Abstract
Background:
Since the introduction of continuous subcutaneous insulin infusion (CSII), the benefits have been numerous. However, adverse events (AEs) are experienced by up to 40% of users per year, exposing them to potentially fatal risks. The available evidence on the variables that trigger AEs associated with CSII remains limited, indicating the importance of studies on the subject.
Aim:
To propose a taxonomy based on the prevalent AEs experienced by patients from a reference diabetes mellitus (DM) center in Brazil using different CSII devices.
Methods:
118 patients participated in an online interview and answered the questions of the data collection instrument. Identifying categories and subcategories of analysis contributed to constructing the AEs taxonomy.
Results:
The five analysis categories identified were: CSII User Interface (n = 45), CSII Alert System (n = 13), CSII Software and Connection (n = 11), CSII Durability (n = 30), and Electrical and Mechanical System of CSII (n = 60) A total of 159 AEs were identified, including conflicting alert messages and error/warning notification failures, errors resulting from engine malfunctions, data loss, patient interface deficiencies, button problems, and battery failure.
Conclusions:
The study describes in a taxonomic format the AEs directly associated with the use of modern CSIIs that may contribute with additional information to the Food and Drug Administration (FDA) Medical Device Report (MDR) adverse event codes. In addition to guiding educational actions in the treatment of DM and providing information for health professionals and medical device developers, prospective studies examining the frequency of such problems, including the potential psychosocial impact of this technologically advanced therapy, are needed.
Keywords
Introduction
The number of people affected by diabetes mellitus (DM) has grown by more than 46% in the last ten years. The world population is estimated to be 537 million people, reaching 783 million in 2045. 1 In Brazil, about 15.7 million people with DM represent about 7.4% of the population, growing exponentially and placing the country in the sixth position in the world ranking of people with type 1 diabetes mellitus (T1D). 1 The lack of glycemic control caused by the disease can lead to the risk of developing microvascular complications such as blindness, amputation, and renal failure,2,3.
The search for a better glycemic control stimulated researchers to study the effects caused by different forms of insulin administration. The Diabetes Control Complications Trial study 4 demonstrated that intensive therapy with continuous subcutaneous insulin infusion (CSII), when compared to conventional treatment with multiple doses of insulin (MDI), provides a significant reduction in the incidence and progression of microvascular complications.5,6 Therefore, using a CSII is an evolutionary form of therapy, mainly used to treat people with T1D of all age groups. Additionally, these pumps present self-monitoring blood glucose meters and continuous glucose monitoring (CGM) sensors. 7
However, modern technology and technological advances in the treatment of T1D do not minimize the occurrence of adverse events (AEs) related to the use of the device and classify them as everyday situations experienced by users at some point (40% of users per year). 8 The unexpected interruption of insulin infusion promotes hyperglycemia and ketoacidosis, while excessive administration of insulin causes severe hypoglycemia, and those are inherent risks to the use of the technology. 7
AEs with CSII occur during the use of the device, unlike device performance evaluations that usually take place under ideal laboratory conditions.
9
Conceptually, AEs can be “Nonmetabolic AEs” related to events in the catheter infusion set; the device itself (software or screen issue, keyboard issue, battery or component shortage, insulin leakage, water damage); or skin complications. “Metabolic AEs” resulting from diabetic ketoacidosis and severe hypoglycemia due, among other reasons, to failures in the infusion set or errors regarding “bolus” calculation.
8
The primary language in the regulation about an AE, according to Food and Drug Administration (FDA), Code of Federal Regulations (CFR) 21 part 803
10
definition is: Caused or contributed means that death or serious injury was or may have been attributed to a medical device, or that a medical device was or may have been a factor in death or serious injury, including events occurring as a result of (1) Failure, (2) Malfunction, (3) Improper or inadequate design, (4) Manufacture, (5) Labeling, or (6) User error.
However, available evidence on the safety and efficacy of CSII remains limited. Thus, it is interesting to develop a taxonomy for AEs in order to locate information through ordering and labeling data.9,10. Campos and Gomes 11 complement this view by stating taxonomy enables the organization and retrieval of information as in a conceptual map thematic. The present study aims to propose a taxonomy based on the prevalent AEs experienced by patients from a reference DM center in Brazil using different CSII devices.
Methods
The Ethics and Research Committee (CEP) of the Federal University of São Paulo (UNIFESP) approved the study (approval number: CEP/UNIFESP Project number: 1242/2019), and the data collection period was from January to June 2021. The study inclusion criteria involved children, adolescents, and adults with T1D, using any brand of CSII for at least six months, with an online signature of the Free and Informed Consent Term, and regular attendance at the Ambulatory of Insulin Pump of Federal University of São Paulo Diabetes Center.
The study consisted of a semi-structured online interview based on a data collection instrument (Supplemental Appendix 1) to investigate the potential AEs resulting from the use of three devices, Medtronic [United States of America, device 1]—MiniMed models Paradigm 722 (model A), 640G (model B), 715 (model C), Veo 754 (model D); Accu-Check Combo [Roche Diabetes Care AG, Burgdorf, Switzerland, device 2]; and Tandem Diabetes Care [United States of America, device 3].
The authors developed the questionnaire and submitted it to content validation by researchers and professionals in the research area. The analysis and documentation of the AEs were developed according to the following steps. Initially, a “collection of terms” was carried out, consisting of a consultation of the specialized literature to understand the existing expressions on the subject. Subsequently, to unify the language, these terms were standardized, and the messages sent by the patients were classified as “not relevant to the object of study,” “with distant relevance to the object of study,” or “directly related to the object of study.” In the next step, the categories and subcategories of interpretation emerged by relationships of relevance and thematic similarity, and, for that, the relationships of semantic equivalence (synonymous terms) were considered. 12
From there, five categories of analysis and their subcategories emerged: CSII User Interface (n = 45), CSII Alert System (n = 13), CSII Software and Connection (n = 11), CSII Durability (n = 30), and Electrical and Mechanical System of CSII (n = 60). The taxonomic structure was consolidated by applying a systemic view on the classification of its components. A complete mapping occurred, highlighting the links among the various categories and subcategories. Finally, we sought to compare the categories that responded to the proposed objective with the available scientific literature.
Results and Discussion
The survey involved 118 patients with T1D (71 women/47 men) aged 2 to 55 (
Demographic Data of the Study Patients Treated at the Ambulatory of Insulin Pump of Federal University of São Paulo Diabetes Center (São Paulo, Brazil, 2021).
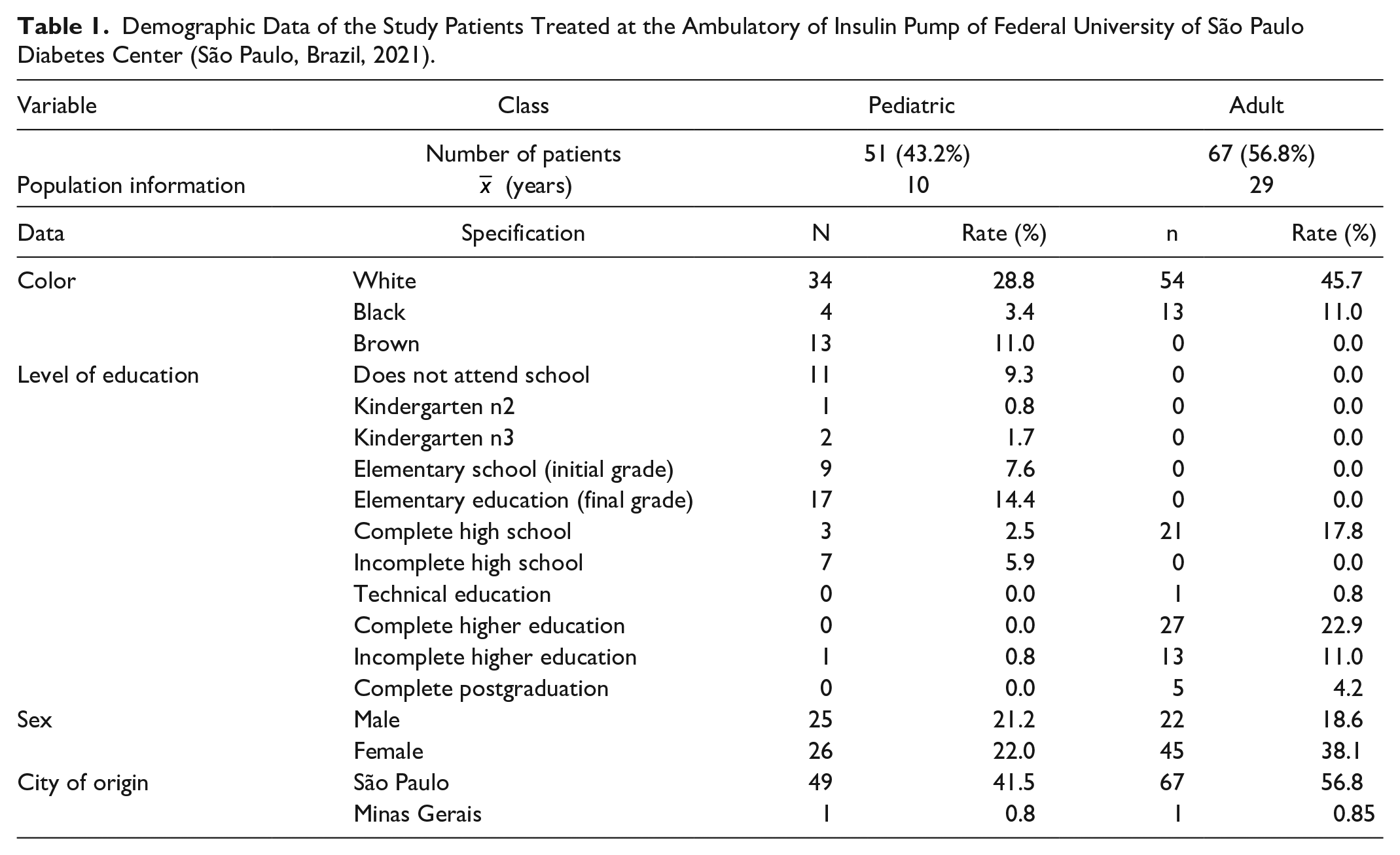
Regarding the T1D diagnostic time, 3.5% were diagnosed approximately 38 years ago; 22% 32 years, and 74.5% 22 years. The mean time of use of CSII for the pediatric and adult population were 5 years and 9 years, respectively. The time between diagnosis of T1D and the beginning of CSII was 1 to 15 years and 7 to 37 years for pediatric and adult patients, respectively (Figure 1).
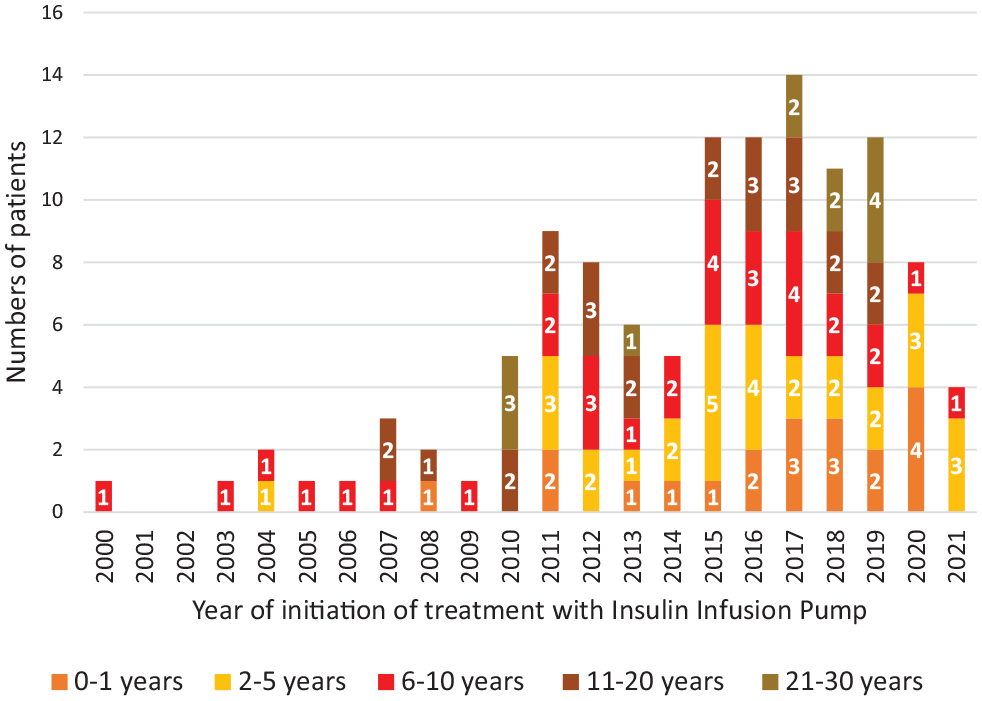
Intervals between the diagnosis of type 1 diabetes mellitus and the installation of the device for the treatment (São Paulo, Brazil, 2021).
The insulin type used in CSII was Lispro (Humalog) in 64.4% of cases; Aspart (NovoRapid) in 27.1%; 6.8% did not remember the name of the insulin at the time of the interview; 0.85% used Faster Aspart (FIASP), and 0.85% (n = 1) Glulisina (APIDRA) (Table 2). Pickup et al. 13 also showed more significant use of the Aspart and Lispro in their study.
Distribution of Patients Using Different Models and Brands of Continuous Subcutaneous Insulin Infusion Device (São Paulo, Brazil, 2021).

FDA works with the description of AEs through a set of codes organized in a hierarchical tree-like structure, where the higher-level codes (closer to the root) are more generic while the lower-level codes (leaf) are more specific. More specific parent and child codes can be considered members of the set of issues or observations described by the parent code. Thus, each set of codes attempts to accurately represent the relationship between different but similar codes (Table 3). 14
Food and Drug Administration Medical Device Report Adverse Event Codes and the Division Into the Seven Categories.

Source: https://www.fda.gov/medical-devices/mandatory-reporting-requirements-manufacturers-importers-and-device-user-facilities/mdr-adverse-event-codes (accessed on May 10, 2022).
In the present study, we are presenting five categories of analysis: CSII User Interface (n = 45), CSII Alert System (n = 13), CSII Software and Connection (n = 11), CSII Durability (n = 30), and Electrical and Mechanical System of the CSII (n = 60). A total of 159 AEs were identified, including conflicting alert messages and error/warning notification failures, errors due to engine malfunction, data loss, patient interface deficiencies, button issues, and device battery failure, the last two being very prevalent occurrences (Figure 2).

Taxonomy of device failures reported by patients treated at the Ambulatory of Insulin Pump of Federal University of São Paulo Diabetes Center (São Paulo, Brazil, 2021).
The taxonomy presented in the present investigation deals with AEs explicitly related to the CSII device. It is organized into categories of analysis in a concise, and peculiar way for the first time in the scientific literature. Among the FDA categories, two refer to the Taxonomy proposed in the present study, “Medical Device Problem” and “Medical Device Component” (malfunction, deterioration of function, failure of medical devices, and the parts and components involved or affected by the adverse event/incident of the medical device).
CSII Alert System
The following are failures related to alert messages issued by CSII, including 13 AEs, repeated message engine error (unspecified reason) (n = 3); persistent occlusion message (n = 1); error messages on the CSII screen repeatedly (unspecified reason) (n = 1); mechanical error message (abrupt interruption of operation) (n = 1); no insulin end alert (n = 2); no low battery message until CSII goes out (n = 1); motor error message due to cannula obstruction (n = 1); insulin reservoir not recognized after changing supplies (n = 2); and insulin reservoir not recognized during volume filling (n = 1).
The reports of non-occurrence of specific alarms may be related to the failure of the CSII or the user’s inattention in not realizing the alert, which leads to AEs. It is necessary to verify if this is a one-off or regular occurrence for greater understanding. In the reports of “no adm” and “engine error” alarms, probably a malfunction of the device was occurring, requiring evaluation by the manufacturer. According to FDA, malfunction means a device fails to meet its performance specifications or perform as intended. Performance specifications include all claims made in labeling the device. 10
Mechanical errors also require this assessment. Persistent occlusion messages requires immediate intervention at the focus of the problem, which may be infusion set obstruction. In these situations, it is necessary users training to identify the causal factors of the occurrence and resolution of the obstruction problem.
CSIIs Durability
In this category there are two subcategories, “Faults related to CSII physical damage by external factors” with 16 reports by patients and “Faults related to CSII physical damage by material factors” with 14 descriptions, totaling 30 AEs.
The first subcategory involves broken battery compartment (n = 3), broken display after falling (n = 2), cracked display after falling (n = 1), cracked CSII after falling (n = 1), fall with interruption of CSII operation (n = 1), CSII bottom shift after fall (n = 1), broken clip used to secure the CSII to the pants (n = 1), CSII abruptly stopped working (n = 1), CSII in improper contact with water (n = 2), cracked snap-on screen (n = 1), and battery cover deforming due to improper coin closing procedure (n = 2).
These failures are related to external factors such as humidity, dropping with physical damage to the device, or using the device within a magnetic field. Using a CSII cover to protect the equipment is recommended. The manufacturer’s recommendation is to remove the device to perform any imaging examination that has radiation. Damage to the worn cover is currently minimized by using a specific part supplied with the device and is very useful in opening the cover, avoiding unnecessary local wear.
The second subcategory refers to events related to the battery cover that does not close properly (n = 5), spring came loose in the battery compartment (n = 4), battery compartment being rusty (n = 1), cracked CSII housing (n = 2), and broken reservoir piston (n = 2). In this subclass, it was impossible to identify the real cause of the event, either due to improper use (external interference) of the device or product failure, or even the total time of use, which is an expected consequence. The patient must follow the manufacturer’s recommendation on the correct use of components for battery replacement and steps for replacing the insulin reservoir and infusion set. Nowadays, one of the CSII manufacturers has improved the battery change system, probably minimizing this problem.
However, CSIIs are electronic devices subject to errors and possible breakage or damage. Patients should always have an emergency kit with syringes and an insulin pen available for the proper treatment of DM in these circumstances.
CSII User Interface
In this category, there are two subcategories, “Failures related to damage buttons” totaling 31 reports and “Failures related to display visibility” with 14 descriptions, totaling 45 AEs.
Regarding the specification of the first subcategory, the problems were button with worn material (n = 7), excessively hard button (n = 6), button with bad contact (n = 6), loose CSII button (n = 2), broken button (n = 1), and the button does not work (n = 9). It is helpful to handle the buttons with a short nail, and cleaning the equipment buttons should be a routine of care. Routine use of the model A/device 2 would be helpful to avoid wearing out the buttons, as it has a control that the user will constantly manipulate.
The 14 faults related to visibility were poor screen visibility (scratches after CSII fall) (n = 5 ), poor screen visibility ( unspecified reason) (n = 2), poor screen visibility (display off) (n = 2), poor screen visibility (screen smudge near battery change period) (n = 1), and CSII panel crashed (button did not accept manual commands) (n = 4). The prevalent failures in this category refer to the difficulty of viewing the screen due to damage caused by dropping the device (scratches), screen with invisible information, or sudden panel lock. To minimize these occurrences and increase the device’s protection, it is recommended to use a CSII cover or a money belt to wrap the equipment.
CSII Electrical and Mechanical System
This category is composed of two subcategories of AEs, the “Faults related to the CSII mechanism” (n = 14) and “CSII battery power failure” (n = 46).
The first subcategory AEs was engine error message with CSII crash (n = 1); engine error message with lower cartridge ejection (n = 1); motor in continuous rotation without insulin volume in the reservoir (n = 1); mechanical problem (unspecified reason) (n = 2); engine malfunction interrupted insulin infusion (n = 2); locked piston (n = 3); and insulin syringe piston not rewound (n = 4). Problems related to the CSII motor can be related to the CSII time of use and pump misuse; in these situations, contact the manufacturer to perform tests to evaluate the pump operation.
CSII battery power failure (expected life battery low) (n = 37), CSII battery power failure (new battery with low battery message) (n = 6), CSII battery power failure ((using a different brand than recommended) (n = 1), CSII battery power failure due to dirt in the battery compartment (n = 1), and impaired insulin ejection due to poor CSII battery power (n = 1) are AEs reported in the second subcategory. Problems related to the CSII battery were, generally very frequent, mainly related to the low expected life battery.
According to FDA, 10 the expected life of a device means the time a device is expected to remain functional after being put into use. Power One battery (device 2) has an expected life of 30 to 60 days, and Energizer (device 1) 20 to 30 days (CSII with CGM sensor). In another study, the end of battery expected life within 72 hours was reported most frequently (50%) with 201 members of the Association for Innovative Diabetes Treatment in Japan. 15 In this study, the mean number of CSII exchanges due to battery failure per patient was 1.37. This corresponds to almost one-third of the data reported in a New Zealand center study in which 84% of subjects reported one or more AEs, with an overall AE incidence due to battery failure equivalent to 3.42 per person/year. 16
The low battery was one of the most reported issues in this study, and among the most observed errors are power interruption, empty battery, and empty cartridge. A 2017 study evaluated the impact of targeted education on CSII warnings and errors in improving competence to resolve alarms and found educational utility in improving response to pump alarms, 17 highlighting the importance of the interdisciplinary educational work with DM patients.
CSII Software and Connection
Error when saving data (n = 2), unexpected automatic data reset (n = 2), information reset when changing the battery (n = 1), data loss after an abrupt shutdown of the pump (n = 1), the problem of Bluetooth connection between CSII and CSII controller (model A—device 2) (n = 1), and connection problem via radio frequency between the CGM sensor and the CSII (device 1) (n = 4) were failures related to the transfer of information.
In general, they are problems related to failures in the Bluetooth connection or problems with equipment with the CGM sensor or even the device’s control (device 2—model A). It can also be a problem related to sudden loss of data, which requires immediate evaluation by the manufacturer. In 2019, one study looked at the behavior of two sensors, CSIIs and two CGMs, and observed that with Dexcom CGMs, users complained about the inaccurate result. Regarding Abbott Freestyle, the most frequent AE was an allergic skin reaction. 18 Failures in the radio frequency connection can happen due to battery power failure, sensor bending, outdated transmitter (generally, the durability is around 12 months), or external frequency interference.
Conclusions
As far as we know, this is the first taxonomy in the scientific literature on the subject presented in a concise and organized way. The data can guide future actions in the education and treatment of DM and serve as a guide to improve the list of potential AEs associated with using the device. Additionally, it provides critical information for researchers, practitioners, and medical device developers, directing them to device enclosures that need more strength and adjustment to minimize user-reported issues. However, this process does not end there. It is necessary to maintain and periodically review the taxonomy, verify the inclusion of new terms and new classification needs, and not lose the benefits generated by the organization of information.
It is worth mentioning that the research objective is not limited to the mentioned AEs, as new scenarios, new technologies, and new consumption requirements bring with them new flaws. Among the study’s limitations, one is related to the absence of description regarding the clinical outcomes due to the AEs. In addition, a prospective study of AEs resulting from the use of the device would be valuable to better clarify the frequency of such problems, including the potential psychosocial impact of technologically advanced therapy. Finally, it is impossible to quantify how many of the AEs required user action to solve the problem with the data identified in this research.
Supplemental Material
sj-docx-1-dst-10.1177_19322968221106196 – Supplemental material for Insulin Pump-Associated Adverse Events in a Brazilian Reference Center for the Treatment of Diabetes Mellitus: Proposal for a Taxonomy of Device Failures in Adults, Adolescents, and Children
Supplemental material, sj-docx-1-dst-10.1177_19322968221106196 for Insulin Pump-Associated Adverse Events in a Brazilian Reference Center for the Treatment of Diabetes Mellitus: Proposal for a Taxonomy of Device Failures in Adults, Adolescents, and Children by Ana Lúcia Domingues Neves, Luiz Eduardo Galvão Martins, Mônica Andrade Lima Gabbay, Gabriela Cavicchioli, Fernanda Silva Tenorio and Tatiana Sousa Cunha in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
We would like to thank all patients with Type 1 diabetes mellitus and the team for their unrestricted support in data collection.
Abbreviations
AEs, adverse events; CEP, Ethics and Research Committee; CFR, Code of Federal Regulations; CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; DM, diabetes mellitus; FDA, Food and Drug Administration; MDI, multiple doses of insulin; MDR, medical device report; T1D, type 1 diabetes mellitus; UNIFESP, Federal University of São Paulo;
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Coordination for the Improvement of Higher Education Personnel - Brazil (CAPES, Finance Code 001), Research Support Foundation of the State of São Paulo (FAPESP nº 2018/22094-0) and National Council for Scientific and Technological Development (CNPq 308721/2020-9).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
