Abstract
Background:
We evaluated the feasibility of real-time continuous glucose monitoring (CGM) for titrating continuous intravenous insulin infusion (CII) to manage hyperglycemia in postoperative individuals in the cardiovascular intensive care unit and assessed their accuracy, nursing acceptance, and postoperative individual satisfaction.
Methods:
Dexcom G6 CGM devices were applied to 59 postsurgical patients with hyperglycemia receiving CII. A hybrid approach combining CGM with periodic point-of-care blood glucose (POC-BG) tests with two phases (initial-ongoing) of validation was used to determine CGM accuracy. Mean and median absolute relative differences and Clarke Error Grid were plotted to evaluate the CGM accuracy. Surveys of nurses and patients on the use of CGMs experience were conducted and results were analyzed.
Results:
In this cohort (mean age 64, 32% female, 32% with diabetes) with 864 paired POC-BG and CGM values analyzed, mean and median absolute relative difference between POC-BG and CGM values were 13.2% and 9.8%, respectively. 99.7% of paired CGM and POC-BG were in Zones A and B of the Clarke Error Grid. Responses from nurses reported CGMs being very or quite convenient (n = 28; 93%) and it was favored over POC-BG testing (n = 28; 93%). Majority of patients (n = 42; 93%) reported their care process using CGM as being good or very good.
Conclusion:
This pilot study demonstrates the feasibility, accuracy, and nursing convenience of adopting CGM via a hybrid approach for insulin titration in postoperative settings. These findings provide robust rationale for larger confirmatory studies to evaluate the benefit of CGM in postoperative care to improve workflow, enhance health outcomes, and cost-effectiveness.
Keywords
Introduction
Postcardiac surgical patients in the cardiovascular intensive care unit (CV-ICU) often require continuous intravenous insulin infusion (CII) for managing hyperglycemia and thus require close glucose monitoring.1,2 Hyperglycemia, in both individuals with and without diabetes, has been associated with increased rates of major complications including wound infections, acute renal failure, extended hospitalization, and higher perioperative mortality.3,4 Furnary et al demonstrated that CII reduced the risk of absolute mortality by 57% and risk-adjusted mortality by 50% in individuals who underwent cardiac bypass surgery in the intensive care unit (ICU). 5 Based on prior evidence, professional clinical guidelines recommend CII for managing hyperglycemia in the ICU as standard of care.6,7 However, CII requires hourly point-of-care blood glucose (POC-BG) testing7-9 and increases the risk of hypoglycemia (up to 32%) in individuals in the ICU.10,11 Hypoglycemia in critically ill patients is also linked to an increased risk of complications, prolonged hospitalization, and higher mortality rates.11,12 Additional difficulties arise due to the discomfort associated with hourly blood glucose checks, particularly in the dynamic setting of critically ill patients. Furthermore, these POC-BG checks consume valuable resources such as blood, nursing time, test strips, lancets, and more.
Current available continuous glucose monitoring (CGM) devices measure glucose levels in interstitial fluid every 5 minutes and are factory calibrated with pre-validated accuracy or mean absolute relative difference (MARD) of less than 10%. 13 CGM devices monitor glucose trends and therefore detect glycemic changes. Their predictive alarms for impending hypoglycemia and hyperglycemia help reduce these rates. These functionalities and their improving access have increased CGM use steadily in the outpatient settings.9,14 Current CGM devices are not approved by the U.S. Food and Drug Administration (FDA) for inpatient use. However, during the coronavirus disease 2019 (COVID-19) pandemic, the FDA granted an emergency authorization for their hospital use to reserve personal protective equipment and reduce the COVID-19 exposure. 15 Since then, inpatient use is gaining popularity for both individuals with and without COVID.16-22 However, limited data exist in the use of CGM in postoperative individuals, including those using CII and requiring high frequency glucose monitoring. There are no standard practices or guidelines for initiating CGM use in the inpatient setting.
The objective of this pilot quality and process initiative is to assess the feasibility and performance of real-time CGM for titrating CII via (1) evaluation of CGM glucose accuracy in postoperative ICU population and (2) assessing both nursing acceptance/convenience and postoperative patient satisfaction with CGM use.
Methods
Protocol and Staff Training
In this pilot quality improvement (QI) initiative project, we adapted and made a minor modification to initial validation phase of the hybrid protocol (refer to the protocol in the Supplementary Material) originally developed by Faulds et al.22,23 to integrate CGM with periodic POC-BG tests to monitor and ensure the ongoing accuracy of CGM measurements (available at http://www.covidindiabetes.org). We developed comprehensive nursing education materials (in Supplementary Material) to help prepare ICU nurses to set-up and apply CGM sensors to postoperative patients with hyperglycemia managed by the CII protocol. We trained “super user” ICU registered nurses. The clinical nurse specialists and super users subsequently conducted training sessions for nurses in the CV-ICU. In the context of this resource-constrained QI pilot project, the Hospital Intensive Insulin Program (HIIP) team, consisting of five Advanced Practice Providers, played a crucial role by assisting with the CGM setup and application of sensors for the individuals involved. Ongoing support was provided by nursing super users, the clinical nurse specialists, and HIIP team members, who collaborated to guide the nurses through protocol adherence, address inquiries, and troubleshoot any challenges encountered.
The nursing-driven hybrid protocol consisted of two distinct phases.
Initial CGM validation phase
During this phase, we validated the accuracy of CGM values by comparing them with the POC-BG readings. After CGMs were placed (on the abdomen in all patients) and completed their warm-up period, we compared CGM and POC-BG readings every 1 to 2 hours using institutional blood glucose meters. This was done in accordance with our prior validated CII calculator, 24 instead of the Fauld et al protocol (which described readings every 1 hour). Validation was considered successful when two consecutive CGM readings were within 20% of the POC-BG when BG exceeded 100 mg/dL, or within 20 mg/dL if the POC-BG was below 100 mg/dL. The electronic health records (EHR) utilized the CGM glucose value entered in the patient flowsheet row for comparison with POC-BG results using the algorithm above to calculate, confirm, and record the success of the validation.
Ongoing validation phase for the sustained use of CGM
After the initial CGM validation, POC-BG checks and the CGM validation were reduced to every 6 hours, and CII titration in the interim was guided by CGM values until the next POC-BG assessment. In cases where a follow-up CGM value could not be validated against POC-BG checks every 6 hours, the protocol recommended the nurses to revert to the 1- to 2-hour POC-BG testing schedule as directed by the insulin calculator. If two consecutive readings were validated once again, POC-BG checks were adjusted to every 6 hours. In addition, one-time POC-BG measurement was obtained under the following circumstances:
When the CGM receiver showed no glucose readings or trend indicators.
When an impending “urgent low” or a low threshold alert was predicted.
If there were inconsistencies between the CGM values and observed signs or symptoms (e.g., hypoglycemia signs or symptoms).
In the event of variations in clinical status, such as intubation, hemodynamic instability, or adjustments to nutritional intake.
The CGM was discontinued when patients were transferred out of the CV-ICU or when CII was no longer required.
Outcomes Evaluations
Patients’ characteristics were collected through the EHR. We developed the study-purposed survey questionnaires in Qualtrics (Qualtrics, Seattle, WA, USA) and conducted separate surveys for both nurses and patients to assess nursing burden, acceptability, patients’ satisfaction, and overall experience with care. Nurses were advised to complete a survey before starting the project and again after being involved in the initial and ongoing validation phases of CGM at the end of the project. The purpose was to report their convenience with using CGM and their preferred glucose monitoring method, which included POC arterial blood, POC finger sticks, and CGM. When appropriate (e.g., not in critical medical phase), patients were approached by the HIIP team members to inquire about the willingness to provide feedback on the CGM use experiences when the CII was completed and the CGM was taken off. The questionnaire included assessment of experiences of care with CGM (options: very good, good, fair, poor), glucose check without pain and disruptions of sleep (yes/no), and overall confidence of care with CGM process (very confident, quite confident, somewhat confident, little confident). Nursing surveys were administrated electronically to nursing staff and the results were uploaded automatically into Qualtrics. However, patient surveys were handed out by the HIIP team members, and the results were subsequently entered into Qualtrics. (See both nursing and patient surveys in Supplementary Material.)
Initiative Logistics
Our quality initiative project received approval from the Institutional Review Board at our institution, ensuring compliance with ethical and regulatory standards. This initiative was conducted from March to May 2022, with Dexcom G6 CGMs (Dexcom, Inc., San Diego, CA, USA) being applied to 59 postsurgical patients who developed postoperative hyperglycemia and received CII in the CV-ICU at the at the University of Michigan University Hospital.
Statistical Analysis
Descriptive analyses were conducted to summarize postoperative patient characteristics, POC-BG, CGM data, and survey data. To evaluate the accuracy of real-time CGM for titrating CII, we paired CGM and POC-BG data from both validation phases (arterial, venous, capillary) and plotted them on the Clarke Error Grid. The error grid analysis was performed with the R package ega (https://cran.r-project.org/web/packages/ega/index.html), which is based upon the Clarke methodology. 25 We based our reference on the POC Inform II (Roche Diagnostics Corporation, Indianapolis, USA) results. Any gaps in the POC Inform II data were supplemented with arterial blood gas ABL800 instruments (Radiometer, Copenhagen, Denmark), and any remaining missing values were completed using venous ABL800 blood gas measurements. The median absolute relative difference was calculated as the median of the absolute differences between the CGM and POC-BG results expressed as a percentage of the POC-BG result. Within this grid, paired values that fell within Zone A were categorized as “accurate,” while those within Zone B were deemed “acceptable.” Values outside of these zones, falling in Zones C, D, or E, were considered “not acceptable.” In addition, we reported a descriptive analysis of the clinical characteristics of the patients and nurses involved. For CGM values, we calculated each individual’s time proportion under each glucose target category. Then, summary statistics like median (Q1, Q3) and mean (SD) were calculated across the cohort. CGM glucose values were categorized into the CII blood glucose target range of 110 to 150 mg/dL, CGM time in range (glucose values between 70 and 180 mg/dL), glucose values between 54-69 mg/dL, below 54 mg/dL, glucose between 181-250 mg/dL, and above 250 mg/dL. Individual times in these ranges were calculated. For each patient, we calculated the standard deviation of the glucose level, and the coefficient of variation by the ratio of the standard deviation and mean of glucose level. Survey results were summarized with descriptive analyses.
Results
Patient Characteristics
We placed Dexcom G6 sensors on the abdomen of 59 postoperative patients experiencing hyperglycemia and requiring CII in the CV-ICU. The clinical characteristics of patients are outlined in Table 1. The mean age of the patients was 64 years, with 32% being females and 32% having a prior diagnosis of diabetes. The majority of patients underwent cardiovascular procedures, with one having a left lung transplant and another undergoing a vascular procedure along with right radical nephrectomy.
Selected Baseline Clinical Characteristics of Individuals With Hyperglycemia (n = 59).

Data reported as mean ± standard deviation or n (%).
Abbreviations: BMI, body mass index; bpm, beats per minutes; BP, blood pressure; CRRT, continuous renal replacement therapy; eGFR, estimated glomerular filtration rate; EN, enteral nutrition; HR, heart rate; IV, intravenous; SOFA, sequential organ failure assessment; TPN, total parenteral nutrition.
CGM Glucose Accuracy
Analyzing 864 paired POC-BG and CGM values, the mean and median absolute relative differences (MARD and median ARD) between readings were 13.2% and 9.8%, respectively. Notably, 99.7% of the paired CGM and POC-BG values fell within Zones A and B of the Clarke Error Grid (Figure 1). Further breakdown reveals that 79% of the values are in Zone A, 20.6% in Zone B, and a minimal 0.35% in Zone D (Figure 1). The average duration for the initial phase time to validation, defined by the time difference between (1) the initial POC-BG and CGM paired time instant and (2) the second consecutive POC-BG and CGM glucose validation time instant, was 172 minutes (with a standard deviation of 144 minutes).
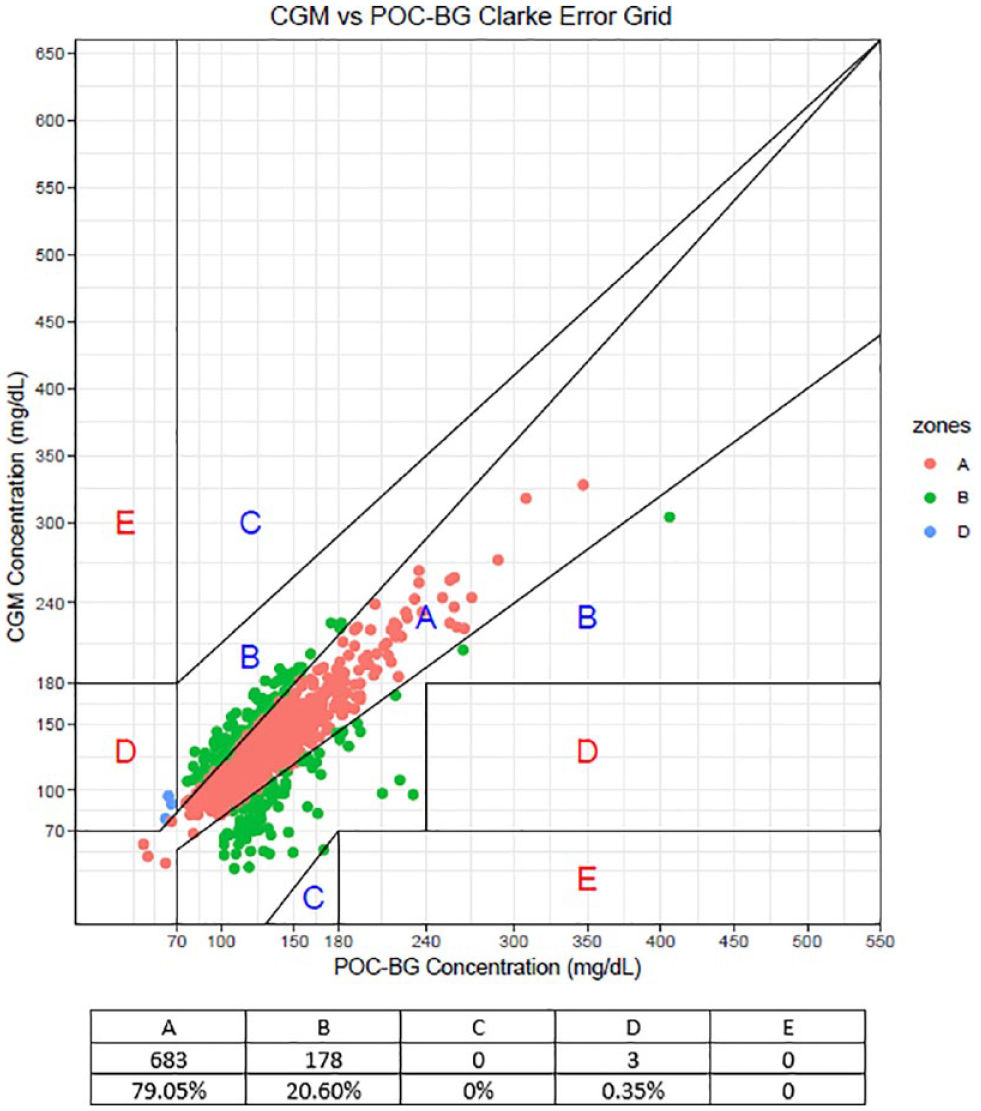
Clarke error grid of CGM versus POC-BG values.
Nurse and Postoperative Patient Surveys
Nursing surveys were distributed to approximately 120 nurses in the CV-ICU. Thirty-six responses were received, six surveys were incomplete, and thus 30 surveys were included in the analysis. The majority of nurses who responded found CGMs quite or very convenient (n = 28; 93%) and favored it over POC-BG testing (n = 28; 93%) (Figure 2). None preferred using finger sticks POC-BG. A complete set of survey responses was obtained from 45 postoperative patients (76%) involved in the QI project, while nonrespondents were unable to participate in the survey due to factors such as altered mental or intubation status. Forty-five completed survey responses were included in the analysis. Majority of patients (n = 42; 93%) reported their care process using CGM as good or very good, while reduced pain from finger sticks (n = 39; 87%) and sleep disruption (n = 39; 87%) was also reported. Most patients (n = 40; 89%) also reported to be quite or very confident of the care with CGM (Figure 3).
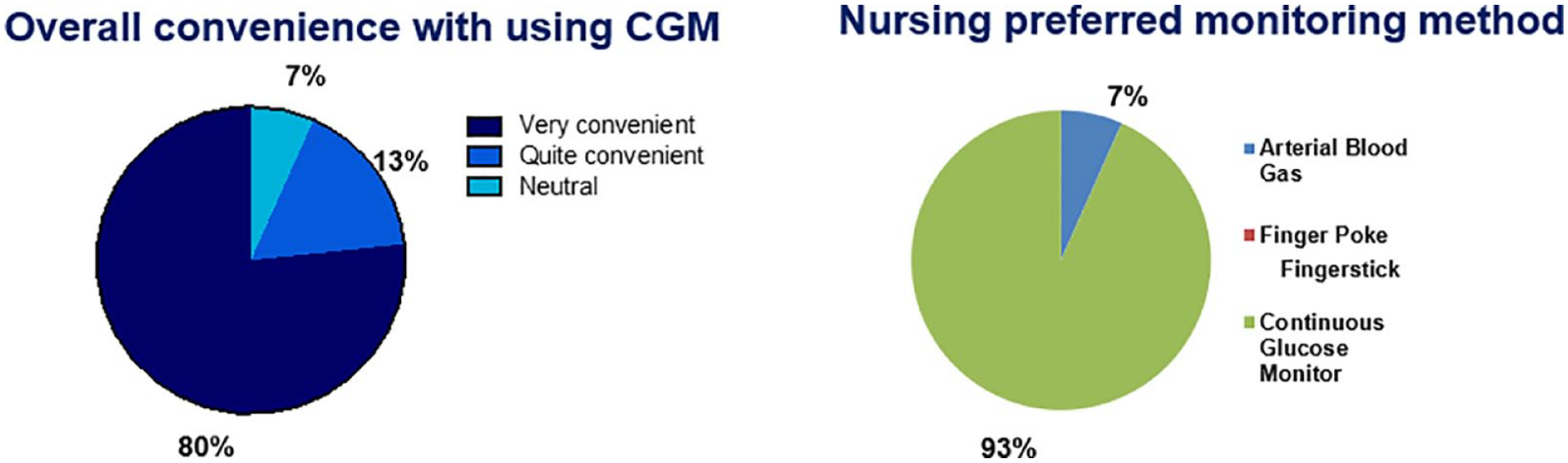
Nurse reporting of convenience with CGM use and glucose monitoring preferences.
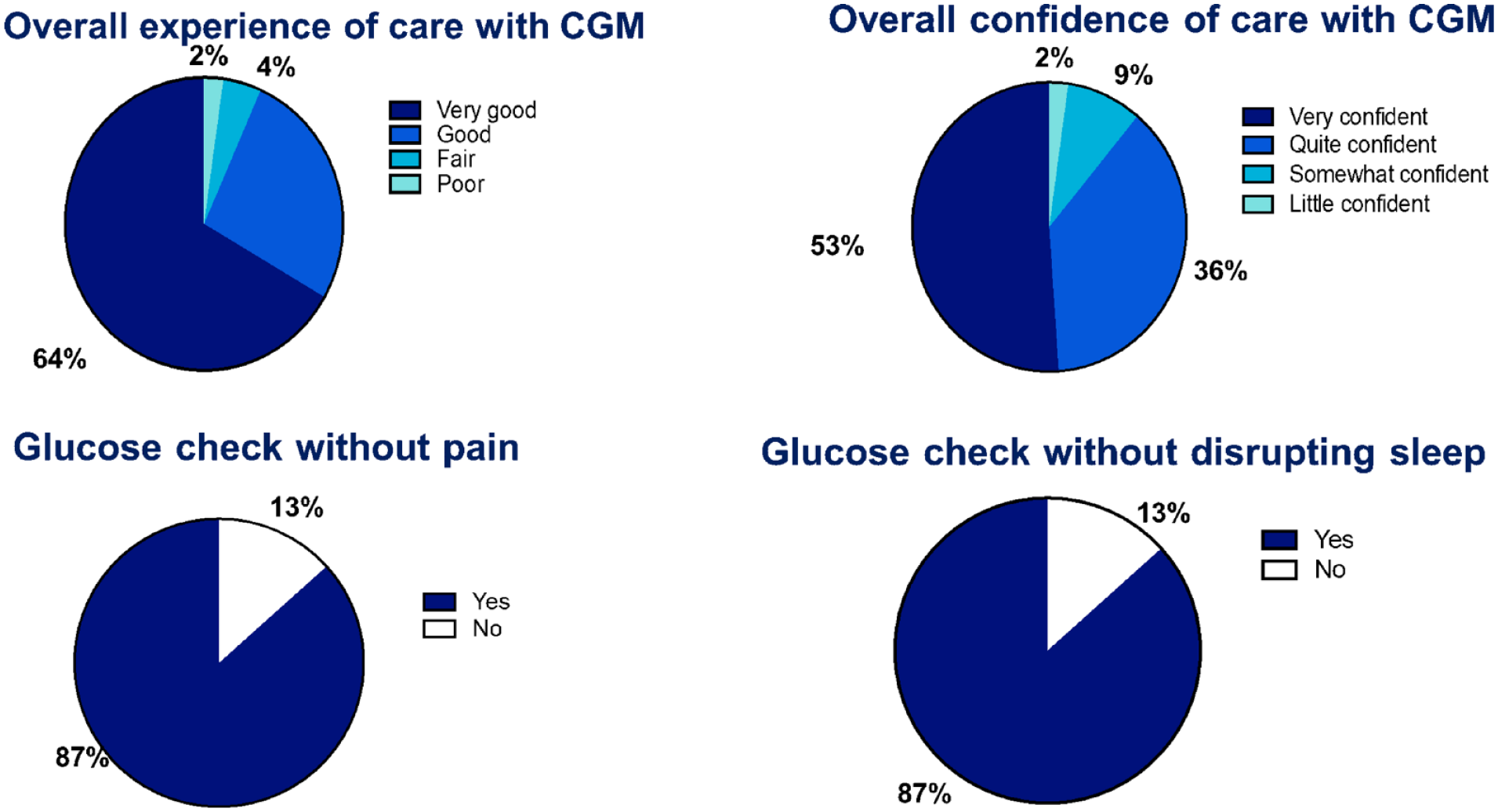
Patient reports of experiences and confidence in CGM utilization.
Glycemic Measures
Subsequently, we explored into various CGM metrics (Table 2) in addition to the CII blood glucose target (110-150 mg/dL) established for the CII calculator. We observed 57% of readings within this target range. Time spent within CGM glucose in range of 70-180 mg/dL was 90%. CGM glucose levels between 54 and 69 mg/dL and below the clinically significant threshold of 54 mg/dL were observed at 1.4% and 1.3%, respectively. Conversely, the time spent glucose between 181 and 250 mg/dL represented 6.8%, while instances of severe hyperglycemia (above 250 mg/dL) were notably low at 0.8%.
CGM Glucose Values (n = 58).
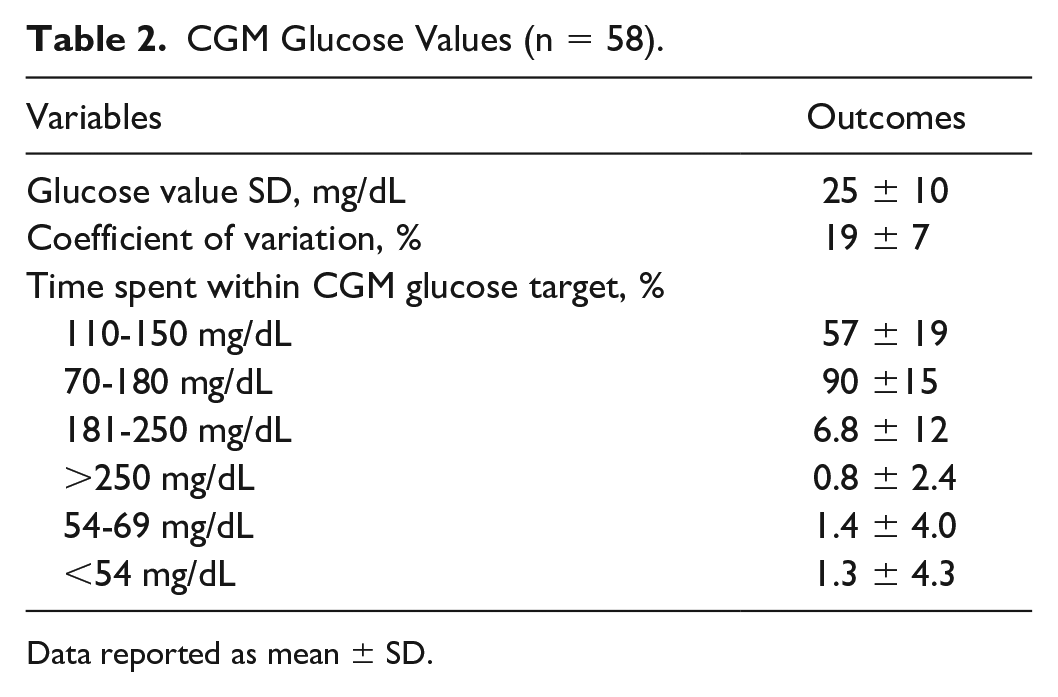
Data reported as mean ± SD.
Discussion
This study presents our institutional experience with real-time CGM utilizing a hybrid approach for postoperative patients requiring CII in the CV-ICU setting. Our pilot QI initiative demonstrated a high-accuracy CGM values in this critically ill postsurgical population. The CII was titrated using a hybrid method with CGM and POC-BG checks which reduced fingerstick burden. Feedback reported by nursing staff and patients in the CV-ICU through surveys was highly positive. These findings emphasize the efficacy and feasibility of using CGM with real-time monitoring within the unique context of CV-ICU care.
To discuss our findings in context of exiting literature, the accuracy data, with 99.7% falling within Zone A and B of the Clarke Error Grid, aligns closely with studies conducted in the ICU setting.18,26 Our pilot’s MARD and median ARD of 13.2 and 9.8% values are consistent with those presented by Perez-Guzman et al 18 and Sadhu et al. 26 An earlier, relatively smaller study by Ancona et al investigated the accuracy of Freestyle Libre in critically ill patients, demonstrating a similar MARD of 14%. In another study comparing CGM devices to arterial blood glucose levels as the reference, showed 97.8% of readings fell within Zones A and B of the Clarke Error Grid. 27 The consistency observed in CGM accuracy across these studies, including ours, highlights the reliability of CGM technology, especially in the challenging and dynamic environment of critically ill patients.
The implementation process was smooth, possibly attributing to (1) the welcomed changes as suggested by the favorable patient and nurse survey results, (2) the support of the HIIP team who have already established a working relationship with nursing staff, and (3) nursing staff’s familiarity with the existing CII protocols, and (4) the EHR-integrated CGM validation system. After receiving 15 to 30 minutes of education on sensors, monitors, alarms, documentation of readings, and the validation process, the nursing staff smoothly adapted the newly learned technology to the existing CII protocol. The reduced burden of checking POC-BG levels every one to two hours, transitioning from spending five and seven minutes on drawing labs/POC-BG checks to quickly reviewing CGM receivers, was a welcome reduction in the workload for the nurses. The transition to validating the CGM with POC-BG minimally every six hours was easily accepted by nursing staff after their first exposure. While a prolonged validation process of CGM readings with POC-BG readings occasionally occurred when inconsistent glucose values were identified, the POC-BG evaluation and entry process, which already existed prior to CGM use, did not increase nursing burden. However, concerns about data loss during this process were limited or nonexistent. The majority of nursing discussions revolved around when this process would be adapted on a more widespread scale. These reports and findings were accurately reflected in the surveys of the nursing staff. In addition, the acceptability of implementing CGM devices in the CV-ICU was also supported by the feedback provided by patients. In a previous pilot study, the use of CGM in critically ill individuals with COVID-19, notable nursing acceptance was reported, despite the absence of formal nursing surveys. 26 Our survey provided detailed insights into nursing satisfaction and workflow, demonstrating high levels of satisfaction. This includes the reported convenience and improved resource utilization of CGM over traditional methods such as finger stick and arterial line draws. Furthermore, most nursing staff found the educational material to be helpful. Similarly, our survey data with patients also contribute evidence about the high acceptance and confidence of postsurgical patients in the use of CGM especially convenience to effectively measure blood glucose after surgeries. Of the 7% reported neutral nursing experiences with using CGM from nurses and one postoperative patient reporting poor experience of care with CGM, our pilot QI study did not incorporate additional data collection which may be considered a limitation. A future mixed-methods approach, incorporating qualitative interviews to assess the reasons for dissatisfaction with CGM use, should be incorporated to identify and address potential care gaps.
Our glucometric evaluation validates the effectiveness of our computerized CII algorithm, titrated by the hybrid CGM process. We observed a higher time in the range of glucose between 70 and 180 mg/dL compared with other studies, while noting an increased duration of glucose between 54 and 69 mg/dL and <54 mg/dL.21,23 These variations may be attributed to differences in the specific populations included. Prior studies have primarily focused on COVID populations who might have been exposed to steroids, high inflammatory states, and extreme physiological stress. Although these individuals presented with a lower time in the desired glucose range, they were also afforded protection against hypoglycemia development. Individuals with a longer time in the targeted glucose range (i.e., tighter glycemic control) may be at risk for a higher incidence of hypoglycemia.
The use of CGM in the inpatient setting lacks formal FDA approval at present. With the FDA’s emergency authorization, the use of CGM in the inpatient setting has gained popularity in hospitals. Despite the absence of formal approval, there are several encouraging studies including ours highlighting the accuracy and performance of CGM in various inpatient settings, including ICUs.18,19,21,26-31 Notably, a recent study at a large academic hospital demonstrated an acceptable accuracy, assessed by MARD and Clarke Error Grid, for both noncritically ill and critically ill patients in real-world fashion. 32 The Endocrine Society’s expert panels have endorsed the initiation of CGM in the hospital settings, particular in individuals with an increased risk of hypoglycemia. The recommended approach involves a hybrid strategy, wherein CGM usage is integrated with periodic POC-BG measurements. 9
The strengths of our pilot QI initiative project include its relevance in the post-COVID era which could support future generalizability. The implementation of a standardized survey sampling methodology was a powerful feedback data on its acceptability. In addition, the extensive nurse training before the study’s commencement and the active involvement of the HIIP team contribute positively to its overall acceptance and impact. Several limitations existed in our project. Due to the nature of being a pilot, our sample size was limited. We evaluated only one available CGM and were not able to evaluate time motion studies, cost-effectiveness, and the protocol’s applicability to other ICUs with varying nurse-to-patient ratios and other critically ill populations. In addition, the study-purposed surveys in this project have not been validated. Finally, as a natural limitation of a QI project, the absence of a control group does not allow full assessments of the impacts of CGMs on health outcomes and changes in ICU and hospital burden. Further research is needed to evaluate whether implementing CGM in ICU care improves patient outcomes, satisfaction, and enhances hospital resource efficiency, including staff time and financial implications.
Conclusions
This pilot quality improvement initiative provides robust evidence on the accuracy, feasibility, convenience, and acceptance of titrating CII using CGM devices in the postoperative CV-ICU individuals. This provides robust data for conducting large prospective trials in varied ICU populations utilizing current or newer ICU dedicated CGM devices.
Supplemental Material
sj-docx-1-dst-10.1177_19322968241241005 – Supplemental material for Feasibility and Performance of Continuous Glucose Monitoring to Guide Computerized Insulin Infusion Therapy in Cardiovascular Intensive Care Unit
Supplemental material, sj-docx-1-dst-10.1177_19322968241241005 for Feasibility and Performance of Continuous Glucose Monitoring to Guide Computerized Insulin Infusion Therapy in Cardiovascular Intensive Care Unit by Lynn Ang, Yu Kuei Lin, Lee F. Schroeder, Yiyuan Huang, Christina A. DeGeorge, Patrick Arnold, Folake Akanbi, Sharon Knotts, Elizabeth DuBois, Nicole Desbrough, Yunyan Qu, Regi Freeman, Nazanene H. Esfandiari, Rodica Pop-Busui and Roma Gianchandani in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-2-dst-10.1177_19322968241241005 – Supplemental material for Feasibility and Performance of Continuous Glucose Monitoring to Guide Computerized Insulin Infusion Therapy in Cardiovascular Intensive Care Unit
Supplemental material, sj-docx-2-dst-10.1177_19322968241241005 for Feasibility and Performance of Continuous Glucose Monitoring to Guide Computerized Insulin Infusion Therapy in Cardiovascular Intensive Care Unit by Lynn Ang, Yu Kuei Lin, Lee F. Schroeder, Yiyuan Huang, Christina A. DeGeorge, Patrick Arnold, Folake Akanbi, Sharon Knotts, Elizabeth DuBois, Nicole Desbrough, Yunyan Qu, Regi Freeman, Nazanene H. Esfandiari, Rodica Pop-Busui and Roma Gianchandani in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-3-dst-10.1177_19322968241241005 – Supplemental material for Feasibility and Performance of Continuous Glucose Monitoring to Guide Computerized Insulin Infusion Therapy in Cardiovascular Intensive Care Unit
Supplemental material, sj-docx-3-dst-10.1177_19322968241241005 for Feasibility and Performance of Continuous Glucose Monitoring to Guide Computerized Insulin Infusion Therapy in Cardiovascular Intensive Care Unit by Lynn Ang, Yu Kuei Lin, Lee F. Schroeder, Yiyuan Huang, Christina A. DeGeorge, Patrick Arnold, Folake Akanbi, Sharon Knotts, Elizabeth DuBois, Nicole Desbrough, Yunyan Qu, Regi Freeman, Nazanene H. Esfandiari, Rodica Pop-Busui and Roma Gianchandani in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-4-dst-10.1177_19322968241241005 – Supplemental material for Feasibility and Performance of Continuous Glucose Monitoring to Guide Computerized Insulin Infusion Therapy in Cardiovascular Intensive Care Unit
Supplemental material, sj-docx-4-dst-10.1177_19322968241241005 for Feasibility and Performance of Continuous Glucose Monitoring to Guide Computerized Insulin Infusion Therapy in Cardiovascular Intensive Care Unit by Lynn Ang, Yu Kuei Lin, Lee F. Schroeder, Yiyuan Huang, Christina A. DeGeorge, Patrick Arnold, Folake Akanbi, Sharon Knotts, Elizabeth DuBois, Nicole Desbrough, Yunyan Qu, Regi Freeman, Nazanene H. Esfandiari, Rodica Pop-Busui and Roma Gianchandani in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-5-dst-10.1177_19322968241241005 – Supplemental material for Feasibility and Performance of Continuous Glucose Monitoring to Guide Computerized Insulin Infusion Therapy in Cardiovascular Intensive Care Unit
Supplemental material, sj-docx-5-dst-10.1177_19322968241241005 for Feasibility and Performance of Continuous Glucose Monitoring to Guide Computerized Insulin Infusion Therapy in Cardiovascular Intensive Care Unit by Lynn Ang, Yu Kuei Lin, Lee F. Schroeder, Yiyuan Huang, Christina A. DeGeorge, Patrick Arnold, Folake Akanbi, Sharon Knotts, Elizabeth DuBois, Nicole Desbrough, Yunyan Qu, Regi Freeman, Nazanene H. Esfandiari, Rodica Pop-Busui and Roma Gianchandani in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
BMI, body mass index; bpm, beats per minutes; BP, blood pressure; CGM, continuous glucose monitoring; CII, continuous intravenous insulin infusion; CV-ICU, cardiovascular intensive care unit; COVID-19, coronavirus disease 2019; CRRT, continuous renal replacement therapy; EHR, electronic health records; eGFR, estimated glomerular filtration rate; EN, enteral nutrition; FDA, U.S. Food and Drug Administration; HIIP, Hospital Intensive Insulin Program; HR, heart rate; ICU, intensive care unit; MARD, mean absolute relative difference; POC-BG, point-of-care blood glucose; QI, quality improvement; SD, standard deviation; SOFA, sequential organ failure assessment; TPN, total parenteral nutrition.
Authors’ Contributions
LA, and RG designed the quality improvement project, and were involved in analysis, and interpretation of the data. LA drafted the initial manuscript and revised the manuscript to the final version. YH and LFS contributed to analysis and interpretation of the data and critically reviewed and revised the manuscript. LA and YKL involved in the development of surveys, data analysis and interpretation. YKL, AK, CAD, SK, ND, YQ, PA, ED, RF, NHE, and RPB critically reviewed and revised the manuscript. CAD, SK, ND, YQ, and ED conducted the project. LA and RG are the guarantors of this work and, as such, had full access to all the data in the project and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LA, YKL, LFS, YH, CAD, PA, FA, SK, ED, ND, YQ, RF, NHE, and RG have no conflicting interest to disclose. RPB received grant support to University of Michigan from Dexcom.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LA is supported by the JDRF Center of Excellence at University of Michigan. YKL is supported by National Institute of Diabetes and Digestive and Kidney Diseases (K23DK129724). RPB is supported by R01DK107956, R01DK11672, U01DK119083 and the JDRF Center of Excellence at University of Michigan.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
