Abstract
Background:
The SENZA-PDN study evaluated high-frequency 10-kHz spinal cord stimulation (SCS) for the treatment of painful diabetic neuropathy (PDN). Over 24 months, 10-kHz SCS provided sustained pain relief and improved health-related quality of life. This report presents additional outcomes from the SENZA-PDN study, focusing on diabetes-related pain and quality of life outcomes.
Methods:
The SENZA-PDN study randomized 216 participants with refractory PDN to receive either conventional medical management (CMM) or 10-kHz SCS plus CMM (10-kHz SCS + CMM), allowing crossover after six months if pain relief was insufficient. Postimplantation assessments at 24 months were completed by 142 participants with a permanent 10-kHz SCS implant, comprising 84 initial and 58 crossover recipients. Measures included the Brief Pain Inventory for Diabetic Peripheral Neuropathy (BPI-DPN), Diabetes-Related Quality of Life (DQOL), Global Assessment of Functioning (GAF), and treatment satisfaction.
Results:
Over 24 months, 10-kHz SCS treatment significantly reduced pain severity by 66.9% (P < .001; BPI-DPN) and pain interference with mood and daily activities by 65.8% (P < .001; BPI-DPN). Significant improvements were also observed in overall DQOL score (P < .001) and GAF score (P < .001), and 91.5% of participants reported satisfaction with treatment.
Conclusions:
High-frequency 10-kHz SCS significantly decreased pain severity and provided additional clinically meaningful improvements in DQOL and overall functioning for patients with PDN. The robust and sustained benefits over 24 months, coupled with high participant satisfaction, highlight that 10-kHz SCS is an efficacious and comprehensive therapy for patients with PDN.
Keywords
Introduction
Diabetes is a major global health issue, with an estimated 537 million people affected worldwide. 1 By 2045, this number is projected to rise to 783 million. 1 Approximately half of individuals with diabetes will develop diabetic neuropathy (DN), 2 predominantly in the form of distal symmetric polyneuropathy,2,3 with symptoms typically starting in the feet and progressing to the lower limbs.3-5
Up to 25% of people with diabetes will develop DN accompanied by neuropathic pain symptoms, 6 often described as burning, tingling, and/or akin to stabbing and electric shock-like sensations. 4 This severe, persistent pain substantially interferes with the enjoyment of life, mobility, daily activities, and sleep, leading to depression, anxiety, and poor health-related quality of life (HRQoL).7-10 Comparatively, patients with painful diabetic neuropathy (PDN) report significantly worse HRQoL than those with painless DN. 11 In addition, some evidence indicates a higher body mass index (BMI) and poorer glycemic control in those with PDN relative to those with painless DN, suggesting a detrimental impact of pain on overall diabetes management.12-14
Despite the availability of numerous pharmacological options for PDN pain management, the long-term safety and efficacy of commonly prescribed neuropathic pain medications, such as pregabalin, gabapentin, and duloxetine/venlafaxine, have not been systematically evaluated. The available short-term systematic evaluations indicate that the efficacy of these medications is limited.15-19 In addition, these medications are often associated with intolerable side effects at therapeutic doses.20,21 There is a clear unmet need for efficacious and well-tolerated alternative treatment options to improve the lives of patients with PDN.
Spinal cord stimulation (SCS) is a well-established therapy for neuropathic pain, delivering mild electrical impulses directly to the spinal cord via leads placed in the epidural space that connect to an implantable pulse generator. Low-frequency stimulation at 40-60 Hz (low-frequency spinal cord stimulation [LF-SCS]) masks pain by evoking paresthesia over the affected area. The therapy has been shown to provide modest pain relief in patients with PDN (41%-55%),22,23 although longer-term evidence suggests a waning of the initial effect, 24 likely due to central nervous system tolerance. 25
High-frequency 10-kHz SCS, a paresthesia-free therapy, was shown to be a safe and effective treatment for PDN in the recent SENZA-PDN study, with 24-month results demonstrating an average pain reduction of 80% and a remarkably high proportion of study participants (90%) achieving at least 50% pain relief. 26 In addition, investigators assessed that a majority of study participants (66%) had a clinically meaningful improvement in neurological status, most often in sensory function in the feet.26,27
Here, we extend the presentation of our results to include additional 24-month endpoints from the SENZA-PDN study, focusing on diabetes-related pain and quality of life outcomes.
Methods
SENZA-PDN Study
The SENZA-PDN study, detailed in prior publications,26,28-30 was a multicenter, prospective, randomized, open-label clinical trial (ClinicalTrials.gov: NCT03228420) conducted at 18 clinical sites in the United States, all of which received relevant institutional review board approval. Eligible participants were aged 22 years or older with PDN symptoms for at least 12 months that were unresponsive to conventional pharmacological therapies. All patients had lower limb pain ≥5 cm on a 10-cm visual analog scale (VAS), hemoglobin A1c (HbA1c) ≤10% (86 mmol/mol), BMI ≤45 kg/m2, and daily opioids ≤120 mg morphine equivalents. Participants provided written informed consent prior to study participation.
Procedures
Eligible patients were randomized to receive conventional medical management (CMM) for PDN or 10-kHz SCS as an adjunct to CMM (10-kHz SCS + CMM) for six months. 29 Glycemic control for each participant was managed by their treating physician, separately from the study investigators. At six months, participants with <50% lower limb pain relief and dissatisfaction with treatment could cross to the other treatment group if the investigator agreed that crossover was appropriate.
All recipients of 10-kHz SCS, whether initially assigned to receive 10-kHz SCS + CMM or crossing over to this treatment arm after six months, underwent a temporary stimulation trial lasting between five and seven days. Stimulation was applied to the spinal cord via percutaneous leads placed in the epidural space and an external pulse generator. Temporary trial participants experiencing ≥50% pain relief from baseline (ie, trial success) were eligible for permanent implantation of a 10-kHz SCS system (Nevro Corp, Redwood City, California). 31
Assessments were performed at baseline and specified visits up to 24 months, with all recipients of permanent 10-kHz SCS (including the original randomized group and the crossover cohort) followed through 24 months postimplantation.
Outcomes
We used validated instruments to assess clinical outcomes in our study population. The Brief Pain Inventory short form for Diabetic Peripheral Neuropathy (BPI-DPN) was used to evaluate pain severity and pain interference with mood and daily activities. 32 Participants rated their pain severity at its worst, least, average, and current level on a 0 to 10 numerical rating scale (NRS), with 0 indicating “no pain” and 10 representing “pain as bad as you can imagine.” They also rated how much pain interfered with various aspects of their life (general activity, mood, walking ability, normal work, relations with others, sleep, enjoyment of life), using a similar 0 to 10 NRS, with 0 indicating “does not interfere” and 10 representing “completely interferes.” Pain severity and interference items were each averaged to form two composite scores. 32
Participants rated their quality of life using the Diabetes Quality of Life (DQOL) instrument, a 46-item questionnaire comprising four subscales: satisfaction, impact, social/vocational worry, and diabetes-related worry. 33 Responses were given on a 5-point Likert scale for each subscale item, with scores transformed to a 0 to 100 scale (0 = lowest possible quality of life score to 100 = highest possible quality of life score) before running statistical analyses. 33
Investigators assessed participants’ social, occupational, and psychological functioning using the Global Assessment of Functioning (GAF) scale, which ranges from 0 to 100, with higher scores indicating better functioning. 34 Participants also rated their satisfaction with treatment using a 5-point Likert scale (0 = “very dissatisfied” to 5 = “very satisfied”).
Statistical Methods
Our statistical analysis focused on changes in outcomes between (1) baseline and six months, ie, the randomized phase, and (2) preimplantation and 24 months after permanent implantation of a 10-kHz SCS system, ie, the postimplantation phase. We conducted separate statistical analyses for these phases. The randomized phase analysis evaluated changes in outcomes for both the original recipients of 10-kHz SCS (ie, the original 10-kHz + CMM group) and the initial CMM alone group. The postimplantation phase analysis evaluated changes in outcomes in the original 10-kHz + CMM group, those in the CMM only group who crossed over to receive 10-kHz SCS + CMM (ie, the CMM-to-10-kHz SCS + CMM crossover cohort), and all permanently implanted participants combined. For the postimplantation phase analysis, we defined the preimplantation timepoint as the baseline assessment in the original 10-kHz SCS + CMM group and the crossover timepoint at the end of the randomized phase in the CMM-to-10-kHz SCS + CMM crossover cohort. Results are reported for all available data for each follow-up.
Statistical analyses were performed using SAS (Version 9.4, SAS Institute Inc, Cary, North Carolina). For the statistical significance evaluations, each missing data point was accounted for via a multiple imputation procedure using available nonmissing data. The analysis of time and group effects for continuous and ordinal variables used a repeated-measures linear model. The repeated-measures model included time and group as fixed effects and participant as a random effect, specifying a first-order autoregressive correlation structure. We used similar logistic and nonlinear models to analyze proportions and percentage changes over time.
Results
Study Population and Demographics
Investigators assessed 430 patients for eligibility, with 113 randomized to 10-kHz SCS + CMM and 103 to CMM alone. Detailed baseline characteristics have been published previously.26,29 In summary, the randomized population had a mean (standard deviation [SD]) age of 60.8 years (10.7), with a median (interquartile range [IQR]) duration of diabetes of 10.9 (6.3-16.4) years and a median (IQR) duration of PDN symptoms of 5.6 years (3.0-10.1). The cohort had a mean (SD) HbA1c of 7.4% (1.2) and a mean (SD) lower limb VAS score of 7.3 (1.6), indicating moderate-to-severe neuropathic pain. Baseline characteristics did not significantly differ between the randomized groups.
Among the 113 participants initially allocated to 10-kHz SCS + CMM, 104 underwent trial stimulation, with 98 experiencing ≥50% reduction in pain (ie, trial success), 90 of whom received a permanent system. Of these, 88 completed the six-month randomized phase, with 84 completing 24 months of postimplantation follow-up. At six months, there were no crossovers from 10-kHz SCS + CMM to CMM alone.
In the initial CMM alone group, 95 participants completed the six-month randomized phase, 83 were eligible for crossover, and 77 underwent trial stimulation. Of these, 73 had a successful trial, 64 received a permanent system, and 58 completed the 24-month postimplantation assessment. In total, 181 participants underwent trial stimulation, 171 (94.5%) had a successful trial, 154 received a permanent implant, and 142 (92.2%) completed the 24-month postimplantation follow-up.
Pain Severity and Pain Interference with Mood and Daily Activities
Randomized phase
Participants reported pain severity using the Brief Pain Inventory for Diabetic Peripheral Neuropathy (BPI-DPN; 0-10 NRS). During the randomized phase, pain severity in 10-kHz SCS recipients significantly reduced by 62.0% (95% confidence interval [CI] = 55.4%-68.7%; P < .001; Figure 1a). Notably, pain severity was significantly lower for the 10-kHz SCS + CMM group vs the CMM alone group as early as one month after baseline and at all subsequent time points through to six months (P < .001). In contrast, the CMM alone group had no significant change in pain severity over the same period (P = .99).
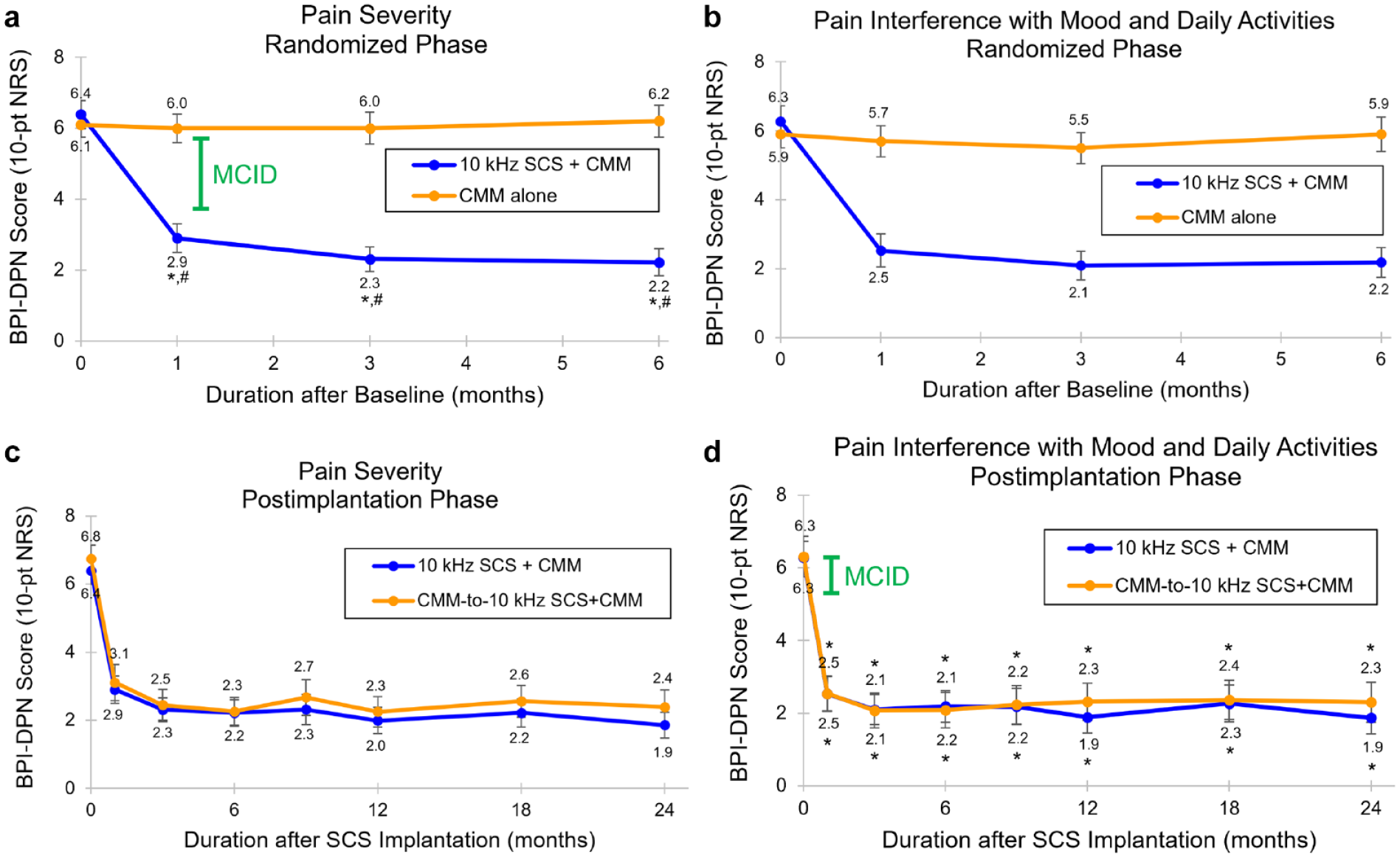
Brief Pain Inventory short form for Diabetic Peripheral Neuropathy (BPI-DPN). Mean pain severity (0-10 numerical rating scale [NRS]), measured using the BPI-DPN, during (a) the six-month randomized phase and (b) the 24-month postimplantation phase. Mean pain interference with mood and daily activities (0-10 NRS), measured using the BPI-DPN, during (c) the six-month randomized phase and (d) the 24-month postimplantation phase. Green bars show the MCID for BPI-DPN pain severity (a and b) 35 and BPI-DPN pain interference (c and d). 36 Error bars indicate 95% CI; *P < .001 vs baseline or preimplantation; #P < .001 vs CMM alone; $P = .045 vs CMM-to-10-kHz SCS + CMM.
Pain interference with mood and daily activities also reduced significantly during the randomized phase among the recipients of 10-kHz SCS, with the BPI-DPN interference score (0-10 NRS) decreasing by 64.3% (95% CI = 57.7%-71.0%; P < .001; Figure 1b). Meanwhile, the CMM alone group had no significant change in their BPI-DPN interference score (P = .58).
Postimplantation phase
After 24 months of 10-kHz SCS, the group of all implanted participants reported a significant decrease of 66.9% (95% CI = 62.4%-71.5%; P < .001) in pain severity, from a preimplantation mean of 6.5 (95% CI = 6.3-6.8) to a mean of 2.1 (95% CI = 1.8-2.4; P < .001) at 24 months (Figure 1c). Pain interference with mood and daily activities also significantly decreased over the same period, with the mean preimplantation interference score reducing by 65.8% (95% CI = 60.2%-71.5%; P < .001) from a preimplantation mean of 6.3 (95% CI = 5.9-6.6) to a mean of 2.0 (95% CI = 1.7-2.4; P < .001) at 24 months (Figure 1d).
Diabetes Quality of Life
Randomized phase
Health-related quality of life was assessed using the DQOL questionnaire, which provided an overall score and four subscale scores: satisfaction, impact, social/vocational worry, and diabetes-related worry. Individuals receiving 10-kHz SCS reported a significant improvement in their overall DQOL score over six months, with an increase of 15.5 points (95% CI = 12.2-18.8; P < .001) at six months (Figure 2a). In addition, significant improvements were observed across all DQOL subscales, with the largest gains observed in the satisfaction and impact domains (Figure 2b). Conversely, the control group had no significant change in their overall DQOL score from baseline (P = .17), and their score was significantly lower than the 10-kHz SCS + CMM group after the baseline visit through to the end of the randomized phase (P < .001). Furthermore, diabetes-related worry in the control group significantly worsened at six months (P = .02).
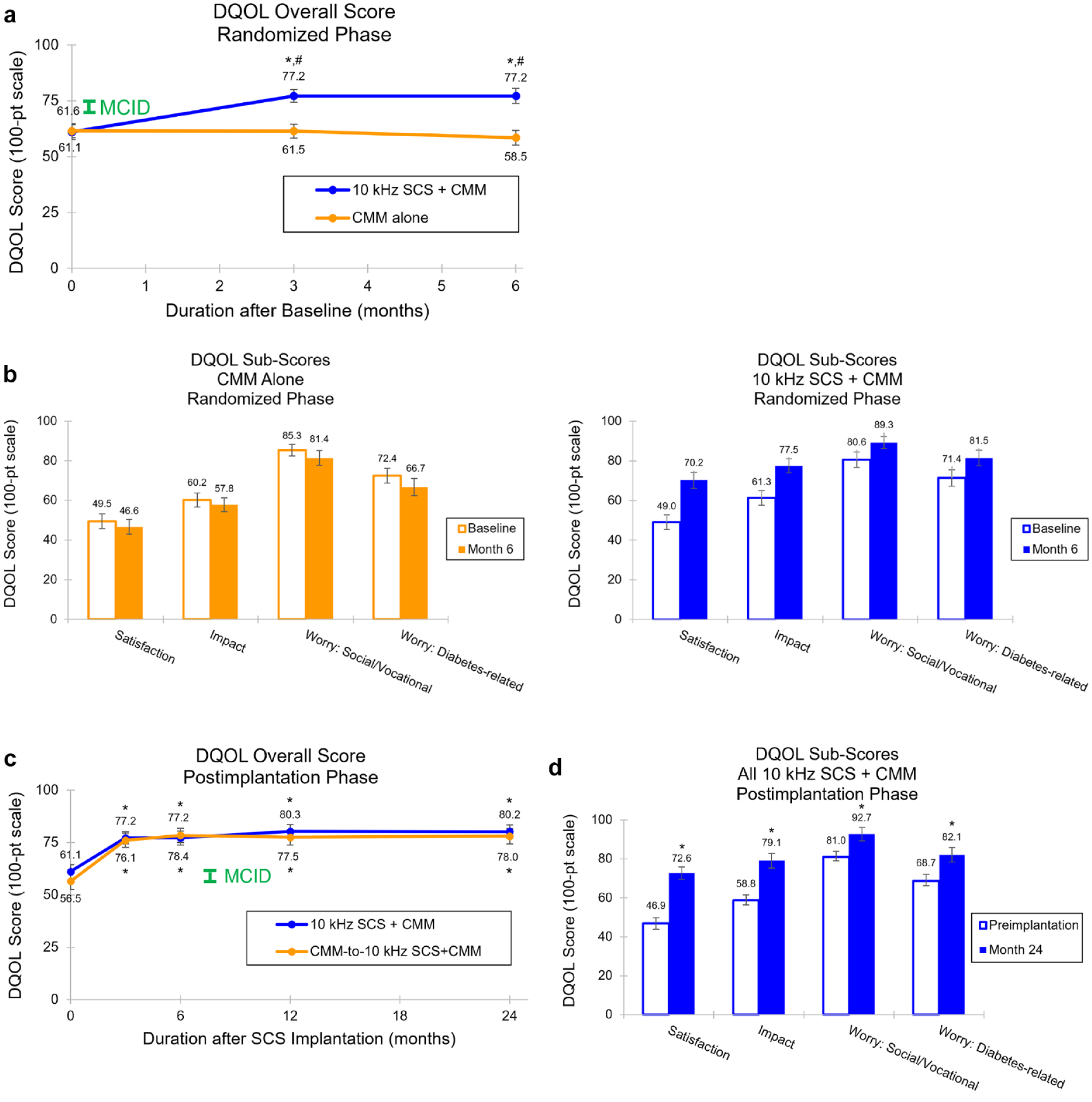
Diabetes-Related Quality of Life (DQOL). Mean DQOL overall score during (a) the six-month randomized phase and (b) the 24-month postimplantation phase. Green bars show the MCID for DQOL overall score (a and b). 37 Mean DQOL subscores during (c) the six-month randomized phase and (d) the 24-month postimplantation phase. Error bars indicate 95% CI; *P <.001 vs baseline or preimplantation; #P < .001 vs CMM alone; $P = .02 vs baseline; &P =.007 vs CMM alone.
Postimplantation phase
Over 24 months, treatment with 10-kHz SCS significantly improved DQOL in implanted study participants, evidenced by an increase of 20.3 points (95% CI = 17.5-23.0) in overall DQOL score, from a mean of 59.2 (95% CI = 56.6-61.7) at preimplantation to a mean of 79.3 (95% CI = 76.9-81.7; P < .001) at 24 months (Figure 2c). Participants also reported significant positive changes across all DQOL subscales, with the satisfaction and impact domains registering the largest improvements (Figure 2d).
Global Assessment of Functioning
Randomized phase
Investigators rated participants’ social, occupational, and psychological functioning using the GAF scale (0-100 points), reporting a similar mean baseline score in the 10-kHz SCS and CMM only groups (P = .85). After six months of treatment, overall functioning in the 10-kHz SCS recipients was significantly improved from baseline, demonstrated by an increase in mean GAF score of 17.8 points (95% CI = 13.9-21.7; P < .001; Figure 3a). Meanwhile, the mean GAF score in the control group trended down and remained significantly lower than the 10-kHz SCS + CMM group through to the end of the randomized phase.
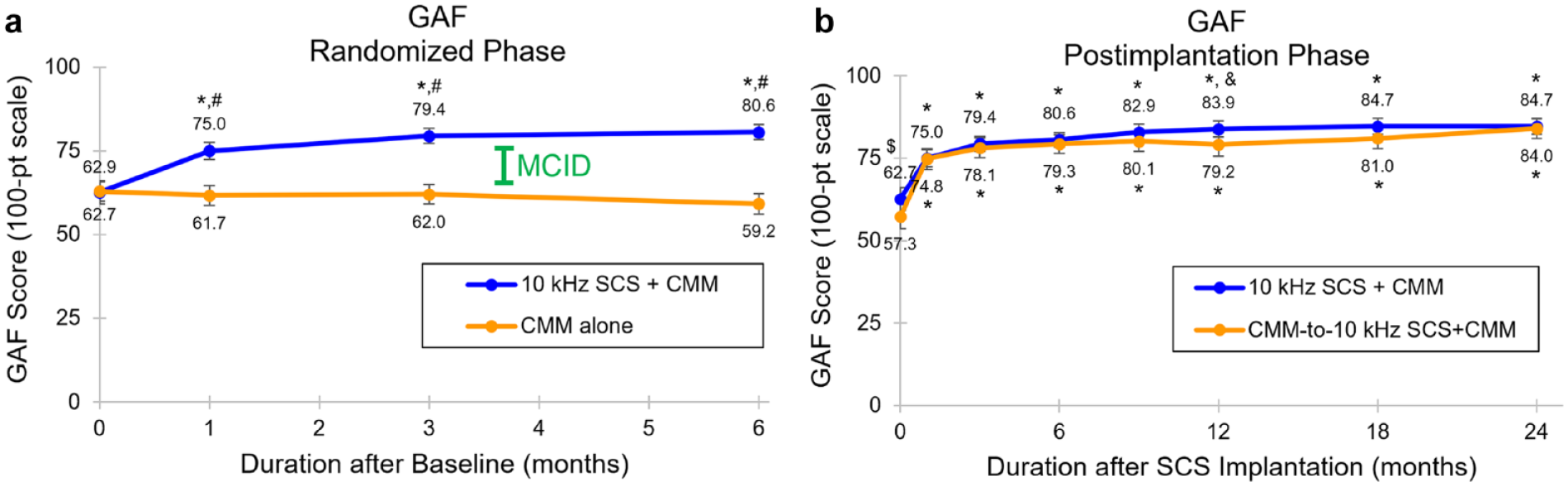
Global Assessment of Function (GAF). Mean GAF score during (a) the six-month randomized phase and (b) the 24-month postimplantation phase. Green bars show the MCID for GAF.38,39 Error bars indicate 95% CI; *P < .001 vs baseline or preimplantation; #P < .001 vs CMM alone; $P = .008 vs CMM-to-10-kHz SCS + CMM; &P = .049 vs CMM-to-10-kHz SCS + CMM.
Postimplantation phase
After 24 months of 10-kHz SCS, the mean GAF score among the group of all implanted participants increased by 23.6 points (95% CI = 20.5-26.6; P < .001) from a mean preimplantation value of 60.4 (95% CI = 57.9-62.9) (Figure 3b).
Satisfaction
Randomized phase
At the end of the randomized phase, 92.0% of 10-kHz SCS recipients (81 of 88) reported being “very satisfied” or “satisfied” with their treatment, whereas, in the CMM alone group, this proportion was only 7.4% (7 of 95; P < .001; Figure 4a).
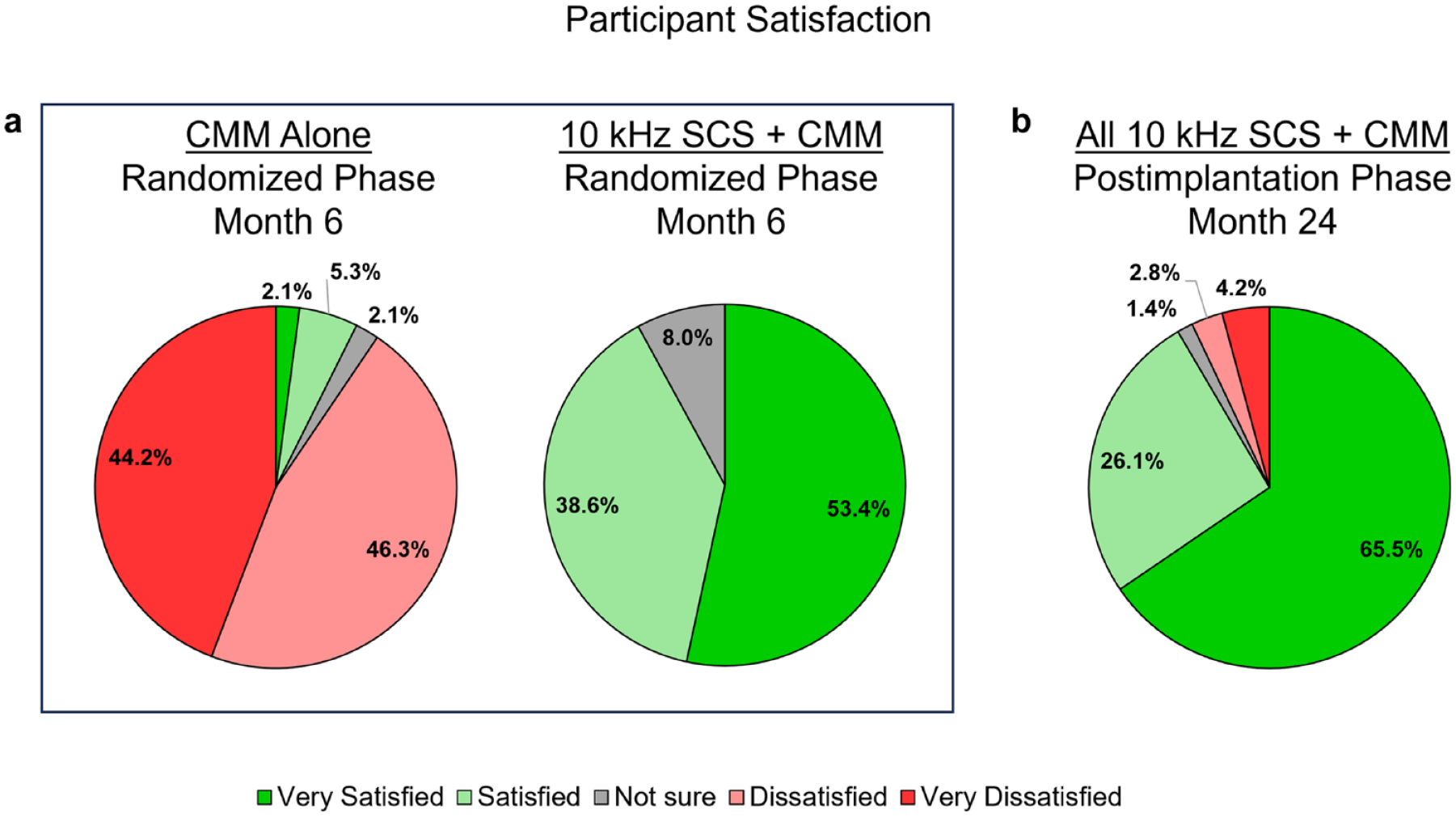
Participant satisfaction. Proportion of participants who were satisfied or very satisfied with their treatment during (a) proportion of participants who rated their satisfaction with treatment in each of the five categories at the end of the randomized phase (month 6) and (b) for all implanted participants at the end of the postimplantation phase (month 24).
Postimplantation phase
Satisfaction with 10-kHz SCS continued to be high after 24 months of treatment, with 91.5% of all implanted participants (130 of 142) reporting being “very satisfied” or “satisfied” with their therapy (Figure 4b).
Discussion
The SENZA-PDN study, the largest randomized controlled trial to date to evaluate SCS for the treatment of PDN, previously demonstrated significant pain relief and additional benefits with 10-kHz SCS, including improved sleep quality, HRQoL, and protective sensation in the feet. 29 The present analysis reinforces the benefits of 10-kHz SCS in patients with PDN, showing a rapid and significant decrease in pain severity, alongside substantial improvements in mood and daily activities, DQOL, and overall functioning. These benefits were sustained over 24 months, with study participants reporting a high level of satisfaction with the therapy.
Whenever feasible, the assessment of treatment benefits from any intervention should extend beyond statistical significance to include an understanding of whether the observed changes are clinically meaningful to patients. This approach helps to assess the real-world impact of treatments and prioritize interventions that offer genuine improvements to patients.
In the present analysis, we observed both statistically significant and clinically meaningful improvements across all evaluated metrics throughout the 24 months of 10-kHz SCS. Specifically, previous research has suggested that a 2-point or 30% reduction in pain severity on the BPI represents the minimum clinically important difference (MCID) for this metric in the context of fibromyalgia, 35 a chronic pain condition that can present with small-fiber neuropathy symptoms.36,37 In our implanted study population, pain severity decreased by 4.4 points (66.9%) over 24 months. For the BPI interference scale, which assesses the impact of pain on mood and daily activities, IMMPACT consensus guidelines (Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials) suggest that a 1-point change is considered a reasonable threshold for an MCID. 38 Our cohort reported a mean change of 4.2 points after 24 months of 10-kHz SCS, highlighting the positive impact of treatment on mood and daily activities. Moreover, the increase in overall DQOL score of 20.3 points at 24 months considerably surpassed the estimated MCID of three to four points in a Taiwanese population with diabetes. 39 Finally, based on a defined MCID of 10 points for the GAF scale,40,41 our analysis found an improvement of more than twice the MCID, with a 23.6-point increase after 24 months of 10-kHz SCS. Overall, our results indicate a substantial, meaningful, and sustained improvement in HRQoL among the SENZA-PDN study participants treated with 10-kHz SCS.
Although some existing pharmacotherapies provide a degree of pain relief for some PDN patients, many continue to suffer from unremitting pain. Traditional paresthesia-based LF-SCS can provide modest alleviation of PDN pain symptoms; however, the initial pain reduction can decrease over time.22-24 In contrast, 10-kHz SCS has demonstrated significantly higher and more enduring pain relief than LF-SCS in this patient population.26,42 Importantly, 10-kHz SCS delivers pain relief without inducing paresthesia, an advantage that may be particularly beneficial for PDN patients experiencing disease-related sensory disturbances, such as tingling or paresthesia in the affected limbs.
The principal strengths of the SENZA-PDN study are its randomized design, large sample size, and long-term follow-up. The study limitations have been detailed in prior publications.26,29 In brief, the nature of the interventions precluded blinding participants and investigators to the treatment allocation, potentially introducing bias and placebo effects. However, the durable results over 24 months support a minimal placebo effect. Some participants in the study had missing data points, although we addressed this issue through a multiple imputation procedure as part of the statistical analysis. Over 90% of permanently implanted participants attended the 24 months visit, and there were no more than 7.8% of datapoints missing in any individual assessment analysis.
It is also important to note that the 24-month assessment was performed relative to the preimplantation visit, which corresponded to baseline in the original 10-kHz SCS + CMM group and six months postbaseline in the crossover cohort (CMM-to-10-kHz SCS + CMM). During these six months, the crossover cohort may have experienced clinical deterioration. However, a comparison of outcomes (ie, pain severity, pain interference with mood and daily activities, overall DQOL, and GAF) at 24 months showed no significant differences between the groups in percentage improvements and final scores, except for a slight difference in the final pain severity score (P = .045), indicating that the six-month implant delay did not disadvantage the crossover cohort.
Although patient-reported outcomes (PROs) are the basis of most of our findings, potentially leading to subjective bias, recall errors, and variability in individual perception, the insights gained from these PROs are invaluable to understanding the patient’s experience. In addition, the clinicians’ assessment of GAF corroborated the findings from the PROs.
Conclusions
High-frequency 10-kHz SCS significantly alleviates pain and provides multiple secondary benefits that improve HRQoL for patients with PDN, with benefits sustained over 24 months. The robustness and durability of these results, coupled with high patient satisfaction, highlight that 10-kHz SCS is an efficacious and comprehensive therapy for patients with PDN.
Footnotes
Acknowledgements
The authors thank Colleen Kelly, PhD, for conducting the statistical analyses. The authors thank Bradford Gliner, MS, and Jeyakumar Subbaroyan, PhD, for their contributions to the study design.
Abbreviations
BMI, body mass index; BPI-DPN, Brief Pain Inventory for diabetic peripheral neuropathy; CI, confidence interval; CMM, conventional medical management; DQOL, Diabetes Quality of Life; DN, diabetic neuropathy; GAF, Global Assessment of functioning; HbA1c, hemoglobin A1c; HRQoL, health-related quality of life; IMMPACT, Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials; IQR, interquartile range; LF-SCS, low-frequency spinal cord stimulation; MCID, minimum clinically important difference; NRS, Numerical Rating Scale; PDN, painful diabetic neuropathy; PRO, patient-reported outcome; SAS, statistical analysis system; SCS, spinal cord stimulation; SD, standard deviation; VAS, visual analog scale.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EAP has received consulting fees from Abbott Laboratories, Biotronik, Boston Scientific, Medtronic Neuromodulation, Nalu Medical, Neuros Medical, Nevro Corp, Presidio Medical, Saluda, and Vertos Medical; research support from Mainstay, Medtronic Neuromodulation, Nalu Medical, Neuros Medical, Nevro Corp, ReNeuron, Saluda, and SPR; and stock options from neuro42 and SynerFuse. TGS has received research support from Nevro Corp. JAS has received research support from Boston Scientific, Nevro Corp, Saluda Medical, and Vertiflex. MJJ is an employee of Nevro Corp. DRE received a fee from Nevro Corp for the preparation of this manuscript in her capacity as an independent medical writer. JLW has received consulting fees from California Institute for Biomedical Research and Eli Lilly and research support from Nevro Corp. SMS has received research support from Nevro Corp. KA has received consulting fees from Biotronik, Medtronic, Nalu Medical, Nevro Corp, and Saluda Medical, as well as research support from Biotronik, IPM Medical Group, Nevro Corp, Saluda Medical, SPR Therapeutics, and Vivex Biologics. MNG has received consulting fees from Abbott Laboratories, Avanos Medical, Avertis Pharmacy, Boston Scientific, Nevro Corp, and Saluda Medical, as well as research support from Abbott Laboratories, Avanos Medical, Boston Scientific, Nalu Medical, Neuros Medical, Nevro Corp, and Saluda Medical. JX has received research support from the Cleveland Clinic Velosano Program, the National Institutes of Health, the Steve and Melody Golding Foundation, and Nevro Corp. CY has received research support from Nevro Corp. AN has received consulting fees from Aurora Spine, Flowonix, and Nevro Corp, as well as research support from Nevro Corp. DGP has received consulting fees from Abbott Laboratories, AIS Healthcare, Allergan, Amgen, Aurora Spine, CornerLoc, Flowonix, Lundbeck, Pajunk Medical, Saluda Medical, Spark Biomedical, and Vertos Medical; research support from Abbott Laboratories, Aurora Spine, Flowonix, Nevro Corp, and Saluda Medical; speakers’ bureau or honoraria from Abbott Laboratories, Allergan, Amgen, CornerLoc, Lundbeck, Saluda Medical, and Vertos Medical; and stock options from CornerLoc. MJC has received research support from Nevro Corp. VG has received research support from Biotronik, Medtronic, Nevro Corp, PainTEQ, SPR Therapeutics, and St. Jude. RHB has received research support from Nevro Corp. NDM has received consulting fees from Averitas, Nevro Corp, and Salix Pharmaceuticals, as well as research support from Boston Scientific and Nevro Corp. DS has received consulting fees from Abbott Laboratories, Boston Scientific, Flowonix, Medtronic, Nevro Corp, Vertiflex, and Vertos Medical, as well as research support from Abbott Laboratories, Biotronik, Nevro Corp, Vertiflex, and Vertos Medical. SPL has received consulting fees from Nevro Corp and research support from Nevro Corp. DJD has received research support from Nevro Corp, as well as funding for serving as principal investigator of a study supported by SPR Therapeutics paid to his institution. KAS has received research support from Nevro Corp. JHG has received consulting fees from Abbott Laboratories, Saluda Medical, and Stratus Medical. PWW has received research support from Nevro Corp. CEA has received consulting fees from AbbVie, Amgen, Biohaven, Clexio Biosciences, Collegium, Eli Lilly, Elsevier, Flowonix, Gene Pharma, Lundbeck, Nevro Corp, Novartis, Pfizer, SK Life Science, Teva Pharmaceutical, and Vertex, as well as research support from AbbVie, Allergan, Amgen, Daiichi Sankyo, Eli Lilly, Novartis, Teva Pharmaceutical, and Vertex Pharmaceuticals. CEN has received consulting fees from Exelixis, Neurogastrx, and Nevro Corp, as well as research support from Nevro Corp. RST has received consulting fees from Nevro Corp, Medtronic, and Saluda Medical. DLC is an employee of Nevro Corp. NAM has received consulting fees from Nevro Corp, Relievant Medsystems, Saluda Medical, Sollis Therapeutics, and Vertos Medical, as well as research support from Avanos Medical, Mesoblast, Neuros Medical, and Nevro Corp.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Nevro Corp.
