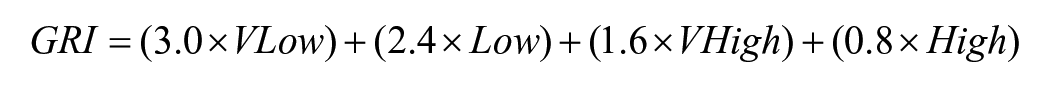Abstract
Background:
The Glycemia Risk Index (GRI) was developed in adults with diabetes and is a validated metric of quality of glycemia. Little is known about the relationship between GRI and type 1 diabetes (T1D) self-management habits, a validated assessment of youths’ engagement in habits associated with glycemic outcomes.
Method:
We retrospectively examined the relationship between GRI and T1D self-management habits in youth with T1D who received care from a Midwest pediatric diabetes clinic network. The GRI was calculated using seven days of continuous glucose monitor (CGM) data, and T1D self-management habits were assessed ±seven days from the GRI score. A mixed-effects Poisson regression model was used to evaluate the total number of habits youth engaged in with GRI, glycated hemoglobin A1c (HbA1c), age, race, ethnicity, and insurance type as fixed effects and participant ID as a random effect to account for multiple clinic visits per individual.
Results:
The cohort included 1182 youth aged 2.5 to 18.0 years (mean = 13.8, SD = 3.5) comprising 50.8% male, 84.6% non-Hispanic White, and 64.8% commercial insurance users across a total of 6029 clinic visits. Glycemia Risk Index scores decreased as total number of habits performed increased, suggesting youth who performed more self-management habits achieved a higher quality of glycemia.
Conclusions:
In youth using CGMs, GRI may serve as an easily obtainable metric to help identify youth with above target glycemia, and engagement/disengagement in the T1D self-management habits may inform clinicians with suitable interventions for improving glycemic outcomes.
Introduction
Individuals with type 1 diabetes (T1D) increasingly report using continuous glucose monitors (CGMs) to track glycemia in real time. 1 Continuous glucose monitors offer insights into glycemia, or the time spent in target range as opposed to time spent above or below range, and optimal glycemia is associated with improved long-term outcomes. 2 A variety of metrics may be used to describe glycemia, many of which involve multiple numbers or components to be evaluated wholistically. The American Diabetes Association endorses seven characteristics to describe glycemia referred to as the Ambulatory Glucose Profile. 3 While these metrics provide a comprehensive overview of glycemia, clinicians and researchers have noted key drawbacks to the Ambulatory Glucose Profile metrics such as the difficulty in monitoring and creating care plans based on seven measures. These barriers have resulted in the development of new metrics to summarize glycemia.
The Glycemia Risk Index (GRI) was developed based on 330 clinicians’ rankings of 2-week CGM tracings from 225 adults with diabetes. 4 Tracings were ranked from most optimal to least optimal glycemia, and analyses of rankings resulted in a composite metric comprised of weighted sums of time spent in very low-glucose hypoglycemia (<54 mg/dL; <3.0 mmol/L), low-glucose hypoglycemia (54 to <70 mg/dL; 3.0-3.9 mmol/L), high-glucose hyperglycemia (>180-250 mg/dL; >10.0-13.9 mmol/L), and very high-glucose hyperglycemia (>250 mg/dL; >13.9 mmol/L). The low component of the GRI—reflecting time spent in low-glucose hypoglycemia and very low-glucose hypoglycemia—was weighted more heavily than the high component of the GRI (i.e., time spent in high-glucose hyperglycemia and very high-glucose hyperglycemia). Low GRI values reflect more optimal quality of glycemia while high values indicate suboptimal quality of glycemia where quality of glycemia refers to the proportions of time spent in very low-glucose hypoglycemia, low-glucose hypoglycemia, high-glucose hyperglycemia, and very high-glucose hyperglycemia concentrations. 4 Altogether, the GRI offers a condensed version of some of the Ambulatory Glucose Profile metrics using at least one or two weeks of CGM data, increasing ease and usability in clinic as well as insights between clinic visits.
The GRI was developed in an adult population; however, patterns of glycemia, as reflected in metrics such as hemoglobin A1c (HbA1c), are known to differ in adults versus children. 5 Studies that have examined the GRI in youth with T1D showed that GRI scores differed between treatment modalities,6-14 so the GRI metric appears to be sensitive to changes in care when assessed as an outcome; however, it is unclear how the GRI relates to other indicators of glycemia. We aimed to examine how GRI values may relate to T1D self-management habits in youth using CGMs. Recently, Lee and colleagues developed a composite metric that summarizes individuals’ engagement with six diabetes self-management behaviors (called the “Six Habits”) that are associated with high quality glycemia. 15 These “Six Habits” include the following: (1) using a CGM or checking blood glucose at least four times/day, (2) giving ≥three rapid-acting user-initiated insulin boluses per day, (3) using an insulin pump, (4) delivering user-initiated boluses prior to meals, (5) reviewing glucose data between clinic visits, and (6) changing insulin doses since the last diabetes clinic visit. Individuals that report consistent engagement with these behaviors show improved glycemia as evidenced by greater time spent in target range (70-180 mg/dL; 3.9-10.0 mmol/L) and lower HbA1c levels. 15
We retrospectively analyzed electronic health record and diabetes data from youth with T1D using CGMs receiving care from a network of pediatric diabetes clinics in the Midwest USA. We analyzed the GRI as a predictor of the total number of habits engaged in a model that includes demographic variables and HbA1c. In this study, we also evaluated consumption of fruits and vegetables as a novel seventh habit, to assess whether self-reported consumption of a healthy diet may also impact the GRI. Altogether, we sought to examine the relationship between the GRI and T1D diabetes self-management habits to evaluate whether the GRI metric is sensitive to or reflective of engagement in the behaviors in children and young adults using CGMs. We hypothesized that lower GRI values would be associated with higher habit scores. The GRI metric may offer a lower-burden and more timely way to track glycemia at an individual and population health level. This may provide heath care providers and individuals with diabetes with insights between regularly scheduled clinic visits, possibly improving timely identification, monitoring, and intervention to reduce dysglycemia and its short- and long-term complications.
Methods
Participants
Data were extracted from an electronic health record database between December 2019 and November 2023 for 2844 individuals receiving care from a network of pediatric diabetes clinics in the Midwest USA. Continuous glucose monitor data were collected via API (application programming interface) or flat file export and stored in a secured cloud storage account. Seven days of CGM data were used to calculate the GRI. To be included in the analysis, individuals had to meet the following criteria: (1) 7-day window of CGM data with ≥50% of wear time (i.e., an individual’s CGM device recorded data for at least half of the total possible glucose readings in the window); (2) T1D duration ≥1 year; (3) age ≤18 years; (4) HbA1c measurement ±21 days from the GRI calculation; (5) assessment of at least three self-management habits completed ±seven days from the GRI calculation. Individuals diagnosed with other types of diabetes (e.g., cystic fibrosis-related diabetes) were excluded from this cohort.
Continuous glucose monitor wear time was calculated by dividing the total number of recorded glucose readings by the total number of possible readings based on the type of technology used. For CGM devices that measure glucose every 5 minutes, the total possible number of glucose readings is 288 (or 2016 per week). For CGM devices that measure glucose every 15 minutes, the total possible number of glucose readings is 96 per day (or 672 per week). Type 1 diabetes duration was calculated by subtracting the clinic encounter date from each individual’s documented T1D diagnosis date.
Figure 1 shows the population for the analysis. Individuals were excluded if they were diagnosed with other types of diabetes such as T2D or cystic fibrosis-related diabetes, resulting in 1182 eligible individuals with T1D with an age range of 2.5 to 18 years. There were a total of 6029 observations. Clinical data were coded and collected in an Institutional Review Board (IRB)-approved research data repository (IRB #11120355) that met the requirements for a waiver of written informed consent. This study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline. 16
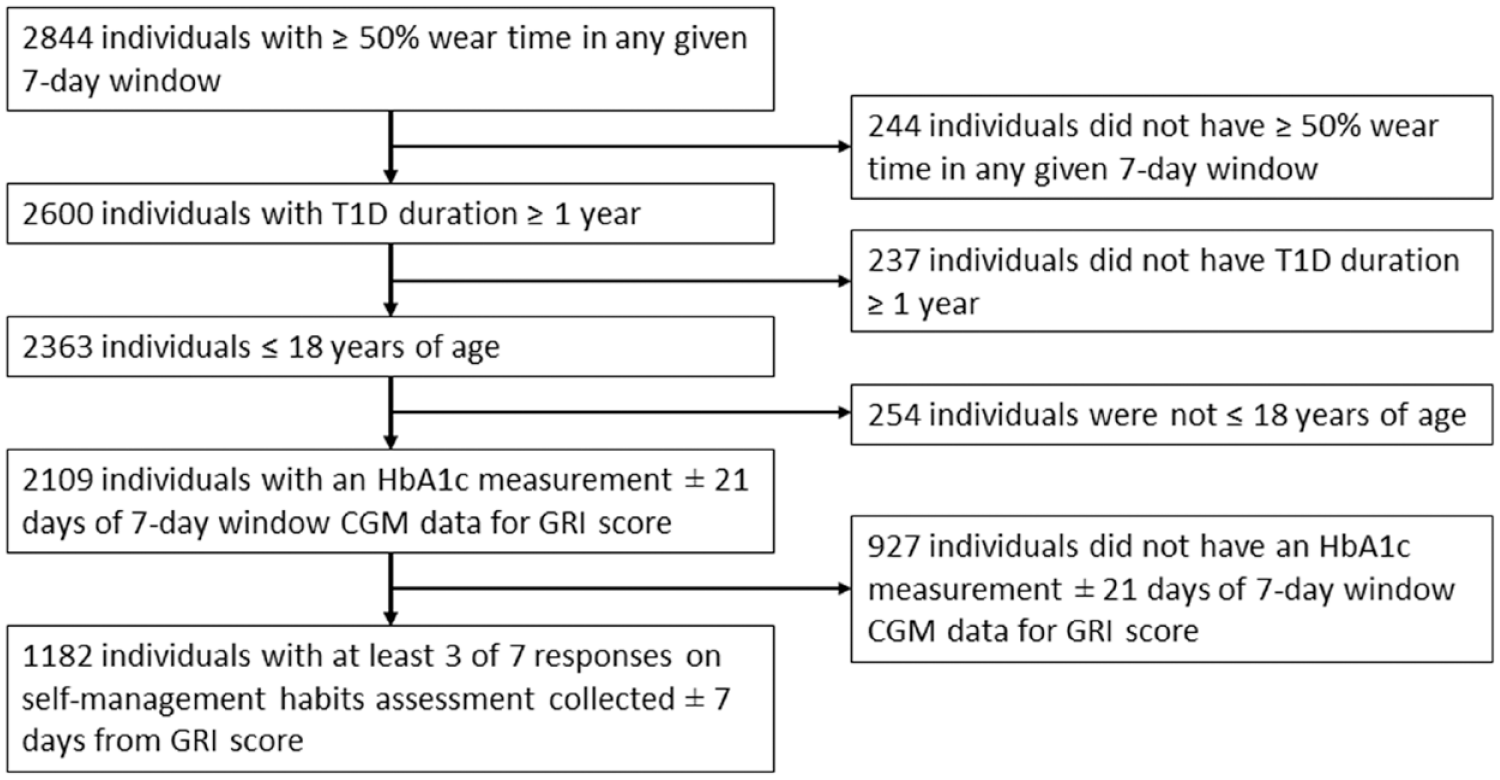
Cohort identification flowchart.
Outcomes
The GRI was calculated according to the following equation:
where VLow represents time spent in very low-glucose hypoglycemia (<54 mg/dL; <3.0 mmol/L), low represents time spent in low-glucose hypoglycemia (54 to <70 mg/dL; 3.0-3.9 mmol/L), High represents time spent in high-glucose hyperglycemia (>180 to 250 mg/dL; >10.0 to 13.9 mmol/L), and VHigh represents time spent in very high-glucose hyperglycemia (>250 mg/dL; >13.9 mmol/L). A GRI score was calculated for each seven-day window of CGM data where all inclusion criteria were met. Low GRI scores reflect optimal quality of glycemia while high GRI scores reflect suboptimal quality of glycemia. Multiple observations per individual were included when data were available and all inclusion criteria were met.
For the self-management habits, each habit was coded as 1 if the individual engaged with the habit and 0 if the individual did not engage with the habit. The “Six Habits” were assessed using the same methodology as that described by Lee et al’s 15 original report. Briefly, performance of Habits 1-3 was determined based on clinicians’ assessments of each individual’s 14-day diabetes device(s) download(s) shared with the clinic. For Habits 4 to 6 (and in cases where device data were not available), habit performance was assessed through self-report (or through proxy-report by a parent). During clinic visits, diabetes educators assessed performance of the seventh (novel) habit by documenting a “yes” or “no” response after asking whether the family engaged in eating a healthy diet. In general, proxy-reporting by parents was used to answer this question; however, older youth who generally managed their own diabetes care could also answer this question. A total habit score was calculated by adding together the number of habits performed by each person at a clinic visit. An individual who performed all seven habits had a total habit score of 7. Because all individuals included in the analysis used a CGM device by definition they were classified as performing Habit 1 (checking blood glucose at least four times/day or use of a CGM), so all individuals had a minimum score of 1 for the total habit score.
Statistical Analysis
Analysis was conducted in R (Version 4.1.2-1.2004.0). 17 A mixed-effects Poisson regression model examined how GRI associated with total habit scores. Glycemia Risk Index, age, race, ethnicity, insurance type, and HbA1c were entered as fixed effects, and participant ID was entered as a random effect to account for multiple observations per individual. Hemoglobin A1c was included as a covariate in the model because we were interested in evaluating GRI’s predictive potential above and beyond HbA1c. Categorical variables (race, ethnicity, insurance type) were encoded using effect coding, and continuous variables (age, HbA1c, GRI) were mean-centered. Race was condensed into a three-level categorical variable (White, Black, or African American, and Other [American Indian or Alaska Native, Asian, Hispanic, Multiracial, Declined, Other]). Ethnicity was condensed into a two-level categorical variable as well (non-Hispanic/non-Latino/Declined and Hispanic/Latino). Insurance type was condensed into a two-level categorical variable (commercial or public/self-pay). We conducted two sensitivity analyses to evaluate the impact of increased CGM wear time and insurance type grouping on the relationships observed between GRI and habit engagement (Supplemental Tables 1 and 2). First, we assessed the impact on GRI and habit engagement when the CGM wear time threshold was increased to ≥70% (Supplemental Table 1), based on the recommendations of previous studies of CGM wear time thresholds.18-20 Then, we assessed the impact on GRI and habit engagement when individuals who used “self-pay” as their primary insurance type were not included in the model, leaving only individuals who used commercial or public insurance types (Supplemental Table 2). The proportion of individuals who were self-pay was relatively small, so the sensitivity analysis examined the impact of grouping this insurance payment type with public insurance.
Results
Demographic characteristics of our cohort (N = 1182) are reported in Table 1. The cohort included 581 females and 601 males with T1D (mean age [SD], 13.8 [3.5] years). There were a mean [SD] of 5.1 [3.8] observations per person and a median [IQR] of 4.0 [5.0] observations per person. The number of observations ranged from 1 to 22 observations per individual included in the analysis. Table 2 contains mean and standard deviation values of the total habit score, the GRI, the GRI high component, the GRI low component, and HbA1c across all observations used in the analysis. Few individuals were engaged in all seven habits. Based on the inclusion criteria, the minimum possible total habit score was 1 because all individuals used CGM devices satisfying the criterion for Habit 1, but we found that the lowest total habit score was 2, indicating that all individuals engaged in at least one other habit in addition to Habit 1. Table 3 shows individual habit engagement and the proportion of observations where the individual was engaged in the habit to observations where the individual was not engaged in the habit or no response was recorded. The proportion of engagement to disengagement/no response was higher for Habits 1-4 than Habits 5 to 7, suggesting individuals were less likely to engage in reviewing glucose data between clinic visits, changing insulin doses between clinic visits, and consuming a healthy diet.
Participant Demographics.

Information was reported as of each individual’s most recent observation (when an individual had multiple observations).
Average Habit, GRI, and HbA1c Values.
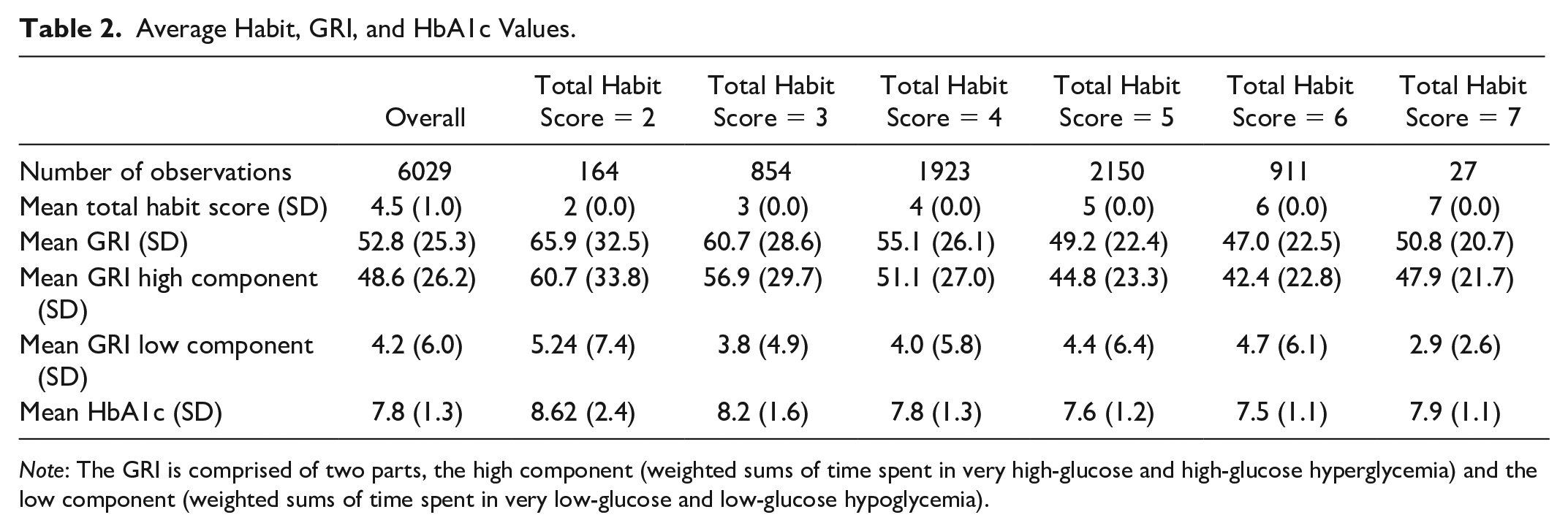
Note: The GRI is comprised of two parts, the high component (weighted sums of time spent in very high-glucose and high-glucose hyperglycemia) and the low component (weighted sums of time spent in very low-glucose and low-glucose hypoglycemia).
Individual Habit Engagement.
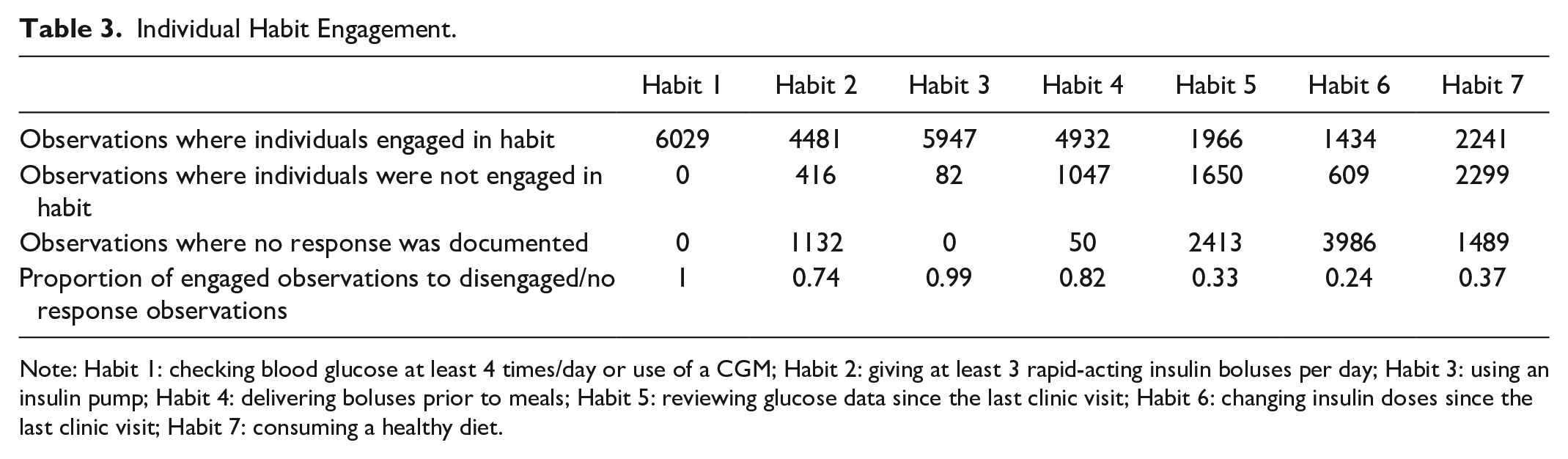
Note: Habit 1: checking blood glucose at least 4 times/day or use of a CGM; Habit 2: giving at least 3 rapid-acting insulin boluses per day; Habit 3: using an insulin pump; Habit 4: delivering boluses prior to meals; Habit 5: reviewing glucose data since the last clinic visit; Habit 6: changing insulin doses since the last clinic visit; Habit 7: consuming a healthy diet.
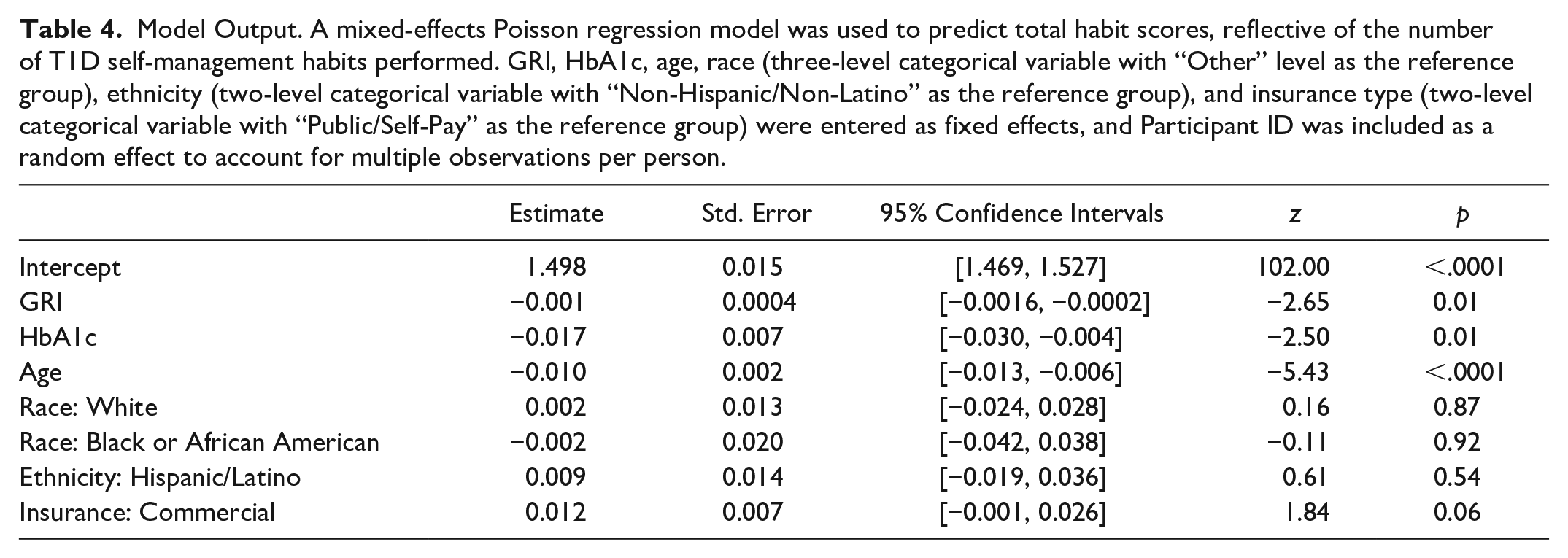
There was a significant main effect of HbA1c (z = −2.50, p = .01). Hemoglobin A1c decreased as habit scores increased (Figure 2). In addition, GRI was a significant predictor of habit scores (z = −2.65, p = .01). As the total habit score increased, the GRI score decreased. Individuals who had optimal quality of glycemia as indicated by a lower GRI score performed more T1D self-management habits, which was consistent with our hypothesis (Figure 3). Altogether, the GRI was predictive of the total habit scores when included in a model with other covariates (Table 4). Sensitivity analyses indicated that GRI remained an independent predictor of the number of self-management habits performed when the CGM wear time threshold was increased to ≥70%, as well as when individuals whose primary insurance type was “self-pay” were dropped from the model (Supplemental Tables 1 and 2).
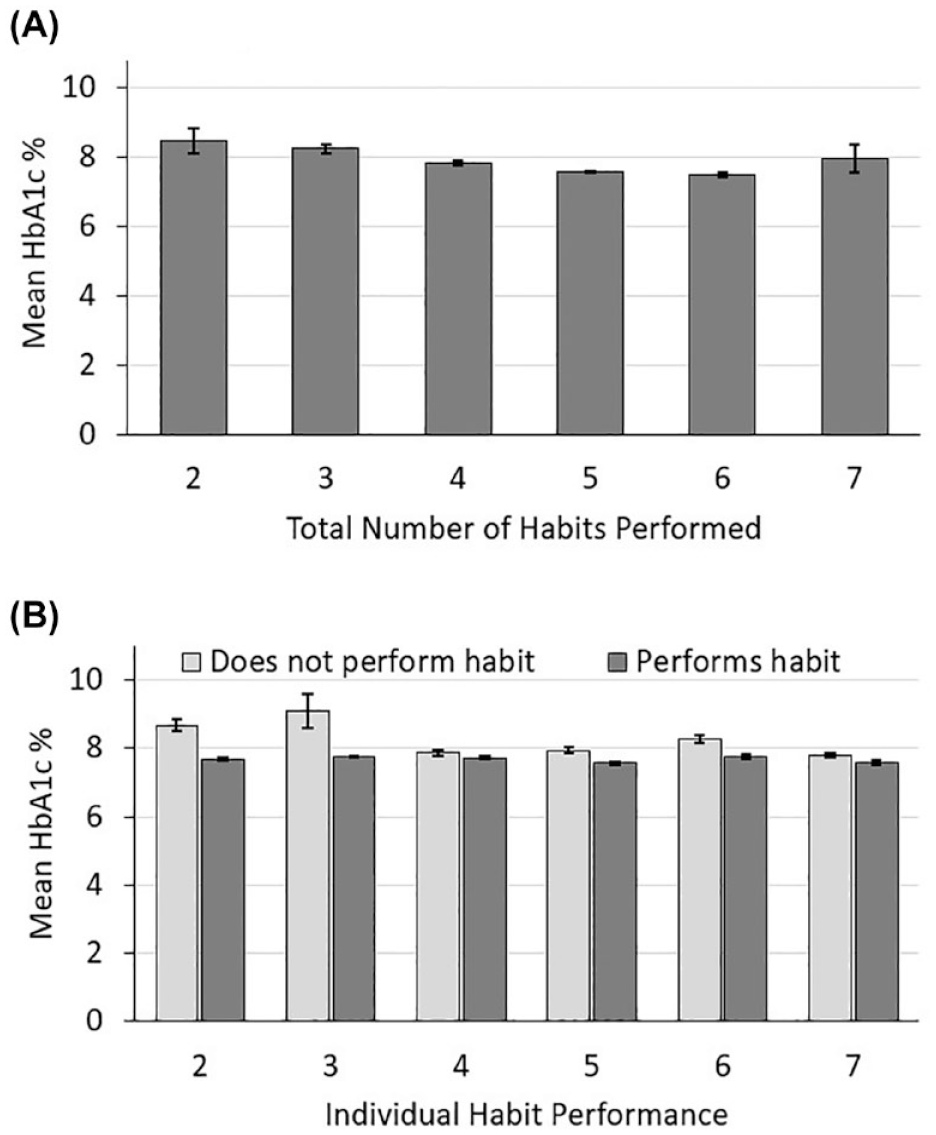
Mean hemoglobin A1c (HbA1c) levels by total habit score or total number of habits performed (A) and by each habit (B). Error bars reflect 95% confidence intervals. Note that all individuals included in analysis engaged in Habit 1, since all individuals used a continuous glucose monitor (CGM). Habit 1: checking blood glucose at least 4 times/day or use of a CGM; Habit 2: giving at least 3 rapid-acting insulin boluses per day; Habit 3: using an insulin pump; Habit 4: delivering boluses prior to meals; Habit 5: reviewing glucose data since the last clinic visit; Habit 6: changing insulin doses since the last clinic visit; Habit 7: consuming a healthy diet.
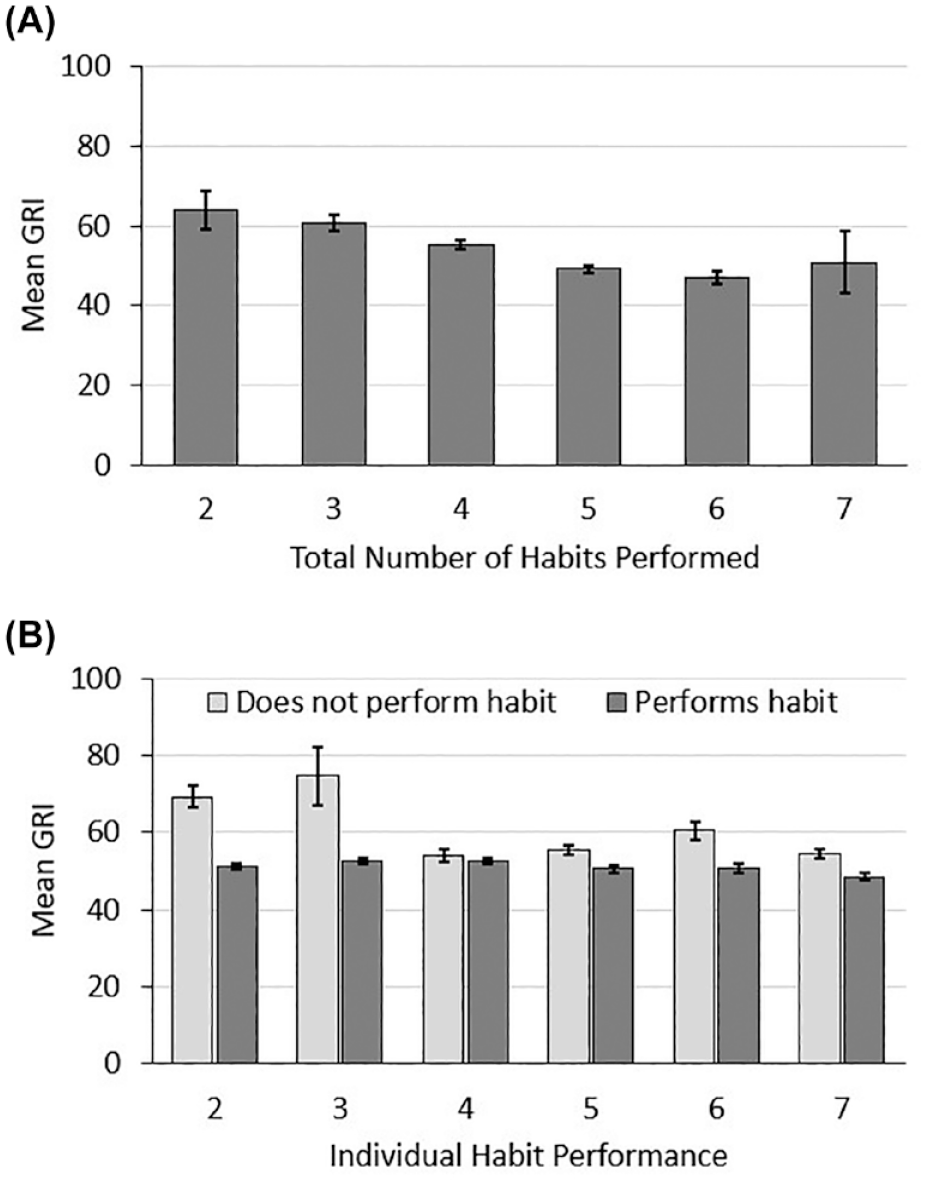
Mean Glycemia Risk Index (GRI) by total habit score or total number of habits performed (A) and by each habit (B). Error bars reflect 95% confidence intervals. Note that all individuals included in analysis engaged in Habit 1, since all individuals used a continuous glucose monitor (CGM). Habit 1: checking blood glucose at least 4 times/day or use of a CGM; Habit 2: giving at least 3 rapid-acting insulin boluses per day; Habit 3: using an insulin pump; Habit 4: delivering boluses prior to meals; Habit 5: reviewing glucose data since the last clinic visit; Habit 6: changing insulin doses since the last clinic visit; Habit 7: consuming a healthy diet.
Model Output. A mixed-effects Poisson regression model was used to predict total habit scores, reflective of the number of T1D self-management habits performed. GRI, HbA1c, age, race (three-level categorical variable with “Other” level as the reference group), ethnicity (two-level categorical variable with “Non-Hispanic/Non-Latino” as the reference group), and insurance type (two-level categorical variable with “Public/Self-Pay” as the reference group) were entered as fixed effects, and Participant ID was included as a random effect to account for multiple observations per person.
Discussion
In a cohort of youth with T1D who used CGMs, the GRI decreased as the total number of habits performed increased, suggesting that youth who performed more T1D self-management habits had a higher quality of glycemia. Likewise, HbA1c decreased as the total number of habits performed increased. We found that few individuals were engaged in all seven habits, but the lowest total habit score was 2, suggesting youth with T1D engaged in at least one other habit in addition to Habit 1 (checking blood glucose at least 4 times/day or using a CGM device). Habits 1 to 4 had higher rates of engagement compared to Habits 5 to 7, meaning fewer individuals reviewed glucose data between clinic visits, changed insulin doses between clinic visits, and consumed a healthy diet. The GRI scores were readily calculated from seven-day windows of CGM data, which is particularly important given the increasing use of CGM devices.1,5 The GRI metric can also be dynamically updated every 7 to 14 days, summarizing one to two weeks of CGM data in contrast to HbA1c which summarizes three to four months of glucose data but at a single time point. In sum, the GRI may serve as an easily obtainable metric to help identify youth using CGMs with above or below target glycemia, and engagement/disengagement in the T1D self-management habits may provide healthcare workers with candidate interventions for improving glycemic outcomes.
The present results support the original report of the T1D self-management habits assessment that demonstrated that engagement in the “Six Habits” was associated with higher time spent in target range and lower HbA1c levels, 15 which we also reported in this study. The original report of the T1D self-management habits showed that a small percentage of individuals performed all six habits and identified Habits 5 and 6 as areas for improvement. 15 In this study, we also found that few individuals engaged in all seven habits, and engagement in Habits 5 to 7 was lower than Habits 1 to 4. We added a seventh habit for consuming a healthy diet to examine the relationships between diet, GRI, and HbA1c. While there were small differences in HbA1c for this novel habit, the GRI appears to be more sensitive, showing a greater differentiation between individuals who performed this habit and those that did not. The GRI may be more sensitive for this habit than HbA1c because HbA1c is typically used to assess glycemia over the previous 3 to 4 months where the GRI reflects glycemia over periods of time as short as seven days or greater. 21 Taken together, the GRI may offer insights into glycemia between regularly scheduled clinic visits and HbA1c measurements, and the T1D self-management habits may be ideal interventions for improving glycemia.
Since GRI can only be calculated in individuals using CGM, all individuals included in this study used CGM. Most individuals in this cohort also used automated insulin delivery (AID) systems. In the original “Six Habits” report, sensitivity analyses showed similar habit engagement patterns in individuals who did versus did not use AID systems. 15 Even so, the use of advanced technologies such as AID is known to impact behaviors related to bolusing. 15 Automated insulin delivery systems’ timing and dosing of insulin boluses may provide added glycemic benefits and tend to make delivery of insulin easier, compared to having no insulin pump.22,23 Our results show that individuals engaged in Habit 2 or Habit 3 experienced improved HbA1c and GRI compared to individuals that did not engage in these habits. Future research may consider the extent to which different AID technologies may impact performance of Habit 2.
Our results suggest that bolus delivery (i.e., giving a needed bolus) has a larger impact on glycemic outcomes than bolus timing. We noted little difference in mean HbA1c and mean GRI between those that reported delivering boluses prior to meals (Habit 4) and those that did not. We observed larger differences in these glycemic outcomes in individuals that reported giving, versus not giving, at least three rapid-acting boluses per day (Habit 2). Of note, performance of Habit 4 is not predicated on giving boluses a certain amount of time prior to meals. The onset of action of rapid-acting insulin is approximately 15 minutes; therefore, the timing of bolus delivery (e.g., giving insulin right before a meal, versus 15 minutes prior) does impact glycemic outcomes. Future research is needed to evaluate whether Habit 4 should be further refined to better account for the impact of bolus timing on postprandial glycemia.
We acknowledge several limitations of this study. All individuals included in the analysis were using a CGM device so that the GRI could be calculated from the CGM data, so this analysis does not capture the entire pediatric population. Similarly, most individuals in this cohort were using advanced technologies for diabetes management, which is not necessarily reflective of the entire population of pediatric individuals with T1D. Stark disparities in access to and continued use of these technologies in youth with T1D remain prevalent in the United States.24,25 Our findings may not generalize to individuals who do not use or have access to the advanced technologies that were so frequently used by the cohort evaluated in this study. We also chose to include individuals with a CGM wear time of at least 50% (equivalent to 3.5 days of data in a 7-day window); therefore, these results may not generalize to individuals with lower CGM wear time. Sensitivity analyses (see Supplemental Materials) showed that the relationship between GRI and habit engagement did not change when the CGM wear time threshold was increased to ≥70%. In addition to this criterion, we selected individuals with a T1D duration of one year or more. More research is needed to understand how the GRI describes glycemia in the first year following diagnosis, including a “honeymoon period” where symptoms of T1D may decrease. 26 Similarly, to better understand the exploratory insight reported here between a healthy diet and GRI, further research is needed to refine and validate the novel seventh habit pertaining to consumption of a healthy diet.
Conclusions
The GRI associated with the T1D self-management habits assessment, offering further validation for both measures. The GRI may serve clinicians with insights into glycemia beyond HbA1c as it can be calculated from as little as one to two weeks of CGM data and dynamically updated. The GRI metric may be easily obtained between regularly scheduled clinic visits and HbA1c measurements, which could improve efforts to identify and offer more timely interventions to youth using CGMs with above or below target glycemia. Clinicians may consider the T1D self-management habits as suitable intervention candidates for improving quality of glycemia in youth with T1D.
Supplemental Material
sj-docx-1-dst-10.1177_19322968241247215 – Supplemental material for The Glycemia Risk Index Predicts Performance of Diabetes Self-Management Habits in Youth With Type 1 Diabetes Mellitus
Supplemental material, sj-docx-1-dst-10.1177_19322968241247215 for The Glycemia Risk Index Predicts Performance of Diabetes Self-Management Habits in Youth With Type 1 Diabetes Mellitus by Kelsey Panfil, Craig A. Vandervelden, Brent Lockee, Erin M. Tallon, David D. Williams and Joyce M. Lee in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-2-dst-10.1177_19322968241247215 – Supplemental material for The Glycemia Risk Index Predicts Performance of Diabetes Self-Management Habits in Youth With Type 1 Diabetes Mellitus
Supplemental material, sj-docx-2-dst-10.1177_19322968241247215 for The Glycemia Risk Index Predicts Performance of Diabetes Self-Management Habits in Youth With Type 1 Diabetes Mellitus by Kelsey Panfil, Craig A. Vandervelden, Brent Lockee, Erin M. Tallon, David D. Williams and Joyce M. Lee in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors thank Susana Patton, PhD (Nemours Children’s Health, Jacksonville, FL, USA) Cinyta Schweisberger, DO (Children’s Mercy Kansas City, Kansas City, MO, USA), Sarah Corathers, MD (Cincinnati Children’s Hospital Medical Center, Cincinnati, OH, USA) and David Klonoff, MD (Mills-Peninsula Medical Center, San Mateo, CA, USA) for providing feedback related to this work.
Abbreviations
CGM, continuous glucose monitor; GRI, Glycemia Risk Index; HbA1c, glycated hemoglobin A1c; T1D, type 1 diabetes.
Author Contributions
KP: Formal Analysis, Writing—Original Draft, Writing—Review & Editing.
CAV: Data Curation, Writing—Review & Editing.
BL: Conceptualization, Writing—Review & Editing, Supervision.
EMT: Writing—Review & Editing.
DDW: Writing—Review & Editing.
JML: Writing—Review & Editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Leona M. and Harry B. Helmsley Charitable Trust foundation (grant nos. G-2017PG-T1D019 and 2008-04043). Dr. Lee is funded by P30DK089503 (MNORC), P30DK020572 (MDRC), and P30DK092926 (MCDTR) from the National Institute of Diabetes and Digestive and Kidney Diseases and the Elizabeth Weiser Caswell Diabetes Institute at the University of Michigan.
ORCID iDs
Data Sharing Statement
The data that support the findings of this study are available from the corresponding author, KP, upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.