit from sound glycemic management of other hyperglycemia-related complications, including retinopathy, diabetic neuropathy, diabetic foot ulcers, and cardiovascular disease. In addition, CGM is likely essential for preventing hypoglycemia in this population.
The CGM may be essential for patients with diabetes and CKD, not only for improving glucose management, but also (of particular importance) for assessing glucose control because anemia, iron deficiency, and other factors may influence the HbA1c level in this population.
Multicenter randomized trials of the effects of CGM in advanced kidney dysfunction are generally lacking in persons with T1D and T2D.
Today, it is unclear to what extent good glucose management prevents a decline in kidney function if established advanced kidney dysfunction exists in patients with diabetes. However, there are also many other reasons for obtaining good glucose control in this population. Persons with diabetes with significant kidney impairment have among the highest risk of diabetic foot ulcers, amputations, retinopathy, neuropathy, cardiovascular disease, and have a clear excess in mortality. These patients therefore will likely benefit from optimized glycemic control, although hallmark studies generally have included only patients without kidney complications or less severe forms of kidney dysfunction when determining the effects of poor glycemic control on complications. In addition, CGM is likely essential in preventing hypoglycemia in patients with DKD.
The optimal minimal frequency of placing a CGM on a person with diabetes is not known and the economic impact of using CGMs for treatment of patients with CKD is also not known. 78 However, studies in patients without advanced CKD show that continuous use of CGM is critical to obtain beneficial effects on HbA1c and hypoglycemia. 135
CGM is likely beneficial in patients with diabetes who have impaired kidney function from several perspectives. CGM has been shown to improve glucose control used both in conjunction with insulin pumps and insulin injections in persons with T1D as well as during treatment in T2D. 121 Two trials of using an automated insulin delivery system in T2D68,139 and observations of automated insulin delivery systems in patients with T1D 140 have demonstrated successful glycemic control. Therefore, as CGM seems to show reasonable accuracy in persons with kidney dysfunction, it is likely that the glucose management will improve also in this population.
Moreover, HbA1c has shown to be inaccurate in depicting the mean glucose level in a significant proportion of patients with diabetes without kidney dysfunction. In a study of patients with T1D, the GOLD, 135 and SILVER trials, 141 10% of patients deviated consistently over time by more than 0.8 HbA1c percentage units (8 mmol/mol) from the general trend between mean glucose and HbA1c. 49 Similar results exist from other studies. 50 These results are likely due to genetic factors influencing glucose transport into erythrocytes and glycation of hemoglobin.
For persons with diabetes and kidney dysfunction, there are additional factors that can disturb HbA1c as a biomarker for estimating the mean glucose level, including anemia and iron deficiency. Hence, CGM may be essential not only for optimizing glucose control but also for determining accurate mean glucose level in persons with diabetes and severe kidney dysfunction.
Although several studies exist indicating that CGM is likely a viable treatment option in patients with diabetes and kidney dysfunction with sufficient accuracy, randomized clinical trials of CGM use in patients with diabetes with severe kidney dysfunction are overall lacking. Such studies are urgently needed for this vulnerable patient group at high risk of diabetes complications to confirm benefits to glucose control as well as safety in this population. These and other future directions for research on the use of CGM in patients with CKD are shown in Figure 8.

Future directions for research on CGM use in patients with CKD/ESKD, including clinical use, randomized clinical trials, economic benefits, and patient benefits.
Barriers to Continuous Glucose Monitoring Access and Use in Populations With Diabetes and With Chronic Kidney Disease/End-Stage Kidney Disease
Barriers to widespread CGM use stem from four broad categories including the health care system, providers, patients, and CGM developers. The top barrier to CGM initiation relates to cost and insurance coverage.
Beyond financial costs, disparities exist in access to CGM, especially in older or minority populations, which can relate to provider practices and bias or patient factors.
Factory-calibrated sensors contain labeling indicating that their use in dialysis populations has not been validated, which may limit uptake in this population.
Rates of CGM use in populations with diabetes with CKD or ESKD are unclear. A telephone survey of community-dwelling adults in the United States reported that only 4% of individuals with diabetes who were eligible for CGM used one in 2020. 142 The CGM was used less by people who were older, were unemployed, had low income, and who had more comorbidities, including kidney disease. Other studies have also shown racial/ethnic disparities in CGM use with lower adoption in individuals identified as being of Black and/or Hispanic race/ethnicity compared with non-Hispanic White race/ethnicity.143,144 A study by the ADA confirmed these findings and concluded that individuals who are older, poorer, and from racial and ethnic minority groups are the least likely to be prescribed CGM devices. 145 As CKD and ESKD disproportionally affect people who are older, from minority backgrounds, and from lower socioeconomic status, 146 CGM sensor use is likely low in these populations.
Barriers to widespread use of CGM technology in patients with diabetes and especially those with CKD or ESKD can be grouped into four categories: (1) the health care system, (2) providers, (3) patients, and (4) CGM developers as shown in Figure 9. From surveys of patients with T1D, the most common barriers to CGM initiation are the cost and lack of insurance coverage.147,148 A requirement for preauthorization also limits access and creates a burden for providers and patients. In addition, defining CGM devices as durable medical equipment versus as a pharmacy benefit has been shown to significantly delay CGM initiation. 149 Other factors include provider practice bias 150 and comfort level with CGM technology. Streamlining the interpretation of CGM reports151,152 and integrating the data with electronic health records 153 could facilitate greater provider uptake of CGM devices. Patients also have reservations about CGMs, which may relate to their level of technology literacy and concerns about data safety and privacy. 154 Contact dermatitis, both irritant and allergic,155,156 as well as bruising 157 can occur with all CGM sensors attached to the skin. Resources are available to limit skin irritation (https://www.pantherprogram.org/skin-solutions), 158 but some patients may need to switch or discontinue CGM devices. 159 Starting patients on CGM can require time with educators and dedicated facilities in health care organizations. 160 For patients with advanced CKD, factory-calibrated CGM devices are not approved but such validation would help support use in this population. 161
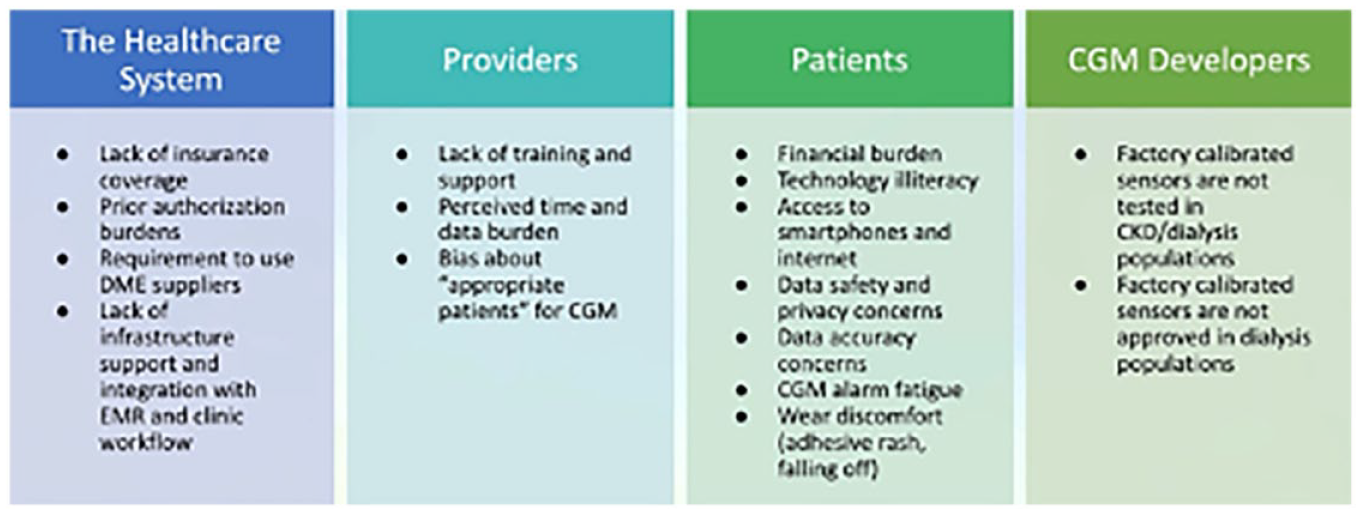
Barriers to widespread use of CGMs.
Discussion
Diabetes and CKD are highly prevalent worldwide, and both have synergistic effects in adversely impacting cardiovascular health, survival, patient-reported outcomes, and health care costs in DKD.17,26,162 In the setting of advanced CKD, glycemic derangements are magnified, and both hypoglycemia and hyperglycemia have been associated with worse outcomes in CKD patients including those receiving dialysis.108,163 It is well known that traditional glycemic metrics in the setting of advanced CKD have limitations in accuracy (HbA1c), are poorly standardized (GA, fructosamine), are inconvenient and provoke/exacerbate pain (fingerstick blood glucose), and fail to provide a comprehensive glycemic picture over an extended period.18,20 In contrast, CGM technology has emerged as a convenient and patient-centered tool that can address these clinical gaps while also informing glycemic control using a preemptive strategy (via alerts) in lieu of a reactive approach.
The uptake of CGM in DKD has been slow, in part because of challenges related to access (ensuing from high costs and non-approval on ESKD patients on dialysis) and limited knowledge and experience among providers and patients. However, there is increasing appreciation that more widespread use of CGM technology in DKD could positively transform glycemic monitoring and control by augmenting or replacing blood glucose testing in this population among primary care and multispecialty providers.17,26,162 In advanced CKD, given the disproportionate burden of hypoglycemia and hyperglycemia which are oftentimes severe and undetected, CGM holds promise as a tool that can ameliorate dysglycemia, improve clinical outcomes, and empower patients living with diabetes and CKD. In the non-CKD population, multiple large-scale randomized controlled trials have demonstrated the efficacy and safety of CGM in adults and children with T1D and T2D.109,164,165
Motivated by these observations, DTS convened a panel of experts in nephrology and diabetes to review and discuss the current state of evidence, challenges, and future research directions centered on the use of CGM technology in patients with diabetes and CKD. Conclusions were based on based on their knowledge of key studies and evidence in the field, rather than a formal systematic review of the literature. Compared to patients with diabetes without underlying CKD, there have been far fewer studies of the accuracy of CGMs or clinical outcomes in those with diabetes and CKD. However, limited existing data on the use of CGMs in patients with DKD, including those on dialysis, are favorable, and there are multiple ongoing well-characterized prospective cohort studies and clinical trials in progress that aim to address these knowledge gaps. However, although further investigations specific to DKD populations are needed, there was consensus among the panelists that this intervention in patients with diabetes who do not have DKD that high-quality research studies and widespread experience have demonstrated benefits of CGM109,164,165. The panel concluded that it is now time to much more broadly introduce this monitoring and managing tool across the DKD population. The group also agreed that, given the lack of other effective tools to avoid the frequent hypoglycemia/hyperglycemia events associated with morbidity and mortality in DKD, there is an urgent need for CGM implementation in parallel with ongoing research studies that will help inform and refine its use in this vulnerable population.
In evaluating the path toward more widespread CGM implementation in DKD, the panel discussed various barriers to widespread use of this technology that were grouped into four categories summarized in Figure 9, specifically (1) difficulties in navigating through the health care system to obtain these devices among providers and patients, (2) lack of knowledge and/or enthusiasm of some health care professionals in championing their use, (3) lack of health care technology literacy or concerns about privacy among some patients, and (4) restrictions in approval/clearance of CGM devices for certain CKD populations (ie, ESKD patients receiving dialysis). 166 In addition, the panel developed 15 key conclusions about clinical implementation and future research directions regarding the use of CGM by diabetes patients with CKD. These conclusions are presented in Table 6. The panel’s conclusions focused on seven areas related to the use of CGM for diabetes patients with CKD including (1) conducting more widespread monitoring of glycemic status using CGM technology worldwide, (2) defining the relationship between CGM metrics/targets and clinical outcomes, (3) determining the effects of CGM on reducing hypoglycemia/hyperglycemia, CKD progression, cardio-kidney-metabolic risk, health-related quality of life, as well as impact on health economic outcomes, (4) using CGM to better understand the natural history of glycemic derangements in CKD/ESKD, AKI, and during the dialytic procedure, (5) applying CGM to inform risk factors for dysglycemia, kidney nutrition interventions, antidiabetic medication use, and the dialysis prescription, (6) understanding the accuracy and potential biases/limitations of CGM (ie, interfering substances) in CKD that will inform further refinement, and (7) identifying the optimal CGM metrics/targets and frequency specific to DKD. It bears mention that among these conclusions, the panel highlighted three major clinical gaps warranting high-prioritization: (1) CGMs are not routinely prescribed for patients with diabetes and CKD, (2) CGMs are not yet approved by the US FDA for patients with diabetes who are on dialysis, and (3) CGMs are not routinely available to all those who need them because of structural barriers in the health care system. There is a compelling need for collectively addressing these gaps with further collaboration among key stakeholders in academia, industry, government agencies, health care systems, and patient advocacy groups, as well as across the disciplines of nephrology, endocrinology, primary care, cardiology, and health care technology, among others.
Fifteen Conclusions for the Use of Continuous Glucose Monitoring in Patients With Diabetes and Kidney Disease Developed by the Diabetes Technology Society Expert Panel on Continuous Glucose Monitoring in People With Chronic Kidney Disease.

In conclusion, CGM is a practical, convenient, and patient-centered technology that has demonstrated substantial benefits in patients with diabetes without CKD. The CGM shows promise in patients with DKD as a transformative tool that can overcome the limitations of traditional glycemic metrics, better inform glycemic management, and lead to improved clinical outcomes.
Footnotes
Acknowledgements
The authors thank Annamarie Sucher-Jones for her expert editorial assistance.
Abbreviations
1,5-AG, 1,5-anhydroglucitol; AKI, acute kidney injury; ASIR, age-standardized incidence rate; AACE, American Association of Clinical Endocrinologists; ADA, American Diabetes Association; BG, blood glucose; BMI, body mass index; CV, cardiovascular; CKD, chronic kidney disease; CAPD, continuous ambulatory peritoneal dialysis; CGM, continuous glucose monitor; CONDOR Trial, Continuous Glucose Monitoring in Dialysis Patients to Overcome Dysglycemia Trial; %CV, coefficient of variation; d, day; DDKT, deceased donor kidney transplantation; DKD, diabetic kidney disease; dL, deciliter; DM, diabetes mellitus; DME, durable medical equipment; DPI, dietary protein intake; DTS, Diabetes Technology Society; EAPC, estimated annual percentage change; EASD, European Association for the Study of Diabetes; EHR, electronic health records; EMR, electronic medical record; ESKD, end-stage kidney disease; eGFR, estimated glomerular filtration rate; FGM, flash glucose monitoring; G3b, stage 3b chronic kidney disease; G4, stage 4 chronic kidney disease; G5, stage 5 chronic kidney disease; GA, glycated albumin; GFR, glomerular filtration rate; GLUT 1, glucose transporter protein type 1; GLUT2, glucose transporter protein type 2; GMI, glucose management indicator; g, grams; HbA1c, Hemoglobin A1C; HD, hemodialysis; HLA, human leukocyte antigen; IV, intravenous line; KDIGO, Kidney Disease Improving Global Outcomes; KDOQI, Kidney Disease Improving Quality Initiative; kg, kilograms; L, liters; MARD, mean absolute relative difference; MBG, mean blood glucose; m, meters; mg, milligrams; min, minutes; mmol, millimoles; mTOR, mammalian target of rapamycin; NDD-CKD, non-dialysis dependent chronic kidney disease; NHANES, US National Health and Nutrition Examination Survey; OAD, oral antidiabetic drug; PEW, protein-energy wasting; PD, peritoneal dialysis; PKD, polycystic kidney disease; PLADO, plant-dominated low-protein diet; PLAFOND, plant-focused low-protein nutrition in diabetic kidney disease; POC, point-of-care; PTH, parathyroid hormone; PTDM, post-transplantation diabetes; RAASi, renin-angiotensin-aldosterone system inhibitors; RTA IV, renal tubular acidosis type 4; SGLT, sodium-glucose cotransporter; SMBG, self-monitored blood glucose; T1D, type 1 diabetes mellitus; T2D, type 2 diabetes mellitus; TAR, time above range; TBR, time below range; TIR, time in range; UACR, urine albumin-to-creatinine ratio; US FDA, United States Food and Drug Administration; vBGM, venous line blood glucose management; WHO-5, World Health Organization-Five.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.M.R. has received honoraria and/or grant support from AstraZeneca, Dexcom, Fresenius, and Vifor. R.Y.G. has nothing to disclose. D.K. has received research support from Abbott Diabetes Care and has been a Consultant to Sanofi, Better Therapeutics, Perfood, and Abbott Diagnostics. A.P.-T. provides consulting and research services to Novo Nordisk, Lilly, Bayer, Dexcom, and Medtronic on behalf of her employer but receives no direct or indirect reimbursement for the services. C.P.K. received consulting feels from Astra Zeneca, Bayer, Boehringer, Ingelheim, Cara Therapeutics, Pharmacosmos, ProKidney, Renibus, and Takeda. R.C.S. has nothing to disclose. A.T.D. has nothing to disclose. R.J.G. has received research support from Novo Nordisk, Dexcom, Boehringer, and Eli Lilly and consulting/advisory/honoraria fees from Abbott, Aztra Zeneca, Bayer, Boehringer, Dexcom, Eli Lilly, Novo Nordisk and Medtronic. R.J.G. is partially supported by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) of the National Institutes of Health (NIH) under Award Numbers P30DK111024 (R.J.G.), K23DK123384 (R.J.G.). and R03DK138255-01. K.K.-Z. has received funding from the NIH to examine CGM in patients with CKD. J.J.N. is partially supported by NIH research grant OT2OD032581 and reports personal fees and other support from Bayer AG, Sanofi, Novo Nordisk, Boehringer Ingelheim, Eli Lilly, Proteomics International, and Dexcom outside the submitted work. I.H.d.B. consults for Alnylam, Boehringer Ingelheim, Lilly, Miter, and Novo Nordisk. I.H.D.B. has also received equipment and supplies for research donated to his institution from Dexcom and Novo Nordisk Research, and funding to his institution from NIDDK and Breakthrough T1D. M.L. has been a consultant or received honoraria from Astra Zeneca, Boehringer Ingelheim, Eli Lilly, Nordic InfuCare, and Novo Nordisk, and has received research grants from Eli Lilly and Novo Nordisk, all outside of the submitted work. S.H.K. reports a relationship with Morphic Medical that includes consulting or advisory. S.H.K. is partially supported by P30DK116074. A.T.A. is a consultant for Liom. C.N.H. is a consultant for Liom. R.E.A. has nothing to disclose. T.T. has nothing to disclose. D.C.K. is a consultant for Afon, embecta, Glucotrack, Lifecare, Novo, Samsung, SynchNeuro, and Thirdwayv.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by grants from Abbott Diabetes Care and Dexcom.