Abstract

Introduction
Pregnancies complicated by type 1 diabetes are subject to variations in insulin sensitivity from fluctuating maternal and placental hormone levels throughout gestation. 1 Hybrid closed-loop (HCL) therapy has been shown to increase time in target glucose range and decrease episodes of hypoglycemia in non-pregnant individuals when compared to sensor-augmented pump therapy (SAPT), 2 but its use has been rarely studied in pregnancy. 3 Some preliminary HCL studies in pregnancy have shown that the therapy appears to help women attain glycemic goals of pregnancy, but its effects on glucose variability (GV) are not well established.3,4 In this analysis, we aim to compare the glycemic risk and continuous glucose monitor (CGM) variability metrics between HCL therapy and SAPT use during pregnancy.
Research Design and Methods
The research design for the Pregnancy Intervention with a Closed-Loop System (PICLS) study has been previously described. 5 Briefly, the PICLS study was a pilot, two-center, open-label, randomized controlled trial comparing HCL therapy and SAPT in pregnancy and early post-partum using the Medtronic 670G insulin pump and Medtronic Guardian 3 sensor, which, at the time of study start, was the only Food and Drug Administration (FDA)-approved HCL system on the market in the United States. Participants were randomized between 14 and 18 weeks of gestation and used their assigned therapy through four to six weeks post-partum.
The PICLS study’s primary objectives were to compare safety parameters and the main glycemic indices (eg, time below, in, and above range) between groups and have been described in detail elsewhere. 5
The secondary objectives of the PICLS study included comparisons of alternative glycemic variables, which we herein present. CGM data were compared between groups for each phase of the study (run-in phase, first trimester, second trimester, third trimester, and post-partum) for the secondary glycemic metrics (low blood glucose index [LBGI], high blood glucose index [HBGI], mean amplitude of glycemic excursions [MAGE], continuous overall net glycemic action [CONGA n ], and J-index).
Results
There were 23 participants who completed the study (n = 12 SAPT, n = 11 HCL). There were no statistically significant differences in measures of glycemic risk (LBGI and HBGI) or in measures of glycemic variability (MAGE, CONGA, or J-index) observed between the HCL and SAPT groups during any point in the study in pregnancy or post-partum (Table 1).
Measures of Glycemic Risk and Glucose Variability of SAPT and HCL Therapy in Pregnancy.
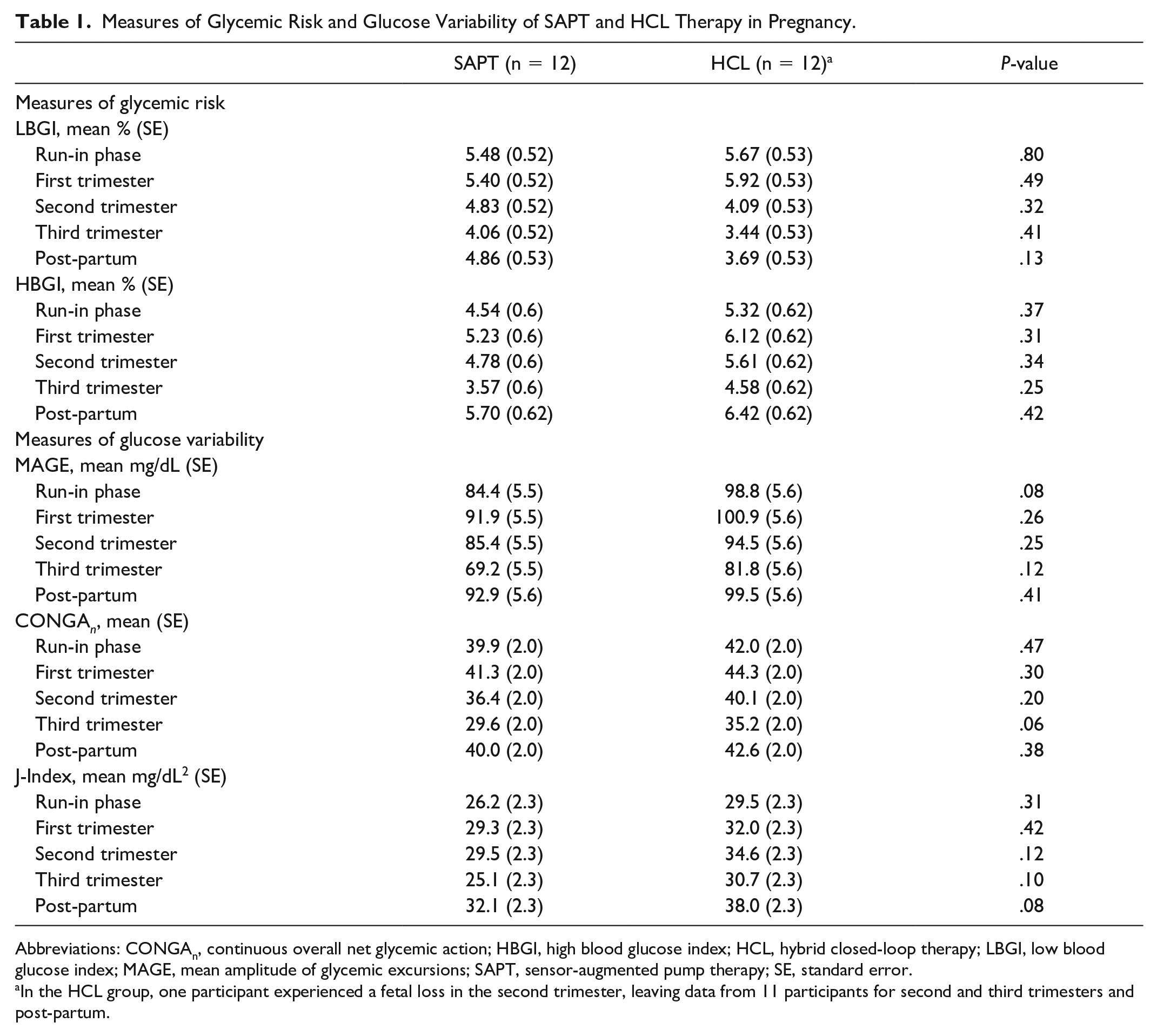
Abbreviations: CONGAn, continuous overall net glycemic action; HBGI, high blood glucose index; HCL, hybrid closed-loop therapy; LBGI, low blood glucose index; MAGE, mean amplitude of glycemic excursions; SAPT, sensor-augmented pump therapy; SE, standard error.
In the HCL group, one participant experienced a fetal loss in the second trimester, leaving data from 11 participants for second and third trimesters and post-partum.
Conclusions
In this pilot study, the fact that glycemic variability of HCL wear in gestation with a system that has a higher-than-recommended glucose target for pregnancy (120 mg/dL study device vs 95 mg/dL fasting glucose target in pregnancy) 6 was not worse than that seen for the same pump and CGM being used without automation (SAPT) is reassuring. However, results should be interpreted with caution given the small sample size. In addition, it is difficult to know if the small clinical differences (that were not statistically significantly different) between groups may be important in terms of adverse pregnancy outcomes. In summary, in the PICLS study, the use of the first HCL system with proper oversight and assistive techniques was not associated with significant differences in secondary metrics of glycemia compared to SAPT.
Footnotes
Acknowledgements
The authors thank the study participants. The authors are appreciative of the funding support from the Juvenile Diabetes Research Foundation, the University of Colorado, and the Children’s Diabetes Foundation. Study data were collected and managed using REDCap electronic data-capture tools hosted at the University of Colorado Anschutz Medical Campus. The authors thank the JAEB Center for administrative support and independent site monitoring. The authors thank the Drug Safety and Monitoring Board for oversight of the study. The authors would also like to thank additional staff at the BDC for their assistance with the PICLS study (Christie Beatson, Tanner Bloks, Jamie Demmitt, Ashleigh Downs, Victoria Gage, Matthew Klein, Emily Malecha, Amanda Rewers, Lisa Sher, and Tim Vigers) and the administrative support staff for both study sites (Jessica Cohen, Kayla McDaniel, and Sandy Nicholls).
Abbreviations
CGM, continuous glucose monitor; CONGA, continuous overall net glycemic action; FDA, Food and Drug Administration; GMI, glucose management indicator; HBGI, high blood glucose index; HCL, hybrid closed-loop; LBGI, low blood glucose index; MAGE, mean amplitude of glucose excursions; SE, standard error.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SP discloses research funding from Dexcom Inc, Eli Lilly, JDRF, Leona & Harry Helmsley Charitable Trust, NIDDK, and Sanofi US Services; research support from Diasome Pharmaceuticals Inc, MiniMed Inc, Sanofi US Services; contributing writer at diatribe; was on the medical advisory board of Medtronic MiniMed Inc (2020). KD discloses receiving research support from Dexcom, Abbott, Insulet, Sanofi, and Viacyte; consulting from Eli Lilly, Dexcom, Insulet, and Oppenheimer; royalties from UpToDate; and honoraria from the Academy for Continued Health Care Learning, Med Learning Group, Medscape, and Elsevier. EB discloses receiving research support from Dexcom, JDRF, and Helmsley Charitable Trust. JK, RG, LP, AB, CJ, EN, and JS-B report no conflicts.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This was an investigator-initiated study supported by JDRF through the Board of Regents at the University of Colorado Denver. Medtronic MiniMed, Inc. provided a discount rate to the study group for the purchase of Medtronic 670G pumps, Guardian sensors, and Guardian transmitters. The Children’s Diabetes Foundation supported the purchase of some study devices for participants through the Helping Little Hands program. Central laboratory testing and nutritional analyses were sponsored by a CCTSI Microgrant through the University of Colorado Denver. This publication was supported by NIH/NCATS Colorado CTSA Grant Number UL1 TR002535. The content of this publication is the authors’ sole responsibility and does not necessarily represent official views of JDRF, Medtronic MiniMed, Inc, Children’s Diabetes Foundation, University of Colorado Denver, or NIH.
