Abstract
Background:
Extended glucose predictions are novel in diabetes management. Currently, there is no solution widely available. People with diabetes mellitus (DM) are offered features like trend arrows and limited predictions linked to predefined situations. Thus, the impact of extended glucose predictions on the burden of diabetes and person-reported outcomes (PROs) is unclear.
Methods:
In this online survey, 206 people with type 1 and type 2 diabetes (T1D and T2D), 70.9% and 29.1%, respectively, who participated in the dia·link online panel and were current continuous glucose monitoring (CGM) users, were presented with different scenarios of hypothetical extended glucose predictions. They were asked to imagine how low glucose predictions of 30 minutes and overnight as well as glucose predictions up to 2 hours would influence their diabetes management. Subsequently, they completed the Hypoglycemia Fear Survey II (HFS-II) and the T1 Diabetes Distress Scale (T1-DDS) by rating each item on a 5-point scale (−2: strong deterioration to +2: strong improvement) according to the potential change due to using glucose predictions.
Results:
For all glucose prediction periods, 30 minutes, up to 2 hours, and at nighttime, the surveyed participants expected moderate improvements in both fear of hypoglycemia (HFS-II: 0.57 ± 0.49) and overall diabetes distress (T1-DDS = 0.44 ± 0.49). The T1-DDS did not differ for type of therapy or diabetes.
Conclusions:
People with T1D and T2D would see glucose predictions as a potential improvement regarding reduced fear of hypoglycemia and diabetes distress. Therefore, glucose predictions represent a value for them in lowering the burden of diabetes and its management.
Keywords
Introduction
Commonly, the efficacy and safety of diabetes treatments are evaluated by clinically measurable outcomes such as levels of glycated hemoglobin (HbA1c), time in range (TIR), fasting glucose, or the number of hypoglycemic events.1,2 However, these endpoints neglect the World Health Organization’s (WHO) holistic concept of health in particular when it comes to digital health solutions such as continuous glucose monitoring (CGM).3-5 The latter may influence outcomes related to people with diabetes mellitus’ (DM) health and well-being that are beyond measurable metabolic parameters as, eg, their empowerment, quality of life, health literacy, or the perceived burdens of the disease.5,6 Therefore, assessing person-reported outcome (PRO) can provide a better understanding of an intervention’s whole impact and can support informed decision-making, eg, if and how a specific intervention could be implemented in diabetes management.4,6
It is particularly important to evaluate the burden of living with diabetes and its associated distress that may have multi-faceted sources such as feeling powerless, as well as worries about hypoglycemia and diabetes management in general. Diabetes distress and the fear of hypoglycemia are both associated with lower levels of self-care, adherence, and general emotional well-being as well as with sub-optimal glycemic control as people affected may tend to maintain higher glucose levels.7-13 Subsequently, family members can also be affected, and there may be various negative socio-economic consequences such as rising health costs, incapacity to work, and loss of productivity.12-14
The results of several studies among people with type 1 diabetes (T1D) suggest that CGM can improve PRO compared with conventional self-monitoring of blood glucose especially with regard to hypoglycemia-associated distress. Accordingly, the additional information provided by CGM (eg, alarms and trend arrows) allows people with diabetes engage in preventive action to avoid hypoglycemia or allows the proactive management. 10 ,15-19 In addition, existing evidence shows that people with type 2 diabetes (T2D), not using insulin, may benefit from CGM due to improvements in, eg, satisfaction, quality of life, and diabetes distress. 20
Common features of available CGM systems include trend arrows that allow predicting short-term glucose trends based on recent glucose levels. In general, trend arrows are very simple forms of glucose forecasts by plainly continuing linearly the glucose excursion of the ultra-short-term past. They differ from a true glucose prediction, among other things, in terms of the length of the forecast horizon and the lack of input parameters other than glucose excursion in the past minutes. In clinical practice, use of trend arrows can support decision-making, eg, regarding meals and insulin therapy.21-24 It has been shown that using trend arrows for diabetes management can improve glycemic outcomes and reduce insulin doses. 23 Although it is assumed that the use of trend arrows could improve the motivation of people with DM, little is known about the impact of trend arrows or glucose prediction on PRO. 22
Nevertheless, trend arrows are a step in the right direction as they deliver some valuable information, but which is not quite sufficient to improve burden and to lift quality of life. Thus, extended glucose predictions need to be implemented, and their value must be understood. A current example of such an extended glucose prediction can be seen in Herrero et al in the same special issue of this journal. 25
With the emergence of machine and deep learning, great progress has been made in predicting glucose, and especially hypoglycemia, for extended periods of time.26-30 A more detailed discussion on extended glucose prediction is provided by Herrero et al. 25
Despite the progress made in diabetes technology such as CGM, there are still some unmet needs. Among others, for some users, the vast amount of data may be overwhelming, and despite CGM use, the fear of hypoglycemia remains, whereas glucose threshold alarms are perceived as intrusive, especially when they occur in public.9,13,15,16 The lack of knowledge about the future course of glucose can lead to hypoglycemia events in critical situations including associated social stigmata and professional discrimination. Impeding hypoglycemia may also impair personal spontaneity, whereas sudden hypoglycemia alarms disturb night’s rest of the person with DM as well as her or his potential partner/family. In summary, the lives of people with DM today might be defined by constantly firefighting instead of a basic feeling of calm and sustainable control. 9 There is a lack of knowledge on how glucose prediction affects diabetes-associated burden and related PRO. This applies in particular to the novel concept of long-term glucose prediction based on CGM algorithms. Possibly, glucose prediction could provide relief for people with DM struggling with the unknown related to their glucose profiles. They could benefit from more certainty in general while being better prepared in meaningful social and professional situations, or being less bothered by hypoglycemia-associated sleep interruptions. Particularly, nocturnal hypoglycemia has great clinical and psychosocial relevance that may benefit from extended glucose prediction as discussed by Kulzer et al in the same special issue of this journal. 31
More evidence is needed to demonstrate that glucose predictions serve these unmet needs. Therefore, we conducted this PRO survey to explore the perceived benefits of a 30-minute and an overnight low glucose prediction, as well as a glucose prediction for up to 2 hours, particularly with regard to fear of hypoglycemia and diabetes distress.
Methods
Study Design
The objective of this online survey was to assess the potential benefits of an entire hypothetical system consisting of low glucose predictions of 30 minutes and overnight, as well as glucose predictions of up to 2 hours, based on CGM algorithms, for diabetes management as well as in terms of diabetes distress and fear of hypoglycemia in people with DM. The survey was conducted between July and September 2022, and a total of 400 members of the dia·link online panel (Forschungsinstitut Diabetes-Akademie Bad Mergentheim [FIDAM], Germany, www.dialink-diabetes.de) were invited to attend. The dia·link panel has been previously described.32,33 Members of the panel are people with DM who gave their informed consent to be invited to online surveys. Ethical approval from the German Psychological Society for the panel and the surveys within the panel was obtained. Those invited to take part in this survey were people with T1D or T2D who used a CGM system. All participants were informed about the survey and provided informed consent before starting the survey.
First, they were presented to four hypothetical scenarios:
Special non-routine situations: Participants were instructed to imagine real-life situations outside of their routine life in which it would not be easy to treat impending hypoglycemia, eg, an important presentation or meeting, an exam, and a night at the opera/cinema.
Nightly hypoglycemia: Participants were instructed to imagine the thought and worries regarding nocturnal hypoglycemia before going to bed, having to treat a nocturnal hypoglycemia, or thinking about the next day after a nocturnal hypoglycemia.
Getting along with the CGM data: Participants were instructed to imagine the effort of better understanding glucose trajectories and identifying patterns.
Life with diabetes: Participants were instructed to imagine real-life situations in which glucose control was unpredictable and uncontrollable, a sudden hypoglycemia occurred without any explanation, or in which they do not want to think about diabetes for a certain amount of time.
Then, they were asked to imagine how low glucose predictions of 30 minutes and overnight, and glucose predictions of up to 2 hours would affect their diabetes management in these situations. The Hypoglycemia Fear Survey II (HFS-II) 34 and T1 Diabetes Distress Scale (T1-DDS) 12 questionnaire were used for assessment of the expected impact of these prediction features. For each item, the expected change due to the respective prediction, participants were asked to answer on a 5-point scale: (−2) strong deterioration, (−1) deterioration, (0) no change, (1) improvement, or (2) strong improvement. The HFS-II consists of 33 items and the two subscales Behavior (HFS-B) and Worry (HFS-W). The HFS-B items describe behaviors that people with DM may resort to in order to prevent hypoglycemic events and/or their negative consequences such as keeping blood glucose levels above a certain value or avoiding being alone. The HFS-W items describe specific hypoglycemia-associated worries that people with DM may have as, eg, nocturnal hypoglycemia or having an accident. 34 The T1-DDS contains a total of 28 items distributed between these seven subscales: powerlessness, management distress, hypoglycemia distress, negative social perceptions, eating distress, physician distress, and friend/family distress. 12 The T1-DDS was chosen because of its multi-faceted assessment of different sources of diabetes distress compared with other distress questionnaires. 8
Statistical Analyses
The sample was recruited using a convenience sampling approach. All analyses were based on the full analysis set (FAS) population of all participants who completed the respective questionnaires. Anticipated changes in HFS-II and T1-DDS scales and subscales are depicted using box-plots to illustrate the distribution of expected effects. Differences in anticipated changes between T1D and T2D as well as between different types of insulin therapy and CGM systems were analyzed using
Results
Sample Characteristics
A total of 206 people with DM took part in the survey, with 204 participants completing HFS-II and 206 finishing T1-DDS. The majority (70.9%) were people with T1D, who were on average younger while having lower means for HbA1c and body mass index (BMI) than those with T2D. Although the T1D cohort included roughly equal numbers of women and men, 21.7% of the T2D subgroup were female. Table 1 shows further demographic characteristics of the sample.
Sample Characteristics.
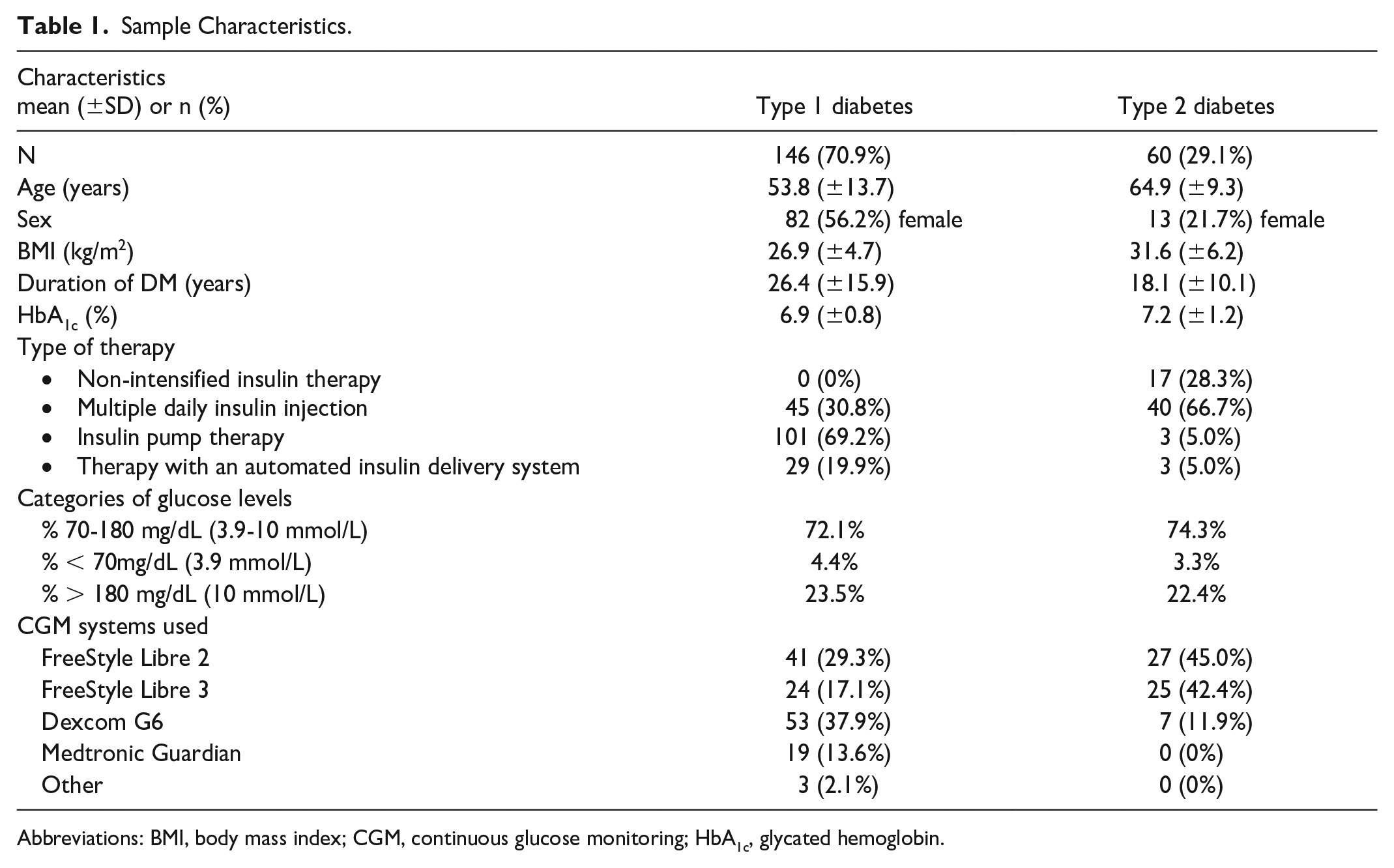
Abbreviations: BMI, body mass index; CGM, continuous glucose monitoring; HbA1c, glycated hemoglobin.
Expected Change in Diabetes Distress
Overall, the respondents expected that diabetes distress will improve as a result of the extended glucose predictions (Figure 1). The overall T1-DDS mean score was 0.44 (standard deviation [SD] = ±0.43), whereas the subscale for hypoglycemic distress reached the highest mean value of 0.81 (SD = ±0.60). Regarding other individual T1-DDS dimensions, noticeable improvements, due to glucose prediction, were in management distress, meaning the feeling of not being sufficiently skilled at managing DM, and in powerlessness, meaning the worries that serious long-term complications will develop no matter how hard people with DM try. In addition, due to glucose prediction, people with T2D showed an improved score for the dimension of eating distress, meaning the feeling that thoughts about food control their lives. Figure 1 shows the improvements in the other T1-DDS subscales.
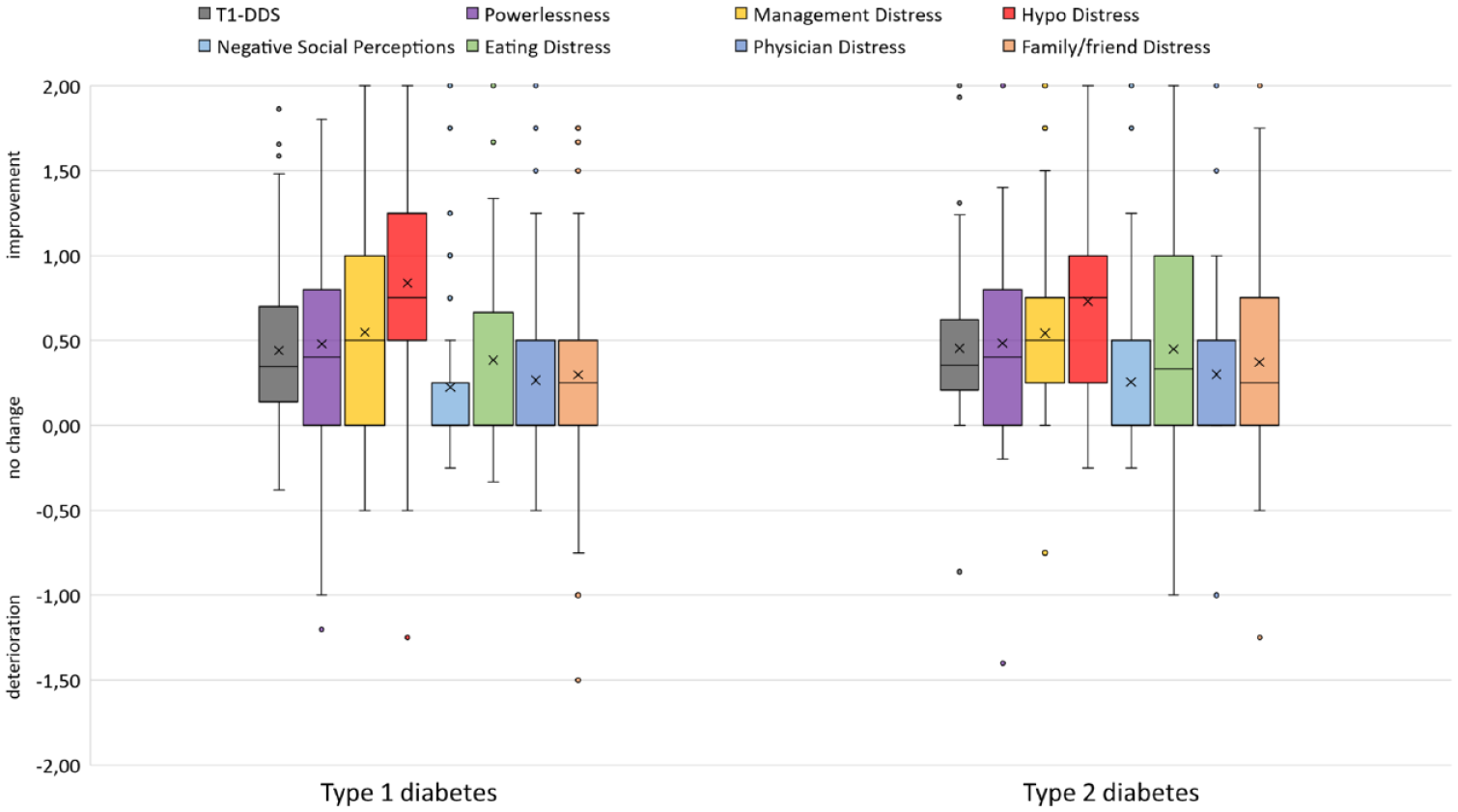
Expected change in diabetes distress (mean T1-DDS scores) stratified by type of diabetes and by T1-DDS subscales (N = 206).
On an item level, the strongest improvements were expected regarding distress about nocturnal hypoglycemia, feeling discouraged when seeing glucose values that are hard to explain, not ever feeling safe about the risk of severe hypoglycemia, and distress that one is not as skilled at managing diabetes as one should be (Supplementary Table S1). These results indicate that extended glucose predictions have the potential to reduce the burden of daily diabetes management and related worries.
The anticipated extent of improvements in diabetes distress did not differ significantly for type of therapy (all
Expected Change in Fear of Hypoglycemia
For the overall sample, the expected fear of hypoglycemia improved moderately with a mean HFS-II score of 0.57 (SD = ±0.49), whereas the HFS-II scores of people with T1D were higher than those in the T2D group (Figure 2). About 86.3% of all participants anticipated that fear of hypoglycemia would improve with the extended glucose prediction and only 2.9% expected a deterioration. There were no significant differences between the types of diabetes (
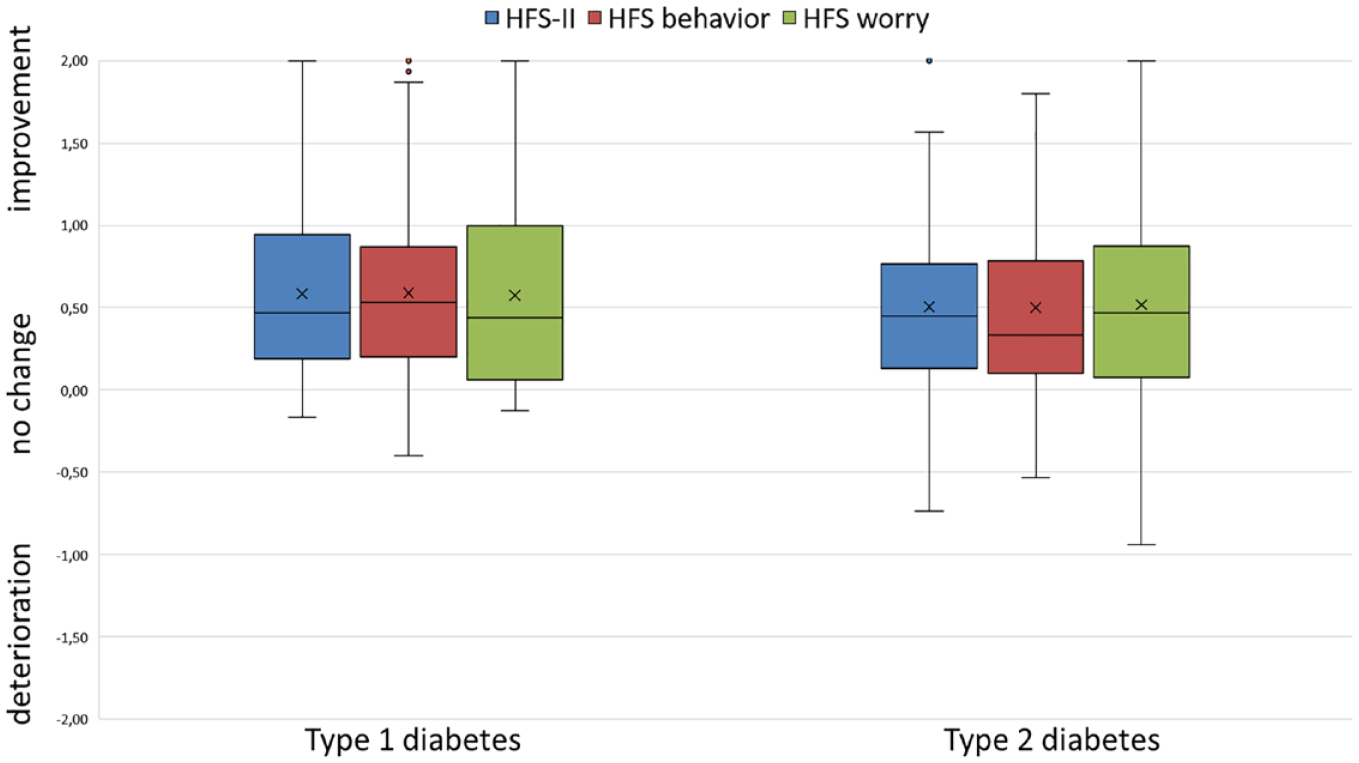
Expected change in fear of hypoglycemia (mean HFS-II scores) stratified by type of diabetes and by HFS-II dimensions (N = 204).
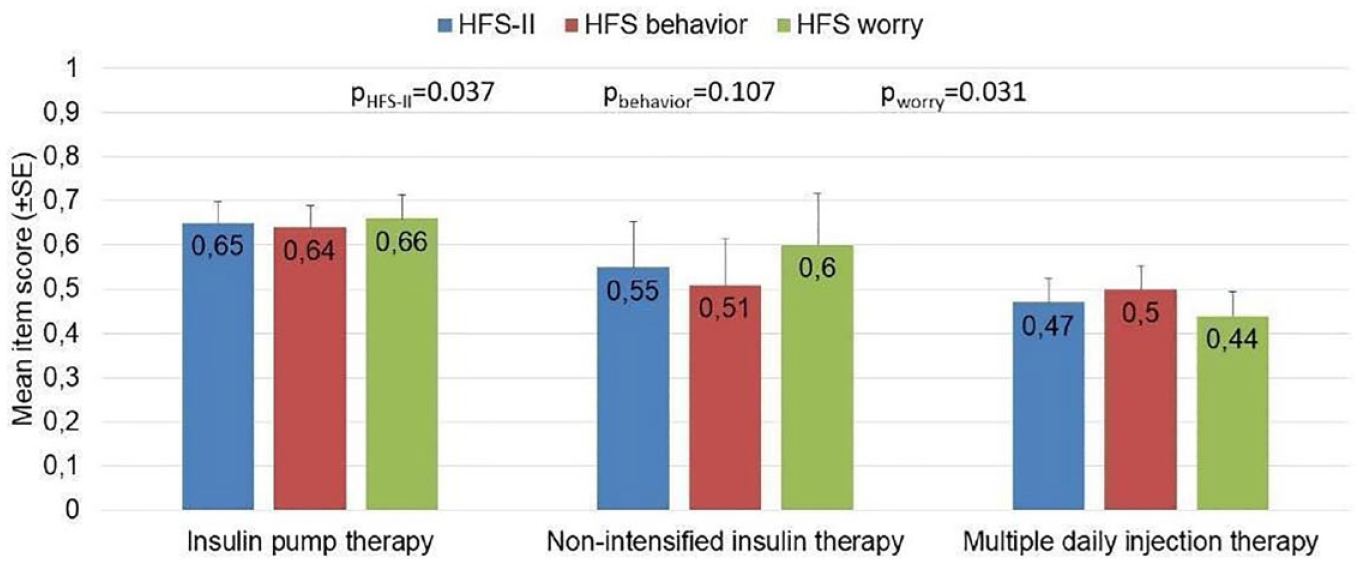
Mean HFS-II scores stratified by types of diabetes therapy and by HFS-II dimensions (N = 204).
With regard to the individual questionnaire items, there were substantial expected improvements due to glucose predictions in some behavioral dimensions (hypoglycemia avoidance strategies) such as eating snacks between meals or avoiding physical activities to prevent hypoglycemia (Supplementary Table S2). Furthermore, participants expected the following behavioral scores to improve: limiting car rides and travels, keeping glucose levels higher in certain social or critical situations, or reducing insulin doses. In terms of the HFS-W items, participants expected fewer worries about hypoglycemia, due to glucose prediction, while driving a car or being responsible for others as well as being impaired in the performance of important activities, making mistakes, or causing an accident due to hypoglycemia (Supplementary Table S2). These results indicate that extended glucose prediction would help people with DM feel more prepared instead of having to firefight glycemic excursions.
With regard to nighttime low glucose prediction and the fear of severe nocturnal hypoglycemia, 75% of those surveyed expected improvements or strong improvements with a rating for the respective item of around 1 (Supplementary Table S1).
There was a statistically significant effect between types of insulin therapy with individuals using an insulin pump showing the greatest expected improvement in fear of hypoglycemia, followed by people with non-intensified insulin therapy and those with multiple daily injection therapy (
Discussion
In this online survey, we investigated the potential impact of a CGM-based prediction suite providing a 30-minute low glucose forecast, a 2-hour glucose forecast, and a nighttime low glucose forecast on fear of hypoglycemia and diabetes distress in people with T1D or T2D who were CGM users.
The results have shown substantial improvement in the worry and behavioral components of fear of hypoglycemia as well as in diabetes distress with its subscales hypoglycemia-related distress, management distress, and powerlessness. Participants indicated that they expect an extended glucose prediction before car rides to be beneficial regarding less worries about hypoglycemia while driving and restricting themselves less in car/bike driving due to this fear.
This is in line with the findings that indicate that glucose predictions could be seen as support according to fear of hypoglycemia and consequences in public and the impact on behavior. Furthermore, participants expect that extended glucose predictions may improve their behavior by keeping glucose levels high in important or social situations to avoid hypoglycemia. This indicates that extended glucose predictions may provide relief for people with DM in situations where they do not want to disclose their condition and its management to the public and where they may intend to avoid diabetes stigma.
Participants stated that a nighttime low glucose prediction would make them less afraid of nighttime hypoglycemia. This indicates that the additional information of a glucose prediction warning of low glucose during the sleeping hours is seen as a valuable tool by people with DM, as it might enable them in a preventive manner. Furthermore, we found that participants anticipated that the prediction of low glucose events could reduce their worries about missing hypoglycemia and improve their spontaneity and social life. So, extended glucose predictions could empower people with DM to live a more physical and socially active life.
Improved quality of life through reduced fear of hypoglycemia and diabetes distress by glucose predictions is of great importance and therefore meaningful in itself.10,15,16 This is also underscored by international diabetes guidelines that define the reduction of fear of hypoglycemia and diabetes distress as key treatment goals.35,36
However, the following limitations of our survey need to be considered. First, it is mainly tech-savvy and highly motivated people who register for the dia·link panel, which can lead to selection bias. Second, the T1-DDS was originally developed for people with T1D due to its focus on aspects of intensified insulin therapy. 12 However, the same questionnaire was needed for people with T1D and T2D to compare the results. As the majority of people with T2D had an intensified insulin therapy, the results can still be considered as valid. Finally, the test scenarios were only hypothetical, and the respondents anticipated the benefits of extended glucose predictions without actually experiencing or testing them.
Limitations and caveats of an extended glucose prediction must also be considered, as no prediction algorithm is without limitations. 26 Besides positive aspects, an extended prediction may also lead to overcorrections or hasty treatment decisions leading to increased glucose fluctuations and dissatisfaction. Thus, training and education for people with diabetes are essential not only to fully understanding the possibilities but also limitations of an extended glucose prediction. Therefore, further research is needed to show improvements in PRO by the extended glucose predictions. Randomized controlled trials are needed to differentiate the glycemic and PRO-related benefits of an extended prediction horizon (>30 minutes, 120 minutes, overnight hypo prediction) beyond the standard prediction (15-30 minutes) of current CGM systems. Furthermore, research on the actual use of such predictive features is needed, also in different populations (T1D vs T2D) with different treatment regimens (eg, basal insulin only, multiple daily injections).
Conclusions
This online survey assessed how hypothetical glucose predictions, based on CGM data, may influence fear of hypoglycemia and diabetes distress in different everyday scenarios among people with T1D or T2D. These results suggest that extended glucose predictions could reduce the diabetes-related burden of people with DM in their everyday life and therefore seem to be a valuable tool in addition to CGM.
Supplemental Material
sj-docx-1-dst-10.1177_19322968241267886 – Supplemental material for Fear of Hypoglycemia and Diabetes Distress: Expected Reduction by Glucose Prediction
Supplemental material, sj-docx-1-dst-10.1177_19322968241267886 for Fear of Hypoglycemia and Diabetes Distress: Expected Reduction by Glucose Prediction by Dominic Ehrmann, Luigi Laviola, Lilli-Sophie Priesterroth, Norbert Hermanns, Nils Babion and Timor Glatzer in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors are grateful to the study participants for making this research possible. The authors thank Dr Guido Freckmann, Professor Lutz Heinemann, Professor Bernhard Kulzer, Professor Oliver Schnell, and Dr Ralph Ziegler for giving advice and input in the course of writing the manuscript. The authors also thank Dr Daniel Militz, Dr Sandra Moceri, and Christine Zepezauer for careful review of the manuscript. The authors thank Hendericus Bos for his contribution in setting up the design of the screens.
Abbreviations
ANOVA, analysis of variance; BMI, body mass index; CGM, continuous glucose monitoring; DM, diabetes mellitus; FAS, full analysis set; HbA1c, glycated hemoglobin; HCP, health care provider; HFS-II, Hypoglycemia Fear Survey II; PRO, person-related outcome; SD, standard deviation; T1D, type 1 diabetes; T1-DDS, T1 Diabetes Distress Scale; T2D, type 2 diabetes; TIR, time in range; WHO, World Health Organization.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.E. reports lecture fees from Roche Diabetes Care, Sanofi-Aventis, and Dexcom Germany and is an Advisory Board member of Roche Diabetes Care and Dexcom Germany. L.L. reports lecture fees from Abbott, AstraZeneca, Boehringer-Ingelheim, Eli Lilly, Merck Sharp & Dohme, Medtronic, Menarini, MOVI SpA, Mundipharma, Novo Nordisk, Roche Diabetes Care, Sanofi-Aventis, and Terumo. He is an Advisory Board member of Abbott, AstraZeneca, Boehringer-Ingelheim, Eli Lilly Italia, Medtronic, MOVI SpA, Novo Nordisk, Roche Diabetes Care, Sanofi-Aventis, and Terumo. L-S.P. reports lecture fees from Ypsomed. N.H. reports Advisory Board member fees from Abbott Diabetes Care and Insulet as well as honoraria for lectures from Berlin Chemie AG, Becton Dickenson, Sanofi Germany, Roche Diabetes Care, and Dexcom Germany. N.B. and T.G. are employees and stockholders of Roche Diabetes Care GmbH.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been funded by Roche Diabetes Care GmbH.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
