Abstract
Introduction:
Automated insulin delivery (AID) systems have improved glycemic control in individuals with type 1 diabetes (T1D) but overweight and increased cardiovascular risk remain a challenge. Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) are associated with improved cardiometabolic profile but are currently not approved for the treatment of T1D.
Material and methods:
Individuals with T1D at Steno Diabetes Center Copenhagen, Denmark, treated with AID and off-label GLP-1 RA for at least six months between January 2017 and May 2024 were included in a retrospective chart review study.
Results:
Nineteen individuals with (median [range]) age 42 (24-60) years were included. At GLP-1 RA initiation, hemoglobin A1c (HbA1c) was 7.3% (6.1%-8.7%), HbA1c 56 (43-72) mmol/mol, body weight 91.5 (78.0-115.0) kg, and body mass index 35.4 (27.0-42.0) kg/m2. Time in range was 74% (29%-82%), time above range 25% (18%-71%) while time below range was 1% (0%-5%). After six months of treatment, body weight changed −11% (−22% to −3%; P = .001) and total daily insulin dose changed −15.1 (−32.5 to −8.2) IU (P = .004). There were no significant changes in HbA1c or other glucose measures. One person developed ketoacidosis caused by infusion set failure, but none reported severe hypoglycemia.
Conclusion:
Glucagon-like peptide-1 receptor agonist as add-on therapy for six months in individuals with obesity and AID-treated T1D led to considerable weight loss and a reduction in insulin dose.
Keywords
Introduction
Automated insulin delivery (AID) systems improve glucose levels in individuals living with type 1 diabetes (T1D).1-5 However, it is well known that intensification of insulin therapy is an important contributor to weight gain.6,7 The Diabetes Control and Complications Trial showed that in the intensively treated group there was a higher prevalence of obesity compared to the control group. 8 Furthermore, in the group receiving intensive treatment, those who experienced the greatest weight gain also showed the most significant increases in blood pressure and lipid levels. 8 In addition, this subgroup of obese individuals lost the preventive benefits of good glycemic control as they exhibited a substantial rise in incidence of cardiovascular disease (CVD) after 12 years of follow-up, comparable to the increase seen in the conventionally treated group. 9
Glucagon-like peptide-1 receptor agonist (GLP-1 RA) is an important glucose-lowering treatment modality in individuals with type 2 diabetes (T2D) and is associated with a reduction in cardiovascular risk.10,11 In overweight and obese individuals with and without diabetes, treatment with a GLP-1 RA is also found to reduce body weight. 12 Glucagon-like peptide-1 receptor agonists are currently not approved for the treatment of T1D. Nevertheless, studies in T1D have reported a reduction in body weight, hemoglobin A1c (HbA1c), and insulin requirements irrespective of insulin regimen. 13
The current study aimed to assess the efficacy of off-label treatment with GLP-1 RA on body weight, body mass index (BMI), glycemic measures, and insulin requirements in AID users with T1D in routine clinic. The secondary aims were to evaluate the safety outcomes such as an effect on blood pressure, renal function, hospital admissions due to GLP-1 RA–induced side effects, retinopathy, hypoglycemia, and diabetic ketoacidosis (DKA).
Methods
This study is a retrospective review of medical records. Individuals with T1D treated with AID at Steno Diabetes Center Copenhagen (SDCC) in Denmark were included if they were >18 years and had been treated with GLP-1 RA for at least six months in the time period from January 2017 to February 2024. Use of GLP-1 RA prior to AID initiation was an exclusion criterion, but user of any other antidiabetic or anti-obesity medications was eligible for inclusion.
Based on coding, people with AID were identified. Data regarding type and dose of GLP-1 RA, height and body weight, glycemic measures (HbA1c, continuous glucose monitoring [CGM]-derived time in range [TIR: 3.9-10.0 mmol/L], time above range [TAR: >10.0 mmol/L], time below range [TBR: <3.9 mmol/L], coefficient of variation), insulin pump data (total daily insulin dose [TDD] and total daily carbohydrate entry), blood pressure, cholesterol levels, renal function, gastro-intestinal symptoms, severe hypoglycemia, and ketoacidosis were extracted from the electronic medical record and software applications were used to upload CGM and insulin pump data. The CGM-derived glycemic metrics and insulin pump data from the last 14 days prior to and after six and 12 months of GLP-1 RA treatment were used.
Statistical Analyses
The comparisons between baseline and six- and 12-month measures were performed using the paired t-test after logarithmic transformation of non-normally distributed data. All data are reported as median (range). Statistical analyses were conducted using SPSS (Version 29.0; The R Foundation for Statistical Computing, Vienna, Austria), with statistical significance set at a two-tailed P-value of <.05. SAS 8.3 (SAS institute Inc, Cary, New York) was used to perform the spaghetti plots.
Results
During the study period, 5898 adults with T1D attended SDCC. Among these, 380 people were treated with or had previously been treated with a GLP-1 RA. Fifty of these were using an AID system.
Nineteen people with AID-treated T1D who were naïve to GLP-1 RAs started treatment with a GLP-1 RA between January 2017 and February 2024 and were treated for at least six months. Among these, 12 people were treated for 12 months or more. Baseline characteristics are presented in Table 1. Two individuals had either tried metformin (n = 1) or were treated with metformin prior to initiation of GLP-1 RA therapy. None had tried other oral antidiabetic or anti-obesity medications.
Baseline Characteristics.

Continuous data are presented as median (range).
Abbreviations: BMI: body mass index; CV: coefficient of variation; eGFR: estimated glomerular filtration rate; HDL: high-density lipoprotein; LDL: low-density lipoprotein; TAR: time above range (>10.0 mmol/L); TBR: time below range (<3.9 mmol/L); TIR: time in range (3.9-10.0 mmol/L); TDD: total daily insulin dose; UACR: urine albumin-creatinine ratio.
The median duration of treatment with GLP-1 RA was 16 (6-38) months. Fifteen individuals were treated with semaglutide, three received liraglutide, and one person started liraglutide treatment, but switched to semaglutide after eight months. One individual stopped GLP-1 RA treatment due to gastro-intestinal side effects after seven months of treatment but resumed the treatment after 18 months. There were no recorded episodes of severe hypoglycemia and no hospitalizations due to GLP-1 RA side effects. One person developed DKA due to infusion set failure. With regard to eye complication, 13 people had retinopathy at baseline (three had mild retinopathy, four had moderate non-proliferative retinopathy, and six had proliferative retinopathy). None of the individuals experienced a progression in retinopathy after initiation of GLP-1 RA.
Body weight, glucose metrics, and insulin pump data after six and 12 months of GLP-1 RA treatment are presented in Table 2. The median dose of semaglutide after six months’ treatment was 1.0 (0.25-1.7) mg/week with three people treated with 1.7 mg/week. Liraglutide dose after six months was 1.8 mg for all three persons. There was a significant body weight loss both after six and 12 months of treatment. Body weight changed −9.0 (−22.1 to −0.5) kg corresponding to a reduction of 11% (3%-22%) after six months’ treatment (P = .001). Only individuals using semaglutide completed 12 months of treatment. After 12 months of semaglutide treatment, the median dose of semaglutide was 1.0 (0.5-1.7) mg. These individuals lost 15.0 (2.5-26.1) kg (P = .002) corresponding to a body weight reduction of 18% (6%-26%) at 12 months. CGM data were available at baseline and six months for 14 of 19 individuals and for eight of 12 individuals at 12 months. The percentage of expected CGM readings available was 85% (59%-97%) at baseline, 91% (66%-96%) after six months, and 89% (71%-96%) after 12 months of treatment. Furthermore, treatment with GLP-1 RA in six and 12 months led to a numerical improvement in glucose measures with a reduction in HbA1c, an increase in TIR, and a decrease in TAR without an increase in TBR (Table 2).
Changes in Weight, Glucose Metrics, and Insulin Doses After Six and 12 Months of Treatment With Glucagon-Like Peptide-1 Receptor Agonist.

Continuous data are presented as median (range).
Abbreviations: BMI: body mass index; CV: coefficient of variation; GLP-1 RA: glucagon-like peptide-1 receptor agonist; TAR: time above range (>10.0 mmol/L); TBR: time below range (<3.9 mmol/L); TIR: time in range (3.9-10.0 mmol/L); TDD: total daily insulin dose.
Comparison between baseline and six months of treatment with GLP-1 RA.
Comparison between baseline and 12 months of treatment with GLP-1 RA.
With respect to insulin pump data after six months of treatment, the TDD changed significantly with −19.3 (−32.5 to −8.2) IU/day (P = .004) while TDD per kg body weight is −0.14 (−0.32 to 0.12) IU/kg body weight (P = .14). Total daily carbohydrate entry was not significantly affected by GLP-1 RA treatment (−39 [−144 to 31] g/day [P = .40]).
Data on lipids, blood pressure, and renal function were available at six months for the majority of participants (79%, 68%, and 89%, respectively). We found numerical but statistically insignificant changes in total cholesterol, low-density lipoprotein cholesterol (LDL-c), and systolic blood pressure. Total cholesterol changed −0.3 (−0.8 to 0.4) mmol/L which was caused by a change in LDL-c of 0.4 (−0.8 to 0.1) mmol/L. No changes in high-density lipoprotein cholesterol, triglycerides, diastolic blood pressure, and renal function were found. After six months of GLP-1 RA treatment, systolic blood pressure changed −10 (−31 to 8) mmHg. No participants were hospitalized due to GLP-1 RA–induced side effects.
The difference between treatment with liraglutide and semaglutide was examined, and the changes are illustrated in Figures 1 and 2. Figure 1 presents the individual changes in body weight after six months of GLP-1 RA treatment, while changes in HbA1c for each individual are demonstrated in Figure 2. Body weight changed −3.7 (−4.5 to −0.5) kg and BMI changed −1.3 (−2.1 to −0.1) kg/m2 after six months of liraglutide treatment; however, these reductions were insignificant. There was no change in glucose metrics and insulin requirements. In contrast, people treated with semaglutide had a weight change of −9.9 (−22.1 to −3.2) kg (P = .009) and a change in BMI of −3.6 (−8.0 to −1.3) kg/m2 (P < .001), but no significant changes were found in glucose metrics.
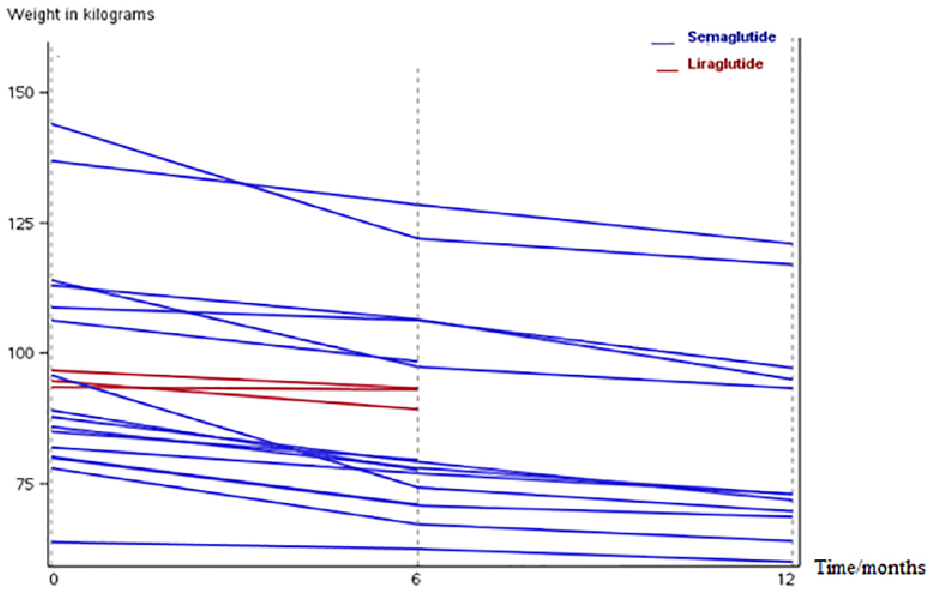
Changes in body weight after 12 months of treatment with a glucagon-like peptide-1 receptor agonist.
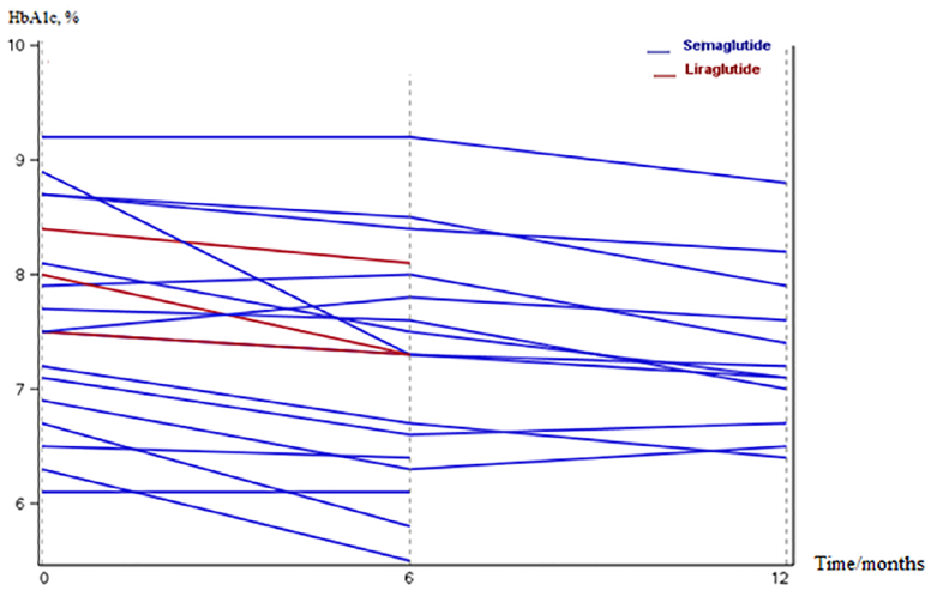
Changes in HbA1c after 12 months of treatment with a glucagon-like peptide-1 receptor agonist.
Discussion
In this retrospective chart review study, we found that six months of off-label treatment with GLP-1 RA in overweight and obese individuals with AID-treated T1D led to a weight loss of 9 kg (11%) and a reduction in TDD of 26%. Furthermore, we noticed a trend toward improved glycemic measures. For the sub-population treated with GLP-1 RA for more than 12 months, the weight loss was even more pronounced (15.0 kg [18.0%]). Finally, we found no increased risk of severe hypoglycemia or hospital admission due to DKA.
This study is to the best of our knowledge the first study of the effects of liraglutide and semaglutide treatment as an add-on to people with T1D using either a MiniMed 670G, MiniMed 780G, or Tandem Control IQ system. We found a larger reduction in body weight compared to previous studies in individuals with T1D using other treatment modalities.13-18 A meta-analysis of studies published between 2013 and 2021 reported a total weight reduction of 3.8 kg. 13 Study participants were treated with liraglutide, exenatide, and albiglutide, but not semaglutide. 13 The larger weight loss observed in our study may be explained by the fact that 15 of 19 individuals were treated with semaglutide which is a more potent anti-obesity medication. 19 The effects of semaglutide in T1D have not been investigated in randomized controlled trials, but more studies are underway (NCT05822609).20,21 In a one-year observational study by Garg et al 15 including individuals with overweight or obesity and T1D treated with either multiple daily injections (MDI) or insulin pump, 50 participants received a median dose of 0.92 mg semaglutide per week. They found a weight reduction of approximately 7.2 kg, thus lower than the weight change of 15 kg in our 12-month cohort. Both baseline weight (96.5 kg) and BMI (33.5 kg/m2) were comparable to our study, but we had a larger proportion of people treated with 1.7 mg (16% compared to 8%) semaglutide per week which may explain the difference.
We found no significant change in HbA1c which is different compared to previous studies with liraglutide and exenatide.14,17,22,23 This may be explained by the relatively low baseline HbA1c, but it could also be related to the retrospective nature of the study, differences in GLP-1 RA dosage, and large variation within our small sample size. The small sample size may also explain the TIR findings.
Most studies of GLP-1 RA in T1D have found an increased risk of hypoglycemia. In contrast, we did not identify any changes in TBR and no severe hypoglycemia events occured. 24 One explanation could be that AID systems mitigate the risk of hypoglycemia related to sudden changes in daily insulin needs induced by GLP-1 RA. 25 Accordingly, the use of GLP-1 RA as an add-on to AID may be preferable to an add-on to non-automated insulin pumps and MDI.
We observed insignificant reductions in systolic blood pressure of 10 mmHg, total cholesterol of 0.3 nmol/L, and LDL-c of 0.4 mmol/L. Liraglutide and semaglutide reduce the risk of CVD in people with T2D and our findings indicate that people with T1D may achieve similar benefits from treatment.10,11 The changes in cholesterol in the current study are larger compared to previous findings in people with T2D, but the difference could be explained by differences between T1D and T2D and our sample size.11,26
There is an urgent need for therapeutic options for lowering cardiometabolic risk in T1D. In this study, we found indications of cardiometabolic benefits of off-label use of liraglutide and semaglutide. A new consensus statement from Diabetes Technology Society recommends GLP-1 RA as an add-on therapy in people with T1D using an AID as they are shown to reduce postprandial glucose peaks, HbA1c, and TDD. 27 In addition to GLP-1 RA that only bind to the GLP-1 receptor, a new treatment modality, tirzepatide, which is a dual glucose-dependent insulinotropic polypeptide and GLP-1 receptor agonist, has shown convincing reductions in glucose levels and body weight in people with T2D. 18 ,28-30 Two observational studies have examined the effect in people with T1D indicating an even larger effect on weight loss compared to semaglutide without risk of hospitalization due to DKA or severe hypoglycemia.31,32 However, this drug was not available in Denmark during the study period.
Conclusions
Treatment with GLP-1 RA, liraglutide, and semaglutide in overweight and obese people with T1D treated with an AID system led to an improvement in weight and glycemic measures without increasing the risk of severe hypoglycemia and DKA. GLP-1 RA as an add-on treatment should be considered in this group of people.
Footnotes
Acknowledgements
The authors would like to thank Antonius Manders, Steno Diabetes Center Copenhagen, for helping with the identification of the people with AID and GLP-1 RA treatment from the EMR.
Abbreviations
AID, automated insulin delivery; CGM, continuous glucose monitoring; CV, coefficient of variation; CVD, cardiovascular disease; DKA, diabetic ketoacidosis; EMR, electronic medical record; GLP-1 RA, glucagon-like peptide-1 receptor agonist; HDL-c, high-density lipoprotein cholesterol; LDL-c, low-density lipoprotein cholesterol; SDCC, Steno Diabetes Center Copenhagen; T1D, type 1 diabetes; T2D, type 2 diabetes; TAR, time above range; TBR, time below range; TDD, total daily insulin dose; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PH and MBC have no conflicts of interest. KN received funding to her institution for participating in advisory boards from Medtronic, Novo Nordisk, and Convatec and for lecturing from Sanofi, Novo Nordisk, Medtronic, and Dexcom. Her institution received funding for studies she performed from Zealand Pharma, RSP Systems, Novo Nordisk, Medtronic, and Dexcom. SS has received speaker’s fee from Novo Nordisk and Nordic Infucare and had been employed with Novo Nordisk.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
