Abstract

Introduction
Manufacturing of a new medical device requires consideration of numerous factors besides whether there will be patient demand. The manufacturer must consider design control, the product development process, and manufacturing design issues, many of which will not become important for several years into the future when product development reaches specific critical stages on the development pathway. Because of these considerations that must be addressed, in addition to patient preferences, we contend that for manufacturing a new medical device, the manufacturer must consider last things first.
New medical devices represent the combined efforts of many different people with different skills. Usually, the most credit goes to the inventors and developers; however, even the greatest new technology/concept will not make it to the market if the device cannot be manufactured adequately.1,2 The question is, what does adequately mean? Primarily this means cost aspects, which are driven by many different considerations such as total addressable and target markets, competitive situation, target selling price, required quality, and quantities. From a more general point of view, it appears as if manufacturing is regarded as a purely technical task that is of limited criticality and can be simply done once a product has been developed, which usually takes several years. However, the “failure” of several medical device developments in the last years/decades indicates the shortcomings of such an approach. If manufacturing aspects are not adequately taken into account in good time while the product is designed and developed, this can be “doomsday” years before this becomes obvious.
This editorial aims to raise awareness of the importance of timely consideration of manufacturing aspects in the early product development stage of a medical device. Interestingly enough, this is of relevance not only for start-ups (which might be the first thought) but also for established medical device companies. These might take over innovative devices from start-ups without paying sufficient attention to manufacturing aspects.
Design Control
Regulatory authorities, for example, the US Food and Drug Administration (FDA), pay much attention to the development process of medical devices to ensure that devices are designed and developed in a controlled process and that the quality and usability of such devices are sufficient and do not represent any risk to the users. In the “FDA’s Design Control Guidance for Medical Device Manufacturers,” the effective implementation of design controls is described in some detail 3 (Figure 1). In brief, requirements from different stakeholders are developed, and a device is designed and developed to meet those requirements. The design is then evaluated, transferred to production, and the device is manufactured and commercialized.
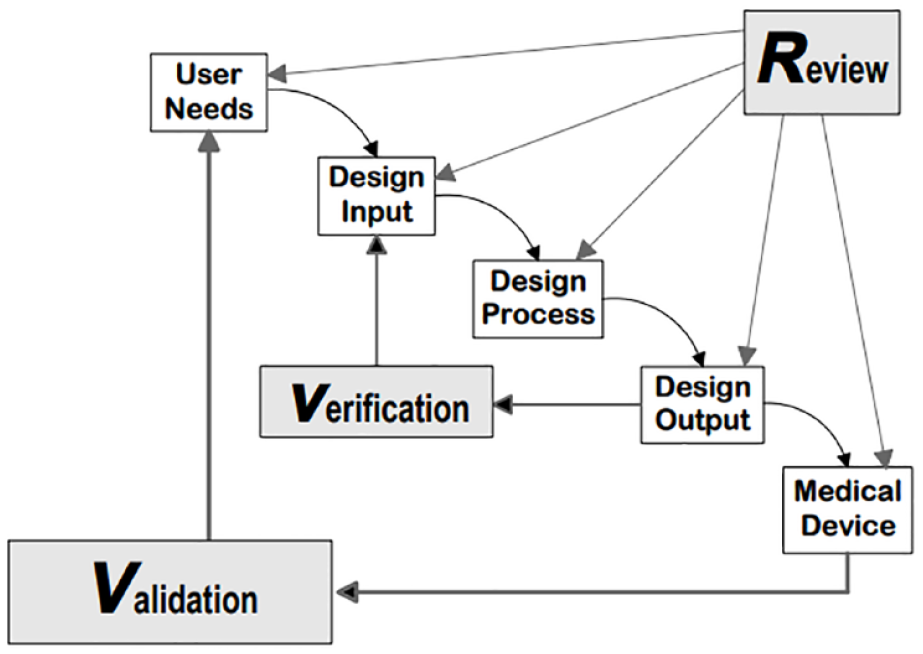
US Food and Drug Administration’s design control guidance for medical device manufacturers. 3
Product Development Process
Manufacturers of medical devices must implement the design control requirements into the product development process, which is part of the organization’s quality management system (QMS). To obtain product approval, compliance must be demonstrated by providing objective evidence that these processes were followed during product development. A very schematic overview of a product development process is shown in Figure 2 (top). Developing and negotiating the requirements of the various stakeholders is an important activity that takes place at the beginning of the project. Customer requirements are derived from unmet user needs and ensure that the product will be liked by customers and users (Figure 2, bottom). Regulatory requirements form the basis for compliance and thus for a successful later product filing. Business requirements ensure that the product will be profitable during routine production and commercialization. The forecasted volumes and target manufacturing costs are important figures and form input for the manufacturing requirements. To estimate these figures, assumptions about key business goals such as target market, competitive situation, selling price, required quality, and quantities for the commercialization phase after the launch of the product must be made. The forecasted volumes and manufacturing costs are then a crucial input for the manufacturing requirements which ensure that the design of the product enables a manufacturing process that allows profitable production at the required costs, quantities, and quality.
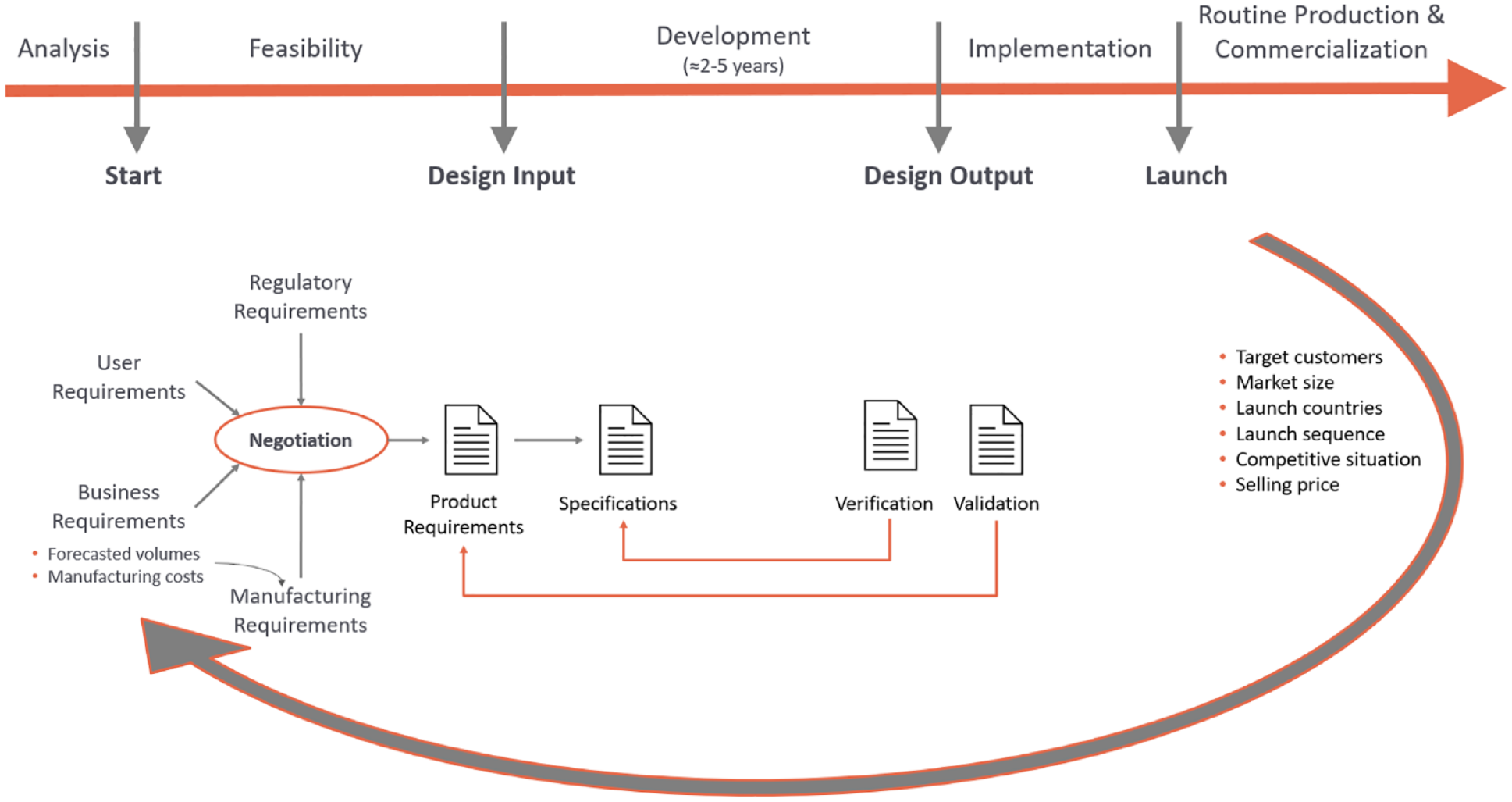
Development process.
Even if the medical device is developed using incremental, evolutionary agile methods, as opposed to the traditional waterfall process shown in Figure 2 (top), the requirements for later quantities and target manufacturing costs are already needed in the early design sprints. Revising the design in later sprints would also be very expensive and time-consuming.
The FDA now mandates a Total Product Life Cycle approach for monitoring medical devices from device design and development to real-world use of the device. This approach informs the FDA’s postmarket and compliance decisions by leveraging knowledge from premarket data and helps with premarket decisions by leveraging knowledge from postmarket data and compliance activities (https://www.fda.gov/about-fda/cdrh-transparency/total-product-life-cycle-medical-devices).
Design for Manufacturing
Design for manufacturing is the general engineering practice of designing devices by integrating product design and process planning into one common activity so that they can be easily produced in the desired quality and quantity and at reasonable costs. Otherwise, the product is not profitable for the manufacturer. The product is designed at the beginning of the development process after all stakeholders have agreed on the product requirements. In other words, at the beginning of product development, you must have the end—the routine production and commercialization phase—in mind to make the right design decisions. This is particularly crucial for big and cost-sensitive markets. If this is not done at the right time, a perfectly functioning product that customers would like and that has just been approved by the regulators may not be manufacturable in the quantities, quality, and cost required to make the product profitable—because the design of the product does not support the required production processes. The later the need for changing product design is noticed in the product development process, the more expensive and time-consuming it is to adapt the design of the product, even though changes early in design which can even have a large impact tend to not add much cost. The relationship between the impact and cost of change on product design and manufacturing complexity at various stages of production and launch of a new product is illustrated in Figure 3. If too many small changes or a significant change is required, it may even be more cost-effective to restart the development process from scratch.
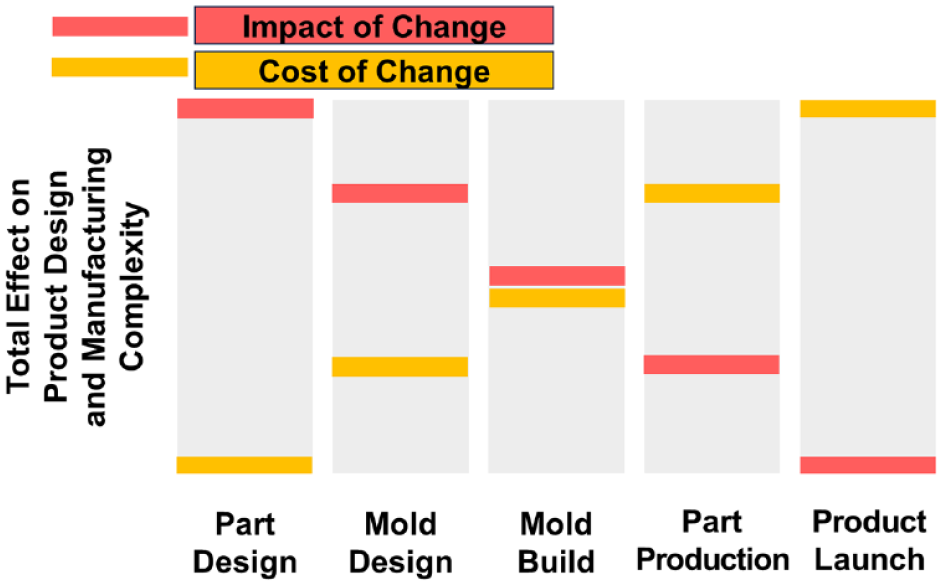
The impact and cost of change on product design and manufacturing complexity at various stages of production and launch of a new product.
The Challenges
Depending on the product, development time can be between two and five years or even longer, and it is not easy to predict how the market and competitive situation will develop over this period. Especially, as the diabetes care market is also changing ever faster, it might be difficult to make the right assumptions about the forecasted quantities and target manufacturing costs at the beginning of the product development. In addition, thorough manufacturing experience is needed to make the right design decisions and production know-how is not one of the typical skills of a start-up by nature. The complexities regarding manufacturing can be different depending on the volume of a given device that shall be manufactured (low volume vs high volume) and the nature of the device (continuous glucose monitoring devices, insulin pumps, infusion sets, patch pumps, insulin pens).
Start-ups often focus on the challenges of the present; they do a great job of identifying unmet user needs and finding innovative technical solutions but tend to put off the challenges of the future, such as the later manufacturing of the product. Well-established manufacturers of medical devices in contrast understand how to refine existing devices because of the established processes for eliciting and negotiating requirements. However, these large companies struggle with innovative devices because the requirements experts are isolated in organizational silos, such as business, regulatory affairs, production, quality, product management, marketing, and R&D, and it is sometimes difficult to bring them together to understand innovative devices and elicit and negotiate appropriate requirements.
Insulin Pumps and the GlucoWatch
In the last decade, several companies have tried to develop patch insulin pumps. It is of interest to note that these are technically very complex devices with high demands on their performance. Miniaturization, robustness, low tolerances, water tightness, power management, insulin compatibility, biocompatibility, sterilization, and friction forces are all aspects that have to be addressed. In turn, high-volume manufacturing and low-price requirements are additional challenges for the product design. The GlucoWatch met a demand for noninvasive glucose monitoring, but design problems led to poor patient acceptance. These were examples of start-ups, which developed innovative solutions for medical devices but failed to consider the importance of product design for manufacturability in a realistic time period. If a start-up is taken over by an established manufacturer, then a necessary costly and time-consuming redesign for manufacturability is often the reason why targeted launch dates have to be postponed. In addition, too many companies involved do not do the product any good. Transfer of know-how and equipment always takes longer than one would assume. In addition, different cultures, mindsets, processes, and QMSs make the transfer of a product from one company to the other tricky. The “Not-invented-here” syndrome is another obstacle to transferring a product from one company to another.
Summary and Outlook
At the very beginning of the product development, the end has to be in mind to make the right design decisions. The importance of a product design that is ready for future scale-up is often underestimated. This holds particularly for large and price-sensitive markets. The product may be loved by customers, may be functional, and meet regulatory requirements, but may not be manufacturable at a reasonable cost and in sufficient quantities.
Footnotes
Acknowledgements
We thank David Klonoff for his excellent comments.
Abbreviations
AID, automated insulin delivery; CGM, continuous glucose monitoring; DFM, design for manufacturing; PwD, patients with diabetes; QMS, quality management system.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MS is an employee of PharmaSens AG. LH is a shareholder of the Profil Institut für Stoffwechselforschung GmbH, Neuss, Germany. LH and MS are consultants for several companies that are developing novel diagnostic and therapeutic options for diabetes treatment.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
