Abstract

Removal of wearable transcutaneous continuous glucose monitoring (CGM) devices is recommended before magnetic resonance imaging (MRI), due to potential safety and accuracy concerns.
We report two cases of in vivo CGM use during MRI, and describe subsequent CGM accuracy.
A 74-year-old woman (case 1) with type 1 diabetes (T1D) inadvertently wore her Dexcom G6 CGM (DexCom Inc, San Diego, CA, USA), located on her abdomen, during a 25-minute cervical spine MRI (Magnetom Aera 1.5T; Siemens, Munich, Germany). Another 54-year-old woman (case 2) with T1D inadvertently wore her FreeStyle Libre 2 CGM (Abbott Diabetes Care, Doncaster, VIC, Australia), located on her arm, during a 15-minute lumbar spine MRI (Magnetom Sola 1.5T; Siemens, Munich, Germany) and subsequent 26-minute brain MRI (Magnetom Vida 3T; Siemens, Munich, Germany). In both cases, CGM devices did not dislodge during MRI, and continued to record continuous glucose readings during, and following, MRI (Figure 1a).
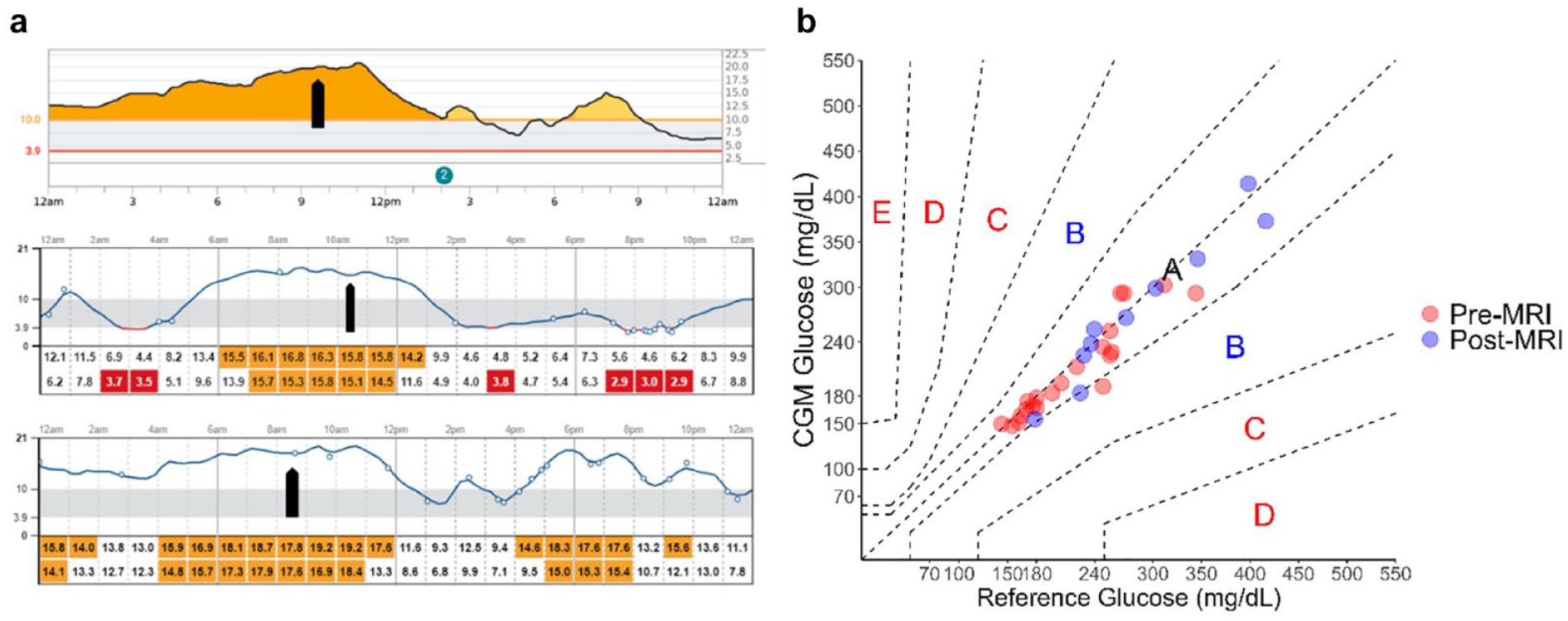
(a) Continuous glucose monitor glucose traces (glucose displayed in mmol/L) from days of MRI scans in cases 1 (first panel) and 2 (second and third panel) (arrows: MRI scan duration); (b) consensus error grid analysis of pooled time-matched continuous glucose monitor and point-of-care glucose pairs from cases 1 and 2 (red circles: before MRI scan; blue circles: after MRI scan).
To assess CGM accuracy, we time-matched (±7 minutes) CGM glucose with point-of-care (POC) capillary blood glucose (BG) measures (Nova StatStrip; Nova Biomedical, Waltham, MA, USA) from the 72 hours before and after MRI, after excluding measures outside device reportable ranges.
Twenty-five time-matched BG pairs (21 pre-MRI; four post-MRI) from case 1 (mean BG 229 mg/dL, SD 65) resulted in mean absolute relative difference (MARD) of 6.7% pre-MRI and 7.9% post-MRI. For case 2, six post-MRI BG pairs (four after first MRI; two after second MRI), but no pre-MRI BG pairs, were obtained (mean BG 282 mg/dL, SD 82), with MARD 5.0% post-MRI. All BG pairs were within Consensus Error Grid zones A/B (93.5% zone A; 6.5% zone B) (Figure 1b). 1
Currently, the only CGM labeled MRI-conditional is the implantable subcutaneous Eversense sensor (Ascensia Diabetes Care, Parsippany, NJ, USA). 2 Wearable transcutaneous CGM devices (including Dexcom G6 and Libre 2) are all labeled MRI-unsafe. Dexcom G6 and Libre 2 simulation studies report preserved CGM function after, and low dislodgement risk during, 1.5T and 3T MRI.3-5 Real-world experience of in vivo transcutaneous CGM use with MRI remains limited.
We report the first cases of in vivo transcutaneous CGM use during MRI. Three concerns exist regarding CGM use during MRI: image artifact, CGM dislodgement, and CGM accuracy. In our cases, magnetic field displacement forces during a 25-minute 1.5T MRI and 26-minute 3T MRI were insufficient to dislodge Dexcom G6 and Libre 2, respectively; CGM accuracy was acceptable following MRI; image artifact was not assessed as CGM was not located within imaged regions.
The main limitation of our report is the limited number of glucose pairs. In addition, our findings may not apply to MRI scans with longer duration, or different scanning conditions.
Continuous glucose monitoring removal before MRI results in glucose monitoring interruptions, along with device wastage and expense. Our cases of continued CGM wear during MRI, together with previous simulation studies, suggest some CGM devices might not dislodge, and function and accuracy may be maintained, under specific MRI conditions. Further studies are required to determine safety and accuracy of CGM use in the setting of MRI.
Footnotes
Abbreviations
BG, blood glucose; CGM, continuous glucose monitoring; MRI, magnetic resonance imaging; POC, point-of-care; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: RW was supported by an Australian Commonwealth Government RTP Scholarship, ACADI PhD Grant, Fred Knight Research Scholarship, Rowden White Scholarship, and Gordon P Castles Scholarship.
