Abstract
Background:
The glycemic characterization of congenital hyperinsulinism (HI), a rare disease causing severe hypoglycemia in childhood, is incomplete. Continuous glucose monitoring (CGM) offers deep glycemic phenotyping to understand disease burden and individualize patient care. Typically, CGM has been restricted to severe HI only, with performance being described in short-term, retrospective studies. We have described CGM-derived phenotyping in a prospective, unselected national cohort providing comprehensive baseline information for future therapeutic trials.
Methods:
Glycemic frequency and trends, point accuracy, and patient experiences were drawn from a prospective, nationwide, observational study of unselected patients with persistent HI using the Dexcom G6 CGM device for 12 months as an additional monitoring tool alongside standard of care self- monitoring blood glucose (SMBG).
Findings:
Among 45 patients with HI, mean age was six years and 53% carried a genetic diagnosis. Data confirmed higher risk of early morning (03:00-07:00 h) hypoglycemia throughout the study period and demonstrated no longitudinal reduction in hypoglycemia with CGM use. Device accuracy was suboptimal; 17 500 glucose levels paired with SMBG demonstrated mean absolute relative difference (MARD) 25% and hypoglycemia detection of 40%. Patient/parent dissatisfaction with CGM was high; 50% of patients discontinued use, citing inaccuracy and pain. However, qualitative feedback was also positive and families reported improved understanding of glycemic patterns to inform changes in behavior to reduce hypoglycemia.
Interpretation:
This comprehensive study provides unbiased insights into glycemic frequency and long-term trends among patients with HI; such data are likely to influence and inform clinical priorities and future therapeutic trials.
Congenital hyperinsulinism (HI) is a rare condition of childhood characterized by excessive and dysregulated secretion of insulin, leading to recurrent and severe hypoglycemia. 1 This hypoketotic hypoglycemia results in a brain damage incidence of 15% to 48%2-4 for patients with HI. Incidence of HI varies by country from 1:50 000 to 1:2500; 5 at least 1:28 000 6 in the United Kingdom, with many more cases lacking a genetic diagnosis. With new genetic variants advancing the etiological basis of disease, 7 there is now a better understanding of both the genetic and pathophysiological basis of congenital HI. 5 However, longitudinal characterization of the patterns and incidence of hypoglycemic episodes in this patient population remains scarce and incomplete, hindering progress in glucose monitoring practices. 8
Medical management to prevent hypoglycemia in HI is complex, with few therapeutic options; 9 many patients experience ongoing hypoglycemia despite optimal medical therapy. Monitoring of glucose to prevent hypoglycemia and resulting brain injury is thus critically important and is recognized as a key challenge for families with HI. 10 Monitoring has traditionally relied on intermittent self-monitoring blood glucose (SMBG) testing with capillary fingerprick samples, 11 but, as the approval of continuous glucose monitoring (CGM) for people living with diabetes, there have been understandable calls from patients, parents, and their advocates for this technology to be made available for those with HI.11,12
Continuous glucose monitoring has suboptimal accuracies in hypoglycemic ranges, confounding their adoption in patients with HI. 13 However, the near-continuous monitoring provides an opportunity for a deeper understanding of glycemic control and dynamics to complement the limited data achieved by intermittent and infrequent fingerprick glucose data.14,15 Continuous glucose monitoring has recently been used for patients with HI as a tool for both group 16 and individual 17 pattern recognition, as well as a behavior change tool to reduce hypoglycemia. 18 However, use of CGM to determine long range cohort incidence, trends, and patterns of hypoglycemia has been limited to restricted analysis of retrospectively collected data from varying devices, with no associated or paired blood glucose confirmatory testing. 8 As CGM is not routinely offered to patients with HI, retrospectively collected data are inherently biased toward those with more severe disease or concentrated in a specific geographical region. Generation of comprehensive, prospectively collected data from an unselected and unbiased cohort is essential to understand the burden of disease, influence patient care, and inform therapeutic trial design.
Recent qualitative research has explored user-experiences of CGM in families with HI, 19 providing an experiential parental perspective. However, results are difficult to generalize as the data set is small and subject to selection bias. There is significant need to investigate the validity of previous findings in larger, less-biased patient groups to aid the design of future clinical trials using CGM in HI.
Objectives
The focus of this study was to characterize deep longitudinal glycemic phenotyping of HI in a nationwide observational cohort through CGM. All patients with HI in the United Kingdom were offered recruitment to this study with the following objectives:
Primary objectives include:
Determine baseline glycemic characteristics of an unselected and comprehensive cohort of patients with HI.
Investigate frequency, patterns, and long-term trends of hypoglycemia in HI to establish a longitudinal phenotype.
Evaluate the experiences and opinions of families with HI using CGM long-term.
Secondary objective include:
Perform accuracy assessments of the Dexcom G6 CGM device in hypoglycemia.
Research Design and Methods
Approval for the study was obtained by the ethics committee of the University of Manchester and the Health Research Authority of the National Health Service (REC reference 07/H1010/88). Adolescents (>12 years of age who wanted to consent) and parents of the younger children (≤12 years of age and those who preferred parental consent) with HI provided written, informed consent for inclusion in the study.
Patients
All patients with persistent HI treated in, or networked with, a British-specialized HI center at study onset were approached for prospective recruitment with the following inclusion criteria:
Diagnosis of persistent hyperinsulinemic hypoglycemia.
Detection of inappropriate insulin secretion or insulin action at the time of hypoglycemia to confirm biochemical basis of hyperinsulinism.
Persistent HI as judged by requirement for persistent therapy, without clinical expectation to reduce therapy intensity over the next 12 months.
Aged under 18 years at the time of study recruitment.
Exclusion criteria were as follows:
Alternative (non-hyperinsulinemic) causes for hypoglycemia.
Unexplained hypoglycemia or hypoglycemia possibly due to HI but without biochemical confirmation.
Syndromic forms of HI such as Turner, Beckwith-Wiedemann, or Kabuki syndrome.
Recruitment
Patients from across Britain were recruited via contact with their regional/supra-regional specialist center (Manchester, Liverpool, Nottingham, Birmingham, London, and Glasgow). As all HI patients in the United Kingdom are at, or networked through, a specialist center (Manchester, Liverpool, and London), this approach identified all eligible cases nationally. Dependent on location, availability, and COVID-19 restrictions at the time of recruitment, patients were either seen in an in-person clinic appointment in the lead center (Manchester) or at a virtual appointment with the research team in Manchester and a specialist nurse practitioner from their regional center. Patients were screened and recruited over a two-month rolling period between December 2021 and January 2022 with a study period of 12 months, finishing in January 2023. If patients were recruited after the initial screening period, their study period still ended in January 2023.
As this study had no precedence, there are no available data regarding change in time spent hypoglycemic over time. As such, power calculations were not undertaken and recruitment targets were pragmatic and aimed to best represent the national stable and persistent HI population.
Study Initiation and Digital Data Collection
A Dexcom G6 receiver and consumables were provided at an in-person appointment or posted to the home address at no cost to patients. Families naive to CGM were offered an additional virtual appointment to explain device setup. Families were shown how to set alarms and adjust parameters. No further, CGM specific, education was provided; the study aim was to characterize baseline glycemic characteristics and dynamics without undue influence from new behaviors formed from expert CGM education. Families were asked to provide CGM data uploads before every sensor change (10 days) via FDA 21 CFR Part 11 compliant secure software. Dexcom Share was not used, consistent with the intention to monitor glycemic trends as close as possible to normal routines, without being influenced by CGM information shared among several family members caring for a patient. Contour Next One glucometers (selected due to high accuracy) 20 were provided for fingerprick blood glucose checking and patients were asked to perform checks twice a day and any time CGM devices reported hypoglycemia, <3.5 mmol/L (63 mg/dL). Data were regularly synchronized to the GlucoContro Website via pseudonymized accounts on a mobile application. Families without computing access were provided device support.
Data Analysis
All data were analyzed using bespoke code developed for this purpose in Python 3.1.1. Hypoglycemia events were defined as per the American Diabetes Association (ADA) criteria for CGM hypoglycemia, requiring three consecutive values below a threshold (chosen as 3.5 mmol/L, as per UK consensus) 21 to trigger both the start and end of an event. 22 Longitudinal trends were analyzed in those contributing ≥90 days of data and, as per ADA guidelines, 22 only for periods where CGM was worn for ≥70% of the time. Trends were analyzed by percentage completion of the study rather than by week of study. This arrangement ensured longitudinal data capture without over-representation of data from the smaller number of individuals still contributing data toward the end of the study.
Accuracy was measured through the comparison of paired CGM and SMBG values. Each SMBG value was paired with the closest CGM value within a 5-minute window either side to allow for point comparisons via metrics such as mean absolute relative difference (MARD) and mean absolute difference (MAD). 13 Hypoglycemia detection rate analyzed all CGM values within a 30 minute window around each SMBG detected hypoglycemia. Finally, paired values were plotted on the hypoglycemia error grid (HEG) to give clinical context to device error. 13 All glycemic data in de-identified form will ultimately be made available in an online repository for use by other research groups.
Questionnaire
Design
Patient and parental perspectives of CGM use in HI were gathered through questionnaire surveys at the end of the study period. Questions were formulated based on themes identified in previous interview studies (with different patient groups)19,23 seeking to validate findings via methodological triangulation. The questionnaire was reviewed by clinicians and specialist nurses in the research team, and wider clinical team, to ensure the questions were acceptable, usable, and minimized bias. The updated and formatted questionnaire was hosted online and piloted with trustees and youth group within “The Children’s Hyperinsulinism Charity” to ensure suitability of language and ease of use.
The questionnaire surveyed the patients’ (if ≥12 years age) and parents’ (in all cases) reasons for starting and, if applicable, stopping CGM; the impact on sleep, fingerprick tests, and anxiety (on 1-5 Likert scale) surrounding hypoglycemic episodes; routine changes; and general positive and negative experiences. In-depth coding and analysis of answers derived from open questions were not undertaken, given previous reports by our group. 19
Dissemination
All participants were eligible to populate a questionnaire, regardless of how long they had remained in the study. This reduced selection bias by including patients both using, and having discontinued, CGM. Ten months after study initiation, families were sent an email with a link to the questionnaire. Initial emails were followed up with reminder emails at two and four weeks and a telephone call at six weeks by the research team. Families unable to access online forms were offered a paper or telephone questionnaire.
Role of the Funding Source
Crinetics Pharmaceuticals provided funding for the study. The study was sponsored by The University of Manchester. CF-C and SB from Crinetics Pharmaceuticals were involved in the design of the study and analysis of data.
Results
CGM Uptake, Demographics, and Persistence
Of 63 patients identified as eligible for inclusion and approached for recruitment, 49 (78%) consented to take part in the study. Of these patients, seven were already using CGM as part of their standard care and agreed to share data. The remaining 42 were not using CGM; devices and training were provided. Of these 42 patients, two withdrew before using CGM, five had used CGM in the past, and 35 were CGM naïve (Figure 1). Two used CGM briefly but did not upload any data. All 46 families of 47 patients (two siblings in one family) who used CGM were sent a link to the online questionnaire and responses were received from 43 (93%).
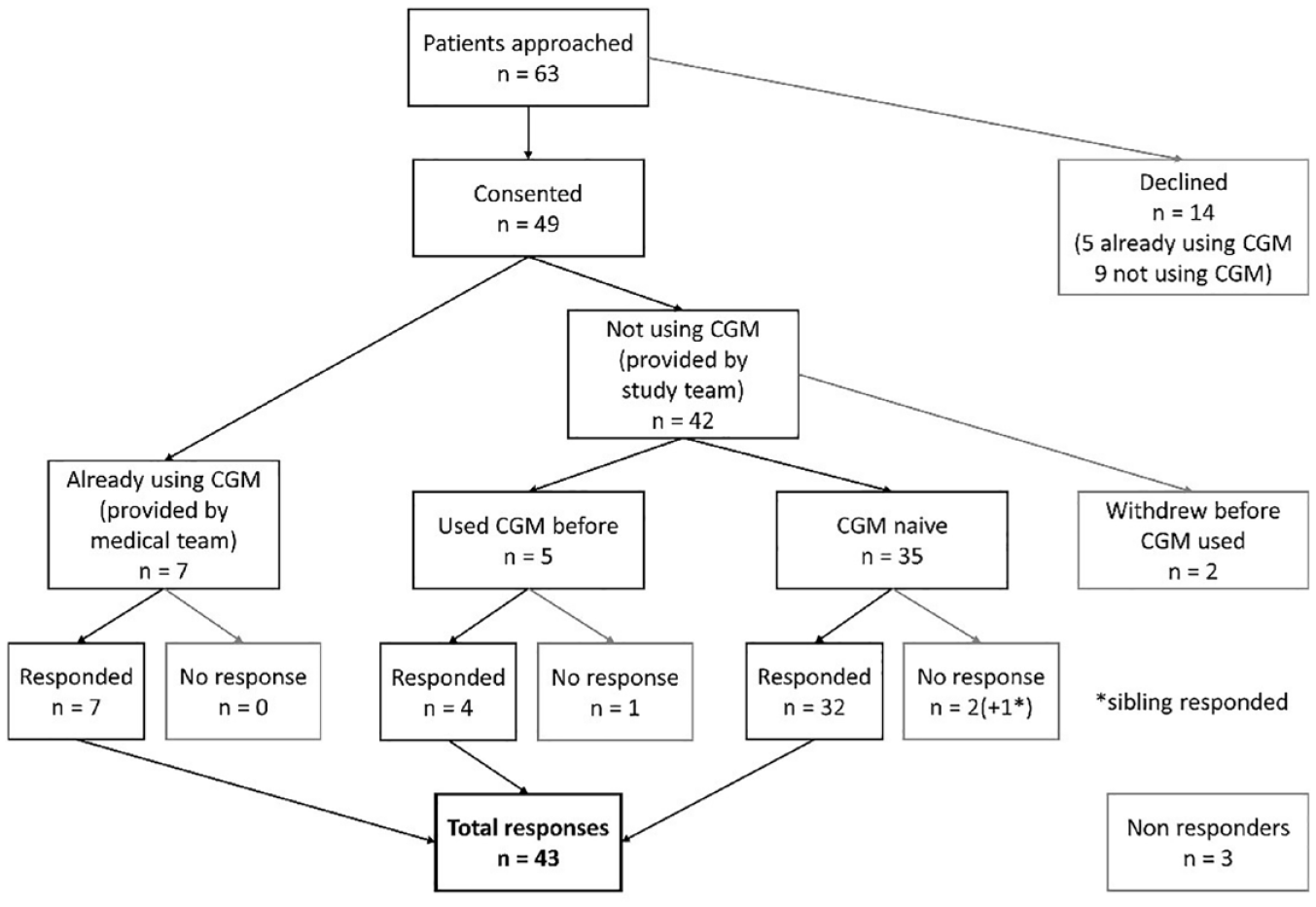
Flow of patients through the study.
Data on demographics, hypoglycemia, and hyperglycemia information are summarized in Table 1.
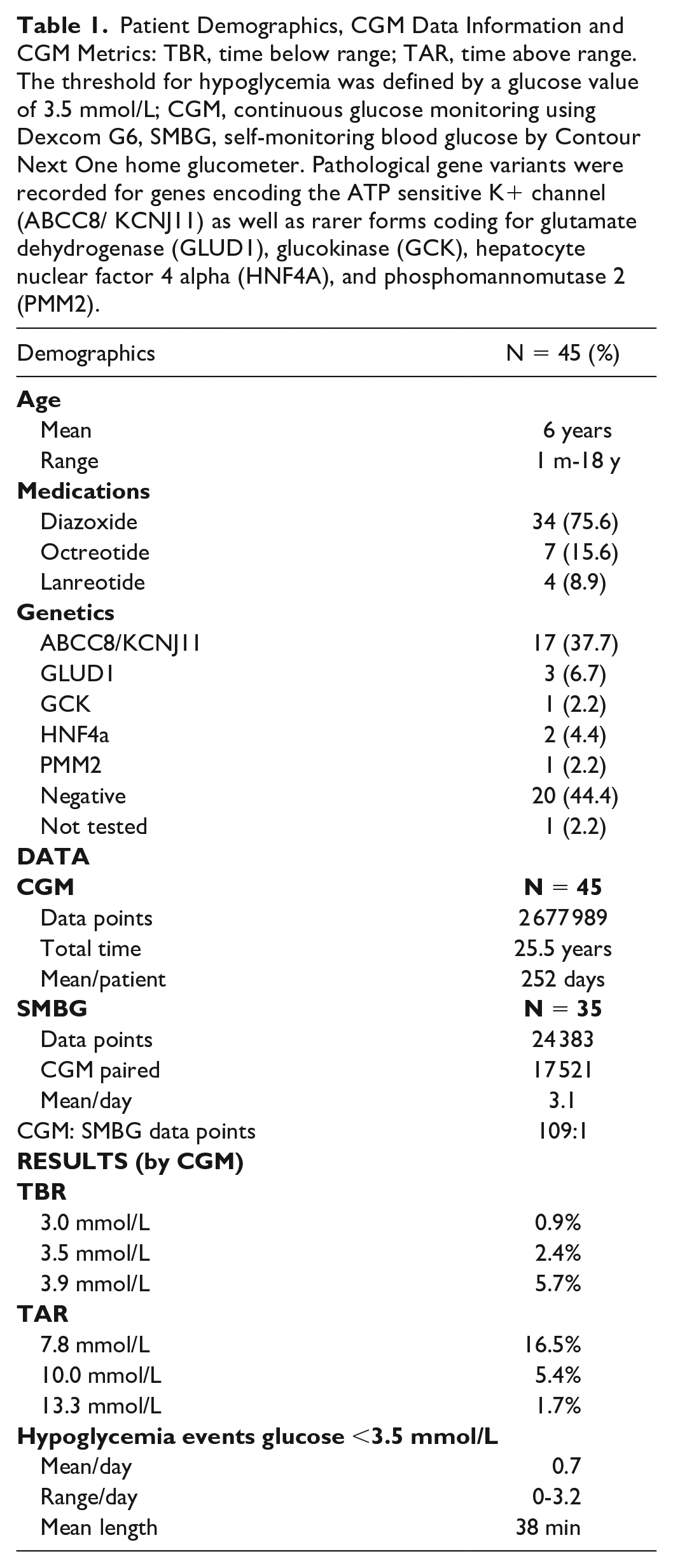
Patient Demographics, CGM Data Information and CGM Metrics: TBR, time below range; TAR, time above range. The threshold for hypoglycemia was defined by a glucose value of 3.5 mmol/L; CGM, continuous glucose monitoring using Dexcom G6, SMBG, self-monitoring blood glucose by Contour Next One home glucometer. Pathological gene variants were recorded for genes encoding the ATP sensitive K+ channel (ABCC8/ KCNJ11) as well as rarer forms coding for glutamate dehydrogenase (GLUD1), glucokinase (GCK), hepatocyte nuclear factor 4 alpha (HNF4A), and phosphomannomutase 2 (PMM2).
As shown in Table 1, patients continued monitoring with CGM for an average of 252 days. There was gradual drop-off in CGM use (Figure 2) with 75% retention at 20 weeks of CGM wear. Many patients (21 of 49; 42%) discontinued use, with 20 of 35 CGM naïve patients (57%) returning CGM devices before the end of the study period (Figure 2). There were no clear commonalities among those discontinuing CGM: discontinuation was not associated with genetic diagnosis, medication type, medication dose, age, or time spent below range.
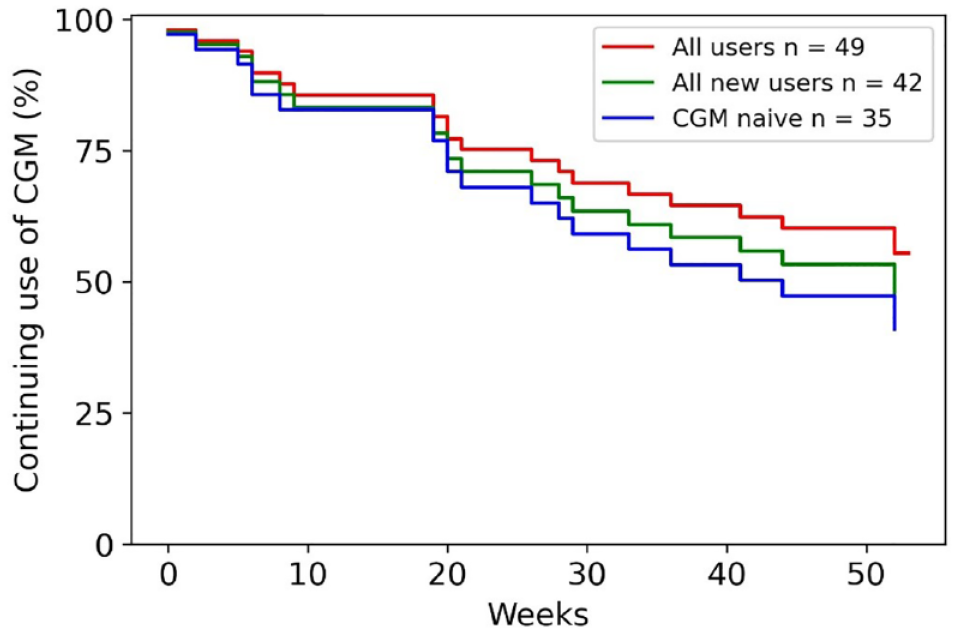
Kaplan-Meier “survival” curve to demonstrate the number of patients still using CGM at the end of the study and discontinuation of CGM as the study progressed. Patients discontinued CGM at different points throughout the 12-month study period. Patients have been categorized into “All users,” “All new users,” and “CGM naïve.” The category “All new users” includes those naïve to CGM and those who had used it in the past but were not using it at the time of study commencement.
Glycemic Phenotype: Frequency and Trends
Percentage time spent hypoglycemic (<3.5 mmol/L) was mean (standard deviation [SD]) 2.4% (2.7) and did not vary by age (P = .2 on Pearson correlation), presence or absence of genetic mutation (P = .6 on t-test), medication type (P = .8 on t-test), or medication dose (P = .38 and P = .69 on Pearson correlation for diazoxide and octreotide, respectively). Multiple regression of all variables confirmed non-significant impact on time hypoglycemic. Patients experienced a mean 0.7 (0.7) hypoglycemia events per day as assessed by CGM, lasting a mean 38 (41) minutes per event.
The frequency of hypoglycemia episodes on CGM fluctuated, with variations observed by time of day. Hypoglycemia was more common in the early hours of the morning, particularly between 04:00 and 06:00 AM (Figure 3), confirming earlier reports8,16 that this period confers particularly high risk to patients with HI. Figure 3 demonstrates a greater than two-fold risk of severe hypoglycemia at 05:00 hours compared to 10:00 hours.
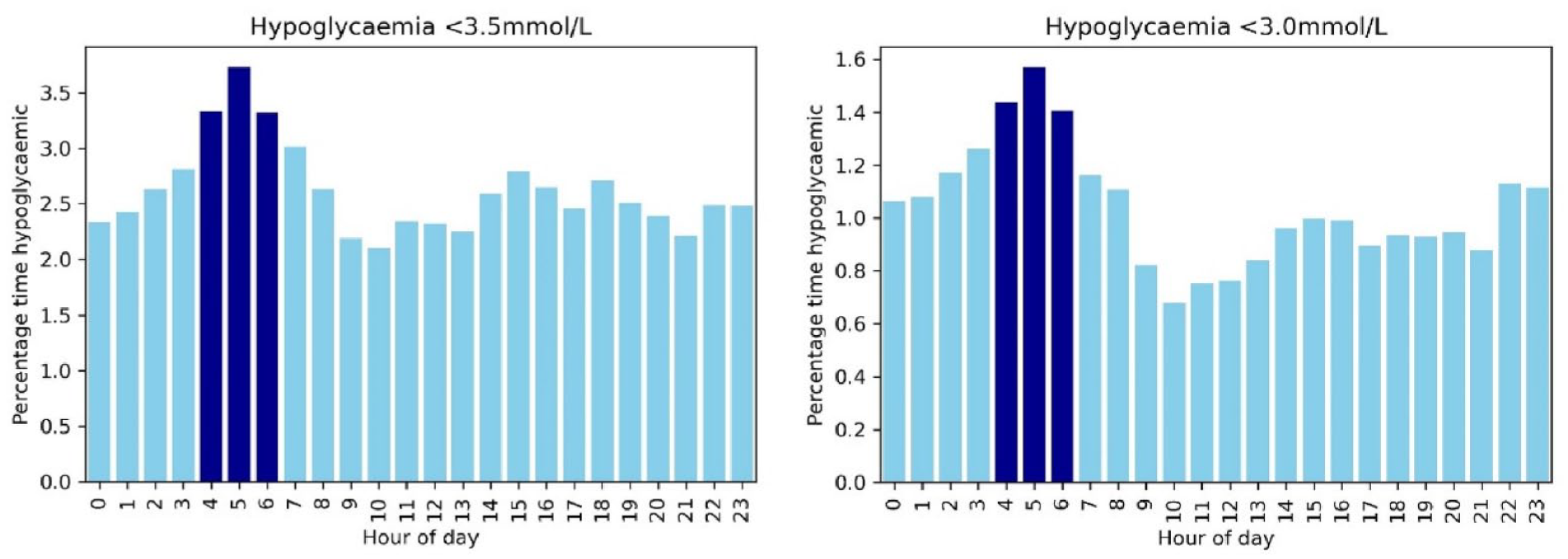
Frequency of hypoglycemia (<3.5 mmol/L) and severe hypoglycemia (<3.0 mmol/L) by time of day based on over 25 years of CGM data from 45 patients with HI. A period of relative high hypoglycemia risk was observed in the early hours of the morning.
Hypoglycemia (<3.5 mmol/L), as measured by CGM, also varied over the course of the study period. For the 34 patients who provided ≥90 days of data, there was a trend to increasing time below range (TBR) over time (Figure 4). This increase in mean TBR was most pronounced within the first half of the study and was followed by a reduction in TBR increment.
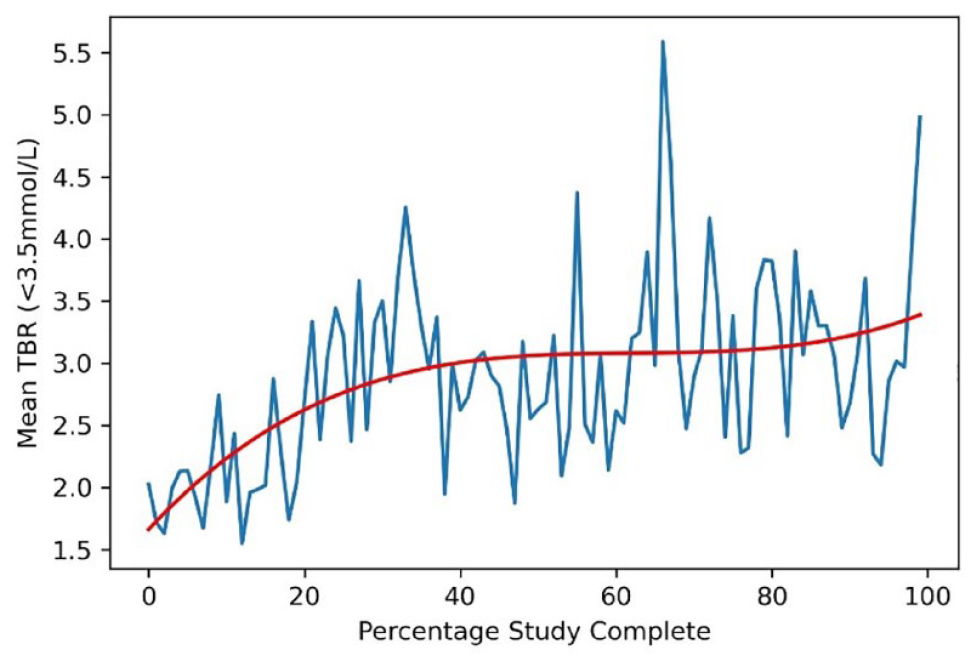
Mean time below range (TBR) over the course of the study period. Red line demonstrates the line of best fit.
When viewed at an individual level, two patients (patient x and patient y) showed a significant increase in TBR over the study period whereas the majority remained stable (Figure 5).
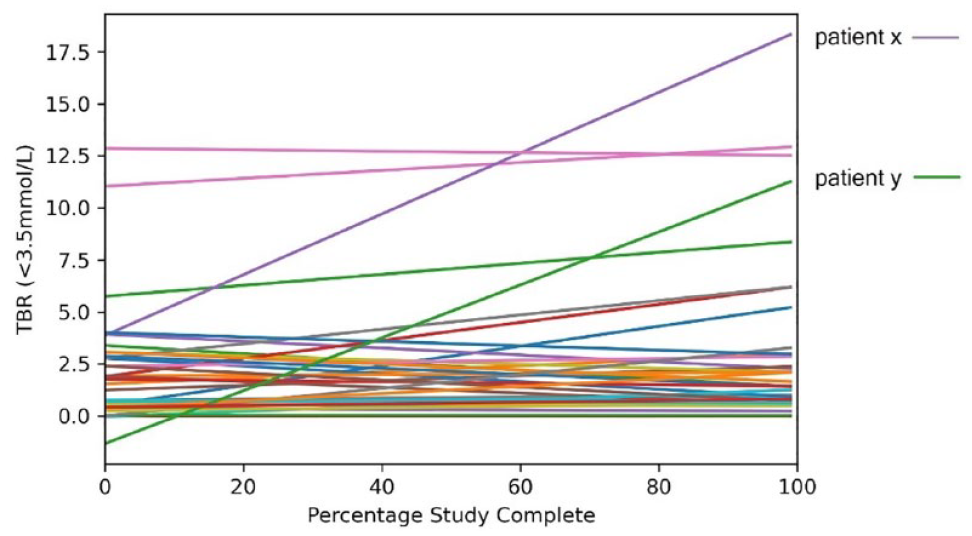
TBR linear line of best fit for individuals over the study period. Two patients (patient x and patient y) showed a linear upward trend to increasing TBR over the study period.
When these two outliers were removed from the analysis, mean TBR over the course of the study was unchanged with an initial period of low hypoglycemia incidence followed by an increase and subsequent stabilization (Figure 6). The families of the two outlying patients were contacted and neither had an explanation for the significant increase in hypoglycemia toward the end of their time in the study. The tendency to early morning hypoglycemia did not change from study start to end, with an ongoing risk of hypoglycemia around 05:00 hours.
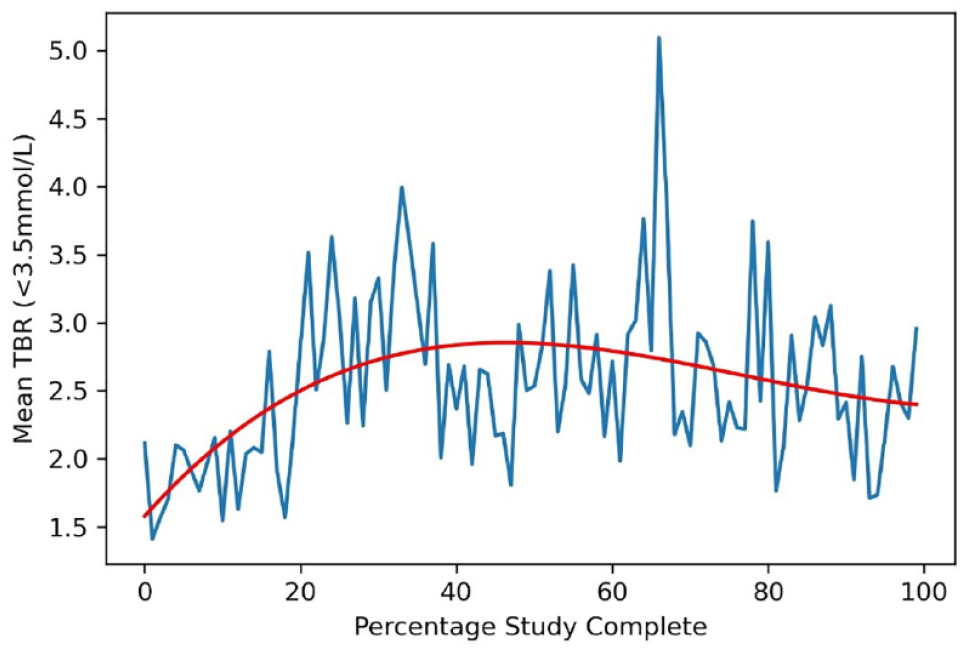
Mean TBR over the course of the study following removal of the two outliers from graphical analysis.
Accuracy Metrics
A total of 17 521 SMBG values were paired with a CGM value (0.7% of all CGM values); of the 8051 hypoglycemic episodes detected by CGM, there were 1237 paired SMBG values (15% of hypoglycemic CGM episodes). Mean absolute relative difference for the group was 25% and was not affected by patient age or presence of genetic mutation (P > .2 for all in analysis of covariance [ANCOVA]). Mean absolute difference was 1.2 mmol/L and mean difference was +0.45 mmol/L, indicating an average higher reading of glucose by the G6 device compared to SMBG values. Point hypoglycemia sensitivity was 32%. Hypoglycemia (<3.5 mmol/L) detection rate was 40% and was also not affected by either age (Pearson correlation coefficient 0.21, P = .3) or presence of genetic mutation (P = .5 on t-test). Owing to the high percentage of patient glucose values ≥4.0 mmol/L (on CGM and SMBG), almost 90% of paired values were classed as posing no risk (Figure 7). However, when the data set was restricted to those values of clinical interest (either CGM or SMBG ≤4 mmol/L), only 70% of these values were classed as no risk by HEG analysis (Figure 7).
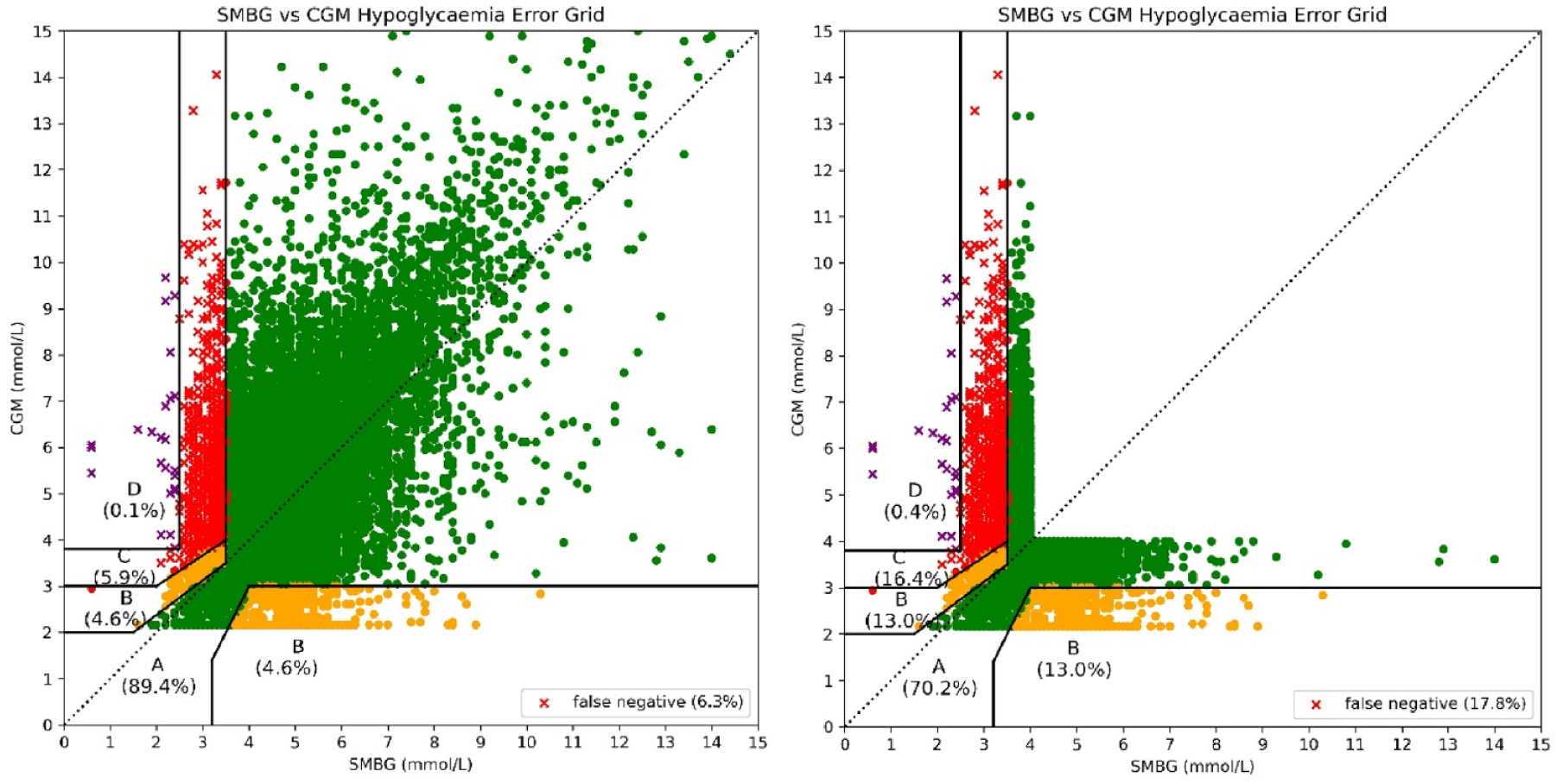
Paired values plotted on the hypoglycemia error grid: all values on the left and values restricted to ≤4 mmol/L on the right. Zones A, B, C, and D reflect the risk of the CGM error as none, slight, moderate, and severe.
Questionnaire Responses
As outlined in Figure 1, 43 of 46 families responded to the questionnaire. In addition, questionnaires were received from all nine adolescents (13–18 years). When asked why they wanted to take part, reasons were varied but most commonly included wanting to understand hypoglycemia better, wanting to reduce fingerpricks, and wanting to reduce hypoglycemia events.
Continuous glucose monitoring impact on patient behaviors
Continuous glucose monitoring wear influenced fingerprick blood glucose testing, with 25 (58%) performing fewer fingerpricks and 12 (28%) performing more. Six (14%) families reported no change in fingerprick testing behavior. Responses from adolescents corroborated parental findings. More parents reported a worsening of sleep (42%) than an improvement in sleep (35%) when using CGM, with adolescents mostly reporting no change (56%). Mean parental anxiety score improved from 4.1 to 2.9 (P < .001 on paired t-test) on Likert scale while that of patients showed a small, and non-significant, increase (2.2 to 2.6, P = .56).
Parents’ median rating of the accuracy of CGM devices was 3/5, with no family reporting devices as “very accurate” and four (9%) reporting 1/5 accuracy (“not accurate at all”). This was mirrored by adolescent patient responses and confirmed by the analysis in section “Accuracy metrics.”
Perceived benefits of continuous glucose monitoring
The majority of parents (79%) reported that CGM had helped them understand glucose trends. This resulted in questionnaire reports of identifying new episodes of hypoglycemia (71%), identifying patterns in hypoglycemia (66%), and a better understanding of the causes of hypoglycemia (42%). Continuous glucose monitoring as a tool for behavior change has been shown previously 18 and was evident in this study, with almost half of parents (47%) reporting that CGM prompted a change in routine. Overall, 63% of parent respondents reported feeling reassured by CGM trends and 66% reported CGM ultimately improving the management of HI for their child.
For certain families, CGM was hugely beneficial with families reporting that, “CGM is essential for children below 5 years”, “fundamental to improving my child’s independence” and “CGM has turned her into a happier and more responsible child”.
Reported problems with continuous glucose monitoring
Reflecting the high discontinuation rate (Figure 2), many families found CGM burdensome with only 19 of the 46 (41%) respondents stating that the positives outweighed the negatives. Of the 42 parents who responded to a question asking for significant problems with CGM, 26/42 (62%) reported pain and 23/42 (55%) cited sensor inaccuracy. Problems with the sensor adhesive were also commonly reported with other problems such as “disruption at school” reported by small numbers of families. These responses did not vary between those who discontinued or continued CGM; pain from sensor insertion and use was the primary reason for discontinuing, with inaccuracy being second most common reason.
Patient respondents also reported inaccuracy (4/9) and pain (4/9) as significant problems with CGM but equally important for patients was the disruption that CGM caused (4/9) at both school and during social activities. This was also recognized as a problem by seven parents, all of older children.
Comments from some families made very clear that CGM was not transformative, with reports such as: “It made an already stressful situation worse”, “I thought at 1st this would be fantastic for the chi kids to try out we found that it was giving us the wrong reading or would say he was stable but after doing my son’s b/m he was having really major hypos”.
Discussion
The use of CGM for patients with HI has been a topic of discussion and debate for over ten years, with a small number of papers describing anecdotal use or retrospective evaluation of glycemic trends and device accuracy. Previous studies have reported data from retrospective cohorts of CHI patients already using CGM; these are likely to be biased toward more severe cases being preferentially provided CGM. Questionnaires and interviews conducted with families using CGM are also subject to positive bias, having excluded those who discontinued CGM.
This study offered long-term CGM to a comprehensive, unselected, and unbiased national cohort of patients with HI, providing the first prospective report of glycemic stability and trends within the condition as well as the only large-scale, unbiased evaluation of families’ experiences with this emergent technology. These data may thus inform design of CGM based studies in hypoglycemia and HI, including clinical trials of novel therapies and testing newer generations of CGM sensors.
Although this study has several strengths in large numbers, unbiased selection and study duration enabling trend analysis, a few weaknesses are also inherent. Investigation and treatment variations at different centers were likely, given the observational nature of this cohort. Similarly, parent interactions with the research team were also variable, influencing individual behavioral responses in the prevention of hypoglycemia. However, such variability also provides wide data latitude assisting future wider study design.
Basic glycemic data on time spent hypoglycemic within this study (2.4%) is similar to previous reports of time hypoglycemic for HI patients on medication (2.5%) 8 and confirms the generally stable glycemic control exhibited by most patients with HI in the United Kingdom. These prospective and heterogeneous data replicate findings from earlier studies 16 suggesting that hypoglycemia is more prevalent in the early hours of the morning. This confirmation should prompt clinical management strategies and trial methodologies to target times of high hypoglycemia risk.
Contrary to the expectation from use of CGM in people living with diabetes, CGM use in HI did not reduce total hypoglycemia episodes in this study, nor reduce the tendency to early morning hypoglycemia. This observation, from our year-long study, apparently incongruent with clinical benefit, suggests the need for alternative approaches to the interpretation of CGM information. Thus, unsupported CGM relying on suboptimal point accuracy of the current generation of CGM sensors requires transformation with additional algorithmic support for meaningful clinical use. Short periods of intermittent CGM use could also be envisaged to provide knowledge of the personal dynamics of HI and allow for individualized adaptations without the burdensome requirement for long periods of continuous use. Such intermittent CGM use contrasts with CGM use in people living with diabetes, where multiple treatment decisions are made every day, based on live glucose data.
Despite studies suggesting a reduction in disease severity over time, 24 we did not observe a reduction in hypoglycemia over the course of this 12-month study. This observation could be explained by change in medication doses over the study period as per clinical preference. The lack of baseline data prior to CGM use is a limitation of the study and it is possible that there was an initial reduction in hypoglycemia during the transition to real-time CGM. Furthermore, hypoglycemia reduction with CGM use may require a longer interval to manifest, not currently captured in the stipulated 12-month study period.
The accuracy of the present generation of CGM devices has been circumspect. This study investigated CGM and SMBG paired data to confirm previous findings 13 of suboptimal accuracy of the Dexcom G6 device in HI. Until accuracy at hypoglycemia is actively addressed by CGM manufacturers, this is likely to remain a major barrier to widespread use in this patient group.
Parental perception is an important consideration in clinical management strategies in HI. 11 This study demonstrates that although CGM was considered a desirable tool (and initial uptake was high), outcomes were suboptimal for many families and that the burdens often outweighed the benefits, resulting in high discontinuation rates. However, many families reported individual ways to use CGM to prevent hypoglycemia.
Conclusion
The deep glycemic phenotyping information generated in this national prospective observational study of CGM in HI demonstrates early morning hypoglycemia risk and absence of long-term reduction in hypoglycemia through CGM wear over the course of 12 months. Family experience of CGM was variable, with more than half of participants discontinuing CGM use during the one-year study period with suboptimal CGM sensor accuracy contributing to loss of confidence, suggesting that CGM use in clinical practice may remain individualized. Nonetheless, insights derived from this comprehensive cohort of patients will inform future studies on the use and application of CGM in HI patients and clinical trial design for novel therapies aiming to reduce hypoglycemia duration in this patient population.
Footnotes
Author Contributions
CW designed and ran the study and wrote the first draft of the manuscript and collated responses from other authors. SA led the design of the questionnaire and the qualitative analysis. SW was the lead nurse for the study and coordinated device supplies, data uploads, and questionnaire completions. EOS and SA aided in the recruitment, consent, and ongoing clinical management of patients. CF-C and SB provided expert industry input to study design and data analysis. MS-E, GS, SS, and AD were local leads for patient recruitment and consent and contributed to manuscript edits. IB was the PI for the study and provided senior support on study design, management, and write up. CW, CFC, and IB directly assessed and verified the underlying data in the manuscript. All authors had full access to the data and accept responsibility for the data and reviewed and approved the final manuscript.
Abbreviations
ADA, American Diabetes Association; CGM, continuous glucose monitoring; HI, hyperinsulinism; MAD, mean absolute difference; MARD, mean absolute relative difference; SMBG, self-monitoring of blood glucose; TBR, time below range.
Data Sharing
Raw and anonymized data will ultimately be shared to a data repository for use by other research teams by contacting the corresponding author and agreement of data usage.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IB has received research and educational grants from Zealand, Merck, Diurnal, and Hanmi Pharmaceuticals; these grants did not have conflict of interest with the study presented herewith. At present, IB is the Chair of the Communications Committee of the European Society of Paediatric Endocrinology (ESPE), the Chair of the National Institute of Health Research (NIHR) Clinical Studies Group (CSG) for the British Society of Paediatric Endocrinology and Diabetes (BSPED), and a member of the Rare Disease Advisory Group (RDAG) in NHS England. He has advised the National Institute of Health and Care Excellence (NICE) on the development of Head Injury guidelines and has advisory roles with the following patient organizations: Congenital Hyperinsulinism International, Congenital Hyperinsulinism Charity, Pituitary Foundation, and Living with Congenital Adrenal Hyperplasia. MS-E is the national clinical lead for pediatric endocrinology in NHS England. She has received research funding from Zealand and Hanmi Pharmaceuticals. MS-E has advisory roles with Congenital Hyperinsulinism International and Congenital Hyperinsulinism Charity. AD has received research funding from Resolute and Hanmi and Zealand Pharmaceuticals. She has advisory roles with Congenital Hyperinsulinism International and Congenital Hyperinsulinism Charity. SS has received research funding from Zealand Pharmaceuticals. He has advisory roles with Congenital Hyperinsulinism International and Congenital Hyperinsulinism Charity. CW is the national trainee lead for the children’s specialty group within the NIHR and has advisory roles with Congenital Hyperinsulinism International and Congenital Hyperinsulinism Charity. SB is a founder and shareholder of Crinetics Pharmaceuticals and is a Board Observer for Radionetics Oncology and Cellular Longevity. CFC is Senior Director of Clinical Research at Crinetics Pharmaceuticals. EOS, SW, SHA, and SA declare no potential conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Crinetics phamaceuticals provided funding for this study.
