Abstract

Continuous glucose monitoring (CGM) systems are a critical part of diabetes management, which must perform well across a wide glucose range and when glucose is rapidly changing to allow nonadjunctive use of CGM to make treatment decisions. To standardize the evaluation of CGM performance, Dexcom collaborated with the Food and Drug Administration (FDA) to establish the integrated CGM (iCGM) category. 1 The special controls for iCGM are based on the study design and performance presented by Dexcom to the FDA. The industry standards for CGM systems were further elucidated in the Clinical and Laboratory Standards Institute (CLSI) document POCT05 2 which was developed by experts in CGM and patient care, including physicians, laboratorians, industry, academics, and government agencies. These standards outline several key attributes of clinical studies to support the performance of CGM systems including (1) evaluating CGM performance in all intended patient populations, including particularly vulnerable populations like children and pregnant individuals; (2) evaluating CGM performance across a wide range of glucose levels with a statistically relevant sample set in the hypoglycemic, euglycemic, and hyperglycemic ranges; and (3) evaluating CGM performance over different rates of change.
Dexcom commercialized the first iCGM device, the Dexcom G6, and has continued to advance the evolution of CGM with the Dexcom G7. “Accuracy and safety of Dexcom G7 CGM in adults with diabetes” 3 describes the pivotal study used to support FDA clearance of the G7. This study thoroughly evaluated the performance of the G7 in over 300 adults with type 1 or type 2 diabetes under highly rigorous conditions as outlined in the FDA iCGM special controls and POCT05, including at all glycemic conditions and when rates were rapidly changing. Data demonstrating that the G7 is highly accurate in adults 3 and children and adolescents 4 are shown in the Table 1.
Performance of the Dexcom CGM in US Pivotal Studies (Arm Placement) Calculated Using mg/dL for Reference Values <100 mg/dL and % for Reference Values ≥100 mg/dL.
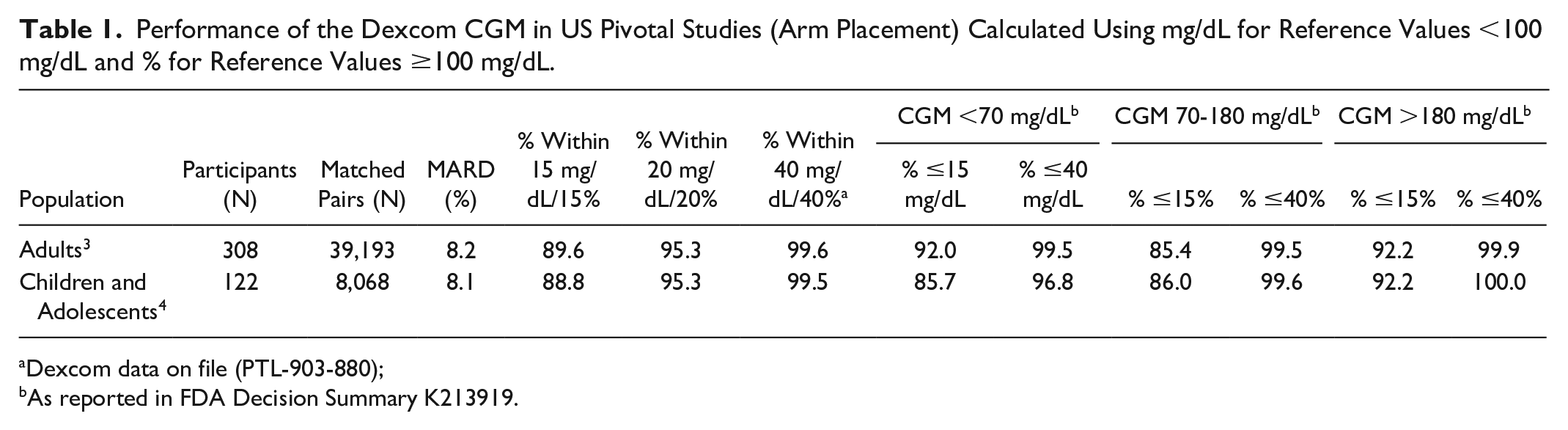
Dexcom data on file (PTL-903-880);
As reported in FDA Decision Summary K213919.
In contrast, “Comparison of Point Accuracy Between Two Widely Used Continuous Glucose Monitoring Systems” by Hanson et al 5 is an unbalanced representation of the accuracy of the Dexcom G7. This study was funded, and the data were analyzed, by Abbott, a direct competitor of Dexcom, which raises the potential of bias. In addition, we are concerned that the study was not registered on ClinicalTrials.gov. The purpose of ClinicalTrials.gov is to provide transparency in clinical research by publicly registering clinical trials and their results. Study registration is commonly required by journals to even be considered for publication.
The study design of the Hanson study was significantly flawed. The study did not incorporate glucose manipulation to evaluate sensor accuracy in hypoglycemic/hyperglycemic ranges or when glucose concentrations were rapidly changing. As a result of this study design flaw, the data included only a single value <54 mg/dL. This study only included 56 participants and was not consistent with the CLSI guidelines or the FDA special controls for iCGM. The described study design did not include a method to facilitate venous glucose to match capillary blood glucose (CBG) more closely. The reference glucometer used in the study is produced by the same manufacturer, Abbott, as one of the sensors tested, the Freestyle Libre 3. Rather than measuring accuracy based on an independent standard, the CBG data simply reflect that one Abbott device correlates more closely with another Abbott device than with a Dexcom device. The article also lacks a detailed description of the analysis including whether any data points were excluded. As noted in the study by Freckmann et al, 6 “data exclusion can have a considerable impact on the resulting performance and should therefore be described in detail.”
The only conclusions that can be drawn from the Hanson article are that details when conducting clinical trials matter, and readers must be cautious about study bias.
Footnotes
Abbreviations
CGM, continuous glucose monitoring; MARD, mean absolute relative difference; iCGM, integrated continuous glucose monitoring system; FDA, Food & Drug Administration; CLSI, Clinical and Laboratory Standards Institute; CBG, capillary blood glucose
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are employees of Dexcom, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dexcom, Inc.
