Abstract
Aim:
Insulin pump, continuous glucose monitoring (CGM), and sensor augmented pump (SAP) technology have evolved continuously leading to the development of automated insulin delivery (AID) systems. Evaluation of the use of diabetes technologies in people with T1D from January 2018 to December 2021.
Methods:
A patient registry (Diabetes Prospective Follow-up Database [DPV]) was analyzed for use of SAP (insulin pump + CGM ≥90 days, no automated dose adjustment) and AID (HCL or LGS/PLGS). In total 46,043 people with T1D aged 0.5 to <26 years treated in 416 diabetes centers (Germany, Austria, Luxemburg, and Switzerland) were included and stratified into 4 groups A-D according to age. Additionally, TiR and HbA1c were analyzed.
Results:
From 2018 to 2021, there was a significant increase from 28.7% to 32.9% (sensor augmented pump [SAP]) and 3.5% to 16.6% (AID) across all age groups, with the most frequent use in group A (<7 years, 38.8%-40.2% and 10.3%-28.5%). A similar increase in SAP and AID use was observed in groups B (7 to <11 years) and C (11 to <16 years): B: +15.8 PP, C: +15.9 PP. HbA1c improved significantly in groups C and D (16 to <26 years) (both P < .01). Time in range (TiR) increased in all groups (A: +3 PP; B: +5 PP; C: +5 PP; D: +5 PP; P < 0.01 for each group). Insulin pumps (61.0% versus 53.4% male) and SAP (33.5% versus 28.9% male) are used more frequently in females.
Conclusion:
In recent years, we found an increasing use of new diabetes technologies and an improvement in metabolic control (TiR) across all age groups.
Keywords
Introduction
Type 1 diabetes (T1D) is an autoimmune disorder that frequently manifests in childhood and adolescence. The goal of every diabetes management is to achieve and maintain targeted glycemia. As recommended by the American Diabetes Association/International Society for Pediatric and Adolescent Diabetes, a glucose target range of 3.8 to 10 mmol/L (70–180 mg/dL) most of the time (≥70%) or an HbA1c ≤7% (53 mmol/mol) should be achieved. Another goal is to avoid both hypoglycemia (<3.8 mmol/L or <70 mg/dL) and hyperglycemia (>10 mmol/L or >180 mg/dL).1,2 People with T1D struggle with the everyday burden of glucose self-monitoring, insulin dosing and administration based on self-measured glucose values, as well as carbohydrate counting before meals. In addition, people with T1D must keep in mind the effects of physical activity on insulin requirements. During different stages of life and development, diabetes management is complicated by age-specific psychological and physiological conditions. 3 Hypoglycemia is a potentially life-threatening complication that limits optimal diabetes control. Thus, automatic algorithms for insulin dosage integrated in insulin pumps (CSII) combined with glucose sensors offer the potential to improve metabolic control. Insulin pumps with low-glucose suspend (LGS) or predictive low-glucose suspend (PLGS) technology are used successfully in many people by stopping insulin delivery based on a low measured or predicted glucose value. 4
CSII and continuous glucose monitoring (CGM) technology has been further developed over the last years and has led to the development of hybrid closed-loop (HCL) insulin delivery systems. Hybrid closed loop or automated insulin delivery (AID) uses a control algorithm that autonomously and continuously adapts subcutaneous basal insulin delivery based on current sensor glucose levels and a predictive calculation of the glucose curve. For carbohydrate intake, bolus insulin must be added manually. 5 Beside these commercial systems do-it-yourself Artificial Pancreas Systems (DIY-APS) are used as an alternative to CSII/HCL systems even in the younger age groups. Do-it-yourself APS is a type of HCL system that uses open-source algorithms. Recent studies have shown that these non-commercial HCL systems can improve diabetes management in children and adults with T1D. 6
The aim of this study was to investigate the use of modern diabetes technologies (SAP and AID) in different age groups with T1D from 2018 to 2021 using a large patient registry from Germany, Austria, Switzerland, and Luxembourg.
Research Design, Methods/Statistical Analysis
DPV Registry
The DPV registry is a prospective multicenter initiative that records demographic and anthropometric characteristics as well as data on diabetes management and diabetes-related comorbidities among children and adults with any type of diabetes. Currently, 515 centers in Germany, Austria, Switzerland, and Luxembourg participate in the DPV initiative using a dedicated electronic health record (DPV software). Twice a year, each center transmits data in pseudonymized form to the University of Ulm for central data aggregation and analysis. Inconsistent data are reported back to the centers for verification or correction. Until December 2021, 158,866 people with T1D were included in the registry. The ethics committee of the University of Ulm, Germany, and local review boards of the participating centers approved the data collection and the analysis of anonymized data from the DPV registry.
Study Population
People living in Germany, Austria, Luxembourg, or Switzerland were eligible for inclusion in the study if they were clinically diagnosed with T1D and their diabetes was being treated with insulin. To rule out neonatal diabetes forms, an age of <6 months at diagnosis was specified as an exclusion criterion. The inclusion criterion was therefore an age of ≥0.5 up to <26 years.
Age was categorized into 4 groups: group A (preschoolers): 0.5 to <7 years, group B (prepubertal): 7 to <11 years, group C (pubertal): 11 to <16 years, and group D (post-pubertal): 16 to <26 years.
Methods/Statistical Analysis
Anthropometric and clinical data were aggregated per subject for each year from 2018 to 2021. Parameters included median age, diabetes duration, sex, body mass index (calculated as weight in kg divided by height in m2), SD score (BMI-SDS), type of insulin therapy, CSII, and SAP including CSII + CGM ≥90 days per year or AID including HCL. The definition for HCL is an insulin pump plus CGM use for 90 days or even longer controlled by an algorithm that can both increase or decrease insulin delivery. The definition for AID is the use of any algorithm which adjusts insulin dose, therefore encomprising both HCL as well as PLGS/LGS. Therefore, HCL is a subgroup of AID. Sensor augmented pump includes patients with insulin pump plus CGM use for at least 90 days of use, but no AID patients. The LGS/PLGS is defined as a subgroup of AID. If several forms of therapy were documented for 1 participant in 1 year (therapy change), the most recent form of therapy was used. In a cross-sectional analysis the use of pump therapy, CGM, AID, and SAP was analyzed per year between 2018 and 2021 in the 4 different age groups.
Parameters as HbA1c (% and mmol/mol), TiR (glucose range of 3.9-10 mmol/L (70-180 mg/dL)), TbR (glucose values below 3.9 mmol/L (70 mg/dL)), and TaR (glucose values above 10 mmol/L (180 mg/dL)) were analyzed. Body mass index values were transformed to SD scores by applying the LMS method. 7 HbA1c values were mathematically standardized to the diabetes control and complications trial (DCCT) reference range (4.05%-6.05%) using the multiple-of-the-mean transformation method to adjust for differences between laboratories.
Results are presented as proportion (categorical values) or median with interquartile range (continuous variables). The Wilcoxon test was used to compare continuous variables, the χ2 test for variables with a binomial distribution. Adjustment for multiple comparisons was performed according to the Holm-Bonferroni step-down procedure.
The trend analysis was performed using a linear regression model adjusted for gender and diabetes duration and stratified by age group. For the sensitivity analysis, we also evaluated results from a linear time trend analysis for HbA1c and TiR with additional adjustment for baseline HbA1c.
All analyses were performed using SAS version 9.4 build TS1M7 on a Windows mainframe computer (SAS Institute). Due to the large sample size, a 2-sided P-value <.01 was considered statistically significant.
Results
Characteristics of the study population are shown in Table 1.
Characteristics of the Study Population Between 2018 and 2021.
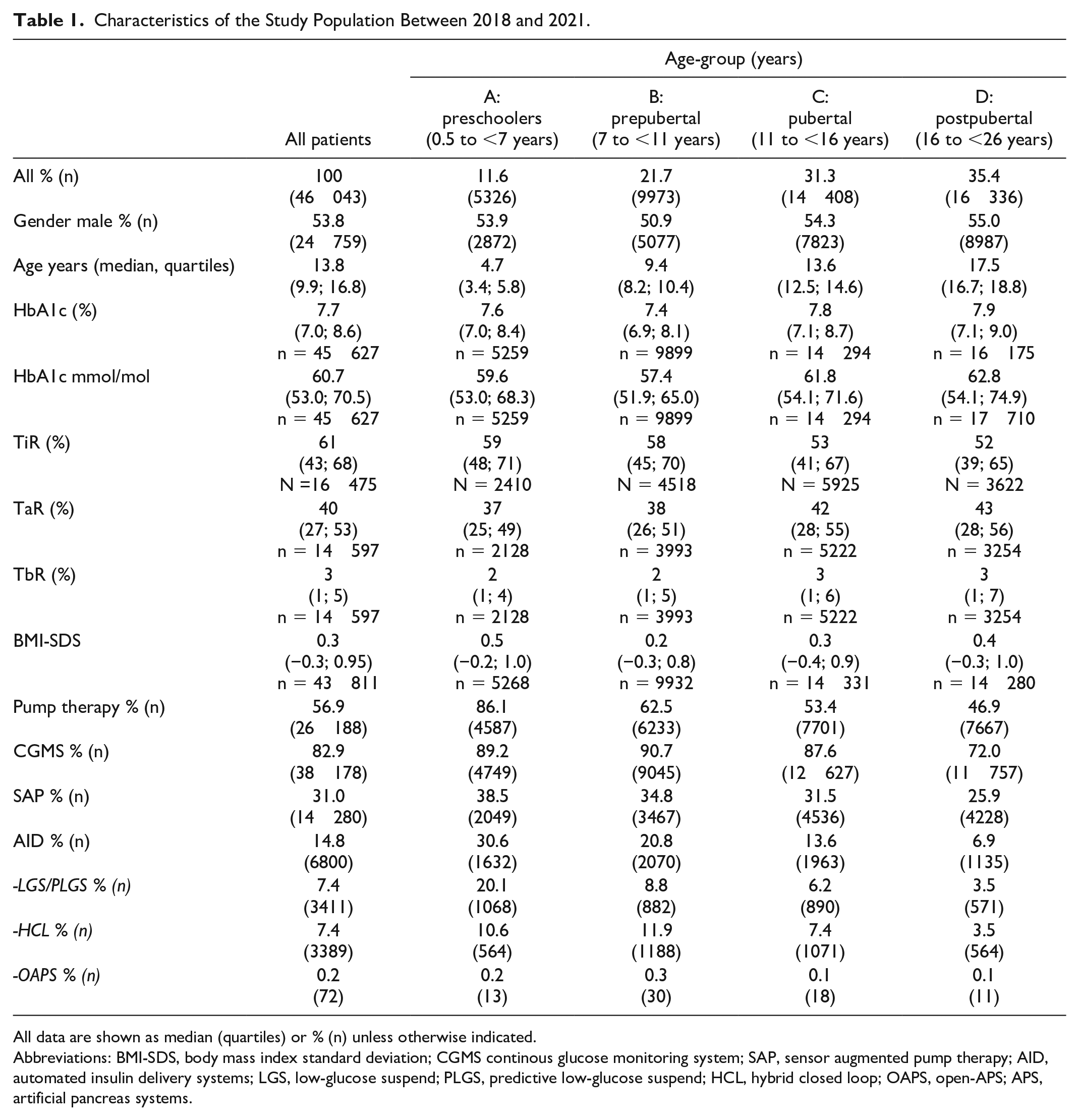
All data are shown as median (quartiles) or % (n) unless otherwise indicated.
Abbreviations: BMI-SDS, body mass index standard deviation; CGMS continous glucose monitoring system; SAP, sensor augmented pump therapy; AID, automated insulin delivery systems; LGS, low-glucose suspend; PLGS, predictive low-glucose suspend; HCL, hybrid closed loop; OAPS, open-APS; APS, artificial pancreas systems.
Data about time in various ranges (TiR, TbR, and TaR) are only available for a portion of participants (see Table 1).
Use of Insulin Pump, CGM, AID and SAP in 2018 to 2021
Technology use increased over time in the entire group (Figure 1) as well as in the 4 age groups (Figure 2).
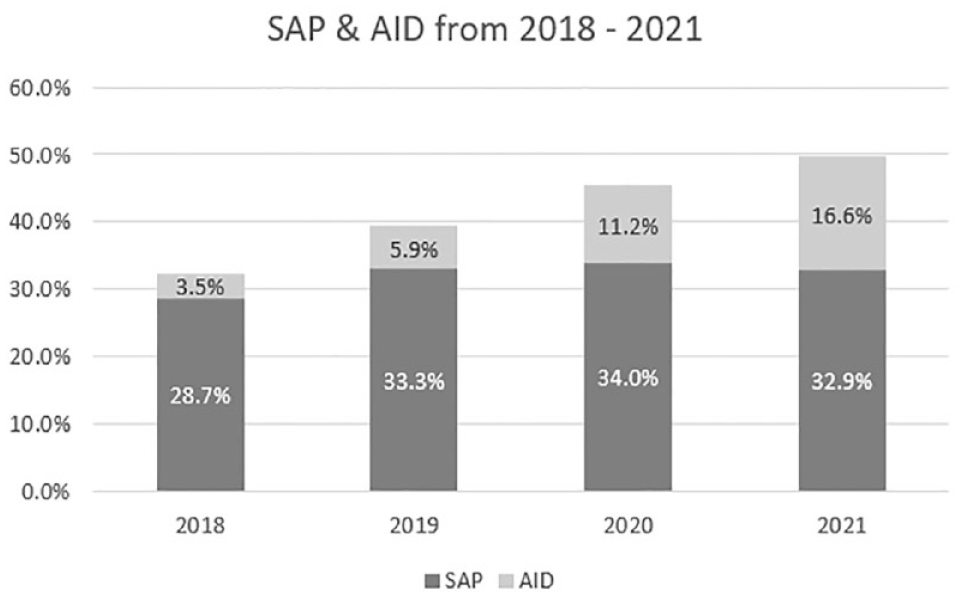
Use of SAP and AID in the entire study population in recent years. Shown are the percentages of participants with T1D using sensor augmented pump therapy or automated insulin delivery systems between 2018 and 2021.
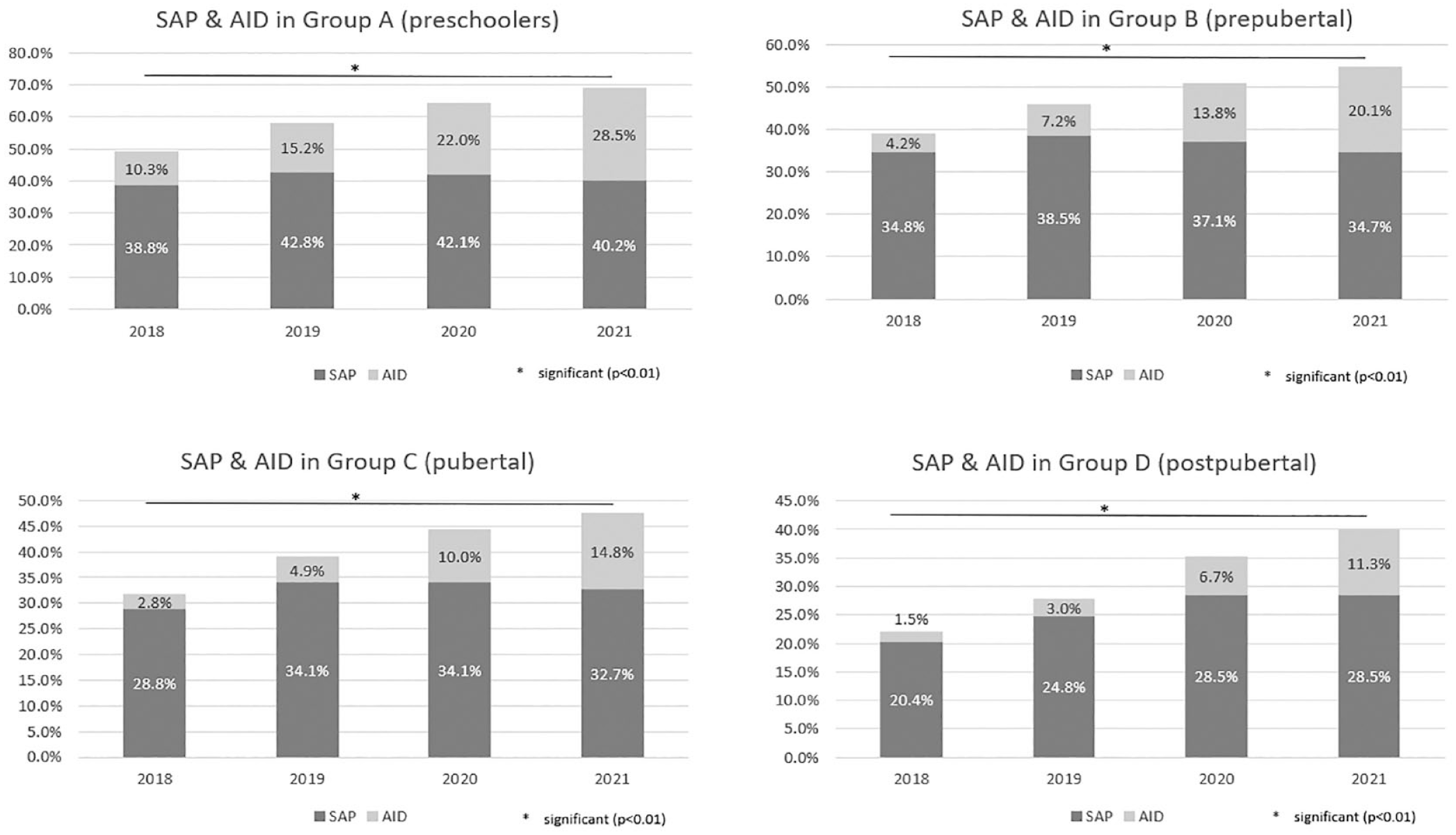
Use of SAP and AID in different age groups from 2018 to 2021. Shown are the percentages of participants with T1D in 4 different age groups using sensor augmented pump therapy or automated insulin delivery systems between 2018 and 2021. Groups: A (preschoolers: 0.5 to <7 years); B (prepubertal): 7 to <11 years; C (pubertal): 11 to <16 years); D (postpubertal): 16 to <26 years). P-value: regression model for trend analysis stratified by age group over the years.
There is a significant difference in the use of SAP and AID technology between the different age groups (P < .01) for the whole observational period.
The highest use of insulin pump (86.1%), SAP (38.5%), and AID (30.6%) is seen in children <7 years of age. The highest CGM use is seen in children 7 to 11 years of age (90.7%) All technologies are used in the younger age groups more frequently (these data are shown in Table 1).
Insulin pumps are used more frequently in female people (61.0% versus 53.4% male; P < .01) and SAP in 33.5% of the female people versus 28.9% male people; P < .01). While there is no sex difference in AID use. Data about technology use in the 4 different age groups from 2018 to 2021 is shown in Table 2.
Participants in Different Age Groups Using Different Technologies, Shown are the Percentages (Numbers) Per Year From 2018-2021.
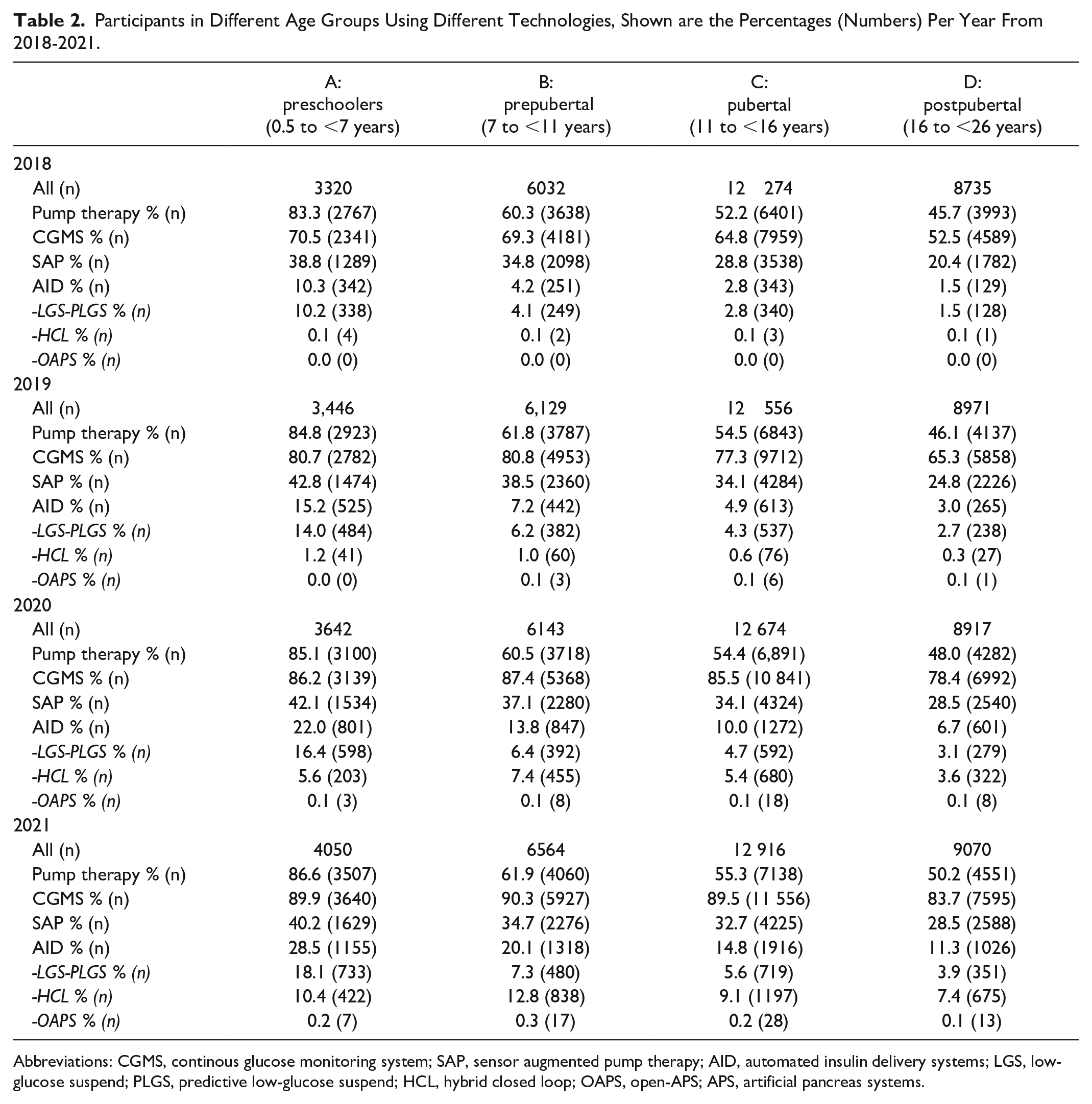
Abbreviations: CGMS, continous glucose monitoring system; SAP, sensor augmented pump therapy; AID, automated insulin delivery systems; LGS, low-glucose suspend; PLGS, predictive low-glucose suspend; HCL, hybrid closed loop; OAPS, open-APS; APS, artificial pancreas systems.
HbA1c, TiR in Different Age Groups From 2018 to 2021
HbA1c change for all groups during the observation period is shown in Figure 3. A significant decrease over the whole period is seen in group C (11 to <16 years) and group D (16 to <26 years) (P < .01). In children < 7 years, there is an increase in HbA1c from 2018 to 2020 (P < .01), then HbA1c remains stable in 2021. In children 7 to <11 years there is an increase in HbA1c from 2018 to 2020 with a reduction in 2021 (P < .01).
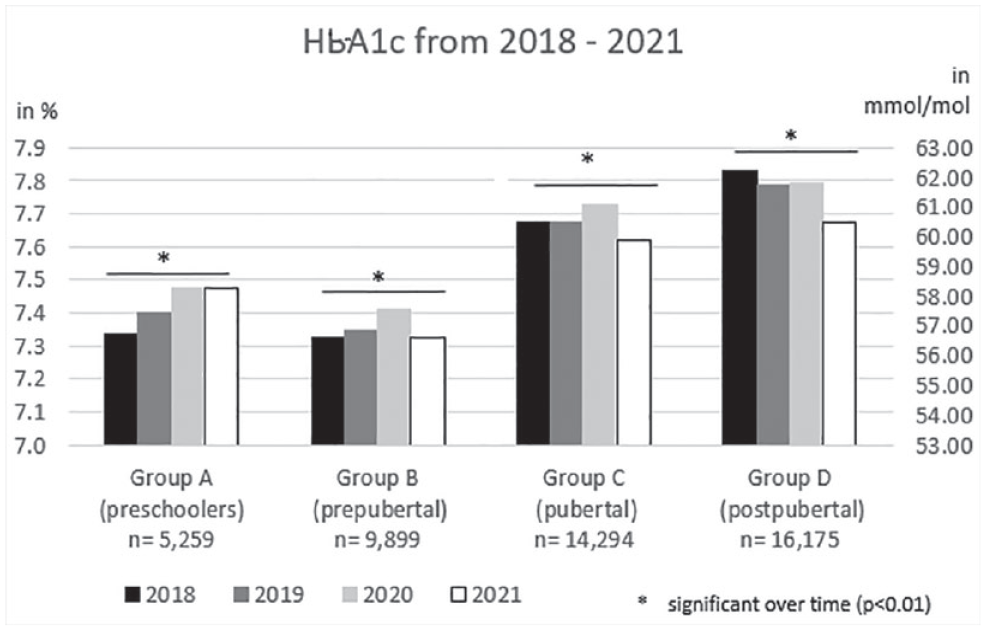
Metabolic control (HbA1c) in different age groups from 2018-2021. Shown are the median HbA1c values in % and in mmol/mol in 4 different age groups between 2018-2021 Groups: A (preschoolers: 0.5-<7 years); B (prepubertal): 7-<11 years; C (pubertal): 11-<16 years); D (postpubertal): 16-<26 years). P-value: regression model for trend analysis stratified by age group over the years.
The percentage of TiR over time is shown in Figure 4. There is an increase in children < 7 years from 56% to 59%; in children 7 to <11 years from 55% to 60%; in children 11 to <16 years from 50% to 55% and in the age-group 16 to <26 years from 48% to 53% (P < .01).
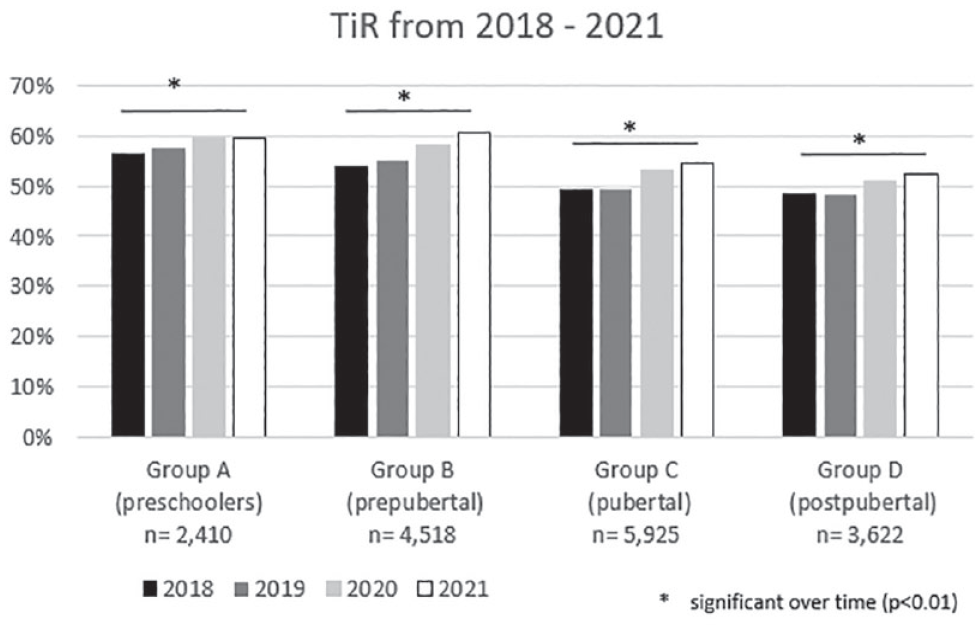
Metabolic control (TiR) in different age groups from 2018 to 2021. Shown are the percentages of TiR (median) in 4 different age groups between 2018-2021. Groups: A (preschoolers: 0.5-<7 years); B (prepubertal): 7-<11 years; C (pubertal): 11-<16 years); D (postpubertal): 16-<26 years). P-value: regression model for trend analysis stratified by age group over the years. Abbreviation: TiR, time in range.
The time trend analysis for HbA1c and time in range (TiR) with adjustment for baseline HbA1c came to the same results, except for an improvement in HbA1c over the whole time period for groups A and B (P < .01).
Discussion
In this study on the use of modern diabetes technologies (AID and SAP), we identified a steady increase in the use of these technologies in all people and in the 4 different age groups from 2018 to 2021, with the highest use in the youngest age group. During the observed period, the number of people using these technologies increased from every third to every second participant.
Over the last years, technologies such as CSII and CGM for the management of T1D at any age have increased worldwide, and open-label studies indicate a positive association with metabolic control and decreased rates of both diabetic ketoacidosis and severe hypoglycemia.8 -14 Insulin pump therapy plus CGM is moving in the direction of AID consisting of an insulin pump, a continuous glucose sensor and algorithms that control insulin delivery. During recent years, developments in T1D technologies like SAP and AID have altered the management of people with T1D.15 -17 Several studies have shown a positive impact of this closed-loop technology on quality of life. Various psychosocial benefits were reported by users. 18 The new diabetes technologies can help people of any age to cope with the everyday burden and distress associated with T1D. 19 AID technology includes (commercial and do-it-yourself) HCL systems as well as CSII with LGS/PLGS. However, 1 limitation of some of the current commercially available closed-loop systems for the age group below 7/6 years is the minimum total daily insulin dose that is required for optimal system performance. Additionally, in 2018 to 2021 all commercially available HCL systems were only approved for people at a certain age and older. This may be 2 reasons why parents of these children and their healthcare professionals (HCPs) choose insulin pumps with LGS/PLGS technology. On the other hand, the oldest group might use AID to a lesser degree as T1D management is regarded as difficult in this age group. People of this age want to make little effort to manage their T1D and be disturbed as little as possible in their daily lives.
A key finding of this study is that we were able to identify a significant gender difference in the use of SAP with females using SAP more frequently than their male counterparts. This difference may be due to a higher CSII use in females.20 -22 One reason for the higher use of CSII/SAP in females could be the higher HbA1c, which is an indication for starting CSII or SAP. 23 A second reason to start CSII or SAP is the availability of different basal rates that can be set on days with different insulin requirements (eg, pre-menstruation, pregnancy, preconception). 24
Insulin pumps using PLGS discontinue insulin delivery when hypoglycemia is predicted by an algorithm. In several studies, the use of PLGS technology reduced the time spent in the hypoglycemic range. These benefits could be achieved at the expense of slightly elevated overnight and fasting glucose levels. Automated insulin delivery therapy is associated with an increased percentage of TiR and reduced TaR and TbR while modestly reducing HbA1c levels in children, adolescents, and adults. TiR is being validated as a new clinical marker to additionally assess the quality of metabolic control. TiR is being validated as a new clinical marker to additionally assess the quality of metabolic control Clinical guidance on TiR targets and data visualization was endorsed by professionals and is now being implemented in daily practice and explained to all persons with T1D. 25 With increasing use of CGM technology during recent years, limitations of HbA1c, which has been the gold standard for assessing glycemic control, have received more attention. HbA1c primarily mirrors hyperglycemia and does not provide information about hypoglycemia, glycemic variability or even the daily glucose pattern.26,27 In our study, HbA1c showed a significant improvement over the observational period only in the pubertal and post-pubertal age groups. In the very young group, we found an increase in HbA1c which, however, started from a low HbA1c. Additionally, diabetes management in the younger age group is mainly in the hands of their parents and care givers and with CGM imminent hypoglycemia could be detected and prevented in advance and this may lead to a higher HbA1c in this age group.
In the pre-pubertal group, an increase in HbA1c was observed until 2020, followed by a significant improvement in 2021. A significant increase in TiR was observed in all age groups. One explanation for this could be the that during the corona lockdown, diabetes care has returned mainly in the hands of parents and more parental supervision leads to target metabolic control.
One strength of our study is its large database with people from many diabetes care centers throughout 4 countries, as well as the prospective documentation of diabetes treatment and outcome. The DPV registry includes an estimated 90% of pediatric people with diabetes in Germany, while the nationwide capture rate of adults with T1D is lower. Moreover, detailed information on demographics and clinical characteristics was available, allowing a comprehensive control of potential confounders.
One limitation of this study is that complete data were not available for each participant, and variability in how clinical characteristics were measured cannot be completely excluded due to the multicenter design. For example, metabolic parameters like HbA1c, TiR, TbR, and TaR were not available for all people in the study population and therefore a clear comparison of these parameters is not possible.
To counteract, we standardized locally measured HbA1c values to the DCCT standard. Furthermore, because of the cross-sectional design, this study does not allow us to draw any conclusions on causality. Finally, the nature of the database does not allow an in-depth analysis of all important determinants (eg, individual socio-economic data).
Conclusion
During recent years, there has been an increase in the overall use of new diabetes technologies (SAP and AID); the increase was more pronounced in the younger age groups. A significant difference in the use of SAP technology between females and males was demonstrated and can be attributed to a higher CSII use among females. In the same time period, we report an improvement in metabolic control (HbA1c, TiR) in the older age groups.
Footnotes
Acknowledgements
Special thanks to A. Hungele and R. Ranz for DPV documentation software support (both clinical data managers, University of Ulm).
The authors are grateful to all participating DPV centers for contributing anonymized data to this study.
Abbreviations
AID, automated insulin delivery systems; BMI, body mass index; BMI-SDS, body mass index standard deviation; CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; DCCT, diabetes control and complications trial; DIY APS, “do-it-yourself” artificial pancreas systems; DPV, diabetes prospective follow-up database; HbA1c, glycated hemoglobin; HCL, hybrid closed loop; HCP, healthcare professionals; LGS, low glucose suspend; OAPS, open-APS; PLGS, predictive low-glucose suspend; PP, percentage points; SAP, sensor augmented pump therapy; T1D, type 1 diabetes; TaR, time above range; TbR, time below range; TiR, time in range.
Authors’ Contributions
All named authors met the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article. LvdB drafted the manuscript, LvdB and BG wrote the manuscript, RWH designed the analysis; he is the principal coordinator of the DPV registry. MA developed the statistical analysis plan, critically discussed this with the authors, analyzed all data, wrote the statistics section and part of the results section, and critically reviewed the entire manuscript. MA is the guarantor for this study. All authors critically reviewed and commented on the article and contributed to the interpretation and approved the final version for submission.
The following centers have contributed to the German/Austrian Diabetes Patient Follow-up (DPV) registry analysis: Aachen—Innere RWTH; Aachen—Uni-Kinderklinik RWTH; Aalen Kinderklinik; Ahlen St. Franziskus Kinderklinik; Aidlingen Praxisgemeinschaft; Altötting Kinderklinik Zentrum Inn-Salzach; Altötting-Burghausen Innere Medizin; Amberg Kinderklinik St. Marien; Amstetten Klinikum Mostviertel Kinderklinik; Arnsberg-Hüsten Karolinenhosp. Kinderabteilung; Asbach Kamillus-Klinik Innere; Aue Helios Kinderklink; Augsburg IV. Med. Uni-Klinik; Augsburg Josefinum Kinderklinik; Augsburg Uni-Kinderklinik; Aurich Kinderklinik; Bad Aibling Internist. Praxis; Bad Driburg / Bad Hermannsborn Innere; Bad Hersfeld Innere; Bad Hersfeld Kinderklinik; Bad Kreuznach Diakonie Kikli; Bad Kreuznach-Viktoriastift; Bad Kösen Median Kinderklinik; Bad Lauterberg Diabeteszentrum Innere; Bad Mergentheim—Diabetesfachklinik; Bad Mergentheim—Kinderdiabetologische Praxis; Bad Oeynhausen Herz- und Diabeteszentrum NRW; Bad Orb Spessart Klinik; Bad Orb Spessart Klinik Reha; Bad Reichenhall Kreisklinik Innere Med.; Bad Salzungen Kinderklinik; Bad Säckingen Hochrheinklinik Innere; Bad Waldsee Kinderarztpraxis; Basel Uni-Kinderspital beider Basel (UKBB); Bautzen Oberlausitz KK; Bayreuth Innere Medizin; Bensheim Heilig Geist Innere; Berchtesgaden CJD; Berchtesgaden CJD-Beruf.REHA; Bergen Gemeinschaftspraxis; Berlin DRK-Kliniken Mitte Innere; Berlin DRK-Kliniken Pädiatrie; Berlin Endokrinologikum; Berlin Evang. Krankenhaus Königin Elisabeth; Berlin Klinik St. Hedwig Innere; Berlin Lichtenberg—Kinderklinik; Berlin Oskar Zieten Krankenhaus Innere; Berlin Parkklinik Weissensee; Berlin Schlosspark-Klinik Innere; Berlin St. Josephskrankenhaus Innere; Berlin Virchow-Kinderklinik; Berlin Vivantes Hellersdorf Innere; Bern Inselspital Kinderklinik; Bern Universitätsklinik für Diabetologie und Endokrinologie; Bielefeld Kinderarztpraxis; Bielefeld Kinderklinik Gilead; Bocholt Kinderklinik; Bochum Universitäts St. Josef; Bochum Universitätskinderklinik St. Josef; Bodnegg—MVZ Wollmarshöhe; Bonn Schwerpunktpraxis; Bonn Uni-Kinderklinik; Bottrop Knappschaftskrankenhaus Innere; Braunschweig Kinderarztpraxis; Bremen—Kinderklinik Nord; Bremen—Mitte Innere; Bremen Zentralkrankenhaus Kinderklinik; Bremerhaven Kinderklinik; Bruchweiler Edelsteinklinik Kinder-Reha; Böblingen Kinderklinik; Castrop-Rauxel Evangelisches Krankenhaus; Castrop-Rauxel Rochus-Hospital; Celle Kinderarztpraxis; Celle Klinik für Kinder- und Jugendmedizin; Chemnitz Kinderklinik; Chemnitz-Hartmannsdorf Innere Medizin—DIAKOMED-1; Coburg Innere Medizin; Coburg Kinderklinik; Coesfeld Kinderklinik; Coesfeld/Dülmen Innere Med.; Darmstadt Innere Medizin; Darmstadt Kinderklinik Prinz. Margaret; Datteln Vestische Kinderklinik; Deggendorf Gemeinschaftspraxis; Deggendorf Medizinische Klinik II; Deggendorf Pädiatrie-Praxis; Delmenhorst JHD Kinderklinik; Dessau amb. Kinderarztzentrum; Detmold Kinderklinik; Dinslaken Kinderklinik; Dornbirn Innere Medizin; Dornbirn Kinderklinik; Dortmund Johannes Hospital; Dortmund Kinderklinik; Dortmund Knappschaftskrankenhaus Innere; Dortmund Medizinische Kliniken Nord; Dortmund-St. Josefshospital Innere; Dortmund-West Innere; Dresden Neustadt Kinderklinik; Dresden Uni-Kinderklinik; Duisburg Evang. und Johanniter Krhs Innere; Duisburg Homberg Helios Rhein-Ruhr Kliniken GmbH; Duisburg Malteser St. Johannes; Duisburg Sana Kinderklinik; Duisburg St. Anna Innere Helios Rhein-Ruhr Kliniken GmbH; Duisburg-Huckingen; Duisburg-St. Johannes Helios; Düren-Birkesdorf Kinderklinik; Düsseldorf Uni-Kinderklinik; Eberswalde Klinikum Barnim Werner Forßmann—Innere; Eckernförde Gem.-Prax; Eisleben Lutherstadt Helios-Klinik; Erfurt Kinderklinik; Erlangen Uni Innere Medizin; Erlangen Uni-Kinderklinik; Essen Diabetes-SPP; Essen Diabetes-Schwerpunktpraxis; Essen Elisabeth Kinderklinik; Essen Kinderarztpraxis; Essen Uni-Kinderklinik; Esslingen Klinik für Kinder und Jugendliche; Eutin Kinderklinik; Feldkirch Kinderklinik; Filderstadt Kinderklinik; Flensburg Diakonissen Kinderklinik; Forchheim Diabeteszentrum SPP; Frankenthal Kinderarztpraxis; Frankfurt Diabeteszentrum Rhein-Main-Erwachsenendiabetologie (Bürgerhospital); Frankfurt Diabeteszentrum Rhein-Main-pädiat. Diabetologie (Clementine-Hospital); Frankfurt Uni-Kinderklinik; Frankfurt Uni-Klinik Innere; Frankfurt Uni-Klinik Innere2; Frankfurt-Höchst, Städtische Kinderklinik; Frankfurt-Sachsenhausen Innere; Frankfurt-Sachsenhausen Innere MVZ; Freiburg Kinder-MVZ; Freiburg St. Josef Kinderklinik; Freiburg Uni Innere; Freiburg Uni-Kinderklinik; Freudenstadt Kinderklinik; Friedberg Innere Klinik; Fulda Kinderklinik; Fürth Kinderklinik; Gaissach Fachklinik der Deutschen Rentenversicherung Bayern Süd; Garmisch-Partenkirchen Kinderklinik; Garmisch-Partenkirchen Klinikum Pädiatrie; Geislingen Klinik Helfenstein Innere; Gelnhausen Kinderklinik; Gelsenkirchen Kinderklinik Marienhospital; Gera Kinderklinik; Gießen Ev. Krankenhaus Mittelhessen; Gießen Uni-Kinderklinik; Graz Uni Innere; Graz Uni-Kinderklinik; Greifswald Uni-Kinderklinik; Gummersbach Oberbergklinikum; Göppingen Innere Medizin; Göppingen Kinderklinik am Eichert; Görlitz Städtische Kinderklinik; Göttingen Uni Gastroenterologie; Göttingen Uni-Kinderklinik; Güstrow Innere; Hachenburg Kinderpraxis; Hagen Kinderklinik; Halberstadt Innere Med. AMEOS Klinik; Halberstadt Kinderklinik AMEOS; Halle Uni-Kinderklinik; Hamburg Altonaer Kinderklinik; Hamburg Endokrinologikum; Hamburg Kinderklinik Wilhelmstift; Hamburg-Nord Kinder-MVZ; Hameln Kinderklinik; Hamm Kinderklinik; Hanau diabetol. Schwerpunktpraxis; Hannover DM-SPP; Hannover Kinderklinik MHH; Hannover Kinderklinik auf der Bult; Haren Kinderarztpraxis; Heide Kinderklinik; Heidelberg St. Josefskrankenhaus; Heidelberg Uni-Kinderklinik; Heidenheim Arztpraxis Allgemeinmed; Heidenheim Kinderklinik; Heilbronn Innere Klinik; Heilbronn Kinderklinik; Herdecke Kinderklinik; Herford Innere Med I; Herford Kinderarztpraxis; Herford Klinikum Kinder & Jugendliche; Heringsdorf Inselklinik; Herne Evan. Krankenhaus Innere; Herten St. Elisabeth Innere Medizin; Hildburghausen Hennebergklinik; Hildesheim Bernward Krks Kinderheilkunde; Hildesheim GmbH—Innere; Hildesheim Kinderarztpraxis; Hildesheim Kinderklinik; Hof Kinderklinik; Hohenmölsen Diabeteszentrum; Homburg Uni-Kinderklinik Saarland; Idar Oberstein Schwerpunktpraxis; Ingolstadt Klinikum Innere; Innsbruck Uni-Kinderklinik; Innsbruck Universitätsklinik Innere; Iserlohn Innere Medizin; Itzehoe Kinderklinik; Jena Kinderarztpraxis; Jena Uni-Kinderklinik; Kaiserslautern Kinderarztpraxis; Kaiserslautern-Westpfalzklinikum Kinderklinik; Kamen Klinikum Westfalen Hellmig Krankenhaus; Kamen MKK—Medizinisches Kompetenzkollegium; Karlsburg Klinik für Diabetes & Stoffwechsel; Karlsruhe Schwerpunktpraxis; Karlsruhe Städtische Kinderklinik; Kassel Klinikum Kinder- und Jugendmedizin; Kaufbeuren Kinderklinik; Kempen Heilig Geist—Innere; Kempen Heilig Geist-KHS—Innere; Kempten Oberallgäu Kinderklinik; Kiel Städtische Kinderklinik; Kiel Universitäts-Kinderklinik; Kirchen DRK Krankenhaus Kinderklinik; Kirchheim-Nürtingen Innere; Klagenfurt Innere Med I; Klagenfurt Kinderklinik; Kleve Innere Medizin; Koblenz Kemperhof 1. Med. Klinik; Koblenz Kemperhof 1. Mediz. Klinik; Koblenz Kinderklinik Kemperhof; Konstanz Innere Klinik; Konstanz Kinderklinik; Krefeld Alexianer Innere; Krefeld Innere Klinik; Krefeld Kinderklinik; Krefeld-Uerdingen St. Josef Innere; Kreischa-Zscheckwitz Klinik Bavaria; Köln Kinderklinik Amsterdamerstrasse; Köln Uni-Kinderklinik; Landau Innere; Landshut Kinderklink; Lappersdorf Kinderarztpraxis; Leer Klinikum—Klinik Kinder & Jugendmedizin; Leipzig Uni-Kinderklinik; Leoben LKH Kinderklinik; Leverkusen Kinderklinik; Lienz BKH Kinderklinik; Lienz BKH Pädiatrie; Lienz Diabetesschwerpunktpraxis für Kinder und Jugendliche; Lilienthal Diabeteszentrum; Limburg Innere Medizin; Lindenfels Luisenkrankenhaus Innere; Lindenfels Luisenkrankenhaus Innere 2; Lindlar DM-Zentrum; Lingen Kinderklinik St. Bonifatius; Linz AKH—2. Med; Linz KUK MedCampus IV Kinderklinik; Linz Krankenhaus Barmherzige Schwestern Kardiologie Abt. Int. II; Linz Krankenhaus der Barmherzigen Schwestern Kinderklinik; Lippstadt Evangelische Kinderklinik; Ludwigsburg Kinderklinik; Ludwigshafen Kinderklinik St. Anna-Stift; Ludwigshafen diabetol. SPP; Luxembourg—Center Hospitalier; Lübeck Uni-Kinderklinik; Lübeck Uni-Klinik Innere Medizin; Lüdenscheid Hilfswerk Kinder & Jugendliche; Lüdenscheid Märkische Kliniken—Kinder & Jugendmedizin; Lünen Klinik am Park; Magdeburg Ki-Klinik St. Marienstift; Magdeburg Städtisches Klinikum Innere; Magdeburg Uni-Kinderklinik; Mainz Uni-Kinderklinik; Manderscheid Rathauspraxis; Mannheim Uni-Kinderklinik; Marburg—UKGM Endokrinologie & Diabetes; Marburg Uni-Kinderklinik; Marktredwitz Innere Medizin; Mechernich Kinderklinik; Meissen Kinderklinik Elblandklinikum; Melk Kinderklinik; Memmingen Internistische Praxis; Memmingen Kinderklinik; Minden Kinderklinik; Moers Kinderklinik; Murnau am Staffelsee—diabetol. SPP; Mutterstadt Kinderarztpraxis; Mödling Kinderklinik; Mönchengladbach Kinderklinik Rheydt Elisabethkrankenhaus; Mühldorf am Inn Kinderarztpraxis; Mühlheim an der Ruhr Evang. Krankenhaus Med. Klin.; München 3. Orden Kinderklinik; München Diabetes-Zentrum Süd; München Kinderarztpraxis diabet. SPP; München Praxiszentrum Saarstrasse; München Schwerpunktpraxis; München von Haunersche Kinderklinik; München-Gauting Kinderarztzentrum; München-Harlaching Kinderklinik; München-Schwabing Kinderklinik; Münster Herz Jesu Innere; Münster Ludgerus-Kliniken GmbH; Münster St. Franziskus Innere Med.; Münster St. Franziskus Kinderklinik; Münster Uni-Kinderklinik; Münster pädiat. Schwerpunktpraxis; Münsterlingen Kinderklinik; Nauen Havellandklinik; Neuburg Kinderklinik; Neumarkt Innere; Neunkirchen Gemeinschaftspraxis Kinderheilkunde; Neunkirchen Innere Medizin; Neunkirchen Marienhausklinik Kohlhof Kinderklinik; Neuruppin Kinderklinik; Neuss Lukas-Krankenhaus Kinderklinik; Neuss Lukaskrankenhaus Kinderklinik; Neuwied Kinderklinik Elisabeth; Neuwied Marienhaus Klinikum St. Elisabeth Innere; Nidda Bad Salzhausen Klinik Rabenstein/Innere-1 Reha; Nürnberg Cnopfsche Kinderklinik; Nürnberg Uniklinik Med. Klinik 4; Nürnberg Uniklinik Zentrum f Neugeb./Kinder & Jugendl.; Oberhausen Innere; Oberhausen Kinderklinik; Oberhausen Kinderpraxis; Oberhausen St. Clemens Hospitale Sterkrade; Oberndorf Gastroenterologische Praxis Schwerpunkt Diabetologie; Oberwart—Burgenländische Krankenanstalten Pädiatrie; Offenbach/Main Innere Medizin; Offenbach/Main Kinderklinik; Offenburg Kinderklinik; Oldenburg Schwerpunktpraxis Pädiatrie; Olpe pädiatrische Gemeinschaftspraxis; Oschersleben MEDIGREIF Bördekrankenhaus; Osnabrück Christliches Kinderhospital; Ottobeuren Kreiskrankenhaus; Oy-Mittelberg Hochgebirgsklinik Kinder-Reha; Paderborn St. Vincenz Kinderklinik; Papenburg Marienkrankenhaus Kinderklinik; Passau Kinderarztpraxis; Passau Kinderklinik; Pforzheim Kinderklinik; Pfullendorf Innere Medizin; Pirmasens Städtisches Krankenhaus Innere; Plauen Vogtlandklinikum; Prenzlau Krankenhaus Innere; Rastatt Kreiskrankenhaus Innere; Ravensburg Kinderklink St. Nikolaus; Regensburg Kinderklinik St. Hedwig; Remscheid Kinderklinik; Rendsburg Kinderklinik; Reutlingen Kinderarztpraxis; Reutlingen Kinderklinik; Reutlingen Klinikum Steinenberg Innere; Reutte Tirol BKH Kinderklinik; Rheine Mathiasspital Kinderklinik; Ried Innkreis Barmherzige Schwestern; Rodalben St. Elisabeth; Rosenheim Innere Medizin; Rosenheim Kinderklinik; Rosenheim Schwerpunktpraxis; Rostock Uni-Kinderklinik; Rostock Universität Innere Medizin; Rotenburg/Wümme Agaplesion Diakonieklinikum Kinderabteilung; Rottweil Gemeinschaftspraxis für Innere Medizin; Rüsselsheim Kinderklinik; Rüsselsheim MVZ; Saaldorf-Surheim Diabetespraxis; Saalfeld Thüringenklinik Kinderklinik; Saarbrücken Kinderklinik Winterberg; Saarlouis Kinderklinik; Salzburg Universität Innere Medizin; Salzburg Universitäts-Kinderklinik; Scheibbs Landesklinikum; Scheidegg Prinzregent Luitpold; Schleswig Heliosklinik Kinderklinik; Schw. Gmünd Stauferklinik Kinderklinik; Schweinfurt Kinderklinik; Schwerin Innere Medizin; Schwerin Kinderklinik; Schwäbisch Hall Diakonie Innere Medizin; Schwäbisch Hall Diakonie Kinderklinik; Siegen Kinderklinik; Singen Hegau Bodensee-Klinikum Kinderklinik; Singen Kinderarztpraxis; Spaichingen Innere; Speyer Diakonissen Stiftungskrankenhaus Pädiatrie; St. Augustin Kinderklinik; St. Johann Tirol Kinderklinik; St. Pölten Universitäts-Kinderklinik; St. Pölten Universitätsklinik Innere; Stade Kinderklinik; Steyr LKH, Kinderklinik; Stockerau Landeskrankenhaus; Stolberg Kinderklinik; Stuttgart Olgahospital Kinderklinik; Suhl Kinderklinik; Sylt Rehaklinik; Tettnang Innere Medizin; Traunstein Kinderklinik; Traunstein diabetol. Schwerpunktpraxis; Trier Kinderklinik der Borromäerinnen; Trostberg Innere; Tübingen Uni-Kinderklinik; Ulm Endokrinologikum; Ulm Endokrinologikum Amedes; Ulm Schwerpunktpraxis Bahnhofsplatz; Ulm Uni Innere Medizin; Ulm Uni-Kinderklinik; Vechta Kinderklinik; Viersen Kinderkrankenhaus St. Nikolaus; Viersen internist. Praxis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LvdB received honoraria for lectures from Medtronic, Insulet and Eli Lilly. The other authors have no disclosures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the German Federal Ministry of Education and Research within the German Center for Diabetes Research (DZD; Grant No. 82DZD14E03), the German Diabetes Association (DDG), and the German Robert Koch Institute (RKI). The funding sources were not involved in the content presented in this article.
