Abstract
Background:
The use of insulin pumps (continuous subcutaneous insulin infusion [CSII]) in individuals living with type 1 diabetes (T1D) improves disease control. However, adverse skin reactions may hamper compliance. We aimed to assess the relationship of insulin pumps, particularly that of infusion set therapy, used in children and adults with T1D and dermatitis including allergic contact dermatitis (ACD).
Methods:
A systematic search of PubMed, and EMBASE, of full-text studies reporting dermatitis in persons with diabetes using a CSII was conducted from 2020 to 2023. The Newcastle-Ottawa Scale was used to assess study quality. The inventory performed at the Department of Occupational and Environmental Dermatology, Malmö, Sweden (YMDA) was also performed highlighting the diagnostic process.
Results:
Among the 391 screened abstracts, 21 studies fulfilled the inclusion criteria. Seven studies included data on children only, four studies were on adults, and nine studies reported data on both children and adults. Participants were exposed to a broad range of pumps. Dermatitis was rarely specified. Up to 60% of those referred to a university hospital due to skin reactions possibly related to insulin pumps had an ACD.
Conclusions:
The review and our findings indicate that there is not sufficient focus on contact allergy in the primary toxicological evaluations of substances used also for insulin pump therapy products and that possible adverse skin reactions are not correctly followed up in the clinical setting.
Keywords
Introduction
Insulin pumps (continuous subcutaneous insulin infusion [CSII]) are commonly used for treatment of individuals living with type 1 diabetes (T1D) with great benefit for diabetes control.1-5 Whereas the first commercial insulin pumps appeared in the 1970s, over the past 30 years diabetes medical device (MD) technology has advanced rapidly. Combining CSIIs with a continuous glucose monitoring (CGM) system is often used to reach even better glucose control. 6 An increasing number of individuals with either T1D or type 2 diabetes are recommended therapy with a variety of insulin pumps. 7 The insulin is delivered through a catheter inserted into the subcutaneous tissue and secured on the skin with adhesive, the infusion set. Some pumps, referred to as “patch pumps,” do not use tubings but adhere directly to the skin. However, adverse skin reactions from these CSII may hamper compliance. When the etiology of such adverse reaction is due to sensitization to substances within the product, it may lead to lifelong contact allergy, thus reducing the quality of life of the patients 8 and increasing health care costs. 9 The pump material and adhesives in tight connection on occluded skin for a prolonged period pose a risk of adverse skin reactions, where particularly allergic contact dermatitis (ACD) has been raised as a concern. The insulin pumps may consist of allergenic substances which differ from substances used in sensors, thus investigating development of sensitivity to the insulin pump, even primarily, is of high interest. In many publications however, authors do not distinguish between reactions to CSII and CGM; in addition, reactions are only patient reported which inhibit correct diagnosis. A correct diagnosis is of the utmost importance as this will enable correct secondary and possibly primary preventive measures. Differentiating between dermatitis due to irritancy or contact allergy is not possible without correct epicutaneous patch testing. 10 The number of users who experience any skin reaction related to CGM or CSII is not fully known. In a recent review where any MDs related to treatment of T1D were investigated, it was estimated that 5% to 10% of patients experienced dermatitis, 11 regardless of age. As the individuals with diabetes are motivated to continue use of the devices and as there is a lack of awareness that the products might cause contact allergy also among clinicians, there is a risk of underreporting adverse skin reactions. Indeed, in questionnaires actively addressing possible skin problems much higher numbers are reported and skin problems have been reported as a major cause of change of product or even interrupted use.12-14 In this systematic review adverse skin reactions with dermatitis and ACD induced by infusion sets (for insulin pump users) or insulin patch pumps are reported. Additionally, since publications are scarce our clinical findings the last three years illustrating the diagnostic difficulties are summarized.
The primary objective of this study was to assess the relationship of CSII, particularly that of infusion set use, in children and adults with insulin-dependent diabetes mellitus (IDDM) and dermatitis including ACD. If possible, the prevalence and incidence of dermatitis including ACD were calculated.
Methods
Following a pre-specified protocol (PROSPERO; registration no. CRD42023483330, https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=483330), studies were included which assessed any dermatitis related to CSIIs in children and adults with IDDM.
Search Strategy
A systematic search of PubMed, EMBASE, Google, and Google Scholar has been performed in addition to hand search in reference lists of included articles and conference reports, from January 1, 2020 to November 24, 2023. Predefined search terms and MeSH (medical subject headings) headings and keywords have been developed in collaboration with a medical librarian. The MeSH terms and searches are described in Supplement I.
Inclusion Criteria
Population-based cross-sectional cohort or nested case-control studies that were published between January 2020 and the time of data extraction and that reported incident and prevalent dermatitis including ACD in adults or children with IDDM using CSIIs were included. Inclusion criteria were epidemiological studies that reported on contact dermatitis, irritant dermatitis, contact allergy, or any dermatitis possibly related to CSII in persons with diagnosed IDDM. For intervention studies, studies were not selected based on treatment, though these data were extracted for comparison purposes. No language restrictions were applied. Studies that included less than ten patients, or studies that did not assess the exposures and outcomes of interest, were excluded. The following outcomes were assessed: dermatitis using a broad definition, contact dermatitis, ACD, contact allergy, and clinical relevance. Descriptive data and details on the study settings were collected. The reasons for exclusion are reported in Supplement II.
Eligibility Assessment and Data Extraction
Two reviewers (LK, EK) independently assessed the eligibility of studies based on the title and abstract. In the second screening phase, full-text articles were assessed independently by the two reviewers (LK, EK). A data extraction form was developed and piloted by reviewers on a random selection of included studies designed to describe the characteristics of studies to be included, as set out in the recommendations in the Cochrane Handbook section 7.3.6. Two reviewers extracted the data from included studies using this form (LK, EK). Extracted data items included characteristics of each study, study methodological quality items, and the outcomes of interest for each study using a standardized data extraction as recommended in the Cochrane Handbook section 5.5. All disagreements were resolved by consensus involving a third reviewer (JU or CS).
Study Quality Assessment
Following the Cochrane Collaboration’s recommendation, a set of quality appraisal items was applied. These include biases in sample selection, validity of measures of disease and educational outcome, appropriateness of statistical analysis, and adjustment for confounders when applicable using the Newcastle-Ottawa quality assessment Scale (NOS). 15 The NOS was used for longitudinal studies and an adopted version for cross-sectional studies.
Statistical Analysis
Articles that met the inclusion criteria were recorded in table format to perform a systematic and narrative synthesis on the available evidence. When numbers were provided in the original articles but not percentages, percentages were calculated. We studied all different CSII systems together and reported combined for any reaction on any CSII system. We also studied each type of CSII individually; if studies included different CSIIs, we distinguished the adverse skin reactions if possible. Data were summarized descriptively, separately for children and adults if possible. Furthermore, we planned to perform a meta-analysis including exploring sources of heterogeneities if possible. This review was reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (Supplement III).
Clinical Patients
In the Department of Occupational and Environmental Dermatology, Malmö, Sweden (YMDA), a total of 900 to 1000 patients are referred annually for possible exogenous dermatitis. The clinic is the referral center for the region of Southern Sweden and referrals come from endocrinologists, general practitioners, and dermatologists. Patch test results for some of the individuals with diabetes have been previously published in detail as case reports.16-19
Clinical Investigations and Patch Tests
Patients investigated for possible ACD were routinely patch tested with the baseline series, an additional MD series 20 and often with their own material as such and in extract. 10 Patch testing and patch test readings were performed according to the recommendations by the International Contact Dermatitis Research Group and the European Society of Contact Dermatitis (ESCD).21,22 We report the proportion of the patients investigated due to suspected ACD to CSII or associated infusion sets with contact allergy to MD-related allergens (allergens patch tested in the department’s MD patch test series, colophonium 20% in petrolatum patch tested in the Swedish baseline series and colophonium 60% in softisan as well as isobornyl acrylate [IBOA] 0.3% in petrolatum patch tested in the extended baseline series).
Ethical Considerations
There is no need for an ethical approval for this review. Ethical approval for the use of the clinical register data was obtained from the Swedish Ethical Review Authority (registration number: 2020-02190).
Results
Among the 391 screened abstracts, 21 studies fulfilled the inclusion criteria (Figure 1 and Table 1).8,22-30,31-41 The results from these studies are presented in Table 1. Several case reports were excluded, as well as abstracts without available full text, letters to the editor, and reviews as they did not contain sufficient original data. The reasons for exclusion are reported in Supplement II. Of the included studies, seven studies reported data of in total 633 children,24,30,31,33-35,41 four studies on 1532 adults,39,29,38,31 and nine studies on both adults and children8,12,23,25-28,37,40 (n = 1211). For one study, the age was unclear. 36 Sixteen studies were cross-sectional,23-30,31-36,38,41 and five were longitudinal.8,37-40 In the longitudinal studies, the follow-up time (the time of observation) ranged between 24 weeks and three years. Of the included studies, there were 16 from Europe,8,23-26,28-30,32-35,38-41 2 from the United States,31,36 1 from Russia, 36 1 from Turkey, 27 and 1 from New Zealand. 37
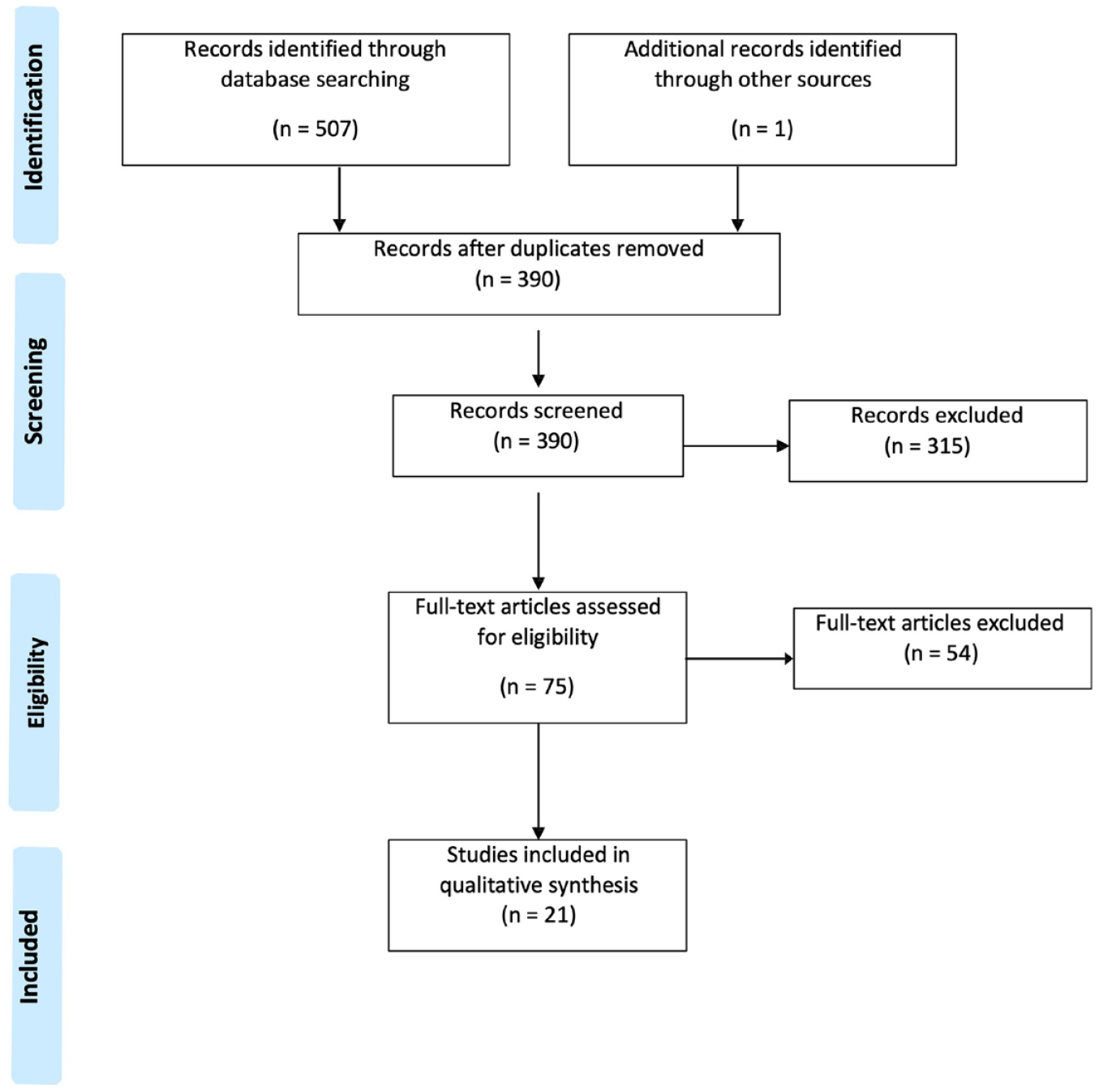
PRISMA flow chart.
Description of Included Studies With Patients With Insulin-Dependent Diabetes Mellitus Using Insulin Pumps.
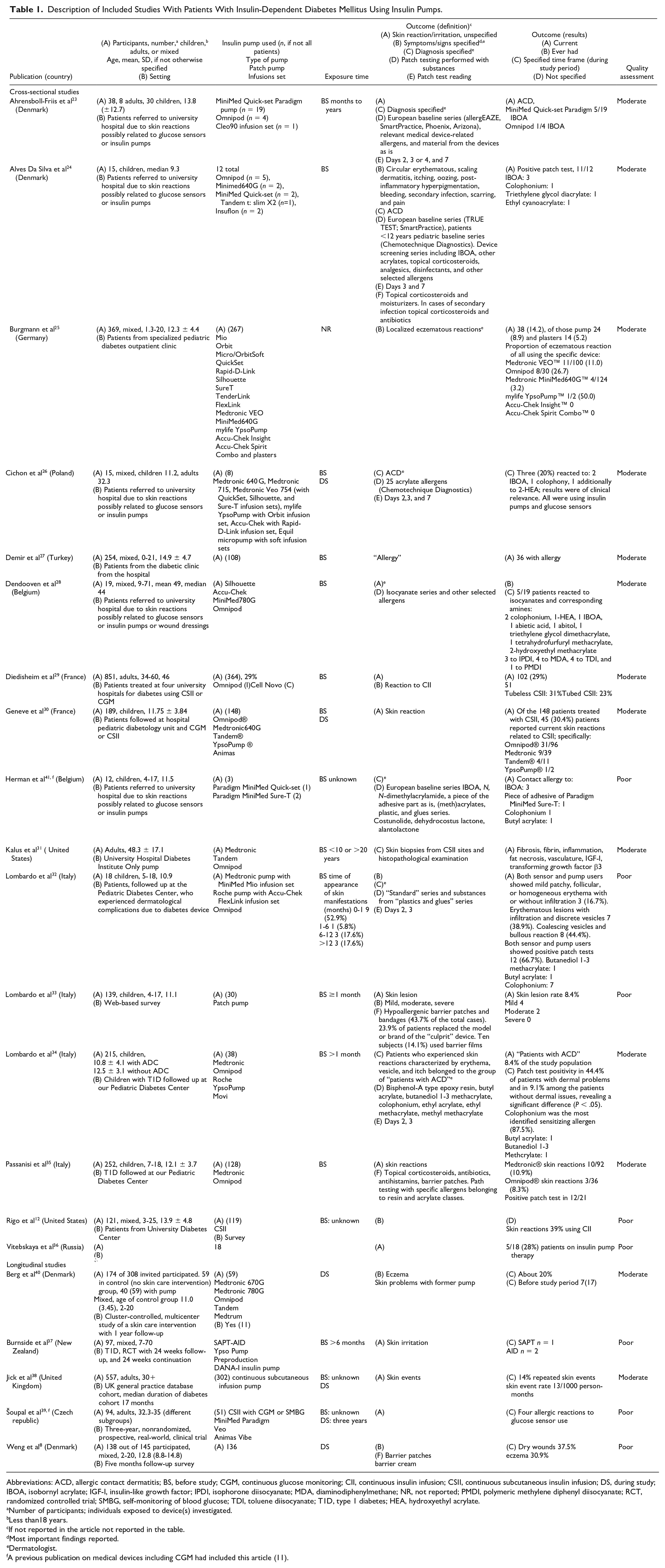
Abbreviations: ACD, allergic contact dermatitis; BS, before study; CGM, continuous glucose monitoring; CII, continuous insulin infusion; CSII, continuous subcutaneous insulin infusion; DS, during study; IBOA, isobornyl acrylate; IGF-I, insulin-like growth factor; IPDI, isophorone diisocyanate; MDA, diaminodiphenylmethane; NR, not reported; PMDI, polymeric methylene diphenyl diisocyanate; RCT, randomized controlled trial; SMBG, self-monitoring of blood glucose; TDI, toluene diisocyanate; T1D, type 1 diabetes; HEA, hydroxyethyl acrylate.
Number of participants; individuals exposed to device(s) investigated.
Less than18 years.
If not reported in the article not reported in the table.
Most important findings reported.
Dermatologist.
A previous publication on medical devices including CGM had included this article (11).
Participants were exposed to different CSII as described in Tables 1 and 2. Most studies did not have an assessment of the prevalence of dermatitis as primary aim and the type of dermatitis was seldom specified. Often, studies did not distinguish between dermatitis related to CGM or CSII. Most studies did not assess comorbid atopic dermatitis. Seven studies published data on patch testing and the prevalence of ACD; of those only one 41 performed patch testing with the devices and adhesives as is, and only three performed a reading at day 7.23,24,26 One study reported the histopathological assessment of CSII sites, 31 and four studies assessed treatment of skin reactions related to CSII.24,33,35 The outcomes were physician diagnosed in 11 studies23-28,31,32,34,39,40 (723,25,26,28,32,34,41 of them by a dermatologist), and self-reported in 78,12,29,30,33,37,38 studies. One study is not known 36 and two studies did not specify the assessment of the outcomes.35,39 Four studies reported a history of atopic dermatitis in their participants.24,25,29,32 In two studies, a history of atopic dermatitis was not separately reported for CSII34,35 while one study did not specifically report a history of atopic dermatitis for children with pumps. 30 Twelve studies did not assess the history.8,12,23,26-28,31,37-41 All studies had a poor or moderate-study quality according to the NOS, mostly because criteria such as selection of participants and persons without exposure to CSII could not be fulfilled (Table 1, Supplement IV). The included studies applied different outcomes and study populations were not homogenous; therefore, a quantitative synthesis and meta-analysis could not be performed.
Description of Included Insulin Pumps and Glucose Sensors.

Abbreviations: —, usage not reported; CSII, continuous subcutaneous insulin infusion; AA, Ascensia Diabetes Care, Basel, Schweiz; A, Abbott Diabetes Care, Witney, United Kingdom; AC, Animas Corporation, West Chester, Pennsylvania; C, Cellnovo Limited, Paris, France; D, Dexcom, Inc, San Diego, California; I, Insulet Corporation, Billerica, Massachusetts; IM, ICU Medical, San Clemente, California; M, Medtronic, Minneapolis, Minnesota; ME, Medtrum, Eschborn, Germany; MEN, Meniarini Diagnostics, Florence, Italy; MT, MicroTech Medical, Hangzhou, China; R, Roche Diabetes Care, Basel, Schweiz; T, Tandem Diabetes Care, San Diego, California; Y, Ypsomed, Burgdorf, Switzerland.
Overall, the prevalence of dermatitis ranged from about 8% to more than 40%. Often, the proportion with ACD to CSII was not assessed due to low numbers, and few studies reported the prevalence across those referred to university hospital due to skin reactions possibly related to glucose sensors or insulin pumps of ACD up to 60%. The proportion of children with any dermatological reaction to CSII was 8% to 40% of those who were patch tested, and the proportion of ACD related to CSII was 40% to more than 60%. The proportion of adults with any form of dermatological reaction to CSIIs was approximately 30%, whereas the proportion of adults with ACD due to CSII was not specifically disclosed. Positive patch test reactions to IBOA and other MD-related allergens are shown in Table 1.
Clinical Study
Over the last three years (2021-2023), the number of referrals for adverse skin reactions to MDs, in the form of either insulin pump or glucose sensor or both, was 123, with an annual increase in referrals each year.29,42,43 The findings for patients referred to YMDA in 2021 to 2023 due to skin reactions to patch pumps or insulin pump infusion sets are summarized in Table 3. In total, 51 patients were referred to YMDA or its branch clinics in 2021 to 2023 due to skin reactions to CSII or their infusion sets. In total, 37 of these patients (18 children and 19 adults) have been patch tested (6 of the patients referred in 2023 are scheduled for patch test investigations in 2024 and 8 patients missed or canceled their clinical appointments). Of the individuals that were patch tested, 16 (43.2%) were allergic to MD-related allergens. In total, 6 of the 16 patients with MD-related contact allergy were allergic to IBOA and/or to colophonium, modified colophonium, or colophony-related substances.
Patients referred to and patch tested at YMDA due to suspected allergic contact dermatitis to insulin pumps or associated infusion set 2021-2023.
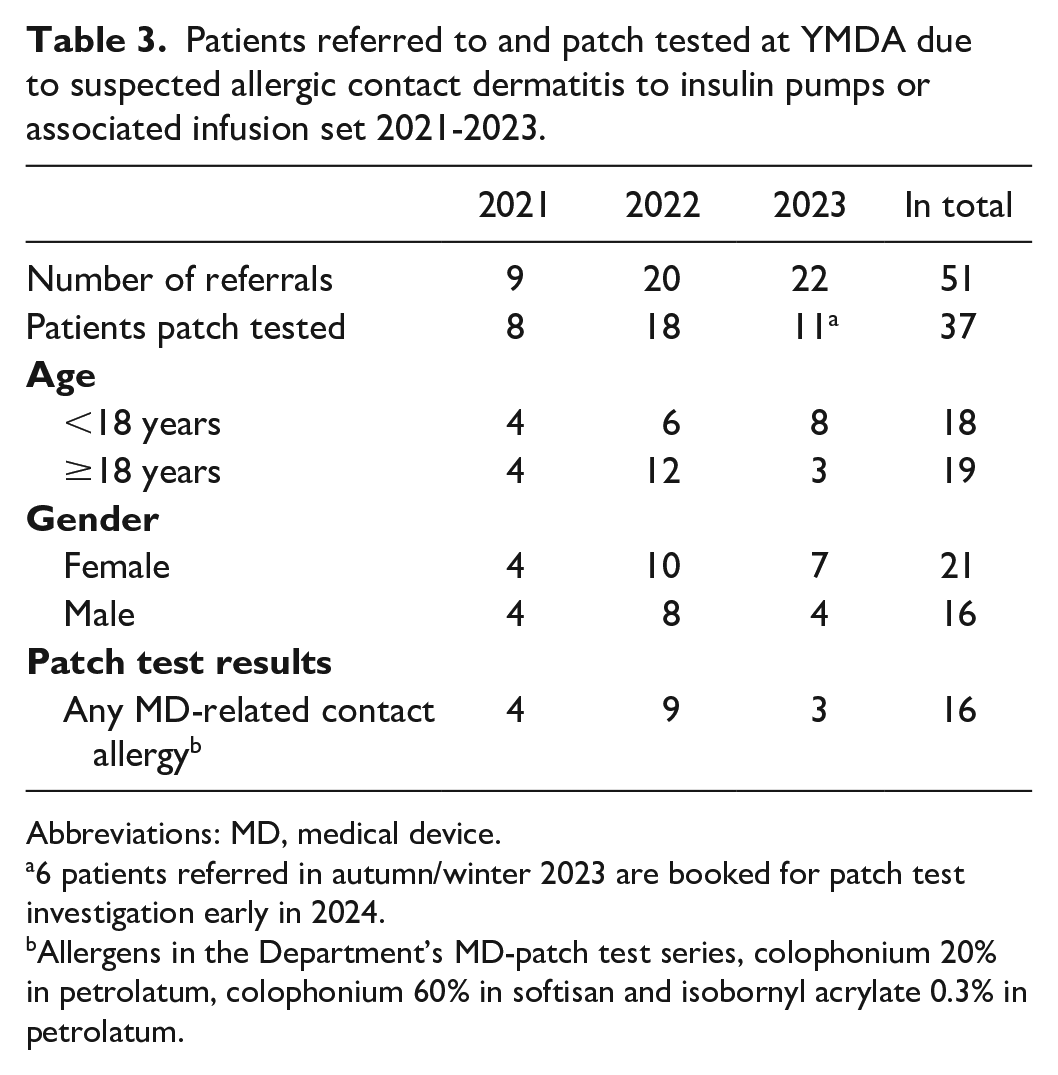
Abbreviations: MD, medical device.
6 patients referred in autumn/winter 2023 are booked for patch test investigation early in 2024.
Allergens in the Department’s MD-patch test series, colophonium 20% in petrolatum, colophonium 60% in softisan and isobornyl acrylate 0.3% in petrolatum.
Discussion
The systematic review performed highlights that the proportion of any skin reaction to CSIIs was high in adults and children with IDDM. There was a high proportion of positive patch tests in children or a mixed population of children and adults with CSII; however, the results need to be interpreted with caution as there were only few studies. For adults alone, no studies were found reporting ACD in users of CSII. Skin reactions due to insulin pump therapy is seldom the main outcome of studies. Studies reporting adverse skin reactions usually do not define the true nature of these cutaneous adverse events and validated outcome assessments are seldom applied. For assessment of ACD, many clinics rely on commercial patch tests probably missing testing for relevant allergens, which is supported by the clinical findings of the current study which showed that many of the patients were allergic to other allergens than those found in the baseline series or commercially available. Furthermore, several studies did not follow the ESCD recommendations. 22
We undertook a comprehensive search using a predefined protocol and assessed study quality. In the included studies, different—if any—definitions were applied for dermatitis. For assessment of ACD, several studies did not follow the recommended reading at day 7,23,24,26 and only one study used test materials from the devices “as is,” 23 thus introducing possible information bias and underassessment of ACD to pumps. Almost all studies included highly selected study populations and might thus be prone to population bias. Most studies were cross-sectional, and possibly missed cases as sensitization often occurs after about six months. However, several studies had a prospective design which enabled assessment of temporality, although most studies did not include a comparison group. This review provides new information on the prevalence of skin reactions and ACD among CSII users.
The findings of our review showed that different skin preserving measures are used, which is in line with a recent publication. 44 It has been suggested that there is probably an underestimation of users reporting skin reactions to the adhesives of both sensors and pumps, 11 and this seems not to have changed in the last years. In our department, the number of referrals has increased the last years. The reason might be an increasing exposure to MDs as such and insulin pumps, but an increased awareness might also be the explanation. The clinical findings do not suggest that insulin pump therapy patients are spared ACD, and the data furthermore suggest that also an insulin pump user, be that an infusion set or a patch pump user, might actually be sensitized to substances in the devices. 19 In Sweden today usually the individual is exposed to both devices, sensor and pump, and if the substance the individual is allergic to is found in both products the history and findings might not allow a definite decision on which was the primary sensitizer.
Contact allergy, being a type IV reaction, is lifelong and an individual once sensitized will experience symptoms if exposed in a sufficient dose on the skin even years after they were initially sensitized. We are surrounded by products and continuously use substances at work and in our spare time that might contain substances that are possible contact allergens. The same substances are found in different settings and not only in MDs. In modern life, almost everyone in all age groups will be exposed to MDs on the skin, not only when in hospital care; this further emphasizes the need to minimize the number sensitized that is produce MDs that are safe regarding at least sensitization. If the substance is regarded ubiquitous and the aim is secondary prevention, the amount used must be below also the threshold for elicitation. Whether a substance, provided it is a possible contact allergen, in a complex product will sensitize or elicit an allergic reaction is defined by the dose per area of skin exposed. 23 Apart from the dose of allergen that comes in contact with the skin also the time of exposure is decisive whether symptoms of ACD will appear. In insulin pump therapy, the exposure time is usually less than that in the sensor systems. Our findings do not however suggest that the patient group has less adverse skin reactions from the insulin pump systems. In MDs, there are several substances used that are already known to be contact allergens9,24,40 and substances through use in MDs in the actual patient setting have been found to be contact allergens.20,25,37 The same has been found true also for infusion set and patch pump users.26-28 Only 6 of the 16 patients with MD-related contact allergy patch tested at YMDA 2021 to 2023 were allergic to IBOA, colophonium, modified colophonium, or colophony-related allergens. As a majority of the cases had contact allergy other than the previously known major MD-related allergens (colophony-related substances and IBOA), this highlights the importance of continuous chemical analyses of MDs to ensure relevant and up-to-date patch testing and relevance assessment of contact allergies found. Large, well-performed studies assessing ACD in individuals with diabetes on CSII therapy are urgently required.
Conclusions
The review and our findings indicate that there is not sufficient focus on contact allergy in the primary toxicological evaluations of substances used also for insulin pump therapy products and that possible adverse skin reactions are not correctly followed up neither in studies nor in the clinical setting.
Supplemental Material
sj-docx-1-dst-10.1177_19322968241252613 – Supplemental material for Insulin Pump Therapy and Adverse Skin Reactions With Focus on Allergic Contact Dermatitis in Individuals Living With Diabetes Mellitus: A Systematic Review and Clinical-Based Update
Supplemental material, sj-docx-1-dst-10.1177_19322968241252613 for Insulin Pump Therapy and Adverse Skin Reactions With Focus on Allergic Contact Dermatitis in Individuals Living With Diabetes Mellitus: A Systematic Review and Clinical-Based Update by Laura Beate von Kobyletzki, Josefin Ulriksdotter, Elisa von Kobyletzki, Martin Mowitz, Johan Jendle and Cecilia Svedman in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-2-dst-10.1177_19322968241252613 – Supplemental material for Insulin Pump Therapy and Adverse Skin Reactions With Focus on Allergic Contact Dermatitis in Individuals Living With Diabetes Mellitus: A Systematic Review and Clinical-Based Update
Supplemental material, sj-docx-2-dst-10.1177_19322968241252613 for Insulin Pump Therapy and Adverse Skin Reactions With Focus on Allergic Contact Dermatitis in Individuals Living With Diabetes Mellitus: A Systematic Review and Clinical-Based Update by Laura Beate von Kobyletzki, Josefin Ulriksdotter, Elisa von Kobyletzki, Martin Mowitz, Johan Jendle and Cecilia Svedman in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-3-dst-10.1177_19322968241252613 – Supplemental material for Insulin Pump Therapy and Adverse Skin Reactions With Focus on Allergic Contact Dermatitis in Individuals Living With Diabetes Mellitus: A Systematic Review and Clinical-Based Update
Supplemental material, sj-docx-3-dst-10.1177_19322968241252613 for Insulin Pump Therapy and Adverse Skin Reactions With Focus on Allergic Contact Dermatitis in Individuals Living With Diabetes Mellitus: A Systematic Review and Clinical-Based Update by Laura Beate von Kobyletzki, Josefin Ulriksdotter, Elisa von Kobyletzki, Martin Mowitz, Johan Jendle and Cecilia Svedman in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-4-dst-10.1177_19322968241252613 – Supplemental material for Insulin Pump Therapy and Adverse Skin Reactions With Focus on Allergic Contact Dermatitis in Individuals Living With Diabetes Mellitus: A Systematic Review and Clinical-Based Update
Supplemental material, sj-docx-4-dst-10.1177_19322968241252613 for Insulin Pump Therapy and Adverse Skin Reactions With Focus on Allergic Contact Dermatitis in Individuals Living With Diabetes Mellitus: A Systematic Review and Clinical-Based Update by Laura Beate von Kobyletzki, Josefin Ulriksdotter, Elisa von Kobyletzki, Martin Mowitz, Johan Jendle and Cecilia Svedman in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors would like to acknowledge the medical librarian Marika Svalstedt for assistance with the literature search.
Abbreviations
ACD, allergic contact dermatitis; CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; IBOA, isobornyl acrylate; IDDM, insulin-dependent diabetes mellitus; IPDI, isophorone diisocyanate; isCGM, intermittently scanned continuous glucose monitoring; MD, medical device; MDA, diaminodiphenylmethane; n, number; NR, not reported; PMDI, polymeric methylene diphenyl diisocyanate; SMBG, self-monitoring of blood glucose; T1D, type 1 diabetes; TDI, toluene diisocyanate.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Cecilia Svedman participates in the IDEA project sponsored by the International Fragrance Association (IFRA).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
