Abstract
Background:
To evaluate the effect of various, everyday intensive care unit (ICU) practices on glucose levels in critically ill pediatric patients with the use of a continuous glucose monitoring system.
Methods:
Seventeen sensors were placed in 16 pediatric patients (8 male). All therapeutic and diagnostic interventions were recorded and 15 minutes later, a flash glucose measurement was obtained by swiping the sensor with a reader. Glucose difference was calculated as the glucose value 15 minutes after the intervention minus the mean daily glucose value for each individual patient. Additionally, the consciousness status of the patient (awake or sedated) was recorded.
Results:
Two hundred and five painful skin interventions were recorded. The mean difference of glucose values was higher by 1.84 ± 14.76 mg/dL (95% CI: −0.19 to 3.87 mg/dL, P = .076). However, when patients were categorized regarding their consciousness level, mean glucose difference was significantly higher in awake state than in sedated patients (4.76 ± 28.07 vs −2.21 ± 15.77 mg/dL, P < .001). Six hundred forty-nine interventions involving the respiratory system were recorded. Glucose difference during washings proved to be significantly higher than the ones during simple suctions (4.74 ± 14.18 mg/dL vs 0.32 ± 18.22 mg/dL, P = .016). Finally, glucose difference in awake patients was higher by 3.66 ± 13.91 mg/dL compared to glucose difference of −2.25 ± 21.07 mg/dL obtained during respiratory intervention in sedated patients.
Conclusions:
Diagnostic and therapeutic procedures in the ICU, especially when performed in an awake state, exacerbate the stress and lead to a significant rise in glucose levels.
Keywords
Introduction
Stress hyperglycemia (SH) is very common among critically ill pediatric subjects. It can be attributed not only to the neuroendocrine response to critical illness but also to several diagnostic and therapeutic interventions in the intensive care unit (ICU). 1 Although hyperglycemia during acute stress is necessary for the host’s survival and aims to restore homeostasis, during critical illness, the counteracting mechanisms become overwhelmed, which leads to a rather prolonged rise in glucose levels.1,2 SH per se and its duration, in particular, have highly been associated with mortality in a wide range of different pathologies in critically ill pediatric subjects, including septic shock, severe burns, trauma, and post cardiac surgery.3-11
Therefore, close monitoring of blood glucose levels in ICU is essential and current protocols include the use of handheld point-of-care glucose meters. However, the main drawback of this method is its intermittent character, which often leads in failure for an accurate detection of either hyper- or hypoglycemia. Continuous glucose monitoring (CGM) systems are constantly gaining ground, as they can overcome this limitation and detect acute changes in blood glucose levels without increasing the burden on nursing staff in terms of time expenditure. 12 FreeStyle Libre (Abbott Diabetes Care, Alameda, CA, USA) is the first flash glucose monitoring (FGM) system that measures interstitial glucose concentration with the use of an enzymatic amperometric three-electrode sensor system and has been approved for use in children aged four years of age and above.13-16 We have recently shown that FreeStyle Libre is well tolerated in a pediatric ICU setting, however with a tendency to underestimate glucose levels. 17
Many practices performed in ICU subjects are accompanied by pain and agitation resulting in physical and psychological complications that may last months after discharge from the hospital. 18 Recently published guidelines on the management of pain and agitation in ICU-treated adult subjects recommend a systemic and rigorous assessment of pain, as pain is consistently underestimated and undertreated in ICU subjects. 19 Pain assessment in noncommunicative subjects is deficit and is carried out through various tools evaluating behavioral or observational parameters. 20 Theoretically, glucose levels rise during pain and stress; however, there is limited evidence to support this. Significantly increased glucose levels have been reported at time of admission compared to time of discharge in adult patients with acute bone fractures, pancreatitis, 21 and acute spinal cord trauma. 22 Higher glucose levels but without reaching statistical significance have also been reported in patients with various painful vascular conditions. 23 Recently, Zhang et al have demonstrated higher plasma glucose values during spontaneous migraine attacks, independent of the presence of aura and the pain intensity. 24
The aim of this study was to evaluate glucose levels in critically ill pediatric subjects during various painful skin practices or during interventions involving the respiratory system with the use of a CGM system using flash sensing technology. Furthermore, changes in glucose levels during these practices would be analyzed with regard to the severity of intervention and the consciousness status of the patient. To our knowledge, no study has previously explored this topic.
Methods
Between May and August 2018, we included in the study consecutive subjects admitted to the pediatric ICU and fulfilling the following criteria: (i) age >4 years, (ii) expected length of stay in the ICU at least 48 hours, (iii) no use of medication that could affect glucose metabolism, (iv) no preceding diagnosis of impaired glucose metabolism. The study was performed in accordance with the Helsinki Declaration of 1975 and was approved by the Scientific and Administrative Council of Hippokration General Hospital of Thessaloniki. Written informed consent was obtained from the parents or legal caregivers of all included subjects.
We collected demographic data (age and sex) and anthropometric measurements of all the subjects (body weight and height) and calculated the body mass index (BMI) as the ratio weight/height2 (kg/m2, Quetelet index). For every anthropometric parameter, Z-scores were calculated according to the reference values by the Center for Disease Control and Prevention. 25 We also collected data regarding medical history, ICU admission diagnosis, medication, date of admission and discharge from the ICU, date and time of sensor placement and removal, reason of removal, and patient outcomes. All the therapeutic interventions, including administration of medication, venous or arterial punctures, fingerpricks, catheter placements, bronchial suctions, and washings, were recorded, and 15 minutes later, a flash glucose measurement was obtained by swiping the sensor with a reader, in order to detect a possible association between the variations of glucose levels and the therapeutic interventions. Additionally, the consciousness status of the patient (awake or sedated) was recorded.
Sensors were placed on the backside of the upper arm of 16 subjects, according to the instructions provided by the manufacturer. After a warm-up period of 1 hour, the user was able to obtain information regarding the current glucose level in the subcutaneous interstitial fluid, the glucose profile over the previous 8 hours, and the glucose trend, while the sensor automatically stored glucose data every 15 minutes. The sensor is factory calibrated and can be worn for up to 14 days.
For statistical analysis and graphical demonstrations, the Microsoft Excel for Mac 2011 version 14.0.0 and the IBM SPSS Statistics version 20 were used. Shapiro–Wilik and Kolmogorov–Smirnov tests were used to assess normality of the data. We performed student’s t-test to test the equality of means of two samples for normally distributed data and Mann–Whitney U and Kruskal–Wallis for nonparametric data. The level of statistical significance was P <.05.
Results
Seventeen sensors were placed in 16 subjects (8 male and 8 female). Two sensors were applied on the upper arms of a 12-year old female patient, in order to closely monitor glycemic variability due to ketogenic diet. Mean decimal age was 8.03 ± 2.91 years and mean height, weight, and BMI were 130 ± 20 cm, 32.06 ± 21.59 kg, and 17.42 ± 6.69 kg/m2, respectively. The subjects remained hospitalized for a mean of 32.35 ± 31.25 days and the sensors remained in situ for a mean of 9.71 ± 5.35 days. Removal of the sensor was attributed to the completion of the predefined life-span of the sensor (14 days) or to the discharge of the patient from the ICU in 13 subjects, while there were also 3 cases of accidental removal and 1 case, where the sensor needed to be removed due to diagnostic procedures (magnetic resonance imaging). There were no adverse events attributed to sensor placement. We observed a mean blood glucose value of 87.35 ± 21.92 mg/dL. In 711 paired measurements with other glucose measurement methods including blood gas analyzer, capillary blood glucose meter, and biochemical serum analysis, FGM measurements were consistently lower with mean absolute relative difference being 28.34%, 25.11%, and 18.99%, respectively. However, surveillance error grid analysis showed 92.04%, 94.67%, and 95.52% of the readings in the none or slight risk zone, respectively, as we have recently published. 17
As expected, states of extreme physical stress were characterized by a rise of glucose levels. A very characteristic example of this phenomenon was the rise in the glucose levels of patient 2 on the evening of the 21st of May, which coincided with an intracerebral hemorrhage and clinical deterioration (Figure 1(a)). Additionally, in most cases there was a rising trend in glucose levels, as recorded by the sensor, following extubation and patient’s awakening. This is particularly evident in the case of patient 14, as shown in Figure 1(b). In 11 subjects, an altered level of consciousness (awake and sedated) was recorded during the period that the sensor remained in situ. Mean glucose values obtained with the FGM were lower when subjects were sedated than mean glucose values obtained when subjects were awake (77.86 ± 28.20 mg/dL vs 91.13 ± 24.16 mg/dL) but this difference was not statistically significant (P = .053, Figure 2).
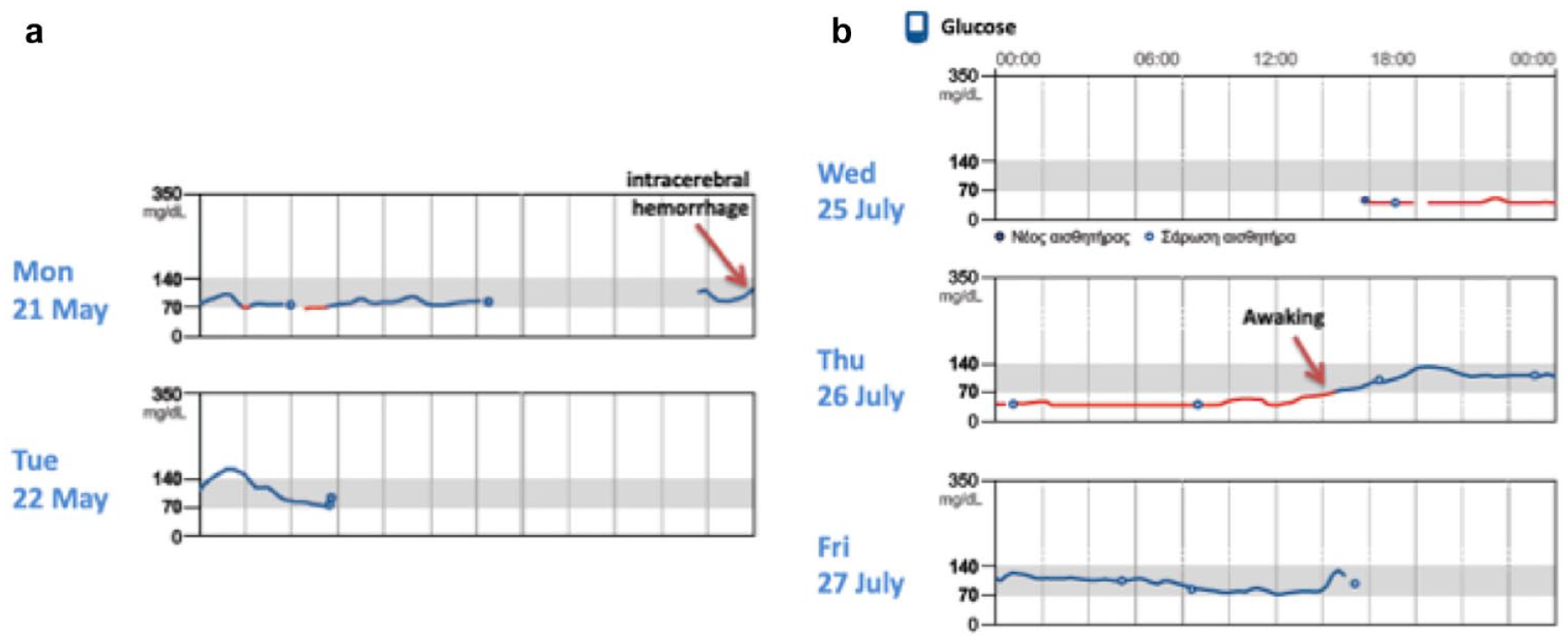
(a) Increased glucose levels associated to an intracerebral hemorrhage and clinical deterioration. (b) Increased glucose levels after extubation and patient awakening.
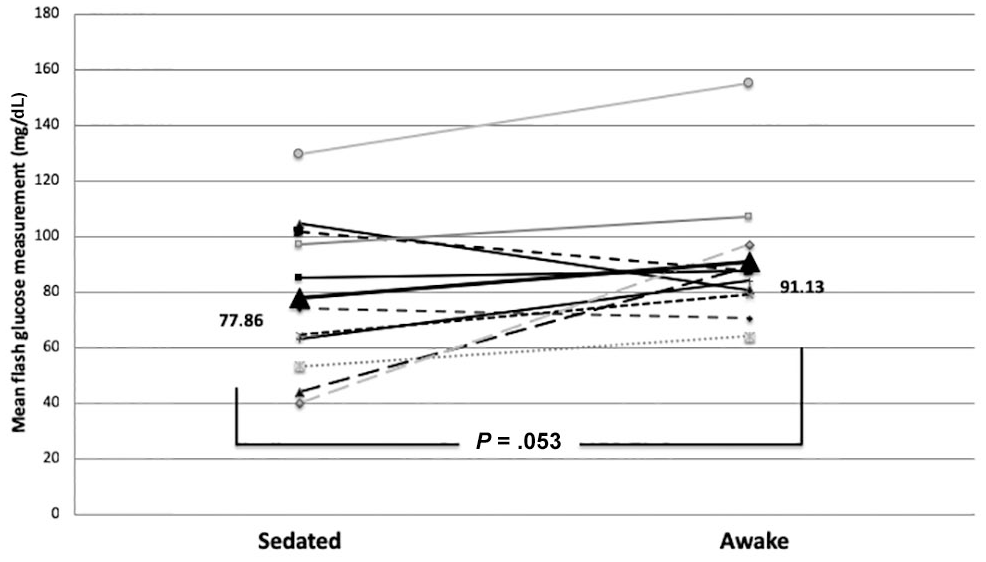
Mean glucose values in sedated and awake state in 11 patients with alteration in level of consciousness during the study period. Mean glucose levels of all patients were lower during sedation with a difference that was approaching significance.
Two hundred and five painful skin interventions were recorded, the vast majority being venipunctures, fingerpricks for measurement of glucose in capillary blood, catheter placements, and paracenteses. Fifteen minutes after every painful skin intervention, a flash glucose measurement was obtained. These values were compared to the mean daily glucose value obtained by the sensor for each individual patient. The mean difference of glucose values at the time of painful interventions minus the mean daily glucose values for each individual patient was higher by 1.84 ± 14.76 mg/dL with 95% CI: −0.19 to 3.87 mg/dL, but without statistically significant difference (mean value of glucose levels at the time of painful interventions: 84.01 ± 24.05 mg/dL vs 82.11 ± 18.95 mg/dL, P = .076). However, when the interventions were categorized based on the patient being conscious or sedated, a significant difference arose. The mean difference of the 119 glucose values, which were obtained when the subjects were conscious minus their individual mean glucose levels, was higher by 4.76 ± 28.07 mg/dL. On the contrary, the mean difference of glucose values for interventions during sedation minus mean daily glucose levels for each individual was lower by −2.21 ± 15.77 mg/dL. This difference was statistically significant (P < .001, Figure 3).
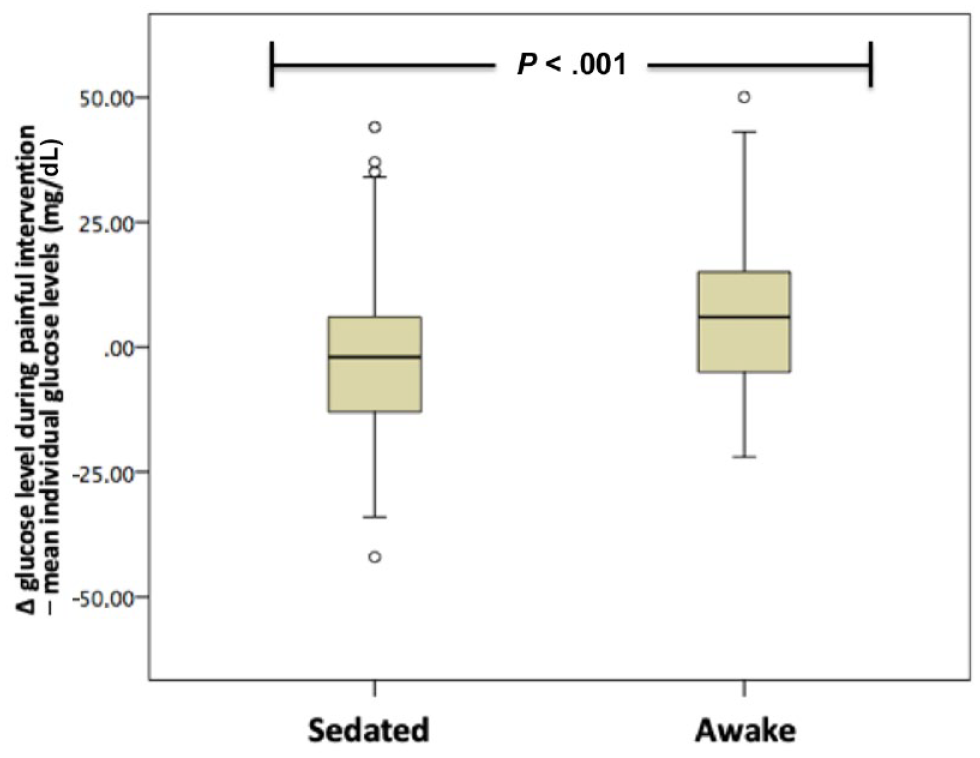
Mean difference of glucose values during painful intervention minus mean daily glucose levels of each individual were significantly lower when subjects were sedated than when subjects were awake.
Six hundred forty-nine interventions involving the respiratory system were recorded, the majority being bronchial suctions or washings, which were also combined with flash glucose measurements. Similarly, these values were also compared to the mean daily glucose values for each individual patient, and their mean value proved to be higher by 0.70 ± 17.95 mg/dL with 95% CI: −0.69 to 2.08 mg/dL, but without reaching statistical significance (mean value of glucose levels at the time of interventions in the respiratory system: 84.58 ± 27.45 mg/dL vs 83.88 ± 20.64 mg/dL, P = .323). However, when these interventions were categorized based on severity (simple bronchial suction as opposed to bronchial washing), 54 bronchial washings and 595 bronchial suctions were recorded. Glucose levels during washings proved to be significantly higher than the ones during simple suctions (4.74 ± 14.18 mg/dL vs 0.32 ± 18.22 mg/dL, P = .016, Figure 4).
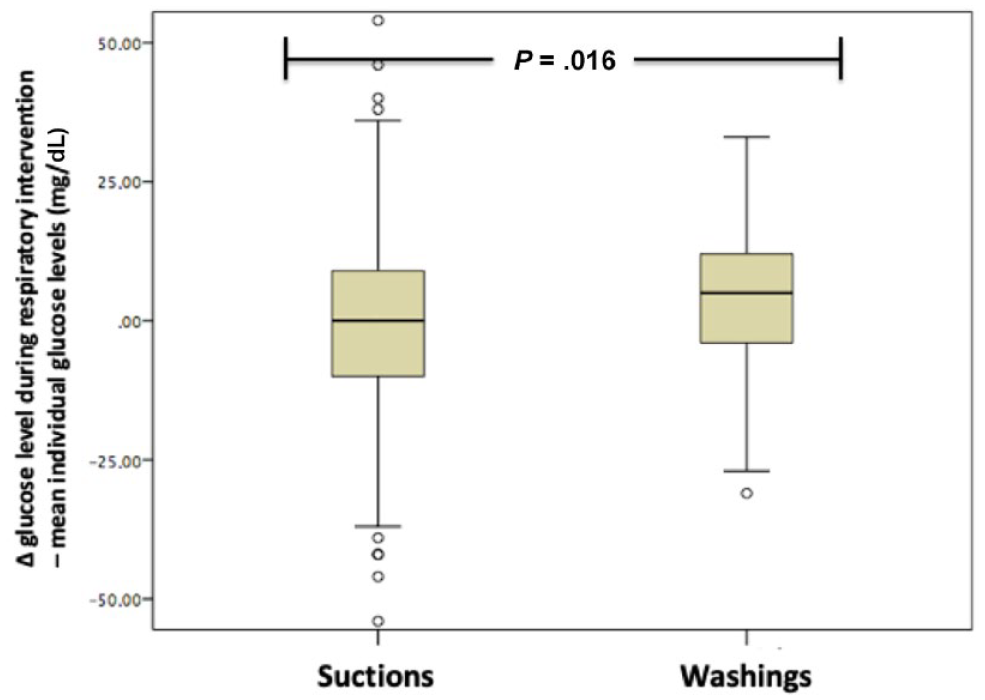
Mean difference of glucose values during respiratory interventions minus mean daily glucose levels of each individual were significantly higher during suctions than simple washings.
Furthermore, when the respiratory interventions were categorized on the basis of the patient being awake or sedated, another significant difference was observed. The mean difference of glucose values recorded during the 339 respiratory interventions on conscious subjects minus mean daily glucose levels for each individual was higher by 3.66 ± 13.91 mg/dL, whereas the mean difference of glucose values during respiratory interventions on sedated subjects minus mean daily glucose levels for each individual was lower by −2.25 ± 21.07 mg/dL. This difference was statistically significant (P < .001, Figure 5).
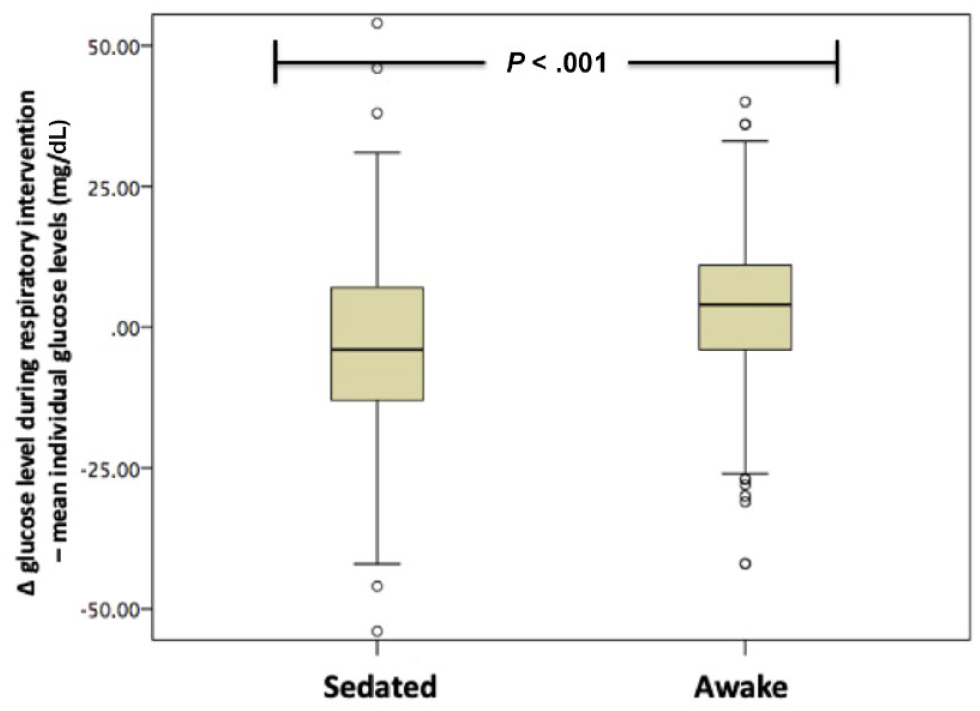
Mean difference of glucose values during respiratory interventions minus mean daily glucose levels of each individual were significantly higher when subjects were awake than when subjects were sedated.
Discussion
Our study is unique, as, to our knowledge, it is the first study to assess the effects of common, everyday therapeutic interventions on the glucose levels of critically ill ICU-treated pediatric subjects with the use of an FGM system. No adverse events, such as allergic reactions, were associated with the application of the sensor and no incompatibility with other devices employed in the ICU was observed. As anticipated, painful interventions, such as venipunctures and respiratory interventions, such as suctions and washings resulted in a rise of glucose levels, which became statistically significant when the subjects were awake. Interestingly, the glucose values were higher for more “severe” interventions. Furthermore, there was a trend of higher glucose values in subjects on awake state versus sedated subjects that was approaching significance.
There is considerable evidence that minimizing the level of sedation in adult patients in the ICU is associated with improved clinical outcomes and this has led to significant changes in sedation practice over the last three decades. 26 On the other hand, pain and discomfort are frequent in critically ill patients, magnified by the inability to communicate and annoying environmental stimuli, which can lead to agitation, which often presents as severe anxiety, delusions, and impaired brain function. It is taken into consideration that all these data derived from studies performed in adults are incorporated in recent guidelines, with a focus on prevention, assessment, and management of pain and discomfort in ICU-treated patients 19 but with cautious extrapolation to pediatric population. Current methods for assessment of pain include self-reported scales, behavioral assessment tools, proxy reports, or physiological measures (eg, vital signs). However, further studies evaluating objective measures, for example, pupillary reflex dilatation response to pain or incorporating new biomarkers associated with pain, are required. Could glucose levels be considered as an objective measure and reflection of pain and discomfort, especially when it is monitored continuously and not intermittently? The option for exploring this hypothesis is now offered with the use of modern CGM systems such as the Libre, which was employed in this study.
The rise observed in glucose levels during therapeutic or diagnostic interventions especially when the patients were conscious and the elevated glucose values of conscious patients could be attributed to greater physical stress. SH is very common among ICU-treated patients, as a result of intrinsic changes associated with critical illness. Hyperglycemia is an evolutionarily preserved response, which is necessary for the host’s survival during critical illness and extreme stress, as it ensures the supply of energy substrates to stressed organs and tissues, as well as adequate endovascular volume through increased osmolarity. 27 SH is the result of the synchronous effect of the secretion of hormones, which are counter-regulatory to insulin, such as glucagon, cortisol, catecholamines, and growth hormone, and proinflammatory cytokines, such as interleukins 1 and 6 and tumor necrosis factor-a. Impaired insulin signaling, which leads to peripheral insulin resistance, increased lipolysis and proteolysis, which provide substrates for gluconeogenesis, increased gluconeogenesis and glycogenolysis in the liver, as well as reduced function of pancreatic beta-cells play a key role in the development of SH.2,27-29 Several investigators have tried to identify the etiology of SH in critically ill children. Recently, El-Sherbini et al 30 and Hacihamdioglu et al 31 demonstrated that impaired beta-cell function is the underlying mechanism of hyperglycemia in the majority of patients by employing the homeostatic model assessment, whereas Verhoeven et al 32 studied pediatric patients with meningococcal disease and found that insulin resistance was accountable for the manifestation of hyperglycemia in 62% of the patients and Ballestero et al 33 found both mechanisms to be accountable for severe hyperglycemia (defined as two blood glucose measurements greater than 180 mg/dL) in half of the study population.
In our small cohort, no patient exhibited glucose levels exceeding the cutoffs for defining hyperglycemia. Expanding our findings, several studies have associated hyperglycemia with various ICU practices. Mosier et al demonstrated that the incidence of hyperglycemia within the first 72 hours of injury was 67% in ventilated pediatric patients with scald burns versus only 29% in the nonventilated group. 34 However, in interpreting these results we should consider whether there is a cause–effect relationship between mechanical ventilation and SH or it is actually the severity of the situation that necessitates ventilation that causes also SH. Additionally, Ognibene et al showed that the duration of mechanical ventilation was a prognostic factor for the risk of SH, 35 whereas Preissig and Rigby associated mechanical ventilation not only with the manifestation but also with the severity and the duration of SH. 36 Extracorporeal life support has also been associated with the severity and/or duration of hyperglycemia in pediatric patients undergoing cardiac surgery,9,36 while renal replacement therapy for the management of acute renal failure, which is a common pathology in the critically ill, constitutes another risk factor.10,37
Several medications commonly administered in the ICU lead to the development of SH. Verhoeven et al showed by using univariate analysis of variance that the main factor associated with hyperglycemia in 49 pediatric patients undergoing cardiac surgery was the administration of glucocorticoids. In a study of 49 ICU-treated children with meningococcal sepsis, the peak glucose value was significantly higher in the group that received inotropes 10 and vasoactive infusions were one of the factors associated with the severity and duration of hyperglycemia in ICU-treated pediatric patients in a study conducted by Preissig and Rigby. 36 Finally, another key contributing factor to hyperglycemia is parenteral nutrition. Taking into consideration that the acute stress response constitutes an uncontrolled catabolic state, 38 the risk of overestimating the patients’ caloric needs arises. Mehta et al studied critically ill ICU-treated pediatric patients, and by the use of targeted indirect calorimetry proved that the majority of patients in the pediatric ICU were indeed hypometabolic and the energy intake was excessive. 39 In a recent study by Tian et al, neonates in a state of extreme stress, as determined by C-reactive protein levels greater than 50 mg/L, with high caloric intake had significantly higher maximum serum glucose levels than the group with low caloric intake (230.33 ± 55.81 vs 135.71 ± 37.97 mg/dL). 40
Our study has some limitations. We evaluated the measurements obtained from 17 sensors placed on 16 subjects. Apart from small sample size the duration of our study was relatively short and we did not evaluate the effect of FGM on glycemic control in subjects with glucose levels outside the normal range. Despite that, our study showed that diagnostic and therapeutic procedures in the ICU, especially when performed on awake subjects, exacerbate the stress and lead to a statistically significant rise in glucose levels, however, without breaching the threshold of hyperglycemia. Further and larger studies should be conducted, in order to evaluate the clinical importance of these glucose spikes, which could justify the development of preventive strategies.
Footnotes
Abbreviations
ICU, Intensive Care Unit; CGM, Continuous Glucose Monitoring; SH, Stress Hyperglycemia; FGM, Flash Glucose Monitoring; BMI, Body Mass Index; MARD, Mean Absolute Relative Difference; SEG, Surveillance Error Grid; HOMA, Homeostatic Model Assessment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
