Abstract

Laboratory hemoglobin A1c (HbA1c) is the gold standard for assessing long-term glycemic control. 1 Point-of-care (POC) assays such as the DCA Vantage Systems HbA1c test (Siemens Healthcare Diagnostics, Tarrytown, New York) provide testing using fingerstick blood samples and results in 6 minutes. Although the ability to obtain rapid HbA1c results in clinic can translate to improved patient outcomes, 2 this is only possible if the test is accurate and reliable. Data from blood glucose monitoring (BGM) and continuous glucose monitoring (CGM) are increasingly important for guiding clinical management, as they provide details on glycemic responses to meals, physical activity, and medications, as well as additional information on hypoglycemia and glucose variability, which are not available from HbA1c.
In January 2024, our team suspected multiple erroneous POC-HbA1c results based on discrepancies with patients’ CGM metrics. The errors were confirmed with simultaneous testing with laboratory HbA1c on several samples. We paused all POC-HbA1c testing and notified the manufacturer. Subsequently, a recall was issued on February 7, 2024 for the Siemens DCA HbA1c Reagent Kit (Material Numbers: 10311134 for lots 9640093, 0645103, 0648103, 0649103, 0654103, 0637093, 0641093, 0642093, 0647103, 0653103, and 0655103). 3 The objective of this report is to quantify the extent of this POC-HbA1c error, to raise awareness, and to emphasize the relevance of CGM metrics.
Retrospective review of the electronic health records showed that the affected reagents were used in our clinics from October 2023 to February 2024 on 406 POC-HbA1c tests in 384 patients. Of these, 52 paired POC-HbA1c and laboratory HbA1c measurements were available within 30-day window from 50 patients. Mean POC-HbA1c was 0.7% higher than laboratory HbA1c (8.7 ± 2.6% vs 8.0 ± 2.3%, P < .001). In addition, 205 paired POC-HbA1c and 14-day Glucose Management Indicator (GMI) measurements were available within 30-day window from 180 patients. Mean POC-HbA1c was 0.6% higher than GMI (8.3 ± 1.9% vs 7.7 ± 1.3%, P < .001).
Scatter plot with linear regression showed positive association between POC-HbA1c and laboratory HbA1c (Figure 1a) and between POC-HbA1c and GMI (Figure 1b). Bland-Altman plots showed an overall high bias of 0.70%-points (95% confidence interval: -1.16, 2.55) between POC-HbA1c and laboratory HbA1c (Figure 1c) and similar high bias of 0.59%-points (95% confidence interval: -1.87, 3.04) between POC-HbA1c and GMI (Figure 1d).
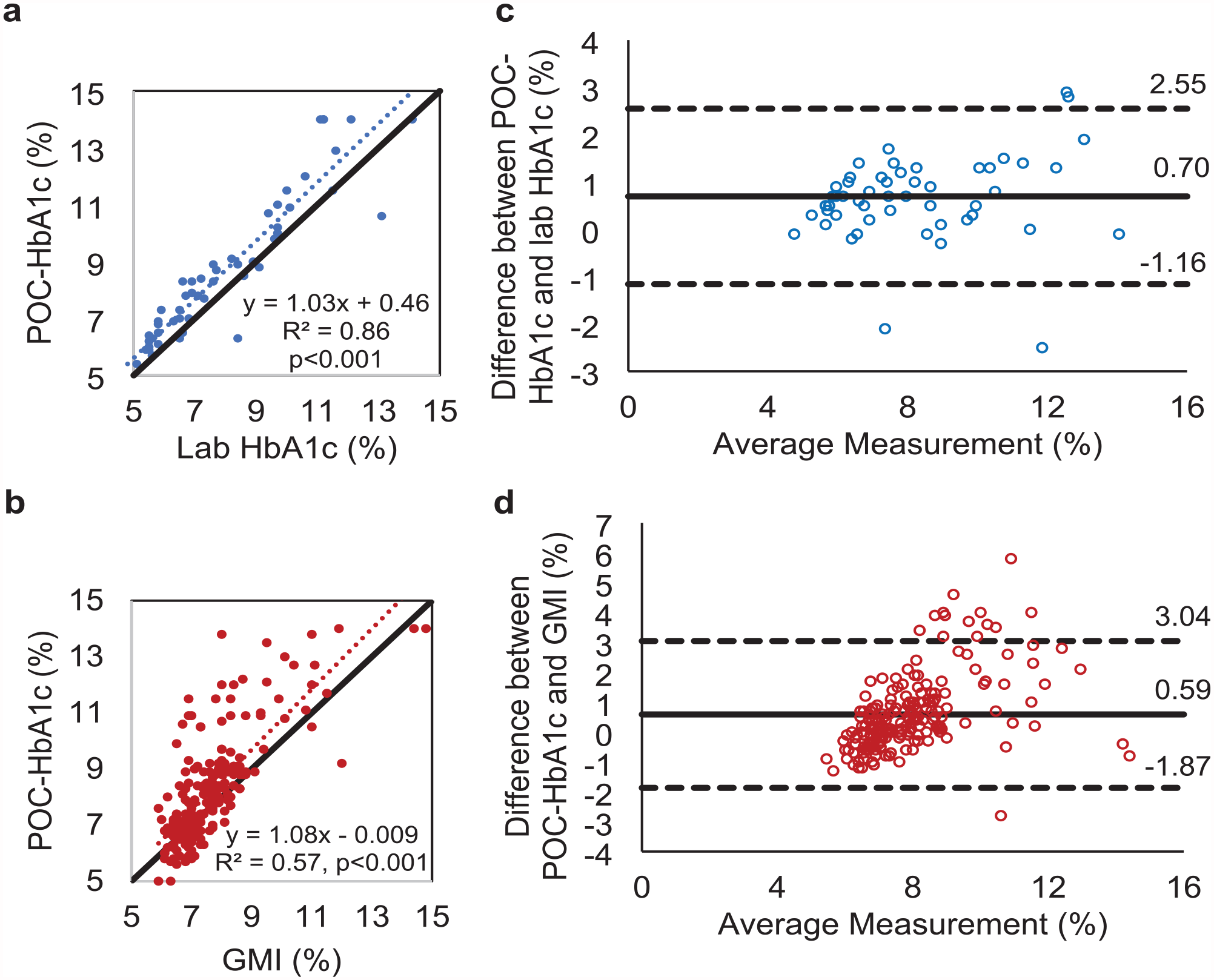
Comparison of paired POC capillary HbA1c vs laboratory venous blood HbA1c or 14-day GMI as reference. Scatter plots of paired POC capillary HbA1c vs laboratory venous blood HbA1c (a) or GMI (b). Black reference line displays x = y, and dotted line displays linear regression. The Bland-Altman plot of paired POC capillary HbA1c vs laboratory venous blood HbA1c (c) or GMI (d). The horizontal solid black line indicates the mean difference, and the horizontal dotted lines mark the limits of the agreement (±1.96 * standard deviation of the difference; 95% confidence interval).
We identified a clinically significant positive bias in the POC-HbA1c test compared with laboratory HbA1c and GMI. This error may have occurred in other clinics and can potentially lead to modifications in diabetes management plans that subsequently increase the patient’s risk of hypoglycemia. In addition to patient care, these results also have implications for registry data, 4 quality improvement, 5 and research programs. 6 This report highlights the need to balance the limitations of POC device accuracy with practicality of clinical workflow to obtain glycemic data in a timely manner to inform clinical decision-making in real-time. We recommend clinics that routinely use POC-HbA1c tests for clinical management to add other markers of glycemic control (eg, CGM metrics or laboratory HbA1c) to corroborate with POC-HbA1c data whenever possible. With increasing accuracy and infrastructure to support widespread CGM technology use, CGM metrics may become the increasingly preferred metrics for glycemia.
Footnotes
Acknowledgements
The authors acknowledge the diabetes clinical staff, clinical laboratory staff, and administration at Stanford Medicine Children’s Health for ongoing quality improvement efforts.
Abbreviations
BGM, blood glucose monitoring; CGM, continuous glucose monitor; GMI, Glucose Management Indicator; HbA1c, hemoglobin A1c; POC, point-of-care
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr DMM has had research support from the National Institute of Health, Breakthrough T1D, National Science Foundation, and the Helmsley Charitable Trust, and his institution has had research support from Medtronic, Dexcom, Insulet, Bigfoot Biomedical, Tandem, and Roche. Dr DMM has consulted for Abbott, Aditxt, the Helmsley Charitable Trust, LifeScan, MannKind, Sanofi, Novo Nordisk, Eli Lilly, Medtronic, Insulet, Dompe, Biospex, Provention Bio, Kriya, Enable Biosciences, and Bayer. The remaining authors do not report any relevant disclosures.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
