Abstract
Background:
Tirzepatide is approved by the United States Food and Drug Administration (FDA) for the management of type 2 diabetes. The efficacy and safety of this drug have not been studied in people with type 1 diabetes (T1D).
Methods:
In this single-center, retrospective, observational study, hemoglobin A1c (HbA1c), weight, body mass index (BMI), and continuous glucose monitoring (CGM) data were collected from electronic health records of adults with T1D at initiation of tirzepatide and at subsequent clinic visits over 8 months. Primary outcomes were reduction in HbA1c and percent change in body weight and secondary outcomes were change in CGM metrics and BMI over 8 months from baseline.
Results:
The mean (±SD) age of the 26 adults (54% female) with T1D was 42 ± 8 years with a mean BMI of 36.7 ± 5.3 kg/m2. There was significant reduction in HbA1c by 0.45% at 3 months and 0.59% at 8 months, and a significant reduction in body weight by 3.4%, 10.5%, and 10.1% at 3, 6, and 8 months after starting tirzepatide. Time in target range (TIR = 70-180 mg/dL) and time in tight target range (TITR = 70-140 mg/dL) increased (+12.6%, P = .002; +10.7%, P = .0016, respectively) and time above range (TAR >180 mg/dL) decreased (−12.6%, P = .002) at 3 months, and these changes were sustained over 8 months. The drug was relatively safe and well tolerated with only 2 patients discontinuing the medication.
Conclusions:
Tirzepatide significantly reduced HbA1c and body weight in adults with T1D. A randomized controlled trial is needed to establish efficacy and safety of this drug in T1D.
Introduction
The prevalence of obesity reached 42% in adults in the United States in 2020. 1 Contrary to conventional belief, the prevalence of obesity is not only common in people with type 2 diabetes (T2D) but is also common in people with type 1 diabetes (T1D). In the Epidemiology of Diabetes Interventions and Complications (EDIC) cohort, 35.9% of the patients with T1D were obese (body mass index [BMI] ≥30) and 42.4% were overweight (BMI = 25-30). 2 This increasing prevalence of obesity in T1D is of particular concern, because insulin resistance and obesity increase the risk of cardiovascular disease (CVD) and is associated with suboptimal glycemic outcomes in T1D. 3 Therefore, investigating use of adjunct medications in T1D such as glucagon-like peptide-1 receptor agonists (GLP-1RA) and sodium-glucose cotransporter-2 inhibitors (SGLT-2i) is necessary.
Many randomized trials have investigated efficacy and safety of SGLT-2i in T1D.4-6 However, this class of drug is not approved by the US Food and Drug Administration (FDA) due to increased diabetic ketoacidosis (DKA) risk in people with T1D.7,8 Previous generation once a day GLP-1RA, such as liraglutide has been investigated for its efficacy and safety in people with T1D.9-12 However, liraglutide was not filed for the FDA approval due to small glycemic benefit, which was countered by increased risk for mild ketosis.9-12 A recent real-life study showed better weight loss efficacy of GLP-1RA, compared with SGLT-2i in patients with T1D, and GLP-1RA use was not associated with increased DKA risk. 13 Similarly, another real-life retrospective study showed significant improvement in glycemic outcomes and weight loss with GLP-1RA in people with T1D. 14 These recent studies, although retrospective, suggest that GLP-1RA use was safe and associated with better glycemic outcomes and weight loss in people with T1D.
Tirzepatide is a once weekly dual agonist to glucose-dependent insulinotropic polypeptide (GIP) and GLP1 receptors. 15 It is currently FDA approved for the treatment of T2D. Several randomized clinical trials showed its efficacy on glycemic control in patients with T2D and a significant weight loss in people with and without diabetes.15-18 Moreover, weight loss efficacy of tirzepatide was superior to GLP-1RA such as liraglutide and semaglutide. 19 Tirzepatide is currently not approved for the management of T1D due to lack of evidence in this population. We investigated the efficacy and safety of tirzepatide in adults with T1D in this retrospective study.
Methods
This was a single-center retrospective study conducted at the Barbara Davis Center for Diabetes Adult Clinic. We collected electronic health records (EHRs) data of adults with T1D who were prescribed tirzepatide in our clinic between June 15, 2022 and January 30, 2023.
HbA1c, weight, and BMI data were collected from EHR on the day of tirzepatide prescription and at their subsequent clinic visits over 8 months. None of the patients were taking other oral antidiabetic medications such as metformin. We also collected their continuous glucose monitoring (CGM) and insulin pump data in CSV format prospectively every month starting from the first prescription of tirzepatide till 8 months or drug discontinuation by the patients. Total insulin dose was extracted from insulin pump download for pump users and from EHR notes for multiple daily injection (MDI) users. Safety data (severe hypoglycemia, diabetic ketoacidosis, or any potential side-effects requiring medical assistance or hospitalization) were also gathered from medical records. This retrospective study was approved by the Colorado Multiple Institutional Review Board under exempt category.
Although dose adjustments varied among patients, the lowest dose of 2.5 mg tirzepatide was the initiation dose for all patients and the dosages were up titrated by their physicians based on individual patient’s glycemic and/or weight loss goal.
Outcomes
Primary outcomes of the study were reduction in HbA1c and percent change in body weight over 8 months. Secondary outcomes were change in time in target range (TIR 70-180 mg/dL), time in tight target range (TITR 70-140 mg/dL), time in hyperglycemia (TAR >180 mg/dL), and change in BMI over time. Safety outcomes included any severe hypoglycemia, DKA, and time spent in CGM glucose below 70 mg/dL (TBR) over 8 months. The efficacy (HbA1c) and safety outcomes data were collected from EHR using the International Statistical Classification of Diseases and Related Health Problems (ICD) codes as described previously. 20
Statistical analysis
Continuous variables are reported as the mean and standard deviation (SD). Categorical variables are reported as the number and percentage of subjects with the characteristic of interest. A repeated measures analysis of variance (ANOVA) model was used for analyses of change in glycemic metrics and percent change in weight outcomes over time adjusting for their baseline value. Time of their subsequent clinic visits was treated as a categorical variable.
All analyses were performed using SAS software version 9.4 (SAS Institute, Cary, North Carolina). P values < .05 were considered significant.
Results
Between June 2022 and January 2023, 26 adults with T1D were prescribed tirzepatide. The baseline characteristics of the participants in presented in Table 1. In brief, mean age of the participants was 42 ± 8 years with BMI of 36.7 ± 5.3 kg/m2 and 54% were female.
Baseline Characteristics of the Participants (n = 26).
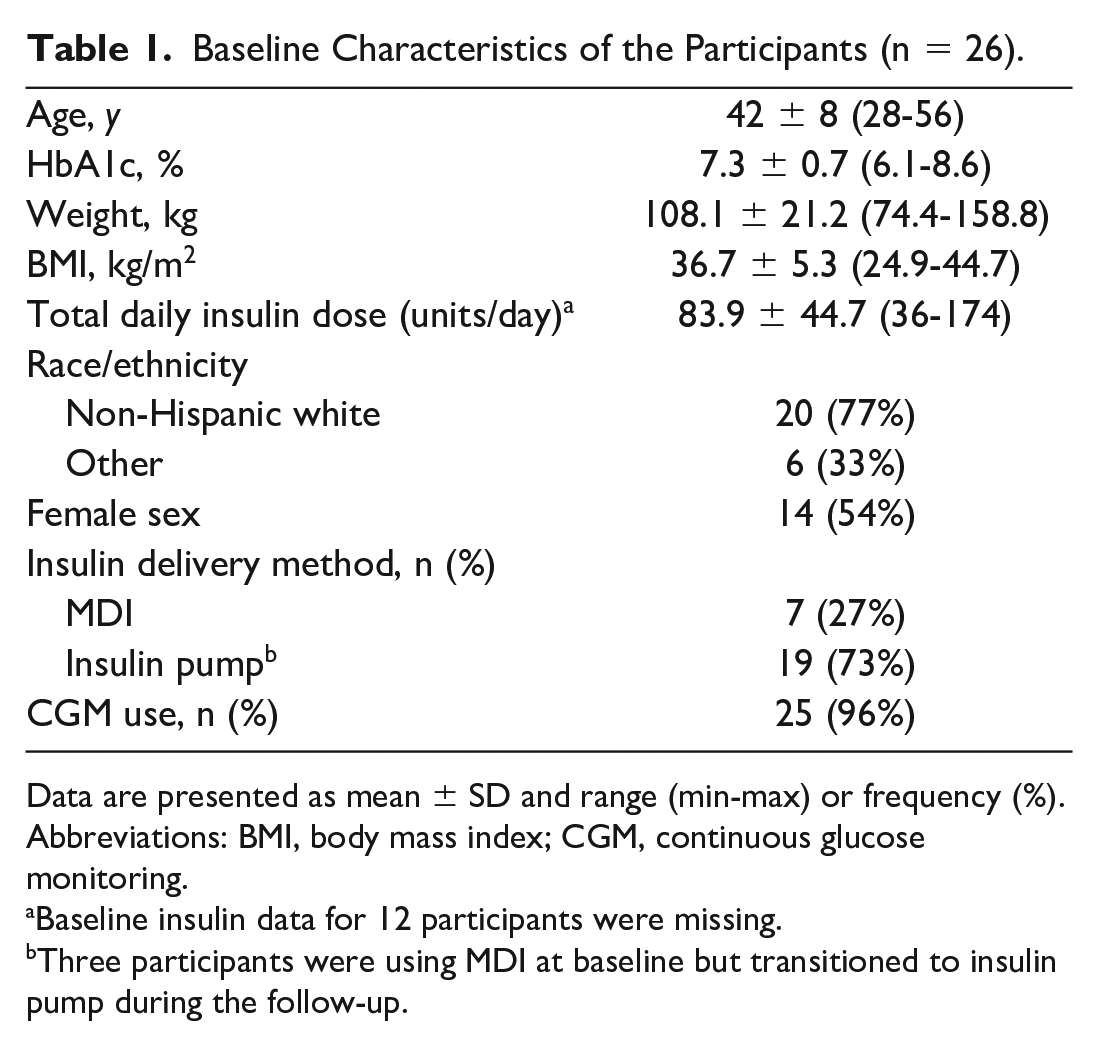
Data are presented as mean ± SD and range (min-max) or frequency (%).
Abbreviations: BMI, body mass index; CGM, continuous glucose monitoring.
Baseline insulin data for 12 participants were missing.
Three participants were using MDI at baseline but transitioned to insulin pump during the follow-up.
Out of 26 patients who started tirzepatide, 25, 25, and 24 patients were still using the medication at 3, 6, and 8 months, respectively. At 3 months, 22 patients were on 2.5 mg and 3 patients were on 5 mg dose. At 6 months, 12 patients were on 7.5 mg dose and others varied from 2.5 to 12.5 mg doses. At 8 months, 7 patients each were on 7.5 mg and 10 mg doses and other patients were on varied doses from 5 to 15 mg.
Primary Outcomes
There was significant reduction in HbA1c by 0.45% at 3 months and sustained over 8 months (Figure 1a and Table 2). The reduction in HbA1c was seen within the first 3 months (0.45%, P = .027), and further significant reduction in HbA1c was very modest. There was a significant reduction in weight over 8 months. From baseline, the percent change in weight was −3.4% at 3 months and −10.5% at 6 months (Figure 1b and Table 2). After 6 months, there was no further reduction in body weight. A mixed-effect model using data from all time points showed a significant association between HbA1c and body weight over time (P = .01). One percent change in body weight was associated with a 0.05% change in HbA1c (P = .004) indicating more weight loss was associated with more reduction in HbA1c. When adjusted for the change in weight, the reduction in HbA1c at 3 and 6 months from baseline was still significant (−0.69%, P = .002; −0.40%, P = .02, respectively).
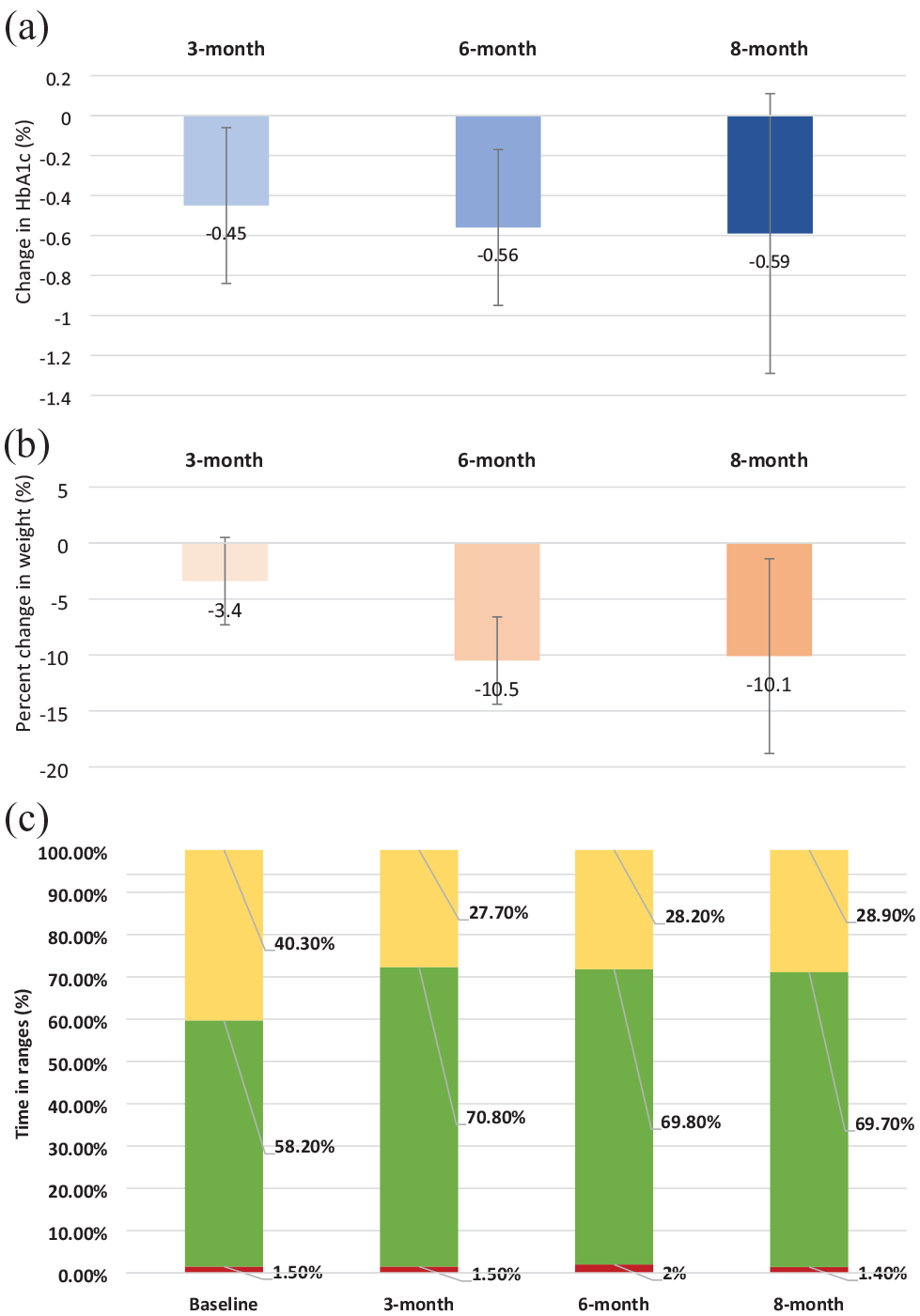
(a) Reduction in HbA1c at 3, 6, and 8 months. (b) Percent change in weight at 3, 6, and 8 months. (c) Time below 70 mg/dL (red), time in 70 to 180 mg/dL (green), time above 180 mg/dL (yellow) at the baseline, 3, 6, and 8 months. Confidence intervals were shown with whiskers on each bar.
Change in Glycemic and Weight Outcomes Over 8 Months.
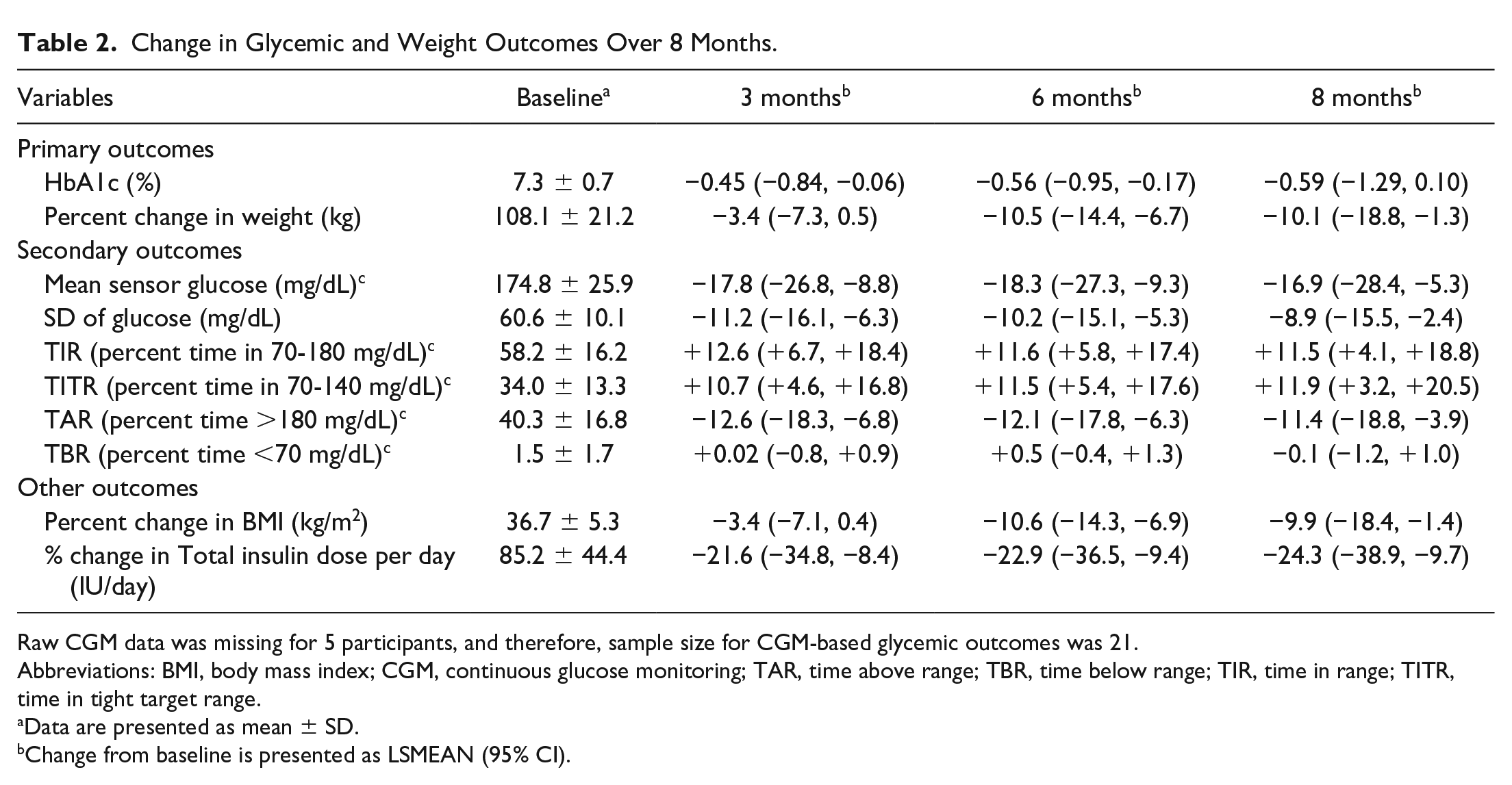
Raw CGM data was missing for 5 participants, and therefore, sample size for CGM-based glycemic outcomes was 21.
Abbreviations: BMI, body mass index; CGM, continuous glucose monitoring; TAR, time above range; TBR, time below range; TIR, time in range; TITR, time in tight target range.
Data are presented as mean ± SD.
Change from baseline is presented as LSMEAN (95% CI).
Secondary Outcomes
There was significant improvement in TIR by 12.6% at 3 months (P = .0002) and sustained over 8 months. Most of the improvement in euglycemia was due to a significant reduction in hyperglycemia, as TAR reduced by 12.6% at 3 months (P = .0002) (Figure 1c and Table 2). Time in tight target range (70–140 mg/dL) improved significantly from baseline by 10.7% at 3 months (P = .0016) and 11.5% at 6 months (P = .0008). Interestingly, improvement in HbA1c as well TIR was observed within the first 3 months of tirzepatide use, without further significant improvement in glycemia from 3 to 8 months despite significant weight loss noted between 3 and 6 months. There was a significant percent change in BMI from baseline at 3 months (−3.35%) and at 6 months (−10.5%). The total daily insulin dose at the baseline (mean = 85.2 IU/day) reduced by 21.6 (95% confidence interval [CI] = −34.8, −8.4) IU/day at 3 months and sustained at 6 and 8 months as −22.9 (95% CI = −36.5, −9.4) and −24.3 (95% CI = −38.9, −9.7) IU/day, respectively (Table 2).
Safety Outcomes
There was no change in TBR despite significant improvement in TIR and reduction in body weight (Figure 1c). Severe hypoglycemia event (n = 1) and severe constipation (n = 1) led to discontinuation of the medication in 2 patients. Peroneal nerve palsy (foot drop) possibly due to rapid weight loss was reported in 1 person. No DKA event was reported during the 8-month follow-up.
Discussion
To our knowledge, this is the first study reporting efficacy and safety of tirzepatide in adults with T1D. Our pilot study found that tirzepatide was associated with significant improvement in glycemic outcomes and reduction in weight. There was no increase in TBR. Glycemic improvements were seen within the first 3 months. There was no clinically significant improvement in HbA1c beyond 3 months, despite continuous reduction in weight. These data suggest that glycemic efficacy can be achieved in a short time frame with 2.5 to 5 mg/week dose without further significant improvement with higher dosage. However, weight loss efficacy of the drug was persisting for a longer time and presumably dose dependent. The change in total daily insulin dose was also significant at 3 months, and minimal change was observed beyond 3 months. We did not collect systematic changes in the basal and/or bolus doses. It would be interesting to evaluate change in basal and bolus doses systematically to generate evidence-based insulin titration guidance for GLP-1RA use in T1D.
Interestingly, we noticed that few individuals did not respond well to tirzepatide with either glucose lowering or weight loss. On further investigation, we found that certain genetic mutations that may be implicated in obesity and less or no response to GLP-1RA or dual agonists like tirzepatide. Detailed investigation on association between potential genetic form of obesity and response to GLP-1RA therapy will be published separately.
Our study provides an initial proof of concept that tirzepatide has potential to improve glycemic outcomes and reduce weight significantly. Tirzepatide was relatively well tolerated. There was 1 severe hypoglycemia event occurred during the 8-month follow-up of 26 people (3.8%). This is lower than the prevalence of severe hypoglycemia (6%) reported in the US T1D Exchange registry. 21 Future clinical trials are warranted to evaluate the benefit-risk ratio of tirzepatide in T1D.
Interpretation of data from our study is limited given it was a retrospective study with a small sample size. Single center, selection bias of participants, lack of controls, and retrospective EHR-based data collection are additional limitations. Dose titration was not uniform, and this indicates physicians may have adjusted dose based on glycemic and/or weight loss goal of the individual and/or dose availability given the shortage of drugs during the time frame of our study. These different drug dose administrations may have confounded the outcomes. Moreover, we did not collect data on blood pressure and lipid levels. Future studies are needed to evaluate the effects of tirzepatide on cardiovascular risk factors in people with T1D.
In summary, tirzepatide in adjunct to insulin therapy may help to improve glycemic control and reduction in weight without excess risk for hypoglycemia in adults with T1D. Randomized clinical trials (phase 2 and phase 3) are warranted to evaluate safety and efficacy of this drug in people with T1D.
Footnotes
Acknowledgements
The authors thank Christie Beatson, Sean Walker and Matthew Klein for help with data collection and data cleaning.
Author Contributions
VNS designed the study and collected the data. FD and JKS-B conducted statistical analysis. VNS, HKA, and KEK drafted the manuscript; all authors edited and approved the manuscript. VNS is the guarantor of this work and, as such, has full access to all the data in the study and takes responsibility for the integrity of the data and accuracy of the data analysis.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HKA received research support through University of Colorado from Dexcom, Tandem Diabetes, Senseonics, Medtronic, Eli Lilly, REMD Biotherapeutics, IM Therapeutics, and IAFMS and received honoraria through University of Colorado from Senseonics and Mannkind for advisory board attendance. VNS received research support from Novo Nordisk, Insulet, and Tandem Diabetes Care and received honoraria from LifeScan for advisory board attendance and from Dexcom, Embecta, and Insulet for speaking arrangements. FD, JKS, and KEK do not report any conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
