Abstract
Background:
As type 2 diabetes (T2D) progresses, intensification to combination therapies, such as iGlarLixi (a fixed-ratio GLP-1 RA and basal insulin combination), may be required. Here a simulation study was used to assess the effect of iGlarLixi administration timing (
Methods:
Models of lixisenatide were built with a selection procedure, optimizing measurement fits and model complexity, and were included in a pre-existing T2D simulation platform containing glargine models. With the resulting tool, a simulated trial was conducted with 100 in-silico participants with T2D. Individuals were given iGLarLixi either before breakfast or before an evening meal for 2 weeks and daily glycemic profiles were analyzed. In the model, breakfast was considered the largest meal of the day.
Results:
A similar percentage of time within 24 hours was spent with blood sugar levels between 70 to 180 mg/dL when iGlarLixi was administered pre-breakfast or pre-evening meal (73% vs 71%, respectively). Overall percent of time with blood glucose levels above 180 mg/dL within a 24-hour period was similar when iGlarLixi was administered pre-breakfast or pre-evening meal (26% vs 28%, respectively). Rates of hypoglycemia were low in both regimens, with a blood glucose concentration of below 70 mg/dL only observed for 1% of the 24-hour time period for either timing of administration.
Conclusions:
Good efficacy was observed when iGlarlixi was administered pre-breakfast; however, administration of iGlarlixi pre-evening meal was also deemed to be effective, even though in the model the size of the evening meal was smaller than that of the breakfast.
Keywords
Introduction
Type 2 diabetes (T2D) is a progressive disease characterized by a decline in β-cell function, which contributes to the worsening of glycemic control and treatment failure.1-3 Most people with T2D will ultimately require pharmacological intervention. This is usually initiated with a single oral agent, followed by sequential addition of oral agents, before intensification to injectable agents.4-6 The American Diabetes Association recommends glucagon-like peptide 1 receptor agonists (GLP-1 RAs) as the first injectable agent after failure of oral antidiabetic agents. 4 However, as T2D progresses, intensification to combination therapies, such as fixed-ratio GLP-1 RA and basal insulin combinations, may be required. iGlarLixi is a titratable, fixed-ratio combination, available in the United States as a 3 U:1 µg ratio of insulin glargine and the GLP-1 RA, lixisenatide, with efficacy demonstrated in the LixiLan clinical program.7-9 Lixisenatide is a short-acting, prandial, GLP-1 RA that has been demonstrated to be equally effective if administered in the evening before a meal or in the morning before breakfast. 10 However, little is known about the effects of administering iGlarLixi before breakfast compared with administration before an evening meal.
Model-based assessments can be used to efficiently quantify and deconstruct the effects of a therapeutic agent.11,12 Indeed, achieving this task in the absence of a model-based method is not straightforward. For instance, lixisenatide delays gastric emptying, and responses to an oral glucose challenge in the presence of lixisenatide would lead to lower postprandial glucose levels versus those that would occur in the absence of lixisenatide, which in turn could lead to lower insulin secretion rates. If the C-peptide measurements were to be viewed independently from the glucose tracings, it could be falsely concluded that lixisenatide reduces the insulin-secretory response, when in fact the decrease in C-peptide results from the delayed delivery of carbohydrates to the bloodstream, which necessitates less insulin production because of lower postprandial glucose. Model-based assessments can be developed to account for the majority of these physiological processes, such as the impact of glucose on insulin secretion. Consequently, most of these confounding factor-based errors can be resolved.
The development of precise, knowledge-based models is the first step toward building or extending simulation platforms. 13 This is the context in which the well-established US Food and Drug Administration-accepted University of Virginia (UVA)/Padova simulator was developed. 14
Apart from the trial by Ahrén et al
10
which demonstrated noninferiority of lixisenatide administered before the main meal of the day versus administration before breakfast, clinical trials of lixisenatide and iGlarLixi use morning dosing. The aim of this analysis was to determine if iGlarLixi could be administered before an evening meal with equal efficacy to administration before breakfast. Thus, an in-silico study was conducted to compare the effect of morning versus evening administration of iGlarLixi on blood sugar profiles. To achieve this, we present compartmental models of lixisenatide pharmacokinetics and pharmacodynamics that quantify and deconstruct the complex mechanisms of action of the drug and demonstrate its use by assessing the timing of administration (
Methods
Data Sources
Models were developed from 2 previously published datasets. The first dataset was used to construct the lixisenatide model and establish the effect on both insulin and glucagon secretion. Data were collected from 2 individual, parallel, single-center, double-blind, 1:1 randomized, single-dose crossover studies (20 healthy adults and 22 adults with T2D using only the data from the patients with T2D for model development in this study). 15 Participants were given 20 µg of lixisenatide or placebo subcutaneously (SC) 2 hours before the administration of intravenous glucose (0.3 g/kg body weight; 50% aqueous solution) over 30 seconds. Plasma concentrations of glucose, glucagon, C-peptide, and free insulin were measured from 30 minutes before to 4 hours after the SC injection in a high-frequency manner. Lixisenatide plasma concentrations were measured from injection until 12 hours thereafter.
The second study was used to quantify the impact of lixisenatide on gastric emptying. This was a single-center, randomized, open-label crossover study during which single doses of lixisenatide 2.5, 5, 10, or 20 µg, or a matched placebo (5 sequences) were administered to 20 healthy participants after an overnight fast of at least 10 hours. 16 Sixty minutes after the injection of lixisenatide or placebo, a standardized liquid meal was administered followed 10 to 15 minutes later by 1000 mg of acetaminophen (paracetamol), absorption of which has been shown to be reliably dependent on the rate of gastric emptying. Acetaminophen concentrations were analyzed from blood samples taken at predefined times from 90 minutes before to 300 minutes after meal intake and were used as markers of gastric emptying.
Model Development
A compartmental model approach was utilized. As the model was based on mass conservation, it is appropriate to model substrate transport. Model choices were influenced by the available literature, most of which came from the UVA/Padova simulator.17,18 The model selection procedure is detailed in Appendix 1, and the model identification procedure and assessment are reported in Appendix 2 and Supplementary Figures S1 to S5. The model was validated against data from 7-point self-monitoring blood glucose profiles from patients with T2D.7,19
Simulation
Selected models were used to augment a previously published T2D simulation platform. 18 An in-silico study with a cohort of 100 participants was conducted using the platform to simulate the daily administration of a combination of lixisenatide and insulin glargine. The same cohort underwent 2 different administration simulations: a pre-breakfast or a pre-evening meal injection of the combination therapy. Meals identical in size and time of administration were generated randomly from a random meal generator. To account for titration, a titration algorithm based on data from patients included in the LixiLan-O randomized trial was included in the model. 9
The simulations allowed for 24-hour blood glucose levels from both treatment administration schedules to be modeled and, therefore, for possible periods of hyper- or hypoglycemia to be anticipated. In addition, insulin and glucagon secretion after intravenous glucose tolerance test (IVGTT) could be simulated.
Results
Models
Model evaluation and model fit. The developed model was evaluated against the previously published datasets by assessing how those data “fitted” into the model. The model was validated against 7-point self-monitoring blood profiles from patients with T2D.7,19 Overall, the current model aligned well with the known effects of lixisenatide, and the correlation between the 2 was considered strong (data not shown).
Parameter values, fit, and residuals of first models satisfying the criteria for lixisenatide pharmacokinetics, insulin secretion, glucagon secretion, and models of gastric emptying are provided in Supplementary Figures S6 to S9, respectively. Residual distributions were available for the first 3 models, and all met the criteria for satisfactory goodness of fit.
The model appropriately reproduced the insulin secretion response to an IVGTT that was higher with lixisenatide included in the model than without (Figure 1). The glucagon response to an IVGTT was also reproduced; a greater reduction in glucagon levels was predicted when lixisenatide was included in the model than without (Figure 2). These curves confirm the model can reproduce the insulin and glucagon responses accurately.
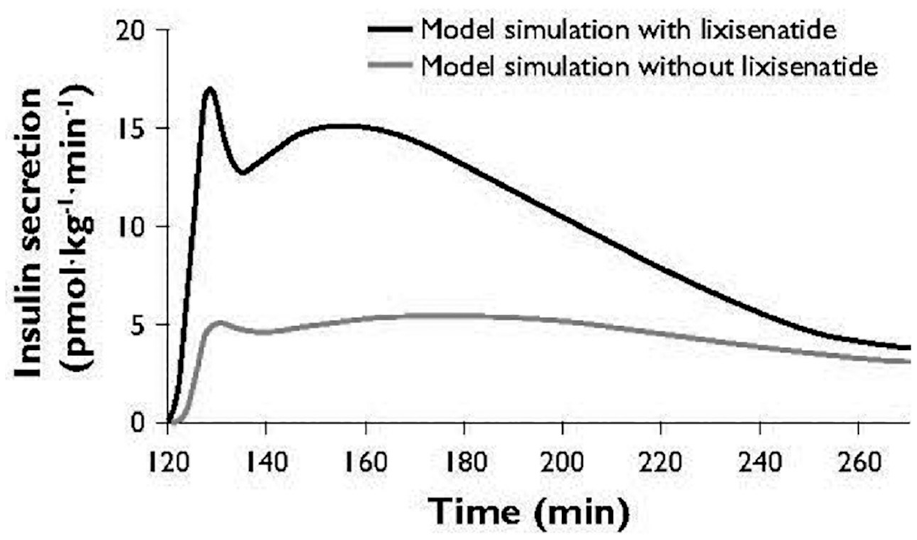
Simulated insulin secretion response to an IVGTT-like scenario. IVGTT, intravenous glucose tolerance test.
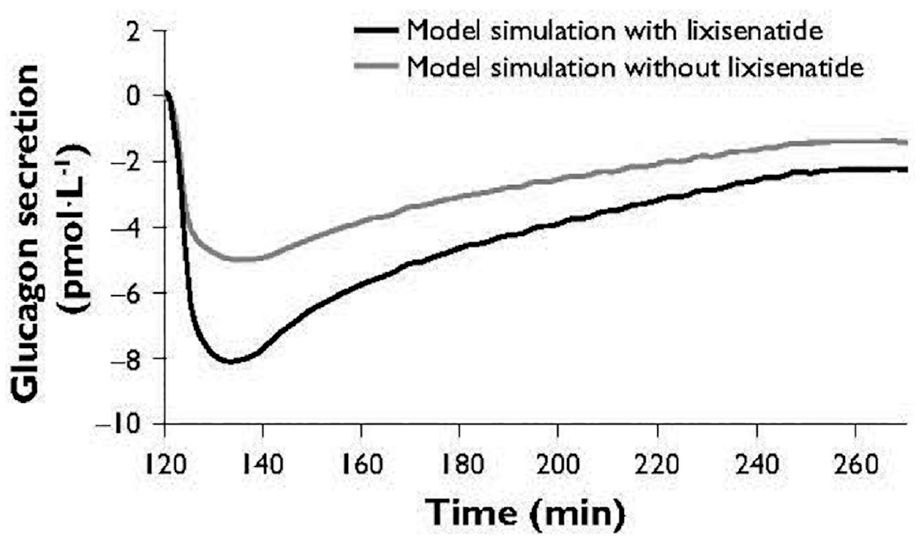
Simulated glucagon response to an IVGTT-like scenario. IVGTT, intravenous glucose tolerance test.
Simulation
Glycemic profiles after both pre-breakfast and pre-evening meal SC injection of iGlarLixi are shown in Figure 3. In the simulation, the 24-hour glucose profile was relatively stable after pre-breakfast administration of iGlarLixi, with the lowest levels of glucose within the 24-hour period observed overnight and before the evening meal. A similar percentage of time over the 24 hours was spent with a blood glucose level between 70 and 180 mg/dL when iGlarLixi was administered pre-breakfast or pre-evening meal (73% vs 71%, respectively). When administered in the evening, glucose levels showed more variability over a 24-hour period, with the largest peaks in blood glucose concentrations observed after breakfast and lunch in those administered iGlarLixi pre-evening meal. However, the overall percentage of time with blood glucose levels above 180 mg/dL within a 24-hour period was similar when iGlarLixi was administered pre-breakfast or pre-evening meal (26% vs 28%, respectively). Additionally, when iGlarLixi was administered pre-evening meal, glucose blood concentrations trended lower overnight than in those administered iGlarLixi pre-breakfast. Rates of hypoglycemia were low in both regimens, with a blood glucose concentration of below 70 mg/dL observed only for 1% of the 24-hour time period for either timing of administration.
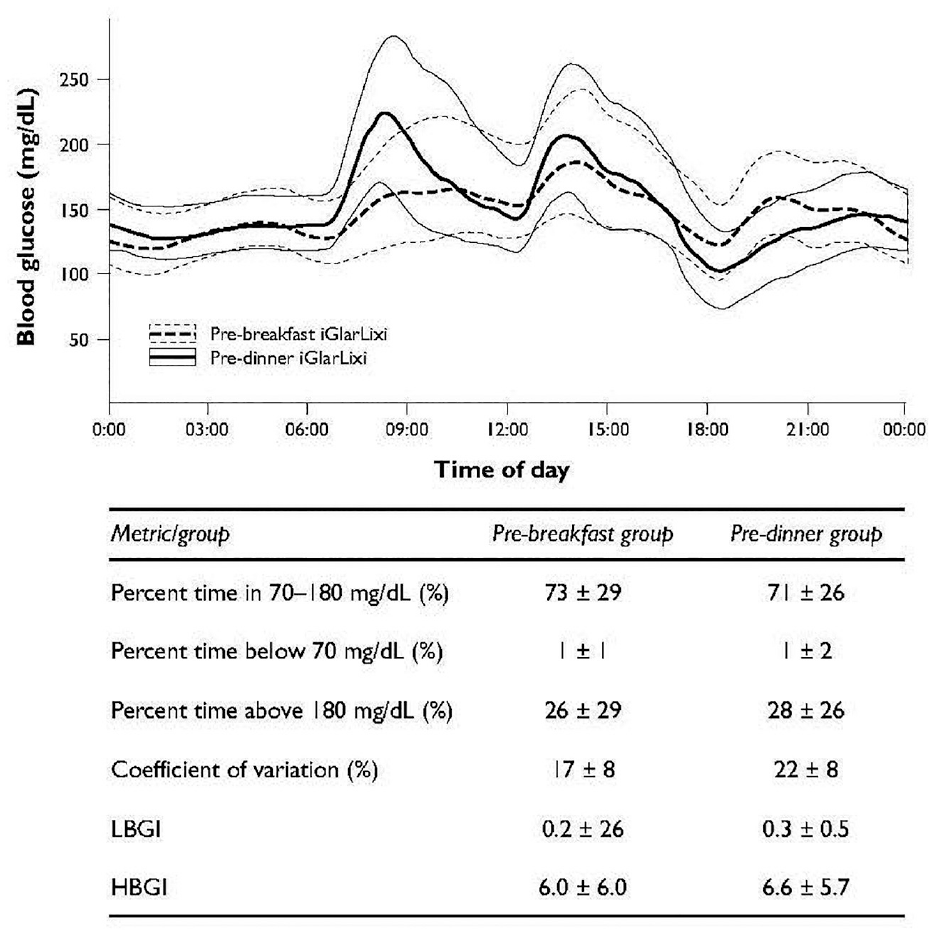
Simulation of the blood glucose values with combination lixisenatide and glargine injected pre-breakfast versus pre-dinner. HBGI, high blood glucose index; LGBI, low blood glucose index.
Discussion
These analyses have allowed for the simulation of iGlarLixi administration before either breakfast or an evening meal. Although both regimens were observed to have acceptable glucose level variability and comparable efficacy, glucose levels showed numerically less variability when iGlarLixi was administered before breakfast. Rates of hypoglycemia were low in the simulation. Full development of the model is discussed in Appendix 2.
By looking at the response to an IVGTT, we could see the model behaved as expected. Assessment of the fit of the curves for insulin and glucagon responses to the same glucose levels reveals that the impact of lixisenatide on glucagon is less pronounced than that on insulin. In this simulation, evening administration of iGlarLixi was associated with increased hyperglycemia in the daytime compared with morning administration; however, both groups had a similar percentage of time within the 24-hour period with a blood glucose of >180 mg/dL. These results suggest that efficacy with administration at either time point was acceptable. Glucose levels showed more variability over the 24-hour period in the evening administration group. The differences observed in these glycemic outcomes are highly related to meal habits. Generated meals were of smaller sizes at dinner than at breakfast, favoring the pre-breakfast administration group, and may explain the difference in the results observed in the 2 regimens. It should be noted that this is contrary to the normal meal routine in the United States, where dinner is the largest meal of the day. This suggests that the developed simulation tool could be leveraged to personalize therapy to patient characteristics, habits, and response to agents.
The US prescribing information for iGlarLixi 20 and lixisenatide 21 recommends that these agents are to be taken within the hour before the first meal of the day, in comparison to the US prescribing information for the other GLP-1 RAs that are administered daily, liraglutide, 22 which is administered once daily, independently of meals, and exenatide 23 which is administered twice daily, 60 minutes before evening and morning meals. The current study supports the findings that lixisenatide is equally effective when administered either before an evening or before a morning meal, 10 which suggests that iGlarLixi can also be administered with flexibility related to mealtime. Thus, the GLP-1 RAs administered daily, lixisenatide and liraglutide, offer greater flexibility in administration timing, satisfying those patients with erratic lifestyles, whereas, by comparison, exenatide must be administered twice daily, requiring a more rigid administration regimen. Flexibility in administration often benefits those people whose lifestyle requires more versatility in administration. A reason often given for nonadherence to insulin injections has been the inflexibility in the timing of injections, 24 whereas improved insulin therapy adherence has been demonstrated in flexible regimens of administration. 25 These simulated results warrant further clinical study in people with T2D.
There are a number of limitations that should be considered in the current study. The generated models are centered exclusively on glucose and insulin concentration as inputs, so the effects of other components such as free fatty acids or other hormones were ignored. Furthermore, models are limited to our a priori knowledge of physiology. In addition, mathematical models can be only a simplified representation of the studied phenomenon. Our method is further limited by the nature of the experiment used to identify these models: the intravenous or oral glucose challenge. This specific “excitation” of the phenomenon dynamics may be too restrictive for a complete understanding of the action of lixisenatide under more therapeutic conditions. In the current study, modeling of gastric emptying after lixisenatide administration was carried out using data from healthy individuals. However, results observed in the current study are similar to data from a recent study by Rayner et al 26 of gastric emptying in people with T2D following lixisenatide administration.
Conclusions
Based on frequent blood measurements from intravenous glucose or meal response studies, starting from models of glucose homeostasis available in the literature, models of lixisenatide, insulin secretion, glucagon, and meal absorption that remain valid in the presence of lixisenatide were constructed. This model-based assessment allowed for the isolation of the major components of these complex responses, and for the quantification and unambiguous interpretation of the metabolic changes triggered by lixisenatide. Estimated parameters of the constructed models were found to be consistent with the published literature describing the impact of the short-acting GLP-1 RA lixisenatide 27 and more broadly of GLP-1 RAs28,29 on glucose metabolism and endocrine secretion, although long-acting GLP-1 RAs have diminished postprandial glucose effects. Through delayed gastric emptying, glucose-dependent inhibition of glucagon, and a reduction in the need for insulin secretion (due to delayed appearance of carbohydrates in the blood after a meal), lixisenatide can achieve better postprandial plasma glucose control. Integrated into a well-established simulation platform, these models allowed for the evaluation of the impact of the administration timing of lixisenatide in combination with insulin glargine 100 U. Efficacy was comparable when iGlarlixi was administered in the morning pre-breakfast or pre-evening meal, even though in the model the size of the evening meal was smaller than that of the breakfast. This suggests that in those patients who need greater flexibility in administration of iGlarLixi or lixisenatide, an evening administration regimen may be acceptable. However, it is important for clinicians to understand the potential for glycemic variability observed after evening administration and to monitor glucose levels in patients accordingly.
Supplemental Material
sj-tif-1-dst-10.1177_19322968211015671 – Supplemental material for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles
Supplemental material, sj-tif-1-dst-10.1177_19322968211015671 for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles by Thibault Gautier, Rupesh Silwal, Aramesh Saremi, Anders Boss and Marc D. Breton in Journal of Diabetes Science and Technology
Supplemental Material
sj-tif-2-dst-10.1177_19322968211015671 – Supplemental material for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles
Supplemental material, sj-tif-2-dst-10.1177_19322968211015671 for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles by Thibault Gautier, Rupesh Silwal, Aramesh Saremi, Anders Boss and Marc D. Breton in Journal of Diabetes Science and Technology
Supplemental Material
sj-tif-3-dst-10.1177_19322968211015671 – Supplemental material for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles
Supplemental material, sj-tif-3-dst-10.1177_19322968211015671 for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles by Thibault Gautier, Rupesh Silwal, Aramesh Saremi, Anders Boss and Marc D. Breton in Journal of Diabetes Science and Technology
Supplemental Material
sj-tif-4-dst-10.1177_19322968211015671 – Supplemental material for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles
Supplemental material, sj-tif-4-dst-10.1177_19322968211015671 for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles by Thibault Gautier, Rupesh Silwal, Aramesh Saremi, Anders Boss and Marc D. Breton in Journal of Diabetes Science and Technology
Supplemental Material
sj-tif-5-dst-10.1177_19322968211015671 – Supplemental material for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles
Supplemental material, sj-tif-5-dst-10.1177_19322968211015671 for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles by Thibault Gautier, Rupesh Silwal, Aramesh Saremi, Anders Boss and Marc D. Breton in Journal of Diabetes Science and Technology
Supplemental Material
sj-tif-6-dst-10.1177_19322968211015671 – Supplemental material for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles
Supplemental material, sj-tif-6-dst-10.1177_19322968211015671 for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles by Thibault Gautier, Rupesh Silwal, Aramesh Saremi, Anders Boss and Marc D. Breton in Journal of Diabetes Science and Technology
Supplemental Material
sj-tif-7-dst-10.1177_19322968211015671 – Supplemental material for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles
Supplemental material, sj-tif-7-dst-10.1177_19322968211015671 for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles by Thibault Gautier, Rupesh Silwal, Aramesh Saremi, Anders Boss and Marc D. Breton in Journal of Diabetes Science and Technology
Supplemental Material
sj-tif-8-dst-10.1177_19322968211015671 – Supplemental material for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles
Supplemental material, sj-tif-8-dst-10.1177_19322968211015671 for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles by Thibault Gautier, Rupesh Silwal, Aramesh Saremi, Anders Boss and Marc D. Breton in Journal of Diabetes Science and Technology
Supplemental Material
sj-tif-9-dst-10.1177_19322968211015671 – Supplemental material for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles
Supplemental material, sj-tif-9-dst-10.1177_19322968211015671 for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles by Thibault Gautier, Rupesh Silwal, Aramesh Saremi, Anders Boss and Marc D. Breton in Journal of Diabetes Science and Technology
Supplemental Material
sj-pdf-10-dst-10.1177_19322968211015671 – Supplemental material for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles
Supplemental material, sj-pdf-10-dst-10.1177_19322968211015671 for Modeling the Effect of Subcutaneous Lixisenatide on Glucoregulatory Endocrine Secretions and Gastric Emptying in Type 2 Diabetes to Simulate the Effect of iGlarLixi Administration Timing on Blood Sugar Profiles by Thibault Gautier, Rupesh Silwal, Aramesh Saremi, Anders Boss and Marc D. Breton in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors received writing/editorial support in the preparation of this manuscript provided by Barrie Anthony, PhD, CMPP, of Evidence Scientific Solutions, funded by Sanofi US, Inc.
Abbreviations
GLP-1 RAs, glucagon-like peptide 1 receptor agonists; IVGTT, intravenous glucose tolerance test; T2D, type 2 diabetes; UVA, University of Virginia.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Research support was provided through the University of Virginia from: Dexcom, Sanofi US, Inc., and Tandem Diabetes Care. Patent royalties were provided through the University of Virginia from: Dexcom, Johnson & Johnson, and Sanofi US, Inc. AS is an employee at Intercept Pharmaceuticals. AB is an employee and stockholder of Sanofi US, Inc. MDB reports having received research support from Arecor ltd, Dexcom, Novo Nordisk, Sanofi US, Inc, and Tandem Diabetes Care; consulting fees and honoraria from Adocia, Air Liquide, Dexcom, and Tandem Diabetes Care; and royalties from intellectual property licenses in this field, managed by the University of Virginia.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Sanofi US, Inc.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
